Abstract
OBJECTIVE
Interleukin-21 (IL-21) is a proinflammatory cytokine that has been shown to affect Treg/Teff balance. However, the mechanism by which IL-21 orchestrates alloimmune response and interplays with Tregs is still unclear.
RESEARCH DESIGN AND METHODS
The interplay between IL-21/IL-21R signaling, FoxP3 expression, and Treg survival and function was evaluated in vitro in immunologically relevant assays and in vivo in allogenic and autoimmune models of islet transplantation.
RESULTS
IL-21R expression decreases on T cells and B cells in vitro and increases in the graft in vivo, while IL-21 levels increase in vitro and in vivo during anti-CD3/anti-CD28 stimulation/allostimulation in the late phase of the alloimmune response. In vitro, IL-21/IL-21R signaling (by using rmIL-21 or genetically modified CD4+ T cells [IL-21 pOrf plasmid–treated or hIL-21-Tg mice]) enhances the T-cell response during anti-CD3/anti-CD28 stimulation/allostimulation, prevents Treg generation, inhibits Treg function, induces Treg apoptosis, and reduces FoxP3 and FoxP3-dependent gene transcripts without affecting FoxP3 methylation status. In vivo targeting of IL-21/IL-21R expands intragraft and peripheral Tregs, promotes Treg neogenesis, and regulates the antidonor immune response, whereas IL-21/IL-21R signaling in Doxa-inducible ROSA-rtTA-IL-21-Tg mice expands Teffs and FoxP3− cells. Treatment with a combination of mIL-21R.Fc and CTLA4-Ig (an inhibitor of the early alloimmune response) leads to robust graft tolerance in a purely alloimmune setting and prolonged islet graft survival in NOD mice.
CONCLUSIONS
IL-21 interferes with different checkpoints of the FoxP3 Treg chain in the late phase of alloimmune response and, thus, acts as an antitolerogenic cytokine. Blockade of the IL-21/IL-21R pathway could be a precondition for tolerogenic protocols in transplantation.
The alloimmune response is a complex phenomenon, based on the activation of the innate and adaptive immune responses, which invariably leads to allograft rejection (1). Autocrine soluble factors, such as cytokines, are able to enhance or alternatively suppress the alloimmune response (1). While interleukin (IL)-2 and γ-interferon (IFN-γ) are among the primary mediators of the early phase of the alloimmune response (2), little is known regarding the late phase of the alloimmune response, during which alloreactive T cells are recruited to the proliferating pool, thus continuing the expansion process, while regulatory T cells (Tregs) are inhibited in exerting their suppressive function (3).
IL-21 is a cytokine produced by activated CD4+ T cells and NK cells that has been demonstrated to directly contribute to the orchestration of the different pathways that regulate the immune response (4,5). IL-21 binds the IL-21 receptor (IL-21R) heterodimer and provides signals to CD8+ naïve T cells to differentiate into cytotoxic effector cells (6) and signals to CD4+ T cells to differentiate into Th17 cells (7–9). It was recently demonstrated that IL-21 has a role in graft-versus-host disease (10,11). The basis of considering IL-21 as an important player in the alloimmune response lies in 1) its robust role in expanding effector T cells (Teffs) (6,12) and 2) its potential inhibitory role on Tregs (8,9). First, IL-21 has been shown to have a role in driving the expansion of Teffs in different disease models (8,13); second, IL-21 has been previously shown to inhibit transforming growth factor (TGF)-β–driven differentiation of naïve T cells into Tregs (8,9), thereby enhancing cytotoxic T-cell generation (14). Tregs control the immune response, and an IL-21–mediated impairment of Treg numbers or function may ultimately result in the impossibility of tolerance induction (15–17). Few data are available thus far on the interplay of IL-21 and Tregs in the alloimmune response (10,11,18).
Our aim is to study how the IL-21/IL-21R pathway interacts with FoxP3 and Tregs during alloimmune response and to explore whether IL-21 may act as an antitolerogenic cytokine, thus representing a barrier to tolerance.
RESEARCH DESIGN AND METHODS
Mice.
The following mice were used: C57BL/6, DBA/1J, BALB/c, Rag1tm1Mom, B6-FoxP3-GFP knock-in, IL-21R−/−, and hIL-21-Tg (15), all in a C57BL/6 background.
Doxycycline-inducible ROSA-rtTA-IL-21-Tg mice.
Transgenic C57BL/6 mice expressing mIL-21 cDNA under the control of a tetracycline-dependent promoter were crossed to ROSA-rtTA mice (see Supplementary Data). Mice were administered streptozotocin (STZ), transplanted with BALB/c islets, and treated the same day with doxycycline (2 mg/mL in drinking water until rejection).
Immunological assays.
Anti-CD3/anti-CD28 (anti-CD3/CD28) stimulation assay and mixed-lymphocyte reaction (MLR) assay were performed as described previously (19). In vitro assays to study generation, survival, and function of CD4+CD25+ Tregs were performed as previously described (17,20,21).
Protocol.
Islet-transplanted mice were treated with 400 μg mIL-21R.Fc (>99% purity; Pfizer, Cambridge, MA) or with 400 μg i.p. of a negative-control IgG2a antibody (anti–Eimeria tenella) every day for 10 days and then three times per week for a total of 4 weeks (22). A group of mice was treated with mIL-21R.Fc in combination with CTLA4-Ig (Abatacept, Orencia; Bristol-Myers Squibb, Princeton, NJ) (500 μg at day 0 and 250 μg at days 2, 4, 6, 8, and 10).
Statistical analyses.
Data are expressed as mean ± standard error. Kaplan-Meier analysis (log-rank test) was performed for survival studies. A P value of <0.05 (by two-tailed testing) was considered an indicator of statistical significance. Analyses of data were performed using an SPSS statistical package for Windows (SPSS Inc., Chicago, IL).
RESULTS
IL-21/IL-21R levels and expression after anti-CD3/CD28 stimulation and allostimulation in vitro.
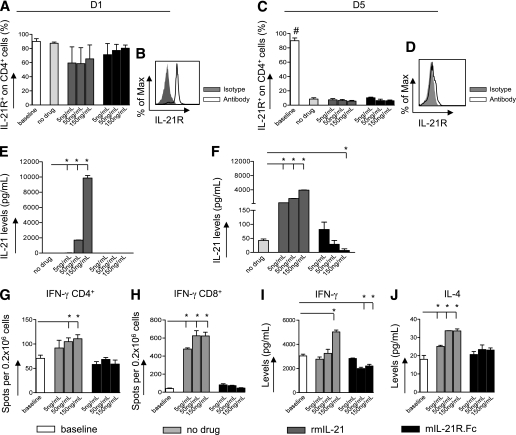
IL-21R is highly expressed on naïve unstimulated CD4+, CD8+, and B220+ cells at baseline, and in contrast with previously published data (23), the percentage of IL-21R+ cells is unchanged on stimulated cells at day 1 and at day 3 (Fig. 1A and B) (some data not shown). It is interesting that after 5 days of anti-CD3/CD28 stimulation, a reduction in the percentage of IL-21R+ T cells and B cells was evident compared with baseline (IL-21R+CD4+ T cells = 8.5 ± 1.7%; IL-21R+CD8+ T cells = 14.9 ± 5.6%; IL-21R+B220+ cells = 21.5 ± 14.5%; baseline vs. all, P < 0.05) (Fig. 1C and D). IL-21 was undetectable in the supernatant at day 1 (Fig. 1E) and at day 3 (data not shown). However, increased levels of IL-21 in the supernatant were found at day 5 compared with day 1 (P < 0.001) in the anti-CD3/CD28 stimulation assay (Fig. 1E and F). When 150 ng/mL of recombinant mouse (rm)IL-21 was added in vitro, IL-21 supernatant levels were higher at day 1 compared with day 5 (P < 0.001) (Fig. 1E and F), possibly for rmIL-21 degradation.
FIG. 1.
