Abstract
The human lipodystrophy gene product Berardinelli-Seip congenital lipodystrophy 2/seipin has been implicated in adipocyte differentiation, lipid droplet (LD) formation, and motor neuron development. However, the molecular function of seipin and its disease-causing mutants remains to be elucidated. Here, we characterize seipin and its mis-sense mutants: N88S/S90L (both linked to motoneuron disorders) and A212P (linked to lipodystrophy) in cultured mammalian cells. Knocking down seipin significantly increases oleate incorporation into triacylglycerol (TAG) and the steady state level of TAG, and induces the proliferation and clustering of small LDs. By contrast, overexpression of seipin reduces TAG synthesis, leading to decreased formation of LDs. Expression of the A212P mutant, however, had little effect on LD biogenesis. Surprisingly, expression of N88S or S90L causes the formation of many small LDs reminiscent of seipin deficient cells. This dominant-negative effect may be due to the N88S/S90L-induced formation of inclusions where wild-type seipin can be trapped. Importantly, coexpression of wild-type seipin and the N88S or S90L mutant can significantly reduce the formation of inclusions. Finally, we demonstrate that seipin can interact with itself and its mutant forms. Our results provide important insights into the biochemical characteristics of seipin and its mis-sense mutants, and suggest that seipin may function to inhibit lipogenesis.
Keywords: Berardinelli-Seip congenital lipodystrophy 2, lipid droplets, adipogenesis, lipodystrophy
Obesity is characterized by the accumulation of fully differentiated adipocytes loaded with lipid droplets (LDs) (1, 2). The increasing prevalence of obesity and its related disorders in both developed and developing countries demands a better understanding of the molecular mechanisms underlying fat storage and adipogenesis (adipocyte differentiation) (2, 3). Of particular relevance to adipogenesis is the rare genetic disease, congenital generalized lipodystrophy (CGL, also known as Berardinelli-Seip congenital lipodystrophy, or BSCL), an autosomal recessive disorder that is characterized by a near total loss of adipose tissue, severe insulin resistance, hypertriglyceridemia, and fatty liver (4). Genome-wide linkage analyses have identified two loci for CGL: CGL type 1 (CGL1) is caused by mutations in the 1-acylglycerol-3-phosphate-O-acyl transferase 2 (AGPAT2) gene and CGL2 by mutations in the BSCL2 gene, which encodes seipin (5, 6). Recently, a homozygous nonsense mutation in caveolin-1 has been linked to CGL in a candidate gene approach, and the malfunction of cavin/PTRF can cause CGL through its effect on caveolins (7–9). Both AGPAT2 and caveolin-1/cavin have clear cellular functions: AGPAT2 catalyzes the formation of phosphatidic acid and appears to control adipogenesis through modulating the synthesis of phospholipids, and caveolin-1/cavin has a defined role in caveolae formation. In contrast, little is known about the molecular function of seipin, despite the fact that CGL2/BSCL2 patients demonstrate the most severe form of fat loss (4).
The human BSCL2/seipin gene has three transcripts: 1.6kb, 1.8kb, and 2.2kb (10). The 1.8kb mRNA is exclusively expressed in the brain and testis but the other two mRNA transcripts are ubiquitously expressed (11). It has been shown recently that seipin is highly expressed in adipose tissue and is strongly induced during adipocyte differentiation (12, 13). The essential role of seipin in adipogenesis has been confirmed in cultured cells, fly and mice (14, 15). Another recent surprising yet exciting finding is that both seipin and the yeast seipin homolog, Fld1p, play a critical role in the cellular dynamics of LDs (16–18). It has been suggested that seipin/Fld1p may regulate the metabolism of fatty acids/phospholipids (phosphatidic acid), and/or the generation of LDs, although the molecular function of seipin remains to be elucidated (19–21).
Seipin is predicted to span the endoplasmic reticulum (ER) membrane twice, with both N- and C termini in the cytoplasm and a large luminal loop (10, 22). Due to alternative translation initiation sites, the human seipin/BSCL2 protein can be 398 or 462 amino acids in length, with the longer isoform having an additional 64 amino acids at the N-terminus. Mis-sense mutations in the luminal loop are linked to autosomal dominant motoneuron disorders (N88S and S90L), and also to the recessive CGL (A212P) (11, 23). It is believed that the N88S and S90L mutations disrupt the glycosylation of seipin, causing protein aggregation, ER stress, and neuronal cell death (22, 24). The A212P mutation probably leads to a partial or complete loss of seipin function, as most seipin mutations associated with CGL cause premature termination of seipin translation (5). In addition, the yeast equivalent of A212P renders the protein unstable (19).
Despite the important role of seipin in both LD formation and adipocyte differentiation, the molecular and cellular function of seipin remains to be elucidated. Here, we choose to characterize wild-type seipin and its three mis-sense mutants (N88S, S90L, and A212P) in nonadipogenic cultured cell lines because seipin is conserved throughout evolution, and because the aim of this study is not to address how mutations of seipin inhibits adipogenesis. Knocking down seipin increases triacylglycerol (TAG) synthesis whereas overexpression of seipin inhibits TAG synthesis and reduces LD formation. Interestingly, overexpression of N88S or S90L but not A212P exerts dominant-negative effects on LD formation. Our results provide important structural and functional insights into seipin and its mutants.
MATERIALS AND METHODS
Plasmid constructions
pmCherry-N1-seipinL (XhoI/XmaI), pmCherry-N1-seipinS (XhoI/XmaI), pmCherry-N1-seipinS 1-280AA (XhoI/XmaI), pmCherry-C1-seipinL (XhoI/XmaI), pmCherry-C1-seipinS (XhoI/XmaI), pEGFP-N1-seipinS (XhoI/XmaI), pCI-neo-Flag-seipinS (XhoI/NotI and NotI/NotI for inserting Flag and seipinS, respectively), pCMV5-seipinL-HA (EcoRI/XmaI) pCMV5-seipinL-Myc (EcoRI/XmaI), and pTRE2 hyg-seipinS-mCherry (NotI/SalI) were generated and used in this study. Plasmids expressing SEC61B-GFP and SEC31A-GFP were a gift from Dr Wanjin Hong (Institute of Molecular and Cell Biology, Singapore). Site-directed mutagenesis of cloned seipinS was performed using KOD DNA Polymerase (Novagen) with forward primer ATCCAGCTGTATGGACCCTACCTCCGCATCCAC and reverse GTGGATGCGGAGGTAGGGTCCATACAGCTGGAT for A212P, forward TCCTTCCCTGTTGCCAGTGTCTCGCTGACTAAG and reverse CTTAGTCAGCGAGACACTGGCAACAGGGAAGGA for N88S, and forward CCTGTTGCCAATGTCTTGCTGACTAAGGGTGGA and reverse TCCACCCTTAGTCAGCAAGACATTGGCAACAGG for S90L.
Cell culture
HeLa and NIH3T3 cells were grown in DMEM supplemented with 10% FBS (Invitrogen). Transient transfections of plasmids were performed using the calcium phosphate method. Small interfering RNA (siRNA) oligos were transfected using the Lipofectamine RNAiMAX reagent (Invitrogen) according to manufacturer's instruction. SiRNAs (SiBscl2-1 and siBscl2-2 against murine Bscl2, siBSCL2-1, and siBSCL2-2 against human BSCL2) were purchased from Sigma and their sequences are shown in supplementary Table I. To induce TAG synthesis and LD formation, cells were treated with 200 µM BSA-coupled oleate (molar ratio of oleate to BSA ∼8:1) 48 h after transfections and incubated.
