Abstract
OBJECTIVE
In humans, it is unclear if insulin resistance at the monocyte level is associated with atherosclerosis in vivo. Here we have studied first-degree relatives of patients with type 2 diabetes to investigate whether a reduction in components of the insulin signal transduction pathways, such as the insulin receptor (InsR) or InsR substrate 1 or 2 (IRS1 or IRS2), or a reduction in genetic modifiers of insulin action, such as the TIMP3/ADAM17 (tissue inhibitor of metalloproteinase 3/A disintegrin and metalloprotease domain 17) pathway, is associated with evidence of atherosclerosis.
RESEARCH DESIGN AND METHODS
Insulin sensitivity was analyzed through euglycemic-hyperinsulinemic clamp, and subclinical atherosclerosis was analyzed through intimal medial thickness. Monocytes were isolated through magnetic cell sorting, and mRNA and proteins were extracted and analyzed by quantitative PCR and pathscan enzyme-linked immunosorbent assays, respectively.
RESULTS
In monocyte cells from human subjects with increased risk for diabetes and atherosclerosis, we found that gene expression, protein levels, and tyrosine phosphorylation of IRS2, but not InsR or IRS1, were decreased. TIMP3 was also reduced, along with insulin resistance, resulting in increased ectodomain shedding activity of the metalloprotease ADAM17.
CONCLUSIONS
Systemic insulin resistance and subclinical atherosclerosis are associated with decreased IRS2 and TIMP3 expression in circulating monocytes.
Insulin resistance and compensatory hyperinsulinemia, both consequences of obesity and lipotoxicity, are well-known risk factors for the development of atherosclerosis (1). Recent findings from animal models outlined that a combination of insulin resistance and hyperinsulinemia promote changes in the liver’s ability to regulate lipoprotein release, which leads to atherogenic dyslipidemia (2). Insulin resistance is causative of unbalanced regulation of transcription factors FoxO1 and FoxA2, which act on the control of VLDL synthesis, whereas hyperinsulinemia may favor lipogenesis through upregulation of transcription factor SREBP2c activity (3). However, atherosclerosis is also an inflammatory process, and how insulin resistance and hyperinsulinemia act in the regulation of innate and adaptive immunity is still debated (4). Data from experimental models have yielded contradictory results. In fact, whereas liver-specific insulin resistance promotes atherosclerosis (5), genetic models of insulin resistance in monocytes, the master regulator of atherosclerotic plaque progression, were found to either promote or prevent lipid accumulation in aortas depending on the strain of mice used (6,7). Nevertheless, the activation of monocytes and their propensity to differentiate into macrophages contributes to insulin resistance in different tissues as well as atherosclerosis (8).
In humans, it is unclear if insulin resistance at the monocyte level is associated with insulin resistance and atherosclerosis in vivo. Here we have used the offspring of patients with type 2 diabetes, a well-characterized model to study how metabolic factors influence vascular homeostasis (9,10), to investigate whether a reduction in components of the insulin signal transduction pathways, such as the insulin receptor (InsR) or InsR substrate 1 or 2 (IRS1 or IRS2), or genetic modifiers of insulin action, such as the TIMP3/ADAM17 (tissue inhibitor of metalloproteinase 3/A disintegrin and metalloprotease domain 17) pathway, is associated with evidence of atherosclerosis.
RESEARCH DESIGN AND METHODS
The study was approved by the local ethics committee and the reported investigations were carried out in accordance with the principles of the Declaration of Helsinki as revised in 2000. An informed written consent was obtained from all participants. A total of 41 unrelated, first-degree relatives (FDRs) of type 2 diabetic patients were enrolled at the Hospital Policlinico Tor Vergata. Inclusion criteria were the eldest offspring in each family with only one parent affected by type 2 diabetes, absence of diabetes (fasting plasma glucose <7.0 mmol/L and/or 2-h post oral glucose tolerance test <11.1 mmol/L), and absence of diseases able to modify glucose metabolism. The clinical characteristics of the parents are included in Supplementary Table 1. On the first day, after 12 h of fasting, FDRs underwent anthropometrical evaluation, including BMI and waist circumference, measurements of clinic blood pressure after 5 min of quiet rest, a 75-g oral glucose tolerance test, measurements of the intima-media thickness (IMT) of the common carotid artery, and evaluation of the endothelial function as previously described (8,9). On the second day, after a 12-h overnight fast, subjects underwent a euglycemic hyperinsulinemic clamp study as previously described (9).