IL-21 is a cytokine of the late phase of the alloimmune response in vitro. The percentage of IL-21R+ CD4+ T cells (obtained from C57BL/6 mice) after 1 day (D1) (A and B) and 5 days (D5) (C and D) in an anti-CD3/CD28 stimulation assay in the presence of increasing concentrations of 5, 50, and 150 ng/mL rmIL-21 or mIL-21R.Fc or medium alone (no drug) (n = 3 experiments/conditions) was determined. IL-21R was highly expressed on CD4+ T cells, without any considerable changes after day 1 (A and B). At day 5, the percentage of IL-21R+CD4+ T cells was reduced compared with baseline in all conditions tested (baseline vs. all, #P < 0.05) (C and D). IL-21 levels in supernatants obtained in the anti-CD3/CD28 stimulation assay were evaluated using the Luminex assay at different time points (n = 3 experiments/conditions). No detectable levels of IL-21 were found in supernatants at day 1 after stimulation (E), whereas an increase was observed at day 5. High concentrations of mIL-21R.Fc (150 ng/mL) significantly reduced IL-21 levels (no drug vs. 150 ng/mL mIL-21R.Fc, *P = 0.01) (F). The addition of serial concentrations of rmIL-21 induced an increase in IL-21 levels at days 1 and 5 (no drug vs. rmIL-21, *P < 0.05) (E). IFN-γ–producing CD4+ and CD8+ T cells extracted from naïve C57BL/6 mice and cultured with anti-CD3/CD28 in the presence of 5, 50, and 150 ng/mL rmIL-21 or mIL-21R.Fc or with medium alone (no drug) were evaluated using the ELISpot assay (G and H) (n = 3 experiments/conditions). An increase in IFN-γ–producing CD4+ and CD8+ T cells was evident on the addition of rmIL-21 (no drug vs. 50 and 150 ng/mL rmIL-21, *P < 0.05) (G and H). The frequencies of IFN-γ–producing CD4+ and CD8+ T cells did not change when mIL-21R.Fc was added to the anti-CD3/CD28 assay (no drug vs. 5, 50, and 150 ng/mL mIL-21R.Fc, NS) (G and H). IFN-γ and IL-4 levels were evaluated in supernatants obtained from anti-CD3/CD28 stimulation assays at day 5 using the Luminex assay (n = 3 experiments/conditions). Increased levels of IFN-γ were detected in the presence of rmIL-21 (no drug vs. 150 ng/mL rmIL-21, *P = 0.0008), whereas IFN-γ levels were reduced in the presence of mIL-21R.Fc (no drug vs. 50 and 150 ng/mL mIL-21R.Fc, *P < 0.05) (I). A dose-dependent increase in IL-4 levels was observed in the presence of rmIL-21 (no drug vs. 5, 50, and 150 ng/mL rmIL-21, *P < 0.05), whereas no changes were evident in the presence of mIL-21R.Fc (no drug vs. 5, 50, and 150 ng/mL mIL-21R.Fc, NS) (J).
At day 5 (but not at days 1 or 3), an overall reduction in the percentage of IL-21R+CD4+/CD8+/B220+ cells was evident (Fig. 1A–D) (some data not shown) regardless of the presence of rmIL-21 or mIL-21R.Fc. The addition of mIL-21R.Fc reduced IL-21 levels in the supernatant at day 5, when IL-21 first became detectable (Fig. 1F). Data obtained from anti-CD3/CD28 stimulation assay data were confirmed in the MLR assay as well (data not shown). To confirm that mIL-21R.Fc binds IL-21 cytokine (thus reducing IL-21 detectable levels in vitro), we added serial concentrations of mIL-21R.Fc (5, 50, and 150 ng/mL) to 150 ng/mL of rmIL-21 and evaluated IL-21 levels by Luminex assay (Millipore, Billerica, MA) at days 1, 3, and 5 after seeding. Reduced IL-21 levels were detected when rmIL-21 and mIL-21R.Fc were cocultured (i.e., IL-21 levels at day 1: no drug = 3290 ± 337 vs. mIL-21R.Fc 150 ng/mL = 1135 ± 284; P < 0.05).
IL-21/IL-21R signaling enhances the T-cell response during anti-CD3/CD28 stimulation and allostimulation in vitro.
We first challenged CD4+ and CD8+ T cells extracted from the spleens of 10-week-old C57BL/6 mice in an anti-CD3/CD28 stimulation assay with the addition of rmIL-21 or mIL-21R.Fc. Addition of rmIL-21 led to a dose-dependent increase of IFN-γ–producing CD4+ T cells (no drug = 70.5 ± 6.5; 5 ng/mL rmIL-21 = 91.7 ± 15.9; 50 ng/mL rmIL-21 = 104.8 ± 7.7; 150 ng/mL rmIL-21 = 110.8 ± 8.3, counted as number of IFN-γ–producing cells per 0.2 × 106 total CD4+ T cells; no drug vs. 50 ng/mL rmIL-21 and 150 ng/mL rmIL-21, P < 0.01) (Fig. 1G) and CD8+ T cells (Fig. 1H). IL-4–producing CD4+ T cells increased in frequency as well when rmIL-21 was added to the anti-CD3/CD28 assay, while no IL-4–producing CD8+ T cells were detected in this assay (data not shown). These data were confirmed in an allostimulation assay (MLR) (data not shown). Addition of mIL-21R.Fc to the anti-CD3/CD28 assay induced a decrease in IFN-γ levels and left IL-4 unchanged (Fig. 1I), while rmIL-21 increased IFN-γ and IL-4 levels (Fig. 1J) 5 days after stimulation.
We then evaluated the effect of genetically overexpressing or knocking down the IL-21/IL-21R pathway. We used CD4+ T cells obtained from IL-21 plasmid–treated mice (injected with IL-21 pOrf plasmid) and from IL-21R−/− mice (with genetic deletion of the IL-21R gene) for in vitro studies, confirming the data obtained with the rmIL-21 and mIL-21R.Fc (Supplementary Fig. 1A–D).
Signaling of IL-21/IL-21R prevents Treg generation in vitro.
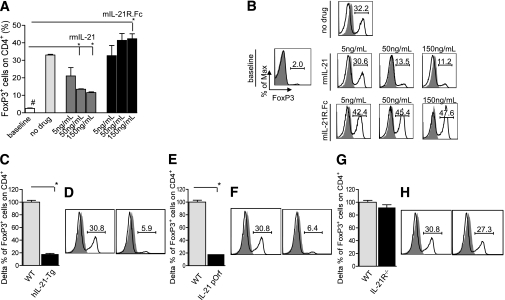
We performed a Treg generation assay (20) to investigate whether conversion from CD4+CD25− cells (and, thus, FoxP3−) into CD4+FoxP3+ cells was affected by rmIL-21 or by the overexpression of IL-21 (through the use of hIL-21-Tg mice or IL-21 plasmid–treated mice). In a complementary experiment, we evaluated the effect of targeting IL-21 chemically (mIL-21R.Fc) or genetically (IL-21R−/− mice) in the same assays. We found a dose-dependent decrease in Treg frequency when rmIL-21 was added compared with the percentage obtained without supplementation (no drug = 33.0 ± 0.4%; 150 ng/mL rmIL-21 = 11.5 ± 0.4%; P < 0.0001) (Fig. 2E and F). An increase in Tregs was evident when mIL-21R.Fc was added at high concentrations only (no drug = 33.0 ± 0.4%; 150 ng/mL mIL-21R.Fc = 42.3 ± 2.8%; P = 0.03) (Fig. 2E and F). When CD4+CD25− cells obtained from hIL-21-Tg or IL-21 plasmid–treated mice were used, fewer Tregs were generated as compared with when using CD4+CD25− cells from wild type (WT) and IL-21R−/− mice (Fig. 2G–L).
FIG. 2.
IL-21/IL-21R signaling and overexpression inhibits Treg generation in vitro. A Treg generation assay was performed in vitro in the presence of rmIL-21 or mIL-21R.Fc or medium alone (no drug) (n = 3 experiments/conditions). CD4+CD25− cells extracted from C57BL/6 mice by magnetic microbeads were cultured with anti-CD3/CD28 antibodies, TGF-β, and increasing concentrations of rmIL-21 or mIL-21R.Fc (both at 5, 50, and 150 ng/mL) or no drug (A and B). After 4 days of culture, the percentage of CD4+FoxP3+ cells was evaluated (Tregs). Although rmIL-21 supplementation suppressed Treg generation (no drug vs. 50 and 150 ng/mL rmIL-21, *P < 0.0001), the addition of mIL-21R.Fc promoted Treg expansion (no drug vs. 150 ng/mL mIL-21R.Fc, *P = 0.03) (n = 3 experiments/conditions) (A and B). Treg generation was performed with WT, hIL-21-Tg, IL-21 plasmid–treated, and extracted IL-21R−/− CD4+CD25− T cells (n = 3 experiments/conditions) (C–H). A reduction of 70% compared with baseline (WT mice) in Treg generation was evident when CD4+ T cells were obtained from IL-21–overexpressing mice (WT vs. IL-21 plasmid treated and hIL-21-Tg, *P < 0.0001) (C–F), whereas no differences were evident when using IL-21R−/− CD4+CD25− (WT vs. IL-21R−/−, NS) (G and H).