Fluorescence microscopy
Fluorescence microscopy was performed under a Leica CTR5500 microscope (Wetzlar, Germany) with an EL6000 fluorescent lamp. Images were taken with a DFC300 FX digital camera and a Leica LAS AF software. Enhanced green fluorescent protein signal was visualized with a Leica GFP filter (a 470/40-nm bandpass excitation filter, a 500-nm dichromatic mirror, and a 525/50-nm bandpass emission filter), and mCherry with a Leica TX2 filter (a 560/40-nm bandpass excitation filter, a 595-nm dichromatic mirror, and a 645/75-nm bandpass emission filter). For observation of LDs, cells were stained with 2 µg/ml BODIPY 493/503 (Invitrogen) and observed with the Leica GFP filter.
Lipid analysis
Mammalian cells were lysed with Lysis buffer A (20 mM Tris-HCl, pH 7.4/150 mM NaCl/1 mM EDTA/1% SDS) containing protease inhibitors. Protein concentrations were determined with a BCA assay kit (Pierce). Subsequently hexane-isopropanol (3:2, v/v) was added to the lysates and lipids were extracted by vigorous vortexing for 2 min. After hexane was collected and evaporated under nitrogen gas, neutral lipids were separated via TLC essentially as described elsewhere (25). For quantitation of TAG and cholesteryl ester, TLC plates were put in an iodine tank for 15 min and scanned. Densitometric analysis was performed using the Image Gauge 4.0 software (Fujifilm Science Lab). For comparison, densitometric units of TAG and cholesteryl ester bands were normalized to protein concentrations.
Lipidomic analysis of cellular phospholipids was performed as described (17).
For the assay of fluorescent fatty acid uptake by cells and its incorporation into TAG, cells were treated with 2 µg/ml BODIPY FL C16 (Invitrogen) and incubated for either 1 h or 16 h followed by observation with the GFP filter. To measure the rate of cellular TAG lipolysis, glycerol released from cells into culture media was determined by a glycerol assay kit (Cayman chemical). Briefly, 48 h after transfections, cells were induced for TAG formation in the presence of 200 µM BSA-coupled oleate for another 16 h, followed immediately by collection of media and assay of glycerol levels. To determine the rate of incorporation of [14C]oleic acid into TAG and cholesteryl ester, 48 h after transfections, cells were pulsed with 0.5 μCi/ml [14C]oleic acid for 2 h. Lipids were extracted by hexane, and separated by TLC. TLC plates were then exposed to a BAS-MS imaging plate (Fujifilm) for 72 h at room temperature. The imaging plate was visualized using the FLA-5100 phosphoimager (Fujifilm). The intensities of bands corresponding to TAG and cholesteryl ester were determined using the Image Gauge 4.0 software (Fujifilm Science Lab), and normalized to protein concentrations.
In vitro diacylglycerol esterification assay
Diacylglycerol acyltransferase (DGAT) assay was performed as described previously with modifications (26). Briefly, 5 µg microsome proteins were assayed in 0.25 M sucrose/1 mM EDTA/150 mM MgCl2/ 100 mM Tris·HCl (pH 7.4) containing 40 µg of BSA, 0.4 mM dioleoylglycerol (which was solubilized in acetone), and 2.5 nmol [14C]oleoyl-CoA (120,000dpm/nmol) (final volume 0.2 ml). Reactions were carried out for 10 min and [14C]triacylglycerols formed were measured by scintillation counting.
Antibodies, immunoprecipitation, immunofluorescence, and immunoblot
DsRed polyclonal antibody was purchased from Clontech for the detection of mCherry and fusion proteins. Flag monoclonal antibody was from Sigma, actin monoclonal antibody from Abcam, and HA and Myc monoclonal antibodies from Covance. For immunoprecipitation, cells were lysed with Lysis buffer B, 50 mM Hepes-KOH, pH 7.4/100 mM NaCl/1.5 mM MgCl2/0.5% (v/v) CHAPSO containing protease inhibitors (27). Cell lysates were passed through a 22-gauge needle 5 times, extracted by rotating for 1 h at 4°C, and clarified by centrifugation at 100,000 g for 15 min. The lysates were precleared by incubation for 1 h at 4°C with 50 μl of protein G sepharose beads (Sigma). Precleared lysates were rotated overnight at 4°C with 10 μg of monoclonal anti-Flag together with 50 μl of protein G sepharose beads. The resulting pelleted beads were washed three times (10 min at 4°C) with 1 ml of Lysis buffer B containing protease inhibitors, resuspended in 80 μl of 2× SDS sample buffer, boiled for 5 min, and subjected to SDS/10% PAGE and immunoblot analysis.
For immunofluorescence, cells were fixed with 4% paraformaldehyde and permeabilized with 0.1% saponin/PBS for 30 min, followed by blocking with 3% BSA/0.05% saponin in PBS for 1 h. Cells were incubated with primary antibodies diluted in 1% BSA/PBS for 1 h, followed by incubation with the Alexa Fluor 568 conjugated secondary antibodies for 1 h.
For immunoblot analysis, cells were lysed with Lysis buffer A and passed through a 22-gauge needle five times and clarified by centrifugation at 3,000 g for 5min. Samples were subjected to SDS/10% PAGE, transferred to nitrocellulose filters and subjected to immunoblot analysis with antibodies. Bound antibodies were visualized by chemiluminescence using the ECL Western blotting detection reagents (GE Healthcare) according to manufacturer's instructions.
For Blue Native PAGE, HeLa cells transfected with wild-type or mutant forms of mCherry- or Flag-tagged seipin were lysed with Lysis buffer B. After clearing unsolubilized debris by centrifugation at 13,000 g for 10 min, lysates were loaded onto 4-16% NativePAGE gels (Invitrogen) for protein separation. Flag- and mCherry-tagged seipin were probed with anti-Flag and anti-mCherry, respectively.
Statistics
For statistics, experiments were performed in triplicates. The results are presented as mean ± SD. Two-tailed Student's t-test was used for comparison.
RESULTS
Knocking down seipin expression increases TAG synthesis
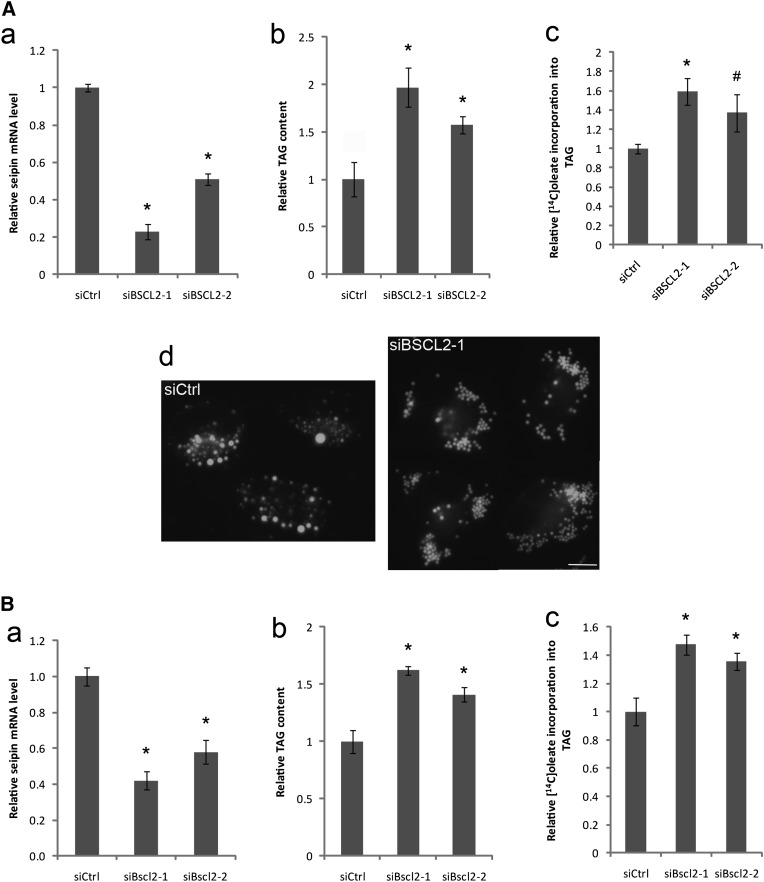
We previously showed that seipin/FLD1 deletion in yeast increased TAG synthesis (17). A recent study in Drosophila also demonstrated that seipin ablation caused TAG accumulation in the salivary gland in a tissue autonomous manner (15). However, the role of seipin in TAG metabolism is somewhat confusing in mammalian cells; a reduction in TAG synthesis was observed in a study using seipin-deficient lymphoblastoid cells (16), whereas proliferation of LDs was observed in seipin deficient fibroblasts (18). We decided to examine the role of seipin in lipid metabolism using common cultured mammalian cell lines. siRNA was employed to first knock down seipin/BSCL2 expression in HeLa cells. Quantitative PCR (qPCR) analysis revealed that siBSCL2-1 reduced seipin mRNA level by ∼80% (Fig. 1A-a), which resulted in a two-fold increase in oleate-induced TAG formation (Fig. 1A-b). In addition, pulse labeling with [14C] oleate displayed a 60% increase in oleate incorporation into TAG (Fig. 1A-c). SiBSCL2-2, which knocked down seipin expression by ∼50%, also induced TAG synthesis, albeit to a lesser extent (Fig. 1A). Interestingly, seipin knockdown by siBSCL2-1 in HeLa cells also resulted in the proliferation and clustering of small LDs (Fig. 1A-d). It should be noted that the proliferation and clustering of LDs has been observed in seipin-deficient human fibroblasts and lymphoblastoid cells and also in yeast cells deleted for the FLD1 gene encoding yeast seipin (16–18). Overall, these results indicate that in cultured mammalian cells, seipin functions to inhibit TAG synthesis consistent with findings from yeast and Drosophila (15).