Enzyme-linked immunosorbent assay.
Serum samples were analyzed by a sandwich enzyme-linked immunosorbent assay (ELISA) method using anti-mouse antibodies for human soluble tumor necrosis factor (TNF) receptor 1, soluble interleukin 6 (IL-6) receptor, soluble vascular cell adhesion molecule 1 (sVCAM-1), soluble intercellular cell adhesion molecule 1 (sICAM-1), soluble chemokine (C-X-C motif) ligand 16, and sCXC3L1 (all from R&D Systems, Minneapolis, MN) according to manufacturer’s instructions.
Monocyte isolation for RNA extraction and real-time quantitative RT-PCR analysis.
Peripheral blood mononuclear cells (PBMCs) were isolated from 40 mL of peripheral blood of FDRs using Ficoll-Plaque Plus (GE Healthcare, Piscataway, NJ) as indicated by the manufacturer. Untouched monocytes were isolated from PBMCs by using an indirect magnetic labeling system (Monocyte Isolation Kit II; Miltenyi Biotec, Auburn, CA). About 2.5–9.7 × 106 monocytes were obtained from 40 mL of peripheral blood. Total RNA was extracted from monocytes with the RNeasy Micro Kit (Qiagen, Valencia, CA) according to the manufacturer’s instructions. Single-strand cDNA was synthesized, according to the Applied Biosystems (Foster City, CA) standard protocol, from 1 μg of total RNA sample using High-Capacity cDNA Archive Kit. Fifty nanograms of cDNA was amplified by real-time PCR RNA expression of TNF-α converting enzyme/ADAM17, ADAM10, TNF-α, TIMP3, lectin-like oxidized low-density lipoprotein receptor-1 (LOX1), chemokine (C-C motif) receptor 2 (CCR2), matrix metalloproteinase 9 (MMP9), InsR, IRS1, IRS2, or sirtuin 1 (SirT1) (primers available upon request) using an ABI PRISM 7900Ht system (Applied Biosystems) and normalized to 18S rRNA as an endogenous control. Each reaction was carried out in duplicate and analysis was performed by 2−ΔΔCt method.
Protein extraction and pathscan sandwich ELISA assay.
For protein extraction, monocytes were isolated as described above. Cells (2.5 × 106) were plated in Dulbecco’s modified Eagle’s medium with 0.2% BSA in the absence of serum for 6 h and then stimulated with insulin (10−7 mol/L) for 10 min. At the end of the treatment, in order to assay insulin signaling components, proteins were extracted and analyzed by pathscan sandwich ELISA kits (Cell Signaling, Beverly, MA) according to the manufacturer’s instructions. The following pathscan sandwich ELISA kits were used: phospho-InsR (PanTyr) and total InsR; phospho-IRS1 (PanTyr) and total IRS1; phospho-IRS2 (PanTyr) and total IRS2; and phospho-v-akt murine thymoma viral oncogene homolog 1 (AKT1) (Ser473) and total Akt1.
ADAM17 activity assay.
Protein extracts were used to determine the ADAM17 activity by the SensoLyte 520 ADAM17 activity assay kit (Fluorimetric; AnaSpec, San Jose, CA) according to the manufacturer’s instructions.
Statistical analysis.
The Kolmogorov-Smirnov test was used to test the normality of distribution, and nonnormally distributed variables, including all mRNA values, were naturally log transformed. Group differences of continuous variables were compared using Student t test or univariate ANOVA test with Bonferroni post-hoc test. Relationships between variables were determined by Pearson correlation coefficient (r). Continuous data are expressed as means ± SD. P < 0.05 was considered statistically significant. All analyses were performed using SPSS software program version 13.0 for Windows. Graphs were designed using GraphPad Prism 5.0 for Windows.
RESULTS
Correlation between insulin resistance, sublinical atherosclerosis, and gene expression in monocytes from offspring of type 2 diabetic subjects.
