Abstract
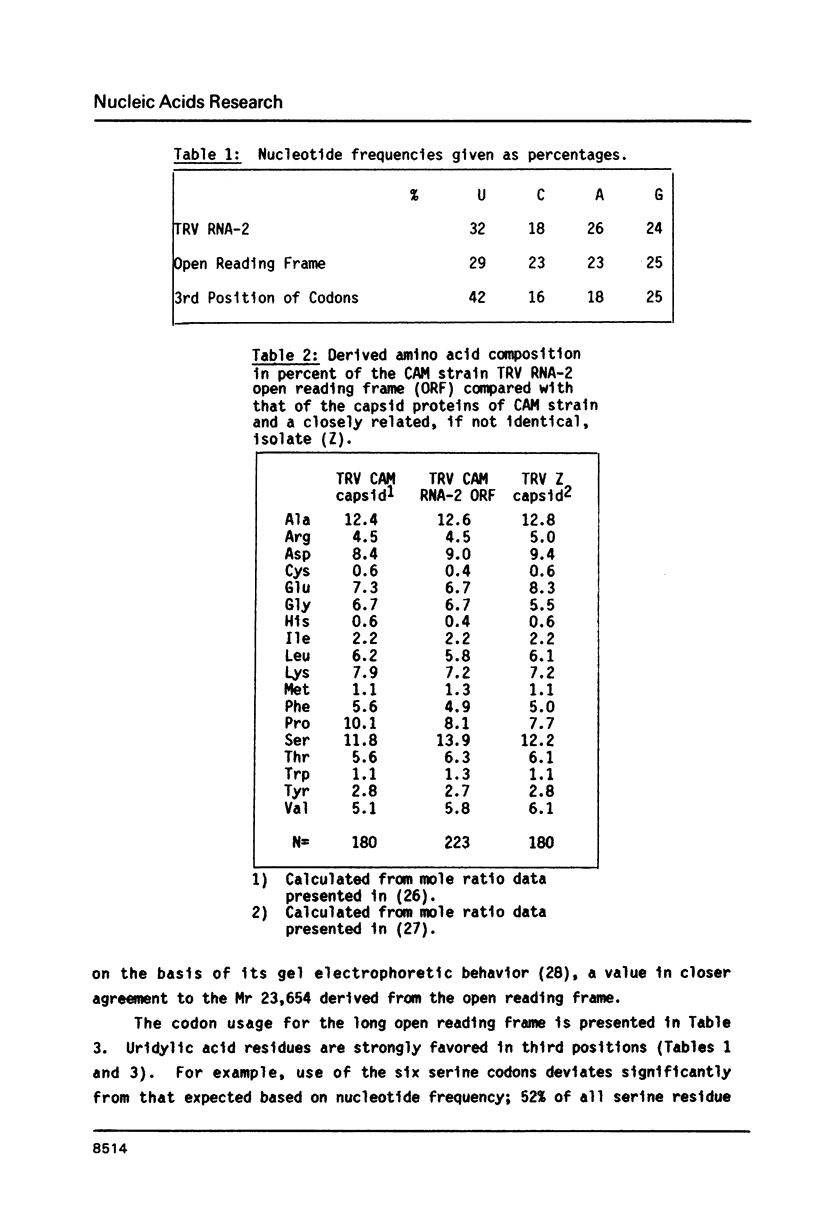