Fig. 1.
Knocking down seipin by siRNA increases TAG synthesis in HeLa and 3T3-L1 cells. HeLa (A) and 3T3-L1 (B) cells were transfected with a control siRNA (siCtrl) or siRNAs against seipin (siBSCL2-1 and siBSCL2-2 to HeLa cells; siBscl2-1 and siBscl2-2 to 3T3-L1 cells) for 48 h. Knockdown efficacy of siRNAs was determined by qPCR (a). SiRNA-transfected cells were treated with 200 µM BSA-coupled oleate for 16 h and relative cellular TAG levels were analyzed via TLC (b). SiRNA-transfected cells were pulsed with [14C]oleic acid for 2 h. Incorporation of [14C]oleic acid into TAG was normalized to protein concentrations (c). * P < 0.01; # P < 0.05, compared with siCtrl. The morphology of LDs was examined in HeLa cells transfected with siCtrl or siBSCL2-1 (A-d). Bar, 10 μm.
We next assessed the effect of seipin knock-down in a different cell line, the 3T3-L1 cells that were not induced to differentiate. SiBscl2-1 and siBscl2-2 knocked down mouse seipin expression by ∼60% and ∼40% in 3T3-L1 cells, respectively (Fig. 1B-a). Knocking down seipin increased oleate-induced TAG synthesis by ∼40% to ∼70% and [14C] oleate incorporation into TAG by 40%–50% (Fig. 1B-b, c). Our data showed that cellular TAG synthesis correlated with the level of seipin, highlighting an important role of seipin in TAG synthesis. We failed to observe the clustering of LDs in 3T3-L1 cells, possibly because these two siRNAs could only achieve less than 60% seipin knockdown. Nevertheless, the differentiation of 3T3-L1 preadipocytes was blocked by introducing these siRNAs (data not shown), in agreement with previous reports that seipin knockdown by ∼50% could block the adipogenic program (12, 13). Together, these data demonstrate that seipin has a clear role in cellular TAG synthesis.
Expression of wild-type and mutant seipin in HeLa cells
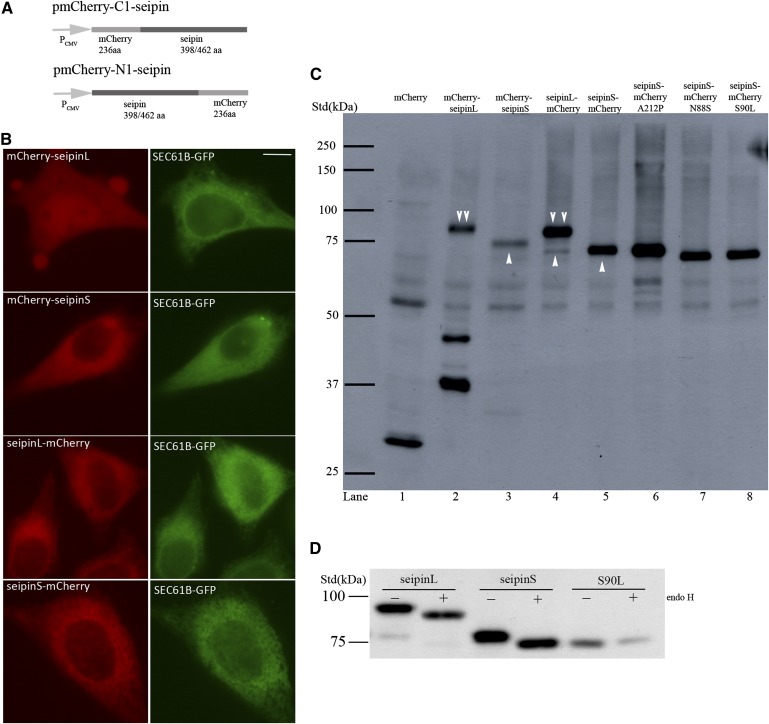
We next decided to examine the effect(s) of seipin overexpression. Conflicting results have been reported on the overexpression of tagged forms of seipin. The original study reported that although seipin N88S and S90L appeared to localize to inclusions when examined by fluorescence microscopy, monomeric forms of seipin were clearly dominant when detected by Western blotting (11). Two recent studies, however, failed to detect the monomeric forms of seipin and suggested that even wild-type seipin may aggregate upon overexpression (12, 13). To better understand the biochemical properties and consequences of overexpressed seipin, we generated constructs expressing either N- or C-terminally mCherry-tagged seipin using the pmCherry-C1 or pmCherry-N1 plasmid, respectively (Fig. 2A), and transiently transfected HeLa cells. As shown in Fig. 2B, for the short isoform of human seipin (seipinS, NP_116056, 398aa), both mCherry-seipinS (mCherry tagged at the N-terminus of seipin) and seipinS-mCherry (mCherry tagged at the C terminus of seipin) displayed a perinuclear reticular distribution of red fluorescence, which colocalized with the ER marker SEC61B-GFP. For the long isoform of human seipin (seipinL, NP_001124174, 462aa), a typical ER pattern was seen when seipinL-mCherry (mCherry at C terminus of seipin) was expressed. In contrast, mCherry-seipinL (mCherry at N-terminus of seipin) appeared to exist throughout the cell but was not restricted to the ER. Immunoblotting analysis using an anti-mCherry antibody revealed a single band of about 75 kDa for both mCherry-seipinS and seipinS-mCherry (the two bands between 50–60 kDa are nonspecific as they are seen in all lanes) (Fig. 2C, lanes 3 and 5). Lysate prepared from cells expressing seipinL-mCherry produced one major band of ∼83 kDa and one minor band of ∼75 kDa, reminiscent of seipinS-mCherry (Fig. 2C, lane 4). This suggested that, among other possibilities, seipinL-mCherry could be cleaved after the methionine encoded by the second start codon. Interestingly, mCherry-seipinL displayed three bands (83 kDa, 45 kDa, and 37 kDa, respectively) (Fig. 2C, lane 2). The largest band was most likely the full length mCherry-seipinL, and the smaller ones (45 and 37 kDa) appeared to be the result of truncations near the N-terminus of seipinL. These data agree with the fact that seipinL-mCherry can also produce a seipinS-mCherry-like band due to N-terminal truncation. In addition, the truncated forms of mCherry-seipinL likely explained the pancellular distribution of the mCherry signal when mCherry-seipinL was expressed (Fig. 2B). Together, these data demonstrate clearly that mCherry-tagged seipin, when overexpressed, can be easily separated and detected by Western blotting and suggest that seipin does not readily form aggregates due to high hydrophobicity.