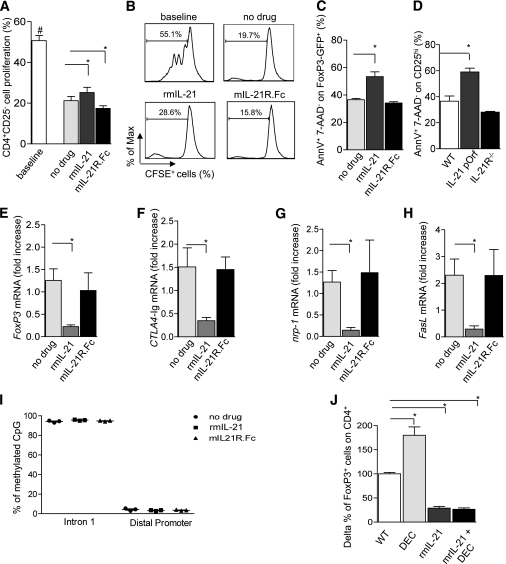
IL-21/IL-21R signaling inhibits Treg function and induces Treg apoptosis in vitro.
To explore whether Tregs generated in the presence of IL-21 are dysfunctional and unable to suppress Teff proliferation in vitro, CD4+CD25− 5-(and 6)-carboxyfluorescein diacetate succinimidyl ester (CFSE)-labeled T cells obtained from WT C57BL/6.Thy1.2 mice were plated with the same number of C57BL/6.Thy1.1 Tregs generated in the presence of high concentrations (150 ng/mL) of rmIL-21 or mIL-21R.Fc or with medium alone. After 72 h of stimulation, cells were harvested, and CFSE dilution was assessed on Thy1.1-negative cells. It is interesting that an increased percentage of Teff proliferation was evident when Tregs generated in the presence of rmIL-21 were added compared with Tregs generated in the absence of drugs or with mIL-21R.Fc (no drug = 19.1 ± 0.5%; rmIL-21 = 27.7 ± 0.9%; mIL-21R.Fc = 16.1 ± 0.3%; rmIL-21 vs. all, P < 0.05) (Fig. 3A and B). This effect may be the result of both a reduced suppressive ability of Tregs and increased Treg apoptosis. Control CFSE-labeled CD4+CD25− T cells stimulated in the absence of Tregs were shown to undergo robust proliferation (Fig. 3A and B). To assess the mechanism through which IL-21 mediates a decrease in in vitro Treg generation, we performed a Treg maintenance assay in which GFP+ cells (sorted by FoxP3-GFP mice) or CD4+CD25hi cells (obtained through cell sorting of splenocytes extracted by IL-21 plasmid–treated or IL-21R−/− mice) were plated with anti-CD3/CD28 monoclonal antibodies as well as IL-2 as a survival stimulus (17). A greater percentage of apoptotic Tregs (AnnexinV+/7-AAD−) was observed in the presence of 150 ng/mL rmIL-21 compared with Tregs cultured in the absence of any drug (no drug = 36.5 ± 0.9%; rmIL-21 = 53.4 ± 3.4%; P = 0.04) (Fig. 3C). In a similar manner, IL-21 overexpressing Tregs (from IL-21 plasmid–treated mice), but not IL-21R−/− Tregs, displayed an increased apoptotic rate in the maintenance assay (Fig. 3D). No differences in mRNA levels of Bcl2, Bcl6, Bax, Caspase 3, Caspase 8, cMyc, Fas, and FasL were detected in Tregs cultured in the presence of rmIL-21 compared with control Tregs (data not shown).
FIG. 3.
IL-21/IL-21R signaling and overexpression reduces Treg function, Treg survival, and FoxP3 gene transcripts without affecting methylation status. A suppression assay was performed in which CFSE-labeled CD4+CD25− cells were stimulated with anti-CD3/CD28 antibodies. Their proliferation was then assessed in the presence of Tregs generated with high concentrations of rmIL-21 or mIL-21R.Fc (both at 150 ng/mL) or without any drug supplementation (n = 3 experiments/conditions) (A and B). The proliferation of stimulated CD4+CD25− in the absence of Tregs was also evaluated (baseline vs. all, #P < 0.01) (A and B). Tregs generated in the presence of rmIL-21 displayed reduced suppression of CD4+CD25− T-cell proliferation compared with control (no drug vs. rmIL-21, *P = 0.01). On the contrary, mIL-21R.Fc–induced Tregs showed an increased ability to suppress CD4+CD25− T-cell proliferation compared with control (no drug vs. mIL-21R.Fc, *P = 0.04) (A and B). The apoptotic rate of FoxP3-GFP+ cells (extracted from FoxP3-GFP-B6 mice) plated with anti-CD3/CD28 antibodies, IL-2, and high concentrations of rmIL-21 or mIL-21R.Fc (both at 150 ng/mL) or with no drug was evaluated in a Treg maintenance assay (n = 3 experiments/conditions) (C). FoxP3-GFP+ cells cultured with rmIL-21 displayed a higher percentage of AnnexinV+/7-AAD− apoptotic cells compared with those cultured with no drug (no drug vs. rmIL-21, *P = 0.04) (C). A greater percentage of apoptotic CD4+CD25hi cells was evident when cells were obtained from IL-21 plasmid–treated mice (WT vs. IL-21 plasmid treated, *P = 0.007) (D), whereas no differences in apoptotic rate were evident when cells were derived from IL-21R−/− mice (WT vs. IL-21R−/−, NS) (n = 3 experiments) (D). There were fewer FoxP3 mRNA transcripts in Tregs generated in the presence of rmIL-21 (n = 3 experiments) (no drug vs. rmIL-21, *P = 0.01) (E). FoxP3-related gene expression (CTLA4 [no drug vs. rmIL-21, *P = 0.03]; Nrp-1 [no drug vs. rmIL-21, *P = 0.01]; and FasL [no drug vs. rmIL-21, *P = 0.02]) was reduced as well (n = 3 experiments) (F–H). To evaluate whether the IL-21 effect on Tregs was mediated by FoxP3 DNA epigenetic reprogramming and hypermethylation, we studied the methylation status of Tregs generated in vitro in the presence of rmIL-21 or mIL-21R.Fc (both used at 150 ng/mL), and a similar percentage of methylation in two FoxP3 DNA regions (Intron 1 and Distal Promoter) was found (no drug vs. rmIL-21 vs. mIL-21R.Fc, NS) (n = 3 experiments) (I). These data were confirmed by adding a hypomethylating agent (decitabine) to rmIL-21 in an in vitro Treg generation assay in which no recovery of FoxP3+ cells was evident (NS), while decitabine (DEC) alone was able to induce an increase in Tregs (WT vs. DEC, *P = 0.01) (n = 3 experiments) (J).
IL-21/IL-21R signaling inhibits FoxP3 transcription without altering the FoxP3 methylation profile in vitro.