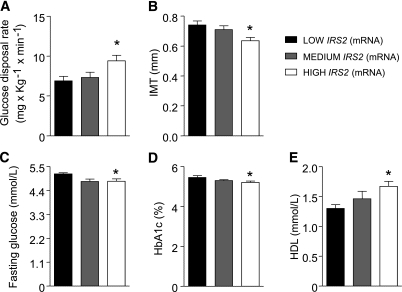
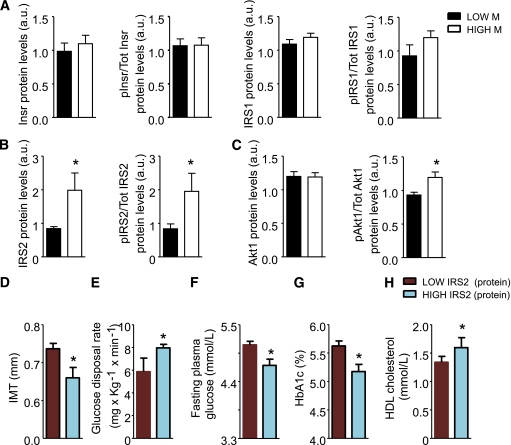
By analyzing clinical characteristics and metabolic parameters of FDRs of type 2 diabetes (anthropometric, biochemical, and clinical parameters are shown in Table 1), we found that insulin resistance, evaluated as glucose disposal rate (M) during a euglycemic hyperinsulinemic clamp, negatively correlates with IMT (r = −0.575; P < 0.0001), and dividing our population into tertiles of insulin resistance assessed by M clamp, we also confirmed that the insulin-resistant tertile has higher IMT values, confirming the validity of this model to study biomarkers linking insulin resistance to atherosclerosis (9,10) (Supplementary Fig. 1A and B). We analyzed the mRNA expression levels of InsR, IRS1, IRS2, and SirT1. No correlation was found between levels of InsR, IRS1, and SirT1 expression with clinical and biochemical parameters (data not shown). However, we found in this group of subjects that IRS2 expression correlated with fasting plasma glucose (r = −0.34; P < 0.05), insulin resistance assessed by M (r = 0.406; P < 0.01), glycated hemoglobin (r = −0.36; P < 0.05), HDL cholesterol (r = 0.28; P < 0.05), and IMT (r = −0.49; P < 0.001) (data not shown). We observed that subjects in the lowest tertiles of expression of IRS2 in monocytes were significantly more insulin resistant and had the highest IMT level (Fig. 1A and B and Supplementary Table 2); accordingly those with low IRS2 had increased fasting plasma glucose and HbA1c and reduced levels of HDL cholesterol (Fig. 1C–E and Supplementary Table 2). Next we analyzed the expression of genes relevant for inflammation, such as TNF-α, CCR2, LOX1. Similarly, we did not find correlation between levels of gene expression and clinical and biochemical parameters in this group of subjects. Because mRNA expression could not reflect protein regulation at the membrane and cytoplasmic level, we analyzed the levels of InsR, IRS1, and IRS2 in the lowest versus the highest tertiles of insulin resistance. Our analysis confirmed that in cultured monocytes of insulin-resistant subjects, IRS2 protein and tyrosine phosporylation levels were significantly decreased (Fig. 2B), whereas no differences were observed for InsR and IRS1 in both protein expression and phopsphorylation (Fig. 2A). Interestingly, whereas Akt1 protein levels were similar in the two groups, Akt serine 473 phosphorylation reflected the reduced IRS2 activation (Fig. 2C). Analysis of the subjects with lowest IRS2 protein levels in monocytes confirmed that these were significantly more insulin resistant and had the highest IMT level (Fig. 2D–H), with results similar to stratification of subjects with IRS2 mRNA levels.
TABLE 1.