Fig. 2.
Expression of mCherry-tagged seipin proteins in HeLa cells. A: Schematic diagrams of seipin-expressing pmCherry-N1 and pmCherry-C1 plasmids. Seipin has one short isoform (seipinS) which encodes 398 amino acids and one long isoform (seipinL), which encodes 462 amino acids. B: HeLa cells were transiently transfected with a plasmid expressing SEC61B-GFP and plasmids expressing either seipinL-mCherry, seipinS-mCherry, mCherry-seipinL, or mCherry-seipinS, and observed for GFP and mCherry signal. Bar, 10 μm. C: Immunoblot analysis of mCherry, mCherry-tagged wild-type and mutant seipin proteins. Arrow head, mCherry-tagged short isoform of seipin; double arrow head, mCherry-tagged long isoform of seipin. D: Lysates prepared from HeLa cells expressing either seipinL-mCherry, seipinS-mCherry, or a mutant of seipinS-mCherry, S90L, were treated with or without endo H and probed with mCherry antibody.
To further investigate the discrepancies between our results and the published ones, we examined seipinL-HA and seipinL-Myc by SDS-PAGE and immunoblotting because the HA and Myc tagged forms of seipin were reported to aggregate (12, 13). Interestingly when protein lysates mixed with Laemmli buffer were loaded for SDS-PAGE without boiling, monomeric forms of seipin were clearly observed for both HA- and Myc-tagged seipin; in contrast, when the same protein samples were boiled at 95°C for 5 min before SDS-PAGE, HA- and Myc-tagged seipin appeared as a complex above the 250 kDa standard (supplementary Fig. I). It is noteworthy that after boiling both seipin-mCherry and mCherry-seipin also appeared as bands above 250 kDa (data not shown). These data indicate that boiling of protein samples causes the aggregation of seipin, which could not be separated as monomeric forms by SDS-PAGE. Therefore, the tagged forms of seipin may not aggregate in vivo and the perceived aggregation is probably due to mis-handling of protein samples containing the integral membrane protein seipin (supplementary Fig. I and personal communications with Dr. Justin Rochford).
We next generated constructs that express the lipodystrophy mutant A212P and the motor neuron disease mutants N88S and S90L by site-directed mutagenesis in seipinS-mCherry. The expression of these mutants was confirmed by immunoblotting (Fig. 2C). Consistent with the finding that seipin has a glycosylation site at amino acids 88-90 (NVS), the N88S and S90L mutants ran slightly faster. The Endo H treatment performed in our study further confirmed that both the long and the short isoforms of seipin are glycosylated (Fig. 2D).
Overexpression of wild-type seipin reduces TAG and lipid droplet biosynthesis
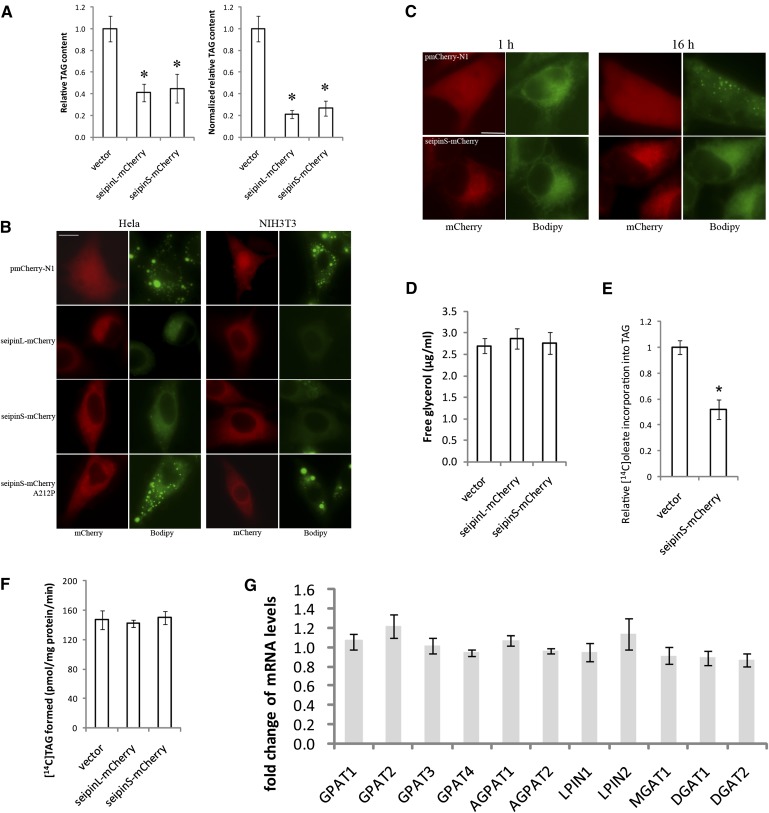
As seipin knockdown increased TAG synthesis and LD proliferation (Fig. 1), we next examined the effect of seipin overexpression on TAG synthesis and LD formation. We used the different forms of tagged seipin for this purpose for the following reasons: 1) We have difficulty detecting the untagged seipin because there is no reliable antibody (data not shown); 2) The tagged seipin localizes to the ER (Fig. 2B); 3) The tagged human seipin can rescue defects in FLD1 deficient yeast, suggesting the fusion protein is functional (17, 18); and 4) We used both GFP/mCherry and myc/HA tagged seipin to minimize the effect of the tag. HeLa cells were transiently transfected with vectors expressing seipinS-mCherry and seipinL-mCherry, and treated with 200 µM BSA-coupled oleate. TLC analysis of TAG revealed that seipin overexpression greatly reduced oleate-induced TAG synthesis; after normalization to the ∼75% transfection rate, cells expressing seipin-mCherry synthesized only about 30% of TAG relative to control cells (Fig. 3A). In contrast, overexpression of seipinS-mCherry did not lead to significant changes in cellular levels of major phospholipids (supplementary Fig. II).
Fig. 3.
Overexpression of mCherry-tagged seipin greatly reduces TAG synthesis and formation of LDs. Cells were transfected with empty or seipin-expressing pmCherry-N1 plasmids for 48 h before assays. A: Transfected HeLa cells were treated with 200 μM oleate for another 16 h and cellular TAG amount was analyzed subsequently. Right panel in A, relative cellular TAG content was normalized to 75% transfection rate [Normalized relative TAG content = (Relative TAG content-25%)/75%]. * P < 0.01, compared with empty vector control. B: Transfected HeLa and NIH3T3 cells were treated with 200 μM oleate for another 16 h. Cells were stained with BODIPY 493/503 and observed for LDs. Bar, 10 μm. C: Seipin overexpression did not affect fatty acid uptake. Transfected HeLa cells were incubated in the presence of 2 µg/ml BODIPY FL C16. At 1 h or 16 h, cells were fixed with 4% formaldehyde and observed for the distribution of BODIPY fluorescence. Bar, 10 μm. D: Seipin overexpression did not enhance TAG lipolysis. Transfected HeLa cells were further treated with 200 μM oleate for another 16 h. Free glycerol released into culture media was analyzed as described in Materials and methods. E: Seipin overexpression reduced oleate incorporation into TAG. Transfected HeLa cells were pulsed with [14C]oleic acid for 2 h. Incorporation of [14C]oleic acid into TAG was normalized to protein concentrations and 75% transfection rate. *, P < 0.01. F: Seipin overexpression does not affect TAG synthesis in vitro. Microsomes were prepared from transfected HeLa cells and incubated in the presence of 0.4 mM dioleoylglycerol and 2.5 nmol [14C]oleoyl-CoA (120,000 dpm/nmol) for 10 min. Lipids were separated via TLC and [14C]TAG formed was quantified by scintillation counting. G: Seipin overexpression does not significantly change the expression level of key lipogenic enzymes. Fold changes of the mRNA levels of genes involved in TAG synthesis (seipinS-mCherry/vector).