To better understand the interplay of IL-21/IL-21R signaling with Tregs, we performed RT-PCR studies to evaluate mRNA transcript levels of FoxP3 and FoxP3-dependent genes in Tregs cultured with 150 ng/mL rmIL-21 or mIL-21R.Fc (17). RT-PCR expression studies confirmed the reduction of FoxP3 mRNA levels (rmIL-21 = 0.2 ± 0.0 fold decrease compared with baseline, P = 0.01) (Fig. 3E) and of FoxP3-targeted genes (CTLA4, Nrp-1, and FasL) in the presence of rmIL-21 (Fig. 3F–H). mIL-21R.Fc did not induce any modifications in FoxP3 and FoxP3-targeted gene expression. To assess whether IL-21 inhibition of TGF-β–driven differentiation of Tregs is mediated by FoxP3 epigenetic reprogramming, we evaluated the DNA methylation profile, through pyrosequencing (see Supplementary Data), of two regions of the FoxP3 gene (Intron 1 and Distal Promoter) known to be highly and poorly methylated, respectively, at basal conditions (16) in Tregs generated in the presence of rmIL-21 or mIL-21R.Fc or without any supplementation (Fig. 3I). No differences were evident in the CpG base pair percentage in the Intron 1 region (mainly hypermethylated) or the Distal Promoter region (mainly hypomethylated) regardless of rmIL-21 or mIL-21R.Fc presence (Fig. 3I).
Further confirmation of a non-FoxP3 methylation–mediated mechanism of IL-21 action was sought by examining the ability of decitabine or 5-Aza-2′-deoxycytidine (a DNA hypomethylating agent), to promote Treg generation in the presence of rmIL-21 (24). As previously shown (24), treatment with decitabine alone was able to induce Treg generation (Δ-percentage compared with no drug: no drug = 100.0 ± 17.7% vs. decitabine = 180.3 ± 17.3%; P = 0.01) (Fig. 3J). Decitabine was, however, unable to increase CD4+FoxP3+ cells in the presence of rmIL-21 (Δ-percentage compared with no drug: rmIL-21 = 29 ± 3.6% vs. rmIL-21 + decitabine = 26 ± 2.9%; NS) (Fig. 3J).
IL-21/IL-21R levels and expression during the alloimmune response in vivo.
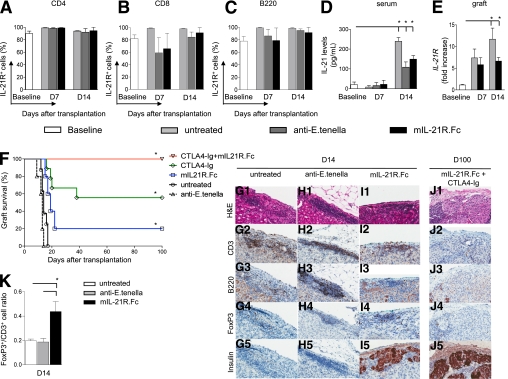
We then evaluated the percentage of IL-21R+ T cells and B cells as well as levels of IL-21 during the alloimmune response in a murine model of islet transplantation in vivo. Islets harvested from BALB/c mice were transplanted under the kidney capsule of hyperglycemic C57BL/6 (STZ) mice and treated with mIL-21-R.Fc, treated with control antibody (anti–E. tenella), or left untreated. No changes in CD4+IL-21R+, CD8+IL-21R+, or B220+IL-21R+ cell percentage were evident in the spleen at days 7 and 14 after transplantation (Fig. 4A–C). IL-21 serum levels analyzed at the same time points as above were not different at day 7 (Fig. 4D) but increased at day 14 after islet transplantation in the three groups of mice (Fig. 4D). The expression of the IL-21/IL-21R pathway was further evaluated in the graft infiltrate of untreated and mIL-21R.Fc–treated islet-transplanted mice at 7 and 14 days after islet transplantation. We found that IL-21R mRNA significantly increased at day 14 compared with baseline (in freshly isolated islets) and at day 7 (day 14 = 11.7 ± 2.5 fold increase compared with baseline; baseline vs. day 14, P = 0.04) (Fig. 4E) in untreated mice, while a lower but not significant intraislet expression of IL-21R mRNA was evident 14 days after islet transplantation in mIL-21R.Fc–treated compared with untreated mice (Fig. 4E).
FIG. 4.
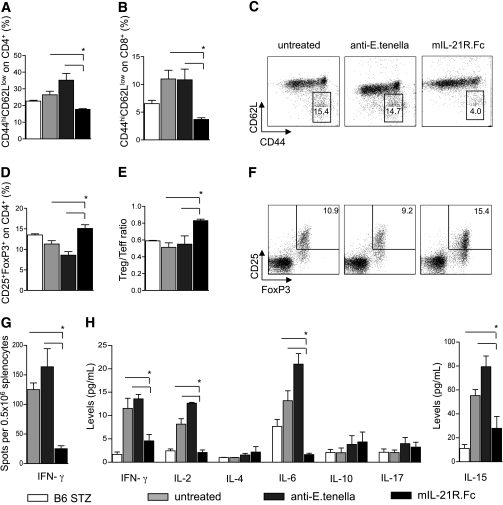
IL-21 is a cytokine of the late phase of the alloimmune response in vivo, and IL-21/IL-21R targeting promotes tolerance during the in vivo alloimmune response. The expression of IL-21R on T cells and B cells as well as IL-21 peripheral levels were analyzed in vivo during the alloimmune response in chemically induced (STZ) diabetic islet-transplanted C57BL/6 mice. IL-21R was highly expressed on CD4+, CD8+, and B220+ cells at baseline, and no differences were evident at the time points tested (days 7 and 14) in all groups of mice studied (n = 4 mice/time point) (A–C). Peripheral IL-21 levels were evaluated using the Luminex assay, which evaluated an increase in IL-21 levels at day 14 after transplant in untreated mice (n = 4) and mice treated with anti–E. tenella (n = 4) and mIL-21R.Fc (n = 4) (albeit to a lower extent in the latter two groups) compared with baseline (baseline vs. all, *P < 0.05) (D). An increase in IL-21R mRNA expression in islet grafts was evident at day 14 in untreated islet-transplanted mice compared with baseline (in freshly isolated islets) (baseline vs. day 14, *P = 0.04) (n = 3) (E), while lower expression was evident in mice treated with mIL-21R.Fc (E). Untreated C57BL/6 mice rapidly rejected islets (MST of 14 days, n = 8) and treatment with anti–E. tenella led to a similar graft survival (MST of 13 days, n = 5) (F). mIL-21R.Fc treatment (n = 5) prolonged graft survival, leading to 20% tolerance (MST >100 days) to islet grafts (MST: untreated = 14 days; anti–E. tenella treated = 13 days; mIL-21R.Fc treated = 19 days; mIL-21R.Fc treated vs. all, *P < 0.05) (F). Treatment with CTLA4-Ig (500 μg at day 0; 250 μg at days 2, 4, 6, 8, and 10) synergized with mIL-21R.Fc treatment (n = 9) to further increase allograft survival compared with either mIL-21R.Fc or with CTLA4-Ig alone treatment, resulting in tolerance in 100% of mice (percentage of tolerance: CTLA4-Ig + mIL-21R.Fc treated = 100%; CTLA4-Ig treated = 55%; mIL-21R.Fc treated = 20%; CTLA4-Ig + mIL-21R.Fc vs. CTLA4-Ig treated [*P = 0.01] and vs. mIL-21R.Fc treated [*P = 0.001]) (F). Histological sections of the graft at day 14 and at day 100 after transplantation (G1–J5) revealed that untreated mice and mice treated with anti–E. tenella at day 14 displayed a complete loss of islet architecture and severe CD3+/B220+ cell infiltration of the graft, with few nested FoxP3+ cells and absent insulin staining (n = 3) (G1–H5). mIL-21R.Fc–treated mice presented preserved islet morphology with mild CD3+/B220+ cell infiltration, primarily constituted of FoxP3+ cells, as well as clearly detectable insulin staining (n = 3) (I1–5). CTLA4-Ig + mIL-21R.Fc–treated tolerant mice at day 100 (n = 3) displayed well-preserved islet architecture with no CD3+/B220+ cell infiltration in the graft and clear insulin staining (J1–5). H&E, hematoxylin-eosin. FoxP3+ and CD3+ cells infiltrating the graft at day 14 were evaluated using Aperio Technology. Greater FoxP3+-to-CD3+ cell ratio was evident in mIL-21R.Fc–treated mice compared with both control groups (mIL-21R.Fc treated vs. untreated and anti–E. tenella treated, *P = 0.04, n = 3 mice per group) (K). D, day. (A high-quality digital representation of this figure is available in the online issue.)
Targeting IL-21/IL-21R expands intragraft Tregs and promotes tolerance in vivo.