Distribution of anthropometric and biochemical parameters and monocyte gene expression data according to tertiles of insulin resistance
| All subjects | Insulin resistance |
P value | |||
|---|---|---|---|---|---|
| Low | Medium | High | |||
| Subjects (n) | 41 | 13 | 14 | 14 | |
| Sex (men/women) | 13/28 | ||||
| Age (years) | 36.5 ± 10.2 | 32.7 ± 10.9 | 35.1 ± 8.8 | 41.6 ± 9.5 | NS |
| BMI (kg/m2) | 25.00 ± 4.3 | 22.1 ± 2.4 | 25.00 ± 3.4 | 27.2 ± 3.4 | 0.001 |
| Waist (cm) | 81.9 ± 11.9 | 73.3 ± 5.0 | 83.9 ± 11.0 | 93.0 ± 11.4 | 0.001 |
| Hypertension (yes/no) | 6/35 | ||||
| NGT/IGT | 35/6 | ||||
| Systolic blood pressure (mmHg) | 119.4 ± 13.3 | 115.9 ± 11.1 | 120.0 ± 15.5 | 120.0 ± 19.1 | NS |
| Diastolic blood pressure (mmHg) | 79.3 ± 8.4 | 75.4 ± 8.2 | 81.2 ± 8.0 | 78.6 ± 9.4 | NS |
| Total cholesterol (mmol/L) | 4.92 ± 1.07 | 4.54 ± 1.02 | 4.86 ± 0.98 | 5.32 ± 1.12 | NS |
| HDL cholesterol (mmol/L) | 1.52 ± 0.40 | 1.79 ± 0.47 | 1.50 ± 0.34 | 1.30 ± 0.24 | 0.005 |
| LDL cholesterol (mmol/L) | 3.08 ± 1.08 | 2.57 ± 0.80 | 2.91 ± 1.01 | 3.72 ± 1.12 | 0.01 |
| Triglycerides (mmol/L) | 1.18 ± 0.77 | 0.77 ± 0.27 | 1.07 ± 0.97 | 1.66 ± 5.09 | 0.005 |
| HbA1c (%) | 5.32 ± 0.27 | 5.23 ± 0.2 | 5.34 ± 0.28 | 5.49 ± 0.28 | 0.05 |
| Fasting plasma glucose (mmol/L) | 5.0 ± 0.4 | 5.0 ± 0.3 | 4.8 ± 0.4 | 5.1 ± 0.4 | NS |
| 2 h p-l plasma glucose (mmol/L) | 6.6 ± 1.3 | 5.4 ± 0.9 | 6.9 ± 1.9 | 7.0 ± 1.4 | 0.01 |
| Fasting plasma insulin (pmol/L) | 60 ± 35 | 44 ± 22 | 47 ± 22 | 89 ± 39 | 0.001 |
| 2 h p-l plasma insulin (pmol/L) | 325 ± 248 | 212 ± 22 | 267 ± 104 | 510 ± 392 | 0.007 |
| HOMA IR index | 2.27 ± 1.42 | 1.65 ± 0.88 | 1.66 ± 0.73 | 3.46 ± 1.62 | 0.0001 |
| Glucose disposal index (mg ⋅ kg−1 ⋅ min−1) | 7.88 ± 2.37 | 10.60 ± 1.41 | 7.875 ± 0.54 | 5.49 ± 1.27 | 0.0001 |
| Fibrinogen (mg/dL) | 304.23 ± 81.29 | 305.92 ± 110.41 | 311.58 ± 68.01 | 299.11 ± 70.47 | NS |
| C-reactive protein (mg/dL) | 4.18 ± 9.14 | 6.8 ± 14.26 | 2.34 ± 3.15 | 2.7 ± 2.29 | NS |
| Free fatty acids (mmol/L) | 0.47 ± 0.21 | 0.46 ± 0.17 | 0.38 ± 0.22 | 0.58 ± 0.13 | NS |
| sVCAM-1 (ng/mL) | 246.01 ± 337.75 | 145.21 ± 163.53 | 267.87 ± 225.82 | 324.93 ± 518.41 | NS |
| sICAM-1 (ng/mL) | 166.43 ± 51.97 | 181.25 ± 66.77 | 152.76 ± 30.79 | 164.24 ± 51.31 | NS |
| sTNF R1 (ng/mL) | 1.27 ± 0.30 | 1.28 ± 0.33 | 1.26 ± 0.13 | 1.28 ± 0.4 | NS |
| sIL6 R (ng/mL) | 26.98 ± 9.27 | 25.01 ± 8.82 | 25.57 ± 10.61 | 30.70 ± 7.86 | NS |
| Soluble chemokine (C-X-C motif) ligand 16 (ng/mL) | 4.11 ± 0.88 | 4.25 ± 1.05 | 4.01 ± 0.59 | 4.08 ± 0.93 | NS |
| Fractalkine (ng/mL) | 3.63 ± 8.98 | 4.94 ± 11.62 | 5.21 ± 10.38 | 0.73 ± 1.32 | NS |
| IMT (mm) | 0.70 ± 0.09 | 0.66 ± 0.09 | 0.67 ± 0.06 | 0.77 ± 0.11 | 0.008 |
| FMD | 12.43 ± 6.84 | 11.22 ± 5.25 | 13.08 ± 8.99 | 12.86 ± 6.71 | NS |