Reduced TAG synthesis upon seipin overexpression led us to examine its effect on LD biogenesis. As shown in Fig. 3B, overexpression of wild-type seipin, both the long and short isoforms, drastically reduced the formation of LDs in both HeLa and NIH3T3 cells. In contrast, even the strongest expression of mCherry alone by the empty vector pmCherry-N1 did not impair the formation of LDs, suggesting that the effect of seipin-mCherry overexpression on LD synthesis was seipin specific. To further rule out the possibility that the mCherry tag may alter the biophysical property and cellular function of seipin, thereby inhibiting TAG synthesis and LD formation, we also transfected HeLa cells with plasmids expressing C-terminally HA- and Myc-tagged seipinL. Our result showed that overexpression of both seipinL-HA and seipinL-Myc strongly blocked LD formation (supplementary Fig. III), which indicates that the inhibition of TAG and LD formation is indeed caused by overexpression of seipin.
To further ensure the effect of overexpressing seipin is physiologically relevant, we also employed an inducible expression system with the Tet response element upstream of a minimal CMV promoter (the Tet Expression Systems, Clontech) (28) to regulate the level of seipin expression (supplementary Fig. IV). Even expressed at a relatively very low level (10 ng/ml doxycycline), seipinS-mCherry was able to significantly inhibit LD formation (supplementary Fig. IV B). Finally and most importantly, it should be noted that the dramatic effect on LD formation was observed in cells overexpressing wild-type seipin, but not seipin-A212P (Fig. 3B), which is a recessive mis-sense mutation associated with congenital generalized lipodystrophy (5). The mRNA and protein expression levels of wild-type seipin and A212P are similar (supplementary Fig. IV C and D). If the effect of overexpressing wild-type seipin is an overexpression artifact, it is highly unlikely that a single amino acid change would abolish that effect.
Reduced TAG synthesis and droplet formation could be due to decreased fatty acid uptake. We therefore examined the uptake and incorporation into LDs of a fluorescent fatty acid, Bodipy FL C16 (Fig. 3C). Seipin-mCherry overexpression did not affect the uptake of Bodipy C16 (Fig. 3C). After 16 h of incubation, Bodipy C16 was seen in cytoplasmic LDs in cells expressing mCherry, but not seipinS-mCherry. Together, these results indicate a specific role of seipin on TAG synthesis and consequently, LD formation.
Reduction in TAG content and LD formation upon seipin overexpression is not due to enhanced lipolysis
The reduction in TAG content and LD formation in cells overexpressing seipin could be attributed to defective synthesis of neutral lipids. However, highly enhanced lipolysis is equally possible. We therefore compared the rate of glycerol release between HeLa cells transfected with empty vector or vectors expressing seipin-mCherry. Expression of both the long and short isoforms of seipin did not significantly increase the glycerol release, indicating that seipin overexpression primarily affected TAG synthesis rather than enhance TAG lipolysis (Fig. 3D). In agreement with this, in vivo [14C] oleate incorporation assay revealed that oleate incorporation into TAG was reduced to about 50% of control cells (Fig. 3E). This was not due to defective cellular uptake of [14C] oleate, because its incorporation into cholesteryl ester was slightly enhanced in cells overexpressing seipin-mCherry (data not shown). Interestingly, the rate of in vitro TAG synthesis from dioleoylglycerol and [14C] oleoyl-CoA of ER microsomes isolated from cells expressing seipin-mCherry was similar to that of control cells (Fig. 3F), indicating that seipin expression did not inhibit the DGAT activities, but possibly affecting the availability of substrates for TAG synthesis. The mRNA levels of genes involved in TAG synthesis (GPAT1-4, AGPAT1-2, LPIN1-2, MGAT1, and DGAT1-2) were also examined by qPCR, and no significant changes were detected (Fig. 3G).
C-terminal tail of seipin is dispensable for its inhibitory role in TAG and LD formation
Fld1p (the yeast seipin protein) has a much shorter C-terminal tail (supplementary Fig. V A). Previously, we showed that the first 280 amino acids of the short isoform of seipin (seipinS 1-280AA) complemented yeast mutant cells deleted for FLD1 gene (17). We therefore determined whether overexpression of mCherry-tagged seipinS 1-280AA could also inhibit LD formation. Similar to the full-length seipinS, mCherry-tagged seipinS 1-280AA also significantly reduced LD formation (supplementary Fig. V B).
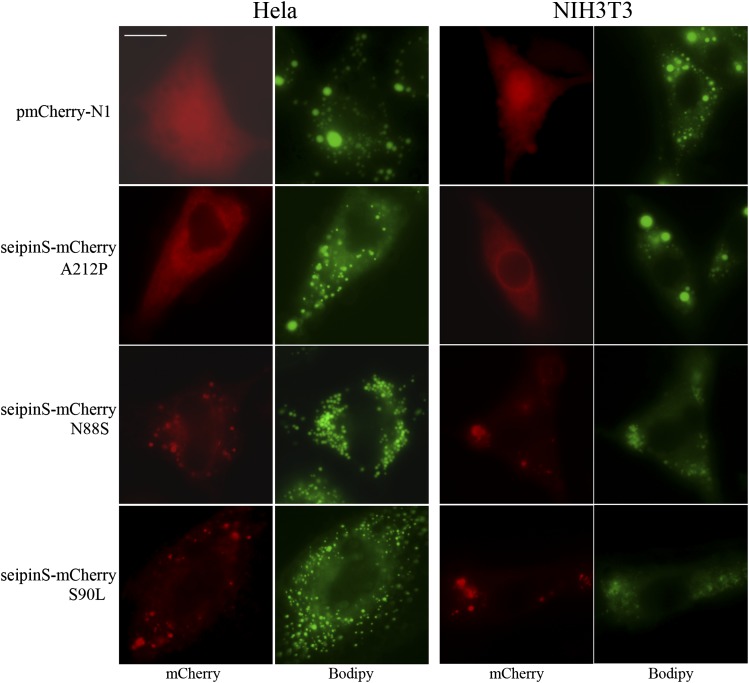
Overexpression of seipin N88S or S90L but not A212P promotes formation of small lipid droplets
The mis-sense mutations of seipin are linked to autosomal dominant motoneuron disorders (N88S and S90L) and the autosomal recessive CGL (A212P). However, their impact on cellular lipid metabolism has not been investigated in detail. Unlike wild-type seipin, overexpression of seipin A212P did not significantly block the formation of LDs (Figs. 3B and 4). A substantial portion of this mutant localized to a perinuclear ring in NIH3T3 cells as reported in murine C3H10T1/2 cells, but not in HeLa cells (12). Surprisingly, unlike the cells expressing mCherry or A212P, which synthesized both large and small LDs, cells expressing N88S or S90L mutants synthesized many small and sometimes clustering LDs reminiscent of the LD morphology observed in seipin knockdown or deficient cells (Fig. 4 and Fig. 1A) (16, 18). These results suggest that the N88S or S90L mutant may exert dominant-negative effects on lipid metabolism upon overexpression. We were not able to biochemically measure TAG level/synthesis in cells expressing the mutants as ∼50% of those cells appeared very unhealthy, presumably due to ER stress (22).
Fig. 4.
The effect of overexpression of mCherry-tagged seipin mutants (A212P, N88S, and S90L) on LD formation. HeLa and NIH3T3 cells were transfected with empty or pmCherry-N1 plasmids that express mutant seipin proteins for 48 h and treated with 200 μM oleate for another 16 h. Cells were stained with BODIPY 493/503 and observed for LDs. Bar, 10 μm.