We then tested the impact of targeting the IL-21/IL-21R axis (using the mIL-21R.Fc compound) on intragraft FoxP3+ cell expansion and ultimately on allograft survival and tolerance induction in a model of the alloimmune response in vivo (i.e., islet transplantation). Untreated C57BL/6 mice rapidly rejected islet allografts (mean survival time [MST] of 14 days, n = 5), and treatment with the negative control antibody anti–E. tenella led to similar graft survival (MST of 13 days, n = 5) (Fig. 4F). mIL-21R.Fc treatment prolonged graft survival, leading to long-term tolerance, defined as MST >100 days, in 20% of treated mice (MST: untreated = 14 days; anti–E. tenella treated = 13 days; mIL-21R.Fc treated = 19 days; mIL-21R.Fc treated vs. all, P < 0.05) (Fig. 4F). We therefore combined mIL-21R.Fc with CTLA4-Ig treatment (CTLA4-Ig: 500 μg at day 0; 250 μg at days 2, 4, 6, 8, and 10), with the aim of suppressing early T-cell activation (which is likely IL-21 independent) through CTLA4-Ig (25). CTLA4-Ig synergized with mIL-21R.Fc to further increase allograft survival compared with treatment with either mIL-21R.Fc or CTLA4-Ig alone, resulting in tolerance induction in 100% of mice (percentage of tolerant mice: CTLA4-Ig + mIL-21R.Fc treated = 100%; CTLA4-Ig alone treated = 55%; mIL-21R.Fc treated = 20%; CTLA4-Ig + mIL-21R.Fc treated vs. all, P ≤ 0.01) (Fig. 4F). Histological sections of the grafts were analyzed at day 14 and at day 100 (Fig. 4G1–J5). Mice treated with anti–E. tenella at day 14 after transplantation displayed a similar severe infiltration of the graft, primarily consisting of CD3+ and B220+ cells (Fig. 4G1–3 and H1–3), with absent insulin staining (Fig. 4G5 and H5) and very few islet-nested FoxP3+ cells (Fig. 4G4 and H4). Conversely, in the mIL-21R.Fc–treated group at day 14, islet morphology was still well preserved with several detectable insulin-producing cells and a mild CD3+ cell infiltrate, primarily made up of islet-nested FoxP3+ cells and very few B cells (Fig. 4I1–5). Islet morphology in tolerant CTLA4-Ig + mIL-21R.Fc–treated mice at 100 days after transplantation showed no T cells or B cells infiltrating the graft and a clear insulin staining (Fig. 4J1–5). An increase in the FoxP3+-to-CD3+ cell ratio, quantified with Aperio Technology (Vista, CA) (19), was observed at day 14 in mIL-21R.Fc–treated mice compared with both untreated mice and mice treated with anti–E. tenella (Fig. 4K). We confirmed our mIL-21R.Fc data and recent data (13) by transplanting BALB/c islets into chemically induced (STZ) hyperglycemic IL-21R−/− mice, and a prolongation of islet graft survival compared with WT control mice was indeed observed (MST: WT = 14 days; IL-21R−/− = 17 days; P = 0.01; data not shown).
FIG. 5.
IL-21/IL-21R targeting expands peripheral Tregs in vivo and regulates the alloimmune response. The effect of targeting IL-21 in the alloimmune response was analyzed in vivo in islet-transplanted mice during the pathogenesis of the alloimmune response. Splenocytes at day 14 after islet transplantation were harvested from untreated mice (n = 4) and mice treated with anti–E. tenella (n = 4) and mIL-21R.Fc (n = 4) and were analyzed by fluorescence-activated cell sorter. The percentages of CD4+ and CD8+ Teffs (CD4+CD44hiCD62Llo and CD8+CD44hiCD62Llo cells) were lower in the mIL-21R.Fc–treated group compared with untreated mice and mice treated with anti–E. tenella (mIL-21R.Fc treated vs. untreated and anti–E. tenella treated, *P < 0.05) (A–C). The percentage of Tregs was greater in the mIL-21R.Fc–treated mice compared with untreated mice and mice treated with anti–E. tenella (mIL-21R.Fc treated vs. untreated and anti–E. tenella treated, *P < 0.05) (D and F), as was the Treg-to-Teff ratio (mIL-21R.Fc treated vs. untreated and anti–E. tenella treated, *P < 0.05) (E). The percentage of IFN-γ–producing cells was evaluated in an ex vivo antidonor immune response using an ELISpot assay. Splenocytes harvested at day 14 after transplant were rechallenged ex vivo with irradiated BALB/c splenocytes, and the number of IFN-γ–producing cells was evaluated. mIL-21R.Fc–treated mice (n = 3) showed a reduced number of IFN-γ–producing cells compared with untreated mice (n = 3) and mice treated with anti–E. tenella (n = 3) (mIL-21R.Fc treated vs. untreated and anti–E. tenella treated, *P < 0.05) (G). The peripheral cytokine profile was studied in serum samples of the three groups of mice at day 14 after transplantation, which revealed an overall suppression of proinflammatory cytokines (IFN-γ, IL-2, IL-6, and IL-15) in the mIL-21R.Fc–treated group (mIL-21R.Fc treated [n = 3] vs. untreated [n = 3] and anti–E. tenella treated [n = 3], *P < 0.05) (H).
Targeting IL-21/IL-21R expands peripheral Tregs and regulates the antidonor immune response in vivo.
A reduction in the percentage of peripheral CD4+CD44hiCD62Llow Teffs was evident in mIL-21R.Fc–treated mice at 14 days after transplant compared with mice treated with anti–E. tenella (controls) and untreated mice and CD8+CD44hiCD62Llow cells (Fig. 5A–C). The percentage of peripheral CD4+CD25+FoxP3+ cells (Tregs) increased in mIL-21R.Fc–treated mice compared with the other two groups (Fig. 5D and F), and a higher Treg-to-Teff ratio was evident in mIL-21R.Fc–treated mice as well (Fig. 5E). Ex vivo rechallenge of splenocytes harvested at day 14 from islet-transplanted mice with donor alloantigen (i.e., BALB/c splenocytes) displayed a profound regulation of the antidonor-specific immune response in mIL-21R.Fc–treated mice, as shown by the reduction of IFN-γ–producing cells in the ELISpot assay (BD Bioscience, San Jose, CA) compared with mice treated with anti–E. tenella and untreated mice (IFN-γ–producing cells per 0.5 × 106: untreated = 125.0 ± 11.3; anti–E. tenella treated = 163.8 ± 30.6; mIL-21R.Fc treated = 25.3 ± 4.9; mIL-21R.Fc treated vs. all, P ≤ 0.01) (Fig. 5G). Of interest, 14 days after islet transplantation, a reshaping of the peripheral cytokine profile was evident in mIL-21R.Fc–treated mice as well, with an overall suppression of proinflammatory cytokines, as shown by the reduction of peripheral serum levels of IFN-γ, IL-2, IL-6, and IL-15 (Fig. 5H) compared with controls and untreated mice. IL-4, IL-10, and IL-17 peripheral levels remained unchanged in response to all treatments (Fig. 5H).
Targeting IL-21/IL-21R promotes Treg neogenesis in vivo.