| Log IR | 0.57 ± 0.41 | 0.71 ± 0.29 | 0.49 ± 0.46 | 0.51 ± 0.44 | NS |
| Log IRS1 | 0.22 ± 0.72 | 0.36 ± 0.47 | 0.14 ± 0.94 | 0.17 ± 0.91 | NS |
| Log IRS2 | 0.09 ± 0.29 | 0.20 ± 0.28 | 0.09 ± 0.31 | 0.02 ± 0.30 | 0.05 |
| Log TIMP3 | −0.12 ± 0.63 | 0.14 ± 0.55 | −0.22 ± 0.70 | −0.26 ± 0.64 | 0.05 |
| Log TACE | −0.11 ± 0.25 | −0.06 ± 0.25 | −0.17 ± 0.26 | −0.10 ± 0.25 | NS |
| Log ADAM10 | 0.48 ± 0.35 | 0.64 ± 0.32 | 0.33 ± 0.32 | 0.56 ± 0.29 | NS |
| Log MMP9 | −0.07 ± 0.30 | −0.07 ± 0.30 | −0.15 ± 0.26 | −0.08 ± 0.27 | NS |
| Log Sirt1 | 0.56 ± 0.35 | 0.62 ± 0.29 | 0.47 ± 0.42 | 0.62 ± 0.29 | NS |
| Log CCR2 | −0.06 ± 0.32 | 0.11 ± 0.29 | −0.12 ± 0.36 | −0.09 ± 0.32 | NS |
| Log TNF-α | −0.52 ± 0.54 | −0.57 ± 0.57 | −0.94 ± 0.26 | −0.26 ± 0.52 | NS |
| Log LOX1 | −0.36 ± 0.41 | −0.34 ± 0.38 | −0.44 ± 0.42 | −0.32 ± 0.46 | NS |
Data are percentage or means ± SD, unless otherwise indicated. P value is reported for significant differences among groups tested with ANOVA with Bonferroni post-hoc test. NGT, normal glucose tolerance; IGT, impaired glucose tolerance; 2 h p-l, 2 h postglucose load; HOMA-IR, homeostasis model assessment of insulin resistance; FMD, flow-mediated dilation; sIL6 R, soluble IL-6 receptor; sTNF R1, soluble TNF receptor 1.
FIG. 1.
mRNA expression of IRS2 in monocytes from FDRs of type 2 diabetic subjects is correlated with in vivo insulin resistance and subclinical atherosclerosis. High log IRS2 mRNA levels are associated with increased levels of M (A), decreased IMT (B), decreased fasting plasma glucose levels (C), decreased glycated hemoglobin (D), and increased HDL cholesterol (E). *P < 0.05 for LOW IRS2 (n = 14) compared with HIGH IRS2 (n = 14) by ANOVA with Bonferroni post-hoc test.
FIG. 2.
InsR and IRS protein levels and phosphorylation status in monocytes from lowest vs. highest insulin resistance tertiles. A–C: InsR and IRS1 protein levels and pan-tyrosine are not different in monocytes from insulin-sensitive (n = 13) vs. insulin-resistant (n = 14) subjects (A). IRS2 protein levels and pan-tyrosine phosphorylation are higher in monocytes from insulin-sensitive (n = 13) vs. insulin-resistant (n = 14) subjects (*P < 0.05, by Student t test) (B). Akt protein levels and serine 473 phosphorylation are higher in monocytes from insulin-sensitive (n = 13) vs. insulin-resistant (n = 14) subjects (*P < 0.05, by Student t test) (C). D–H: FDR subjects characterized by lowest IRS2 protein levels in monocytes exhibit significantly increased IMT (D), decreased M (E), fasting plasma glucose (F), HbA1c (G), and increased HDL (H) (n = 14 and n = 14 for LOW IRS2 and HIGH IRS2, respectively; *P < 0.05, by Student t test). a.u., arbitrary unit.
mRNA TIMP3 expression is reduced in monocytes from insulin-resistant FDRs.
Recent studies using human specimens and animal models suggested that the regulation of the inflammatory behavior of monocytes is controlled by TIMP3, which restrains the release of the soluble fraction of transmembrane proteins through their ectodomain shedding. This process is regulated by sheddases belonging to the ADAM family of metalloproteases, which generate soluble forms of cytokines, growth factors, and receptors, such as TNF-α, IL-6 receptor, VCAM-1, ICAM-1 and selectins, all involved in the chronic inflammatory process. TIMP3 is the physiological inhibitor of the ADAMs involved in the ectodomain shedding process, such as ADAM17 and ADAM10 (11).
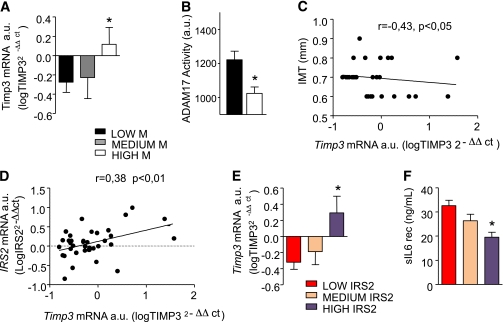
We analyzed the expression of several genes involved in the membrane ectodomain shedding in monocytes extracted from PBMCs of FDRs, such as ADAM17, ADAM10, MMP9, and TIMP3. No correlation was found between gene expression levels and clinical and biochemical parameters. Dividing our population into tertiles of insulin resistance assessed by M clamp, we found that TIMP3 mRNA levels were significantly reduced in insulin-resistant FDRs (Table 1 and Fig. 3A). Insulin-resistant FDRs carrying lower TIMP3 expression were characterized by significantly increased ADAM17 activity (Fig. 3B). TIMP3 mRNA levels were negatively correlated with IMT (r = −0.43; P < 0.05) (Fig. 3C). Further analysis revealed that in monocytes, there is a correlation between IRS2 and TIMP3 expression (Fig. 3D), with subjects in the lowest tertile of IRS2 expression having reduced TIMP3 levels (Fig. 3E) and increased ectodomain shedding activity, as suggested by increased circulating soluble IL-6 receptor and sVCAM-1 (Fig. 3F and Supplementary Table 2).
FIG. 3.
TIMP3 and ADAM17 pathway in monocytes from FDRs. A: TIMP3 expression is higher in insulin-sensitive compared with insulin-resistant FDR subjects (*P < 0.05 for LOW M [n = 14] compared with HIGH M [n = 13] by ANOVA with Bonferroni multiple comparison tests). B: ADAM17 activity is significantly higher in monocyte extracts from insulin-resistant (black bar; n = 14) compared with insulin-sensitive (white bar; n = 13) FDR subjects (*P < 0.05 by Student t test). C: mRNA expression of TIMP3 in monocytes from FDRs of type 2 diabetic subjects is negatively correlated with subclinical atherosclerosis. D: Expression of TIMP3 in monocytes from FDRs of type 2 diabetic subjects is positively correlated with IRS2 mRNA levels. E: Expression of TIMP3 in monocytes from FDRs of type 2 diabetic subjects is reduced according to IRS2 mRNA expressed in tertiles. F: Soluble IL-6 receptor (rec), a marker of increased ectodomain shedding, is significantly higher in subjects with lowest IRS2 mRNA expression levels. *P < 0.05 for LOW IRS2 (n = 14) compared with HIGH IRS2 (n = 14) by Student t test. a.u., arbitrary unit.
DISCUSSION
The contradictory results from monocyte-specific InsR knockouts do not provide definitive evidence for a positive or negative role of myeloid insulin resistance in the pathogenesis of diabetes and atherosclerosis in humans (4–7,12).
Here, we observe no correlation between insulin resistance, subclinical atherosclerosis, and InsR in monocytes from human subjects. In contrast, expression of IRS2, the major transduction element of InsR tyrosine kinase in myeloid cells (13,14), was correlated to both systemic insulin resistance and subclinical atherosclerosis. Overall, in subjects at risk for diabetes, those with insulin resistance and increased IMT carry lower expression of IRS2 in circulating monocytes, suggesting that myeloid-specific insulin resistance at the molecular level facilitates the development of atherosclerosis and diabetes. The reduced IRS2 expression might be explained by the exposure to different insulin levels, since relative hyperinsulinemia and insulin resistance are known to affect IRS2 expression through AkT/FoxO1 activation (15).
The inflammatory behavior of circulating monocytes is dependent on several checkpoints (16). One extracellular factor regulating innate response is TIMP3, through its ability to restrain release of cytokines and chemokines from the plasma membrane (11,17).
Monocytes from subjects characterized by increased insulin resistance and atherosclerosis exhibit reduced mRNA TIMP3 levels. We previously established that TIMP3 is a genetic modifier for insulin resistance in mice; it is reduced in atherosclerotic plaques of subjects with type 2 diabetes, as a result of altered SirT1 activity on vascular smooth muscle cells and monocytes (18,19). Hyperinsulinemia (a compensatory response to peripheral insulin resistance) is known to increase ADAM10 and ADAM17 activity, the inflammatory metalloproteases that are restrained by TIMP3, causing increased release of transmembrane proteins as soluble factors (20,21). Among transmembrane proteins with chemokine activity, we found that in offspring of patients with type 2 diabetes, soluble IL-6 receptor and sVCAM-1 were positively correlated with insulin resistance and markers of atherogenic dyslipidemia, suggesting a role for these chemokines in the early phases of metabolic inflammation.
In conclusion, in monocyte cells from human subjects with increased risk for diabetes and atherosclerosis, we found that gene expression and protein activity of IRS2, but not InsR, negatively correlates with insulin resistance and atherosclerosis. Moreover, TIMP3, a natural checkpoint for inflammation, is reduced along with insulin resistance. Our data suggest that the reduction of TIMP3 may increase the release of inflammatory soluble factors generated through the ectodomain shedding activity of metalloproteases such as ADAM17, a process starting at preclinical levels with release of soluble IL-6 receptor. Intriguingly, data in experimental models suggested that low TIMP3 expression defines a subpopulation of highly invasive foam-cell macrophages with increased proliferation and apoptosis, all properties expected to destabilize atherosclerotic plaques (22).
ACKNOWLEDGMENTS
This study was funded in part by grants from Fondazione Roma 2008, Telethon GGP08065, European Foundation for the Study of Diabetes Clinical Research Grant 2008, and FP7-Health-241913–FLORINASH (all to M.F.)
No potential conflicts of interest relevant to this article were reported.
M.C. performed in vivo clinical studies and in vitro experimental studies, analyzed in vivo and in vitro data, and wrote the manuscript. R.M. performed in vitro experimental studies, analyzed in vivo and in vitro data, and wrote the manuscript. A.L., F.D., I.C., R.D., P.G., S.R., and M.A.M. performed in vivo clinical studies. O.P. performed in vitro experimental studies and wrote the manuscript. D.L., P.S., and R.L. designed the study and wrote the manuscript. M.F. designed the study, performed in vivo clinical studies and in vitro experimental studies, analyzed in vivo and in vitro data, and wrote the manuscript.
Footnotes
This article contains Supplementary Data online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db11-0162/-/DC1.
REFERENCES
- 1.Rocha VZ, Libby P. Obesity, inflammation, and atherosclerosis. Nat Rev Cardiol 2009;6:399–409 [DOI] [PubMed] [Google Scholar]
- 2.Brown MS, Goldstein JL. Selective versus total insulin resistance: a pathogenic paradox. Cell Metab 2008;7:95–96 [DOI] [PubMed] [Google Scholar]
- 3.Stefan N, Häring HU. The metabolically benign and malignant fatty liver. Diabetes 2011;60:2011–2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tabas I, Tall A, Accili D. The impact of macrophage insulin resistance on advanced atherosclerotic plaque progression. Circ Res 2010;106:58–67 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Biddinger SB, Hernandez-Ono A, Rask-Madsen C, et al. Hepatic insulin resistance is sufficient to produce dyslipidemia and susceptibility to atherosclerosis. Cell Metab 2008;7:125–134 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Baumgartl J, Baudler S, Scherner M, et al. Myeloid lineage cell-restricted insulin resistance protects apolipoproteinE-deficient mice against atherosclerosis. Cell Metab 2006;3:247–256 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Han S, Liang CP, DeVries-Seimon T, et al. Macrophage insulin receptor deficiency increases ER stress-induced apoptosis and necrotic core formation in advanced atherosclerotic lesions. Cell Metab 2006;3:257–266 [DOI] [PubMed] [Google Scholar]
- 8.Hotamisligil GS, Erbay E. Nutrient sensing and inflammation in metabolic diseases. Nat Rev Immunol 2008;8:923–934 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rizza S, Tesauro M, Cardillo C, et al. Fish oil supplementation improves endothelial function in normoglycemic offspring of patients with type 2 diabetes. Atherosclerosis 2009;206:569–574 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cardellini M, Marini MA, Frontoni S, et al. Carotid artery intima-media thickness is associated with insulin-mediated glucose disposal in nondiabetic normotensive offspring of type 2 diabetic patients. Am J Physiol Endocrinol Metab 2007;292:E347–E352 [DOI] [PubMed] [Google Scholar]
- 11.Murphy G, Murthy A, Khokha R. Clipping, shedding and RIPping keep immunity on cue. Trends Immunol 2008;29:75–82 [DOI] [PubMed] [Google Scholar]
- 12.Mauer J, Chaurasia B, Plum L, et al. Myeloid cell-restricted insulin receptor deficiency protects against obesity-induced inflammation and systemic insulin resistance. PLoS Genet 2010;6:e1000938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hartman ME, O’Connor JC, Godbout JP, Minor KD, Mazzocco VR, Freund GG. Insulin receptor substrate-2-dependent interleukin-4 signaling in macrophages is impaired in two models of type 2 diabetes mellitus. J Biol Chem 2004;279:28045–28050 [DOI] [PubMed] [Google Scholar]
- 14.Liang CP, Han S, Okamoto H, et al. Increased CD36 protein as a response to defective insulin signaling in macrophages. J Clin Invest 2004;113:764–773 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Guo S, Dunn SL, White MF. The reciprocal stability of FOXO1 and IRS2 creates a regulatory circuit that controls insulin signaling. Mol Endocrinol 2006;20:3389–3399 [DOI] [PubMed] [Google Scholar]
- 16.Liang CP, Han S, Senokuchi T, Tall AR. The macrophage at the crossroads of insulin resistance and atherosclerosis. Circ Res 2007;100:1546–1555 [DOI] [PubMed] [Google Scholar]
- 17.Smookler DS, Mohammed FF, Kassiri Z, Duncan GS, Mak TW, Khokha R. Tissue inhibitor of metalloproteinase 3 regulates TNF-dependent systemic inflammation. J Immunol 2006;176:721–725 [DOI] [PubMed] [Google Scholar]
- 18.Federici M, Hribal ML, Menghini R, et al. Timp3 deficiency in insulin receptor-haploinsufficient mice promotes diabetes and vascular inflammation via increased TNF-alpha. J Clin Invest 2005;115:3494–3505 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cardellini M, Menghini R, Martelli E, et al. TIMP3 is reduced in atherosclerotic plaques from subjects with type 2 diabetes and increased by SirT1. Diabetes 2009;58:2396–2401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chen CD, Podvin S, Gillespie E, Leeman SE, Abraham CR. Insulin stimulates the cleavage and release of the extracellular domain of Klotho by ADAM10 and ADAM17. Proc Natl Acad Sci USA 2007;104:19796–19801 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Fiorentino L, Vivanti A, Cavalera M, et al. Increased tumor necrosis factor alpha-converting enzyme activity induces insulin resistance and hepatosteatosis in mice. Hepatology 2010;51:103–110 [DOI] [PubMed] [Google Scholar]
- 22.Johnson JL, Sala-Newby GB, Ismail Y, Aguilera CM, Newby AC. Low tissue inhibitor of metalloproteinases 3 and high matrix metalloproteinase 14 levels defines a subpopulation of highly invasive foam-cell macrophages. Arterioscler Thromb Vasc Biol 2008;28:1647–1653 [DOI] [PMC free article] [PubMed] [Google Scholar]