The N88S and S90L mutants could recruit the wild-type seipin and vice versa
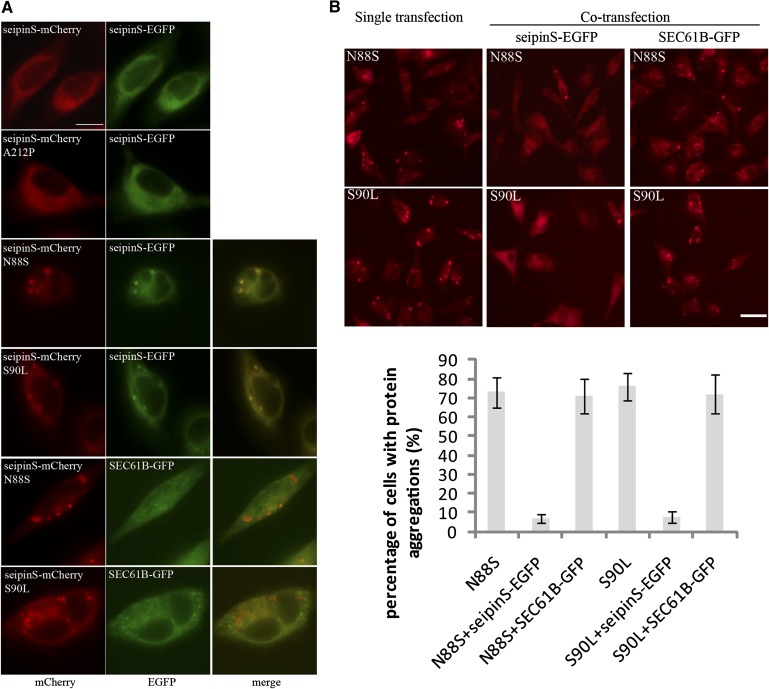
It has been reported that seipin-deficient human fibroblasts and lymphoblastoid cells contain many smaller LDs compared with normal cells (16, 18), which was recapitulated in cultured mammalian cells in this study (Fig. 1). Our finding that overexpression of the N88S and S90L seipin mutants caused cells to synthesize “many and small” LDs led us to speculate that these mutants may “hijack” the wild-type seipin into aggregations, resulting in, in essence, a seipin “loss of function” phenotype. To examine this possibility, we first observed the localization of seipin mis-sense mutants and found that N88S-mCherry and S90L-mCherry localized to punctate structures, similar to the structures identified by previous studies (Fig. 5A) (11, 22). By comparison, seipinS-EGFP localized to the ER, similar to seipinS-mCherry. Interestingly, when HeLa cells were cotransfected with vectors expressing seipinS-EGFP and N88S or S90L-mCherry, respectively, a significant portion of the seipinS-EGFP clearly colocalized with the punctate N88S or S90L aggregates (Fig. 5A). In contrast, when SEC61B-GFP was coexpressed with N88S-mCherry or S90L-mCherry, it was not recruited to the punctate structures (Fig. 5A). These results indicate that N88S and S90L can specifically recruit wild-type seipin to the punctate structures.
Fig. 5.
Seipin mutants defective in glycosylation (N88S and S90L) recruit wild-type seipin into punctate structures, whereas coexpression of wild-type seipin reduces protein aggregation of N88S and S90L seipin mutants in HeLa cells. A: HeLa cells were cotransfected with plasmids expressing either wild-type or mutant seipinS and plasmids expressing either seipinS-EGFP or SEC61B-GFP for 48 h and observed for cellular localization of mCherry and EGFP signals. Bar, 10 μm. B: Cells were either singly transfected with plasmids expressing N88S or S90L seipinS-mCherry, or cotransfected with plasmids expressing seipinS-EGFP or SEC61B-GFP. Forty-eight hours after transfection, cells were observed under a fluorescence microscope. Bar, 40 μm. The percentage of cells that show punctate mCherry signals among all transfected cells (n = 100, in triplicates) was calculated.
Intriguingly, when the wild-type seipinS-EGFP was coexpressed with N88S-mCherry or S90L-mCherry mutants, it significantly prevented the aggregation of N88S and S90L mutants (Fig. 5B). When HeLa cells were transfected with vectors expressing the N88S or S90L alone, about 70% of cells displayed punctate signal, and this percentage decreased to ∼5% when seipinS-EGFP was coexpressed (Fig. 5B). By contrast, the coexpression of SEC61B-GFP did not reduce the aggregation of seipin N88S and S90L. These results suggested that the N88S and S90L mutants could “kidnap” the wild-type seipin into the punctate structures, whereas wild-type seipin could also keep them from aggregating.
Wild-type seipin can interact with itself and the mis-sense mutants
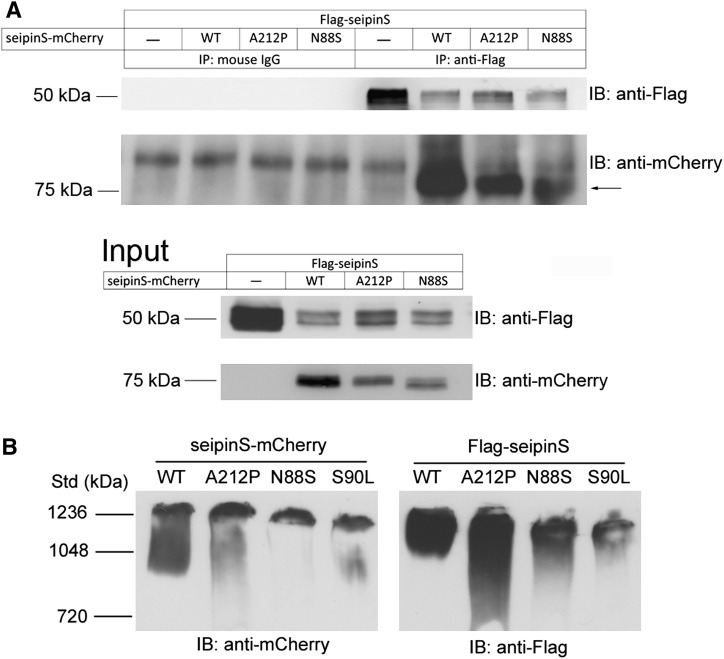
A recent elegant study showed that yeast seipin/Fld1p can form homo-oligomers (19). Based on the functional analysis and the localization results described earlier, we hypothesize that mammalian seipin might interact with itself and N88S or S90L, forming oligomers. To test this, HeLa cells were cotransfected with vectors expressing Flag-seipinS (wild-type) together with wild-type or mutant forms of seipinS-mCherry (A212P and N88S), immunoprecipitated with anti-Flag, and probed with anti-mCherry. Our results indicated that wild-type seipin can interact with itself, and both forms of mutant seipin (A212P and N88S) (Fig. 6A). S90L also interacts with seipin (data not shown). As a control, SEC31A-EGFP and SEC61B-GFP, two ER membrane proteins, did not coimmunoprecipitate with seipin (supplementary Fig. VI). To estimate the size of wild-type and mutant seipin complexes, cell lysates solubilized in 0.5% CHAPSO were separated by blue native PAGE, and tagged seipin and its mutants were detected by specific antibodies (Fig. 6B). Complexes containing wild-type or A212P appear at ∼1236 kDa whereas those containing N88S or S90L are slightly smaller. These data further indicate that seipin and its mutant forms exist in large protein complexes.
Fig. 6.
Human seipin interacts with itself, A212P and N88S. A: Coimmunoprecipitation of A212P and N88S mutants with wild-type seipin. HeLa cells cotransfected with plasmids expressing Flag-seipinS and plasmids expressing mCherry-tagged wild-type seipinS, A212P or N88S mutant were lysed and immunoprecipitated with anti-Flag or with mouse IgG (as a control). The resulting pellets were subjected to SDS/PAGE and immunoblot analysis with anti-Flag and anti-mCherry. Arrow, seipinS-mCherry. B: Both mCherry- and Flag-tagged WT and mutant seipinS form complexes of large molecular weight. HeLa cells were lysed with Lysis buffer B containing 0.5% (v/v) CHAPSO. Proteins were separated by electrophoresis in 4-16% Native PAGE gels.