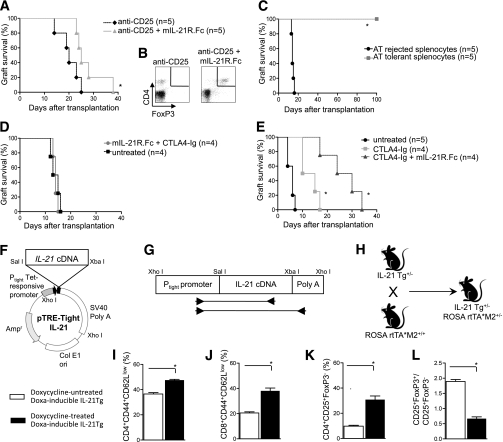
We next asked whether targeting the IL-21/IL-21R axis would be capable of promoting Treg neogenesis in vivo. Thus, we tested whether mIL-21R.Fc treatment could promote the generation of FoxP3 Tregs after depletion of CD25 using an anti–CD25-Ig injection (500 μg at day −6 and at day −1 before islet transplantation). Treatment with anti–CD25-Ig completely depleted CD25+ cells and FoxP3+ cells (data not shown). Targeting of the IL-21/IL-21R axis (with mIL-21R.Fc) during anti–CD25-Ig treatment slightly prolonged islet graft survival compared with anti–CD25-Ig treatment alone (MST: anti–CD25-Ig + mIL-21R.Fc treated = 25; anti–CD25-Ig treated = 20 days; P = 0.04) (Fig. 6A). Moreover, mIL-21R.Fc showed the ability to newly generate splenic CD4+FoxP3+ cells (Fig. 6B). To verify the robustness of the tolerance induced by mIL-21R.Fc + CTLA4-Ig treatment, we recovered 1 × 107 splenocytes from long-term tolerant mice at day 50 after islet transplantation and adoptively transferred them into immunodeficient C57BL/6-RAG mice that had been islet transplanted the day before with BALB/c islets. Splenocytes from rejecting islet-transplanted C57BL/6 mice were used as controls. Of interest, 100% of C57BL/6-RAG mice that were adoptively transferred with splenocytes obtained from long-term tolerant mice did not reject BALB/c islets compared with the control group, which promptly rejected BALB/c islets (MST: long-term tolerant mice = 100 days; rejecting mice = 14 days; P < 0.0001) (Fig. 6C). To evaluate whether mice were immunocompetent, we transplanted skin grafts from third-party DBA/1 J (H2q) donors into mIL-21R.Fc + CTLA4-Ig–treated long-term tolerant mice and into the control group of rejecting mice. Both groups of mice invariably rejected third-party skin grafts at ∼14 days after transplantation, thus confirming that mice treated with mIL-21R.Fc + CTLA4-Ig are immunocompetent (Fig. 6D). Finally, we tested mIL-21R.Fc + CTLA4-Ig treatment in a stringent model of tolerance resistance and in which BALB/c islets were transplanted into NOD mice, confirming a prolongation of graft survival in mice treated with mIL-21R.Fc + CTLA4-Ig compared with both untreated mice and CTLA4-Ig alone–treated mice (Fig. 6E).
FIG. 6.
IL-21/IL-21R targeting generates new Tregs, and IL-21 inducible overexpression expands FoxP3− cells in vivo. To assess whether IL-21 targeting promotes the neogenesis of FoxP3 Tregs, we pretreated C57BL/6 mice with a CD25-depleting agent (i.e., anti–CD25-Ig capable of depleting CD4+CD25+ cells) the day before mice received islets from BALB/c donors. Mice were then treated with mIL-21R.Fc or were left untreated. Mice treated with mIL-21R.Fc displayed slight prolongation in graft survival (MST: anti–CD25-Ig treated = 20 days vs. anti–CD25-Ig + mIL21-R.Fc treated = 25 days, *P = 0.03) (A) but significant neogeneration of CD4+FoxP3+ cells (*P = 0.005) at the day of rejection compared with untreated mice (B). To test the robustness of tolerance toward alloantigen, long-term tolerant splenocytes harvested from mice treated with CTLA4-Ig + mIL-21R.Fc or from rejecting mice as controls were adoptively transferred (AT) into immunodeficient C57BL/6 RAG mice 1 day after BALB/c islet transplantation (C). Of mice receiving splenocytes from long-term tolerant mice, 100% did not reject islet grafts, whereas all mice injected with splenocytes harvested from rejecting mice rejected islet graft with a pattern similar to naïve untreated C57BL/6 mice (MST: tolerant splenocytes = 100 days vs. rejected splenocytes = 14 days, *P < 0.0001) (C). To test immunocompetence of long-term tolerant mice, skin grafts from third-party donors (DBA/1 J [H2q]) were transplanted into mice treated with mIL-21R.Fc + CTLA4-Ig or control rejecting mice (D). All mice rejected donor-derived skin grafts with the same timing, confirming that mice treated with CTLA4-Ig + mIL-21R.Fc are immunocompetent (D). To further test the efficacy of mIL-21R.Fc + CTLA4-Ig treatment, we performed islet transplantation in a stringent model of tolerance resistance. BALB/c islets were transplanted into spontaneously hyperglycemic NOD mice and treated with CTLA4-Ig + mIL-21R.Fc, CTLA4-Ig alone, or were left untreated. Mice treated with CTLA4-Ig + mIL-21R.Fc showed a significant prolongation of graft survival compared with both CTLA4-Ig–treated (*P = 0.01) and untreated mice (*P = 0.008) (E). A transgenic C57BL/6 doxycycline-inducible IL-21-Tg mouse was generated, allowing IL-21 to be induced when needed (F–H). The murine IL-21 cDNA was subcloned into the Sal I and Xba I sites of the pTRE-Tight vector (F). The Xho I restriction fragment, encompassing the Ptight tet-responsive promoter, the IL-21 cDNA, and the SV40 poly A sequence, was excised and used to generate IL-21-Tg founder mice on a C57BL/6 background. The location and relative size of the PCR products used to identify the Tg mice are indicated (G). The IL-21-Tg mice were crossed with ROSA rtTA*M2 transgenic mice to introduce the Doxa-regulated reverse transcriptional activator. The resulting progeny were screened by PCR to identify the IL-21-Tg+ mice used in this study (H). Doxycycline-inducible IL-21-Tg mice (on a C57BL/6 background) were transplanted with BALB/c islets and administered doxycycline at day 0 to induce IL-21. At the time of rejection, doxycycline-induced mice (n = 5) showed an increased percentage of CD4+CD44hiCD62Llow cells (*P = 0.0009) (I), CD8+CD44hiCD62Llow cells (*P = 0.001) (J), and CD4+CD25+FoxP3− cells (*P = 0.002) (K), and a reduced CD25+FoxP3+-to-CD25+FoxP3− cell ratio (*P = 0.0001) (L) in the spleen compared with control mice (n = 4).
IL-21/IL-21R signaling in doxycycline-inducible ROSA-rtTA-IL-21-Tg mice expands Teffs and FoxP3− cells.
To confirm the paramount effect of IL-21 on FoxP3 and Treg function/survival in vivo, we generated the doxycycline-inducible ROSA-rtTA-IL-21-Tg mouse (Doxa-inducible IL-21-Tg). This transgenic C57BL/6 mouse expresses mIL-21 cDNA in a tetracycline-responsive element composed of the tet operator sequence under the control of a tetracycline-controlled transcriptional activator M2 promoter (Fig. 6F–H). We then transplanted BALB/c islets into the Doxa-inducible IL-21-Tg mice, followed by daily doxycycline administration beginning from day 0 until rejection. Doxycycline-treated Doxa-inducible IL-21-Tg mice rejected the grafts with the same timing of Doxa-inducible IL-21-Tg untreated mice (MST: WT doxycycline treated = 14 days vs. Doxa-inducible IL-21-Tg untreated = 17 days, NS) (data not shown). Doxa-inducible IL-21-Tg mice treated with doxycycline showed similar percentage of CD4+CD25+FoxP3+ cells (data not shown), with a clear expansion of CD4+CD44hiCD62Llow cells (Fig. 6I), CD8+CD44hiCD62Llow cells (Fig. 6J), and CD4+CD25+FoxP3− cells (Doxa-inducible IL-21-Tg untreated = 9.7 ± 1.2%; Doxa-inducible IL-21-Tg doxycycline treated = 30.8 ± 5.0%; P = 0.002) (Fig. 6K).
Finally, CD4+CD25+Fox3+-to-CD4+CD25+FoxP3− cell ratio is reduced in Doxa-treated mice compared with controls (Fig. 6L). The absence of CD4+CD25+FoxP3+ cell reduction in Doxa-inducible IL-21-Tg mice observed in the acute model may be related to the fast tempo of acute rejection, which may not fit with the IL-21 role. To further clarify this issue, we have obtained long-term islet function in C57BL/6 islet-transplanted mice with CTLA4-Ig + mIL-21R.Fc, and we have then induced IL-21 by doxycycline administration 40 days after transplantation. Very few Tregs (3.7 ± 0.5%) were detected in mice 20 days after doxycycline administration, when mice started to be sick.
DISCUSSION
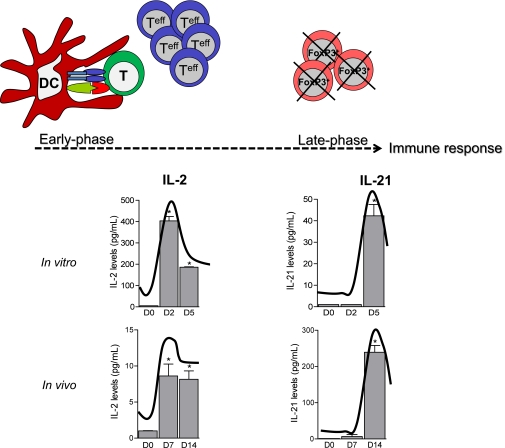
FoxP3+ Tregs play an essential role in maintaining immunological unresponsiveness to alloantigens and in suppressing the immune response (26). Herein, we propose IL-21 as an anti-FoxP3/anti-Treg cytokine of the late-phase alloimmune response that disrupts FoxP3 and alters Treg function, thereby acting as an antitolerogenic factor, thus eliciting an enhanced alloimmune response, and creating a barrier to transplant tolerance (1). In our study, IL-21 was increased in vitro after 5 days in anti-CD3/CD28 and MLR assays and in vivo 14 days after islet transplantation. IL-21R expression decreased in vitro after 5 days of stimulation and was found elevated in the graft infiltrate of rejecting untreated mice at later time points as well. Furthermore, mIL-21R.Fc (a fusion protein that prevents the binding of murine IL-21 to IL-21R) (22) is effective in vitro only in the late phase of allostimulation. These data, combined with the head-to-head comparison of IL-2 and IL-21 levels in vitro and in vivo (Fig. 7), revealed that in a “physiological” alloimmune setting, IL-21 is not responsible for the early response to alloantigen (unlike IL-2), but it does exert its function in the late-phase immune response (Fig. 7) (27). It is possible that IL-21 ensures the redundancy of the system, thus maintaining activation of the alloimmune response and avoiding Treg counterregulation (Fig. 7). IL-21 is capable of enhancing the alloimmune response, as shown by our in vitro experiments with rmIL-21 and with IL-21 plasmid–treated or hIL-21-Tg mice. However, it is likely that IL-21 may have a different role in the physiological alloimmune setting as a mediator of FoxP3 and of Treg dysfunction. The IL-21 interplay with Tregs was tested in vitro, using assays for Treg generation, survival, and function, during the challenge with rmIL-21 or mIL-21R.Fc, or by using hIL-21-Tg, IL-21 plasmid–treated (which overexpress IL-21), or IL-21R−/− mice. We show that TGF-β–driven conversion of CD4+CD25− T cells (FoxP3−), Treg survival, and Treg suppressive function are negatively affected by rmIL-21 but positively affected by mIL-21R.Fc. Indeed, we demonstrate an inhibition of FoxP3 and FoxP3-dependent gene transcripts by IL-21 (28), without evidence of IL-21–mediated epigenetic reprogramming of the FoxP3 gene, at least at the methylation level. These data were further confirmed by the inability of the demethylating agent decitabine to rescue Tregs in the presence of rmIL-21, thus indicating that despite the fact that decitabine is a robust inducer of Tregs in vitro (29), IL-21 is more potent in inhibiting Treg generation. We therefore demonstrate that blockade of the IL-21/IL-21R axis (both chemically and genetically) during the alloimmune response in vivo promotes the expansion of intragraft and peripheral Tregs and induces tolerance when combined with CTLA4-Ig, which targets early T-cell activation and is likely IL-21 independent. We show that mIL-21R.Fc promotes FoxP3 Treg neogenesis in vivo and that the mechanism of tolerance induction is robust and transferable to nude mice. The strong effect of the combination of mIL-21R.Fc and CTLA4-Ig on alloimmunity does not apply to NOD mice, which show the coexistence of strong allo- and autoimmune anti-islet responses (30). Autoimmune response is mediated by several different cell types (e.g., B cells, NK cells, macrophages, and Th17 cells), which may not be targeted by mIL-21R.Fc, but this aspect will be further investigated. Finally, we provide evidence that IL-21 promotes in vivo FoxP3− cell expansion. In fact, in our islet-transplanted Doxa-inducible ROSA-rtTA-IL-21-Tg mice (31,32), in which IL-21 is induced by doxycycline administration, both CD4+ and CD8+ activated/effector cells were found increased compared with controls, while CD25+FoxP3+-to-CD25+FoxP3− cell ratio was reduced. Furthermore, long-term tolerant mice overexpressing IL-21 (by doxycycline administration) showed very few Tregs. Taken together, our data suggest that IL-21 is a potent inhibitor of the FoxP3 Treg chain and an inducer of Teffs and that the blockade of the IL-21/IL-21R pathway may induce a more tolerogenic environment.
FIG. 7.
IL-21 acts as an antitolerogenic cytokine of the late phase of alloimmune response. In the early phase of the alloimmune response, antigen-presenting cells activate T cells through two signals. Proinflammatory cytokines (i.e., IL-2) are locally released, leading to alloreactive T-cell proliferation and destruction of the graft (D0 vs. all, *P < 0.01). In the late phase of the alloimmune response, Teffs release IL-21, thus ablating FoxP3+ Tregs, reducing FoxP3 gene transcripts, decreasing Treg generation and Treg suppressive properties, increasing Treg apoptosis, and inducing Teff expansion (D5 and D14 vs. all, *P < 0.01). D, day. DC, dendritic cells. (A high-quality color representation of this figure is available in the online issue.)
We posit that IL-21, together with other recently identified cytokines (33–35), is part of a network of antitolerogenic cytokines that through induction of Treg apoptosis or dysfunction, could represent a barrier to allograft tolerance. We thus posit that blockade of the IL-21/IL-21R pathway should be a precondition for many tolerogenic protocols in transplantation. Our strategy may be relevant in controlling acute as well as chronic rejection, which causes delayed islet graft loss (36–38) and represents a barrier for long-term allograft survival (39,40), since it abrogates T-cell activation in the early phase of posttransplant alloimmune response (with CTLA4-Ig) and expands/induces Tregs in the late phase (with IL-21 antagonist). We thus propose to start both drugs at the day of transplantation, discontinuing CTLA4-Ig treatment later on and prolonging the administration of IL-21 antagonist.
ACKNOWLEDGMENTS
A.J.Z. has received National Institutes of Health Grant U01-AI-082966. P.F. has received a Juvenile Diabetes Research Foundation Career Development Award and the American Society of Nephrology Career Development Award. W.J.L. is supported by the Intramural Research Program, National Heart, Lung, and Blood Institute, National Institutes of Health.
D.Y. is a paid employee of Pfizer. P.F. has received support from an Independent Investigation Research Proposal from Novartis. W.J.L. is an inventor on patents and patent applications related to IL-21. Pfizer provided the mIL-21R.Fc compound. No other potential conflicts of interest relevant to this article were reported.
A.P. performed research, analyzed data, and wrote the manuscript. M.C. performed research and analyzed data. A.V., K.M.L., S.T., M.D., S.K., L.C., and B.G.M. performed research. P.H., A.S., W.J.L., J.F.M., and A.J.Z. designed research. D.Y. contributed vital new reagents. M.H.S. designed research. P.F. designed research and edited the manuscript.
Parts of this study were presented in abstract form at the 70th Scientific Sessions of the American Diabetes Association, Orlando, Florida, 25–29 June 2010.
The authors are grateful to Pfizer for providing the mIL-21R.Fc compound.
Footnotes
This article contains Supplementary Data online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db11-0880/-/DC1.
REFERENCES
- 1.Walsh PT, Strom TB, Turka LA. Routes to transplant tolerance versus rejection; the role of cytokines. Immunity 2004;20:121–131 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Valujskikh A, Baldwin WM, 3rd, Fairchild RL. Recent progress and new perspectives in studying T cell responses to allografts. Am J Transplant 2010;10:1117–1125 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Demirci G, Gao W, Zheng XX, Malek TR, Strom TB, Li XC. On CD28/CD40 ligand costimulation, common gamma-chain signals, and the alloimmune response. J Immunol 2002;168:4382–4390 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Coquet JM, Kyparissoudis K, Pellicci DG, et al. IL-21 is produced by NKT cells and modulates NKT cell activation and cytokine production. J Immunol 2007;178:2827–2834 [DOI] [PubMed] [Google Scholar]
- 5.Parrish-Novak J, Dillon SR, Nelson A, et al. Interleukin 21 and its receptor are involved in NK cell expansion and regulation of lymphocyte function. Nature 2000;408:57–63 [DOI] [PubMed] [Google Scholar]
- 6.Casey KA, Mescher MF. IL-21 promotes differentiation of naive CD8 T cells to a unique effector phenotype. J Immunol 2007;178:7640–7648 [DOI] [PubMed] [Google Scholar]
- 7.Zhou L, Ivanov II, Spolski R, et al. IL-6 programs T(H)-17 cell differentiation by promoting sequential engagement of the IL-21 and IL-23 pathways. Nat Immunol 2007;8:967–974 [DOI] [PubMed] [Google Scholar]
- 8.Korn T, Bettelli E, Gao W, et al. IL-21 initiates an alternative pathway to induce proinflammatory T(H)17 cells. Nature 2007;448:484–487 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nurieva R, Yang XO, Martinez G, et al. Essential autocrine regulation by IL-21 in the generation of inflammatory T cells. Nature 2007;448:480–483 [DOI] [PubMed] [Google Scholar]
- 10.Bucher C, Koch L, Vogtenhuber C, et al. IL-21 blockade reduces graft-versus-host disease mortality by supporting inducible T regulatory cell generation. Blood 2009;114:5375–5384 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hanash AM, Kappel LW, Yim NL, et al. Abrogation of donor T cell IL-21 signaling leads to tissue-specific modulation of immunity and separation of GVHD from GVL. Blood 2011;118:446–455 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Peluso I, Fantini MC, Fina D, et al. IL-21 counteracts the regulatory T cell-mediated suppression of human CD4+ T lymphocytes. J Immunol 2007;178:732–739 [DOI] [PubMed] [Google Scholar]
- 13.McGuire HM, Walters S, Vogelzang A, et al. Interleukin-21 is critically required in autoimmune and allogeneic responses to islet tissue in murine models. Diabetes 2011;60:867–875 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Li Y, Yee C. IL-21 mediated Foxp3 suppression leads to enhanced generation of antigen-specific CD8+ cytotoxic T lymphocytes. Blood 2008;111:229–235 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Spolski R, Kim HP, Zhu W, Levy DE, Leonard WJ. IL-21 mediates suppressive effects via its induction of IL-10. J Immunol 2009;182:2859–2867 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zheng Q, Xu Y, Liu Y, et al. Induction of Foxp3 demethylation increases regulatory CD4+CD25+ T cells and prevents the occurrence of diabetes in mice. J Mol Med (Berl) 2009;87:1191–1205 [DOI] [PubMed] [Google Scholar]
- 17.Zheng Y, Josefowicz SZ, Kas A, Chu TT, Gavin MA, Rudensky AY. Genome-wide analysis of Foxp3 target genes in developing and mature regulatory T cells. Nature 2007;445:936–940 [DOI] [PubMed] [Google Scholar]
- 18.Baan CC, Balk AH, Dijke IE, et al. Interleukin-21: an interleukin-2 dependent player in rejection processes. Transplantation 2007;83:1485–1492 [DOI] [PubMed] [Google Scholar]
- 19.Vergani A, D’Addio F, Jurewicz M, et al. A novel clinically relevant strategy to abrogate autoimmunity and regulate alloimmunity in NOD mice. Diabetes 2010;59:2253–2264 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fantini MC, Dominitzki S, Rizzo A, Neurath MF, Becker C. In vitro generation of CD4+ CD25+ regulatory cells from murine naive T cells. Nat Protoc 2007;2:1789–1794 [DOI] [PubMed] [Google Scholar]
- 21.Venken K, Thewissen M, Hellings N, et al. A CFSE based assay for measuring CD4+CD25+ regulatory T cell mediated suppression of auto-antigen specific and polyclonal T cell responses. J Immunol Methods 2007;322:1–11 [DOI] [PubMed] [Google Scholar]
- 22.Young DA, Hegen M, Ma HL, et al. Blockade of the interleukin-21/interleukin-21 receptor pathway ameliorates disease in animal models of rheumatoid arthritis. Arthritis Rheum 2007;56:1152–1163 [DOI] [PubMed] [Google Scholar]
- 23.Jin H, Carrio R, Yu A, Malek TR. Distinct activation signals determine whether IL-21 induces B cell costimulation, growth arrest, or Bim-dependent apoptosis. J Immunol 2004;173:657–665 [DOI] [PubMed] [Google Scholar]
- 24.Cejas PJ, Walsh MC, Pearce EL, et al. TRAF6 inhibits Th17 differentiation and TGF-beta-mediated suppression of IL-2. Blood 2010;115:4750–4757 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Vincenti F, Larsen C, Durrbach A, et al. ; Belatacept Study Group Costimulation blockade with belatacept in renal transplantation. N Engl J Med 2005;353:770–781 [DOI] [PubMed] [Google Scholar]
- 26.Cobbold SP, Adams E, Graca L, et al. Immune privilege induced by regulatory T cells in transplantation tolerance. Immunol Rev 2006;213:239–255 [DOI] [PubMed] [Google Scholar]
- 27.Le Moine A, Goldman M. Non-classical pathways of cell-mediated allograft rejection: new challenges for tolerance induction? Am J Transplant 2003;3:101–106 [DOI] [PubMed] [Google Scholar]
- 28.Brady J, Hayakawa Y, Smyth MJ, Nutt SL. IL-21 induces the functional maturation of murine NK cells. J Immunol 2004;172:2048–2058 [DOI] [PubMed] [Google Scholar]
- 29.Choi J, Ritchey J, Prior JL, et al. In vivo administration of hypomethylating agents mitigate graft-versus-host disease without sacrificing graft-versus-leukemia. Blood 2010;116:129–139 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Monti P, Scirpoli M, Maffi P, et al. Islet transplantation in patients with autoimmune diabetes induces homeostatic cytokines that expand autoreactive memory T cells. J Clin Invest 2008;118:1806–1814 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Britsch S, Goerich DE, Riethmacher D, et al. The transcription factor Sox10 is a key regulator of peripheral glial development. Genes Dev 2001;15:66–78 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Gopalkrishnan RV, Christiansen KA, Goldstein NI, DePinho RA, Fisher PB. Use of the human EF-1alpha promoter for expression can significantly increase success in establishing stable cell lines with consistent expression: a study using the tetracycline-inducible system in human cancer cells. Nucleic Acids Res 1999;27:4775–4782 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.O’Sullivan BJ, Thomas HE, Pai S, et al. IL-1 beta breaks tolerance through expansion of CD25+ effector T cells. J Immunol 2006;176:7278–7287 [DOI] [PubMed] [Google Scholar]
- 34.Bettelli E, Carrier Y, Gao W, et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 2006;441:235–238 [DOI] [PubMed] [Google Scholar]
- 35.Huber M, Steinwald V, Guralnik A, et al. IL-27 inhibits the development of regulatory T cells via STAT3. Int Immunol 2008;20:223–234 [DOI] [PubMed] [Google Scholar]
- 36.Davalli AM, Ogawa Y, Ricordi C, Scharp DW, Bonner-Weir S, Weir GC. A selective decrease in the beta cell mass of human islets transplanted into diabetic nude mice. Transplantation 1995;59:817–820 [PubMed] [Google Scholar]
- 37.Ricordi C, Inverardi L, Kenyon NS, Goss J, Bertuzzi F, Alejandro R. Requirements for success in clinical islet transplantation. Transplantation 2005;79:1298–1300 [DOI] [PubMed] [Google Scholar]
- 38.Kessler L, Parissiadis A, Bayle F, et al. ; GRAGIL Study Group Evidence for humoral rejection of a pancreatic islet graft and rescue with rituximab and IV immunoglobulin therapy. Am J Transplant 2009;9:1961–1966 [DOI] [PubMed] [Google Scholar]
- 39.Nankivell BJ, Alexander SI. Rejection of the kidney allograft. N Engl J Med 2010;363:1451–1462 [DOI] [PubMed] [Google Scholar]
- 40.Webber A, Hirose R, Vincenti F. Novel strategies in immunosuppression: issues in perspective. Transplantation 2011;91:1057–1064 [DOI] [PubMed] [Google Scholar]