DISCUSSION
Mutations in seipin are associated with motoneuron disorders and the most severe form of human lipodystrophy (5, 11). Seipin and its yeast homolog have also been demonstrated to regulate the cellular dynamics of LDs, the primary storage form of energy (16–18). Yet, little is known about the biochemical and cellular function of seipin, which resides in the ER through two transmembrane domains but lacks any recognizable functional domains/motifs (10, 23). Here, we established an important role for seipin in TAG metabolism in cultured mammalian cells by knockdown and overexpression studies. We also demonstrate that seipin can interact with itself and its mutant forms. Overexpression of N88S or S90L, but not A212P, appears to exert dominant-negative effects possibly by trapping normal seipin in inclusions. Finally, we provide a possible explanation to the reported aggregation of seipin. Our results offer important insights into the biochemical characteristics and cellular functions of seipin and its mutants.
The role of seipin in adipogenesis has been well established; however, its function in other tissues and especially its molecular function have not been elucidated. Here, a role for seipin in TAG metabolism has been uncovered in cultured mammalian cells: knocking down seipin significantly increased TAG synthesis whereas overexpressing seipin had an opposite effect (Figs. 1 and 3). Whereas results from the knockdown experiments corroborated with observations made in yeast and fly, the overexpression studies were limited by the lack of a reliable seipin antibody. As a result, tagged seipin was used and it was impossible to estimate the fold of protein overexpression relative to the native level in our studies. Despite these difficulties, we believe results from seipin overexpression are likely to be physiologically relevant for the following reasons. First, overexpression of mCherry-, HA-, or myc-tagged seipin, but not the tag (mCherry) alone, all inhibited TAG synthesis, which argues against the possibility that any particular tag may affect seipin function. Second, expression of wild-type seipin but not the mis-sense and loss-of-function form (A212P) reduced TAG synthesis (Fig. 3B), although both the mRNA and protein levels of seipin and its A212P mutant were expressed at a similar level (supplementary Fig. IV). Therefore, the specific effect of wild-type seipin on TAG synthesis is highly unlikely to be an artifact. Third, expressing seipin at a low level by using a Tet-on system also resulted in reduced LD formation (supplementary Fig. IV). Fourth, the effect of seipin overexpression is opposite to that of seipin knockdown. Fifth, the tagged and overexpressed human seipin can rescue defects in seipin/FLD1 deficient yeast, suggesting the fusion protein is functional (17). Finally, although three groups reported the aggregation of tagged seipin upon overexpression (12, 13, 22), our results showed that monomeric seipin can be easily detected on SDS-PAGE. Further analyses revealed that boiling of protein lysates caused tagged seipin to aggregate, and consequently seipin appeared as a band over 250 kDa on SDS-PAGE in earlier studies (supplementary Fig. I). The same phenomenon has also been observed by another group (Justin Rochford, personal communications). Together, these data support seipin's role in regulating TAG synthesis, which should be further examined in other experimental systems, e.g., transgenic mice.
It is not surprising that seipin can interact with itself and the A212P and N88S/S90L mutants because the oligomerization of yeast seipin/Fld1p has been recently demonstrated (19). It is interesting that N88S or S90L but not A212P appears to exert a dominant-negative effect on LD biogenesis upon overexpression. N88S and S90L have been linked to motoneuron disorders and their role in lipid metabolism has never been demonstrated. Our data, for the first time, suggest that N88S/S90L may have an impact on LD formation especially upon overexpression. N88S and S90L have been reported to form large perinuclear inclusions (24), therefore, they may drag wild-type seipin into inclusions upon overexpression so to interfere with normal seipin function and cause a dominant-negative phenotype (Figs. 5 and 6). This may not occur in vivo because the protein level of N88S/S90L could be too low to interfere with normal seipin function. Therefore, patients carrying N88S/S90L show motoneuron defects but not lipodystrophy. The loss-of-function mutant, A212P, localizes to the nuclear envelope when expressed in NIH3T3 cells but this did not affect seipin function and LD morphology. It is possible that the overexpressed A212P can trap seipin in nuclear envelope but the trapped seipin may still be functional. Alternatively, the A212P mutant may be a weak allele of seipin, which retains some normal activity. In this regard, knocking down seipin expression by ∼50% can almost abolish adipogenesis in 3T3-L1 cells (13).
Knocking down seipin increases TAG synthesis in HeLa and 3T3-L1 cells, but inhibits the differentiation of preadipocytes into adipocytes and consequently lipid storage in adipose tissue. How could the loss of seipin function on one hand promote lipogenesis, but on the other hand inhibit adipogenesis and fat accumulation? Recent results from the Farese laboratory provide the most convincing evidence to explain this apparent paradox: mouse embryonic fibroblasts deficient in DGAT activity retain an intact adipogenic program, although TAG synthesis is completely blocked, indicating that lipogenesis and adipognesis are independent events (29). The lack of lipid storage in adipose tissue upon seipin depletion is due to a defect in the differentiation process, but not in lipogenesis. Moreover, massive lipid accumulation is observed in the liver and fibroblasts of BSCL2 patients. The inhibitory role of seipin on cellular lipogenesis is further supported by previous observations that TAG accumulates in the FLD1 (yeast seipin) null mutant and in seipin deficient salivary gland of Drosophila, and that LDs proliferate in seipin-deficient fibroblasts (15, 17, 18). Consistent with results from the current study, seipin has also been implicated in phospholipid and fatty acid metabolism (16, 17). Seipin may function directly in fatty acid metabolism and therefore indirectly impact TAG synthesis. Alternatively, however, the effect of seipin on fatty acids may be secondary as the level and distribution of phospholipids, such as phosphatidylcholine, can have a major impact on lipid acyl chain composition to help maintain membrane integrity (20, 30). Although unlikely, the effect of seipin on TAG/lipid metabolism could be entirely indirect as seipin deletion may increase cellular stresses, such as ER stress, which is known to upregulate TAG synthesis (25). More detailed future analyses with better tools are needed to pinpoint the molecular function of seipin.
In summary, we have demonstrated for the first time that seipin is involved in TAG synthesis in cultured mammalian cell lines under nondifferentiating conditions and we have revealed important biochemical characteristics of seipin and its mutants. These results should facilitate future investigation of the important yet “mysterious” protein, seipin (20, 23).
Supplementary Material
Acknowledgments
We thank Drs. Ian Dawes, Merlin Crossley, Richard Pearson, and members of the Yang laboratory for helpful discussions. We also thank Dr. Justin Rochford for discussing unpublished results.
Footnotes
Abbreviations:
- AGPAT2
- 1-acylglycerol-3-phosphate-O-acyl transferase 2
- BSCL2
- Berardinelli-Seip congenital lipodystrophy 2
- CGL
- congenital generalized lipodystrophy
- DGAT
- diacylglycerol acyltransferase
- ER
- endoplasmic reticulum
- LD
- lipid droplet
- qPCR
- quantitative PCR
- siRNA
- small interfering RNA
- TAG
- triacylglycerol
This work is supported by a research grant from the National Health and Medical Research Council of Australia (#568725). H. Yang is a Future Fellow of the Australian Research Council.
The online version of this article (available at http://www.jlr.org) contains supplementary data in the form of six figures and one table.
REFERENCES
- 1.Farese R. V., Jr, Walther T. C. 2009. Lipid droplets finally get a little R-E-S-P-E-C-T. Cell. 139: 855–860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Park K. W., Halperin D. S., Tontonoz P. 2008. Before they were fat: adipocyte progenitors. Cell Metab. 8: 454–457. [DOI] [PubMed] [Google Scholar]
- 3.Gesta S., Tseng Y. H., Kahn C. R. 2007. Developmental origin of fat: tracking obesity to its source. Cell. 131: 242–256. [DOI] [PubMed] [Google Scholar]
- 4.Agarwal A. K., Garg A. 2006. Genetic basis of lipodystrophies and management of metabolic complications. Annu. Rev. Med. 57: 297–311. [DOI] [PubMed] [Google Scholar]
- 5.Magré J., Delepine M., Khallouf E., Gedde-Dahl T., Jr, Van Maldergem L., Sobel E., Papp J., Meier M., Megarbane A., Bachy A., et al. 2001. Identification of the gene altered in Berardinelli-Seip congenital lipodystrophy on chromosome 11q13. Nat. Genet. 28: 365–370. [DOI] [PubMed] [Google Scholar]
- 6.Agarwal A. K., Arioglu E., De Almeida S., Akkoc N., Taylor S. I., Bowcock A. M., Barnes R. I., Garg A. 2002. AGPAT2 is mutated in congenital generalized lipodystrophy linked to chromosome 9q34. Nat. Genet. 31: 21–23. [DOI] [PubMed] [Google Scholar]
- 7.Kim C. A., Delepine M., Boutet E., El Mourabit H., Le Lay S., Meier M., Nemani M., Bridel E., Leite C. C., Bertola D. R., et al. 2008. Association of a homozygous nonsense caveolin-1 mutation with Berardinelli-Seip congenital lipodystrophy. J. Clin. Endocrinol. Metab. 93: 1129–1134. [DOI] [PubMed] [Google Scholar]
- 8.Hayashi Y. K., Matsuda C., Ogawa M., Goto K., Tominaga K., Mitsuhashi S., Park Y. E., Nonaka I., Hino-Fukuyo N., Haginoya K., et al. 2009. Human PTRF mutations cause secondary deficiency of caveolins resulting in muscular dystrophy with generalized lipodystrophy. J. Clin. Invest. 119: 2623–2633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Liu L., Brown D., McKee M., Lebrasseur N. K., Yang D., Albrecht K. H., Ravid K., Pilch P. F. 2008. Deletion of Cavin/PTRF causes global loss of caveolae, dyslipidemia, and glucose intolerance. Cell Metab. 8: 310–317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lundin C., Nordstrom R., Wagner K., Windpassinger C., Andersson H., von Heijne G., Nilsson I. 2006. Membrane topology of the human seipin protein. FEBS Lett. 580: 2281–2284. [DOI] [PubMed] [Google Scholar]
- 11.Windpassinger C., Auer-Grumbach M., Irobi J., Patel H., Petek E., Horl G., Malli R., Reed J. A., Dierick I., Verpoorten N., et al. 2004. Heterozygous missense mutations in BSCL2 are associated with distal hereditary motor neuropathy and Silver syndrome. Nat. Genet. 36: 271–276. [DOI] [PubMed] [Google Scholar]
- 12.Payne V. A., Grimsey N., Tuthill A., Virtue S., Gray S. L., Dalla Nora E., Semple R. K., O'Rahilly S., Rochford J. J. 2008. The human lipodystrophy gene BSCL2/seipin may be essential for normal adipocyte differentiation. Diabetes. 57: 2055–2060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chen W., Yechoor V. K., Chang B. H., Li M. V., March K. L., Chan L. 2009. The human lipodystrophy gene product BSCL2/seipin plays a key role in adipocyte differentiation. Endocrinology. 150: 4552–4561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cui X., Wang Y., Tang Y., Liu Y., Zhao L., Deng J., Xu G., Peng X., Ju S., Liu G., et al. 2011. Seipin ablation in mice results in severe generalized lipodystrophy. Hum. Mol. Genet. 20: 3022–3030. [DOI] [PubMed] [Google Scholar]
- 15.Tian Y., Bi J., Shui G., Liu Z., Xiang Y., Liu Y., Wenk M. R., Yang H., Huang X. 2011. Tissue-autonomous function of Drosophila seipin in preventing ectopic lipid droplet formation. PLoS Genet. 7: e1001364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Boutet E., El Mourabit H., Prot M., Nemani M., Khallouf E., Colard O., Maurice M., Durand-Schneider A. M., Chretien Y., Gres S., et al. 2009. Seipin deficiency alters fatty acid Delta9 desaturation and lipid droplet formation in Berardinelli-Seip congenital lipodystrophy. Biochimie. 91: 796–803. [DOI] [PubMed] [Google Scholar]
- 17.Fei W., Shui G., Gaeta B., Du X., Kuerschner L., Li P., Brown A. J., Wenk M. R., Parton R. G., Yang H. 2008. Fld1p, a functional homologue of human seipin, regulates the size of lipid droplets in yeast. J. Cell Biol. 180: 473–482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Szymanski K. M., Binns D., Bartz R., Grishin N. V., Li W. P., Agarwal A. K., Garg A., Anderson R. G., Goodman J. M. 2007. The lipodystrophy protein seipin is found at endoplasmic reticulum lipid droplet junctions and is important for droplet morphology. Proc. Natl. Acad. Sci. USA. 104: 20890–20895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Binns D., Lee S., Hilton C. L., Jiang Q. X., Goodman J. M. 2010. Seipin is a discrete homooligomer. Biochemistry. 49: 10747–10755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fei W., Du X., Yang H. 2011. Seipin, adipogenesis and lipid droplets. Trends Endocrinol. Metab. 22: 204–210. [DOI] [PubMed] [Google Scholar]
- 21.Fei W., Shui G., Zhang Y., Krahmer N., Ferguson C., Kapterian T. S., Lin R. C., Dawes I. W., Brown A. J., Li P., et al. 2011. A role for phosphatidic acid in the formation of “supersized” lipid droplets. PLoS Genet. 7: e1002201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ito D., Fujisawa T., Iida H., Suzuki N. 2008. Characterization of seipin/BSCL2, a protein associated with spastic paraplegia 17. Neurobiol. Dis. 31: 266–277. [DOI] [PubMed] [Google Scholar]
- 23.Agarwal A. K., Garg A. 2004. Seipin: a mysterious protein. Trends Mol. Med. 10: 440–444. [DOI] [PubMed] [Google Scholar]
- 24.Ito D., Suzuki N. 2007. Molecular pathogenesis of seipin/BSCL2-related motor neuron diseases. Ann. Neurol. 61: 237–250. [DOI] [PubMed] [Google Scholar]
- 25.Fei W., Wang H., Fu X., Bielby C., Yang H. 2009. Conditions of endoplasmic reticulum stress stimulate lipid droplet formation in Saccharomyces cerevisiae. Biochem. J. 424: 61–67. [DOI] [PubMed] [Google Scholar]
- 26.Cases S., Stone S. J., Zhou P., Yen E., Tow B., Lardizabal K. D., Voelker T., Farese R. V., Jr 2001. Cloning of DGAT2, a second mammalian diacylglycerol acyltransferase, and related family members. J. Biol. Chem. 276: 38870–38876. [DOI] [PubMed] [Google Scholar]
- 27.Feramisco J. D., Radhakrishnan A., Ikeda Y., Reitz J., Brown M. S., Goldstein J. L. 2005. Intramembrane aspartic acid in SCAP protein governs cholesterol-induced conformational change. Proc. Natl. Acad. Sci. USA. 102: 3242–3247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gossen M., Freundlieb S., Bender G., Muller G., Hillen W., Bujard H. 1995. Transcriptional activation by tetracyclines in mammalian cells. Science. 268: 1766–1769. [DOI] [PubMed] [Google Scholar]
- 29.Harris C. A., Haas J. T., Streeper R. S., Stone S. J., Kumari M., Yang K., Han X., Brownell N., Gross R. W., Zechner R., et al. 2011. DGAT enzymes are required for triacylglycerol synthesis and lipid droplets in adipocytes. J. Lipid Res. 52: 657–667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.de Kroon A. I. 2007. Metabolism of phosphatidylcholine and its implications for lipid acyl chain composition in Saccharomyces cerevisiae. Biochim. Biophys. Acta. 1771: 343–352. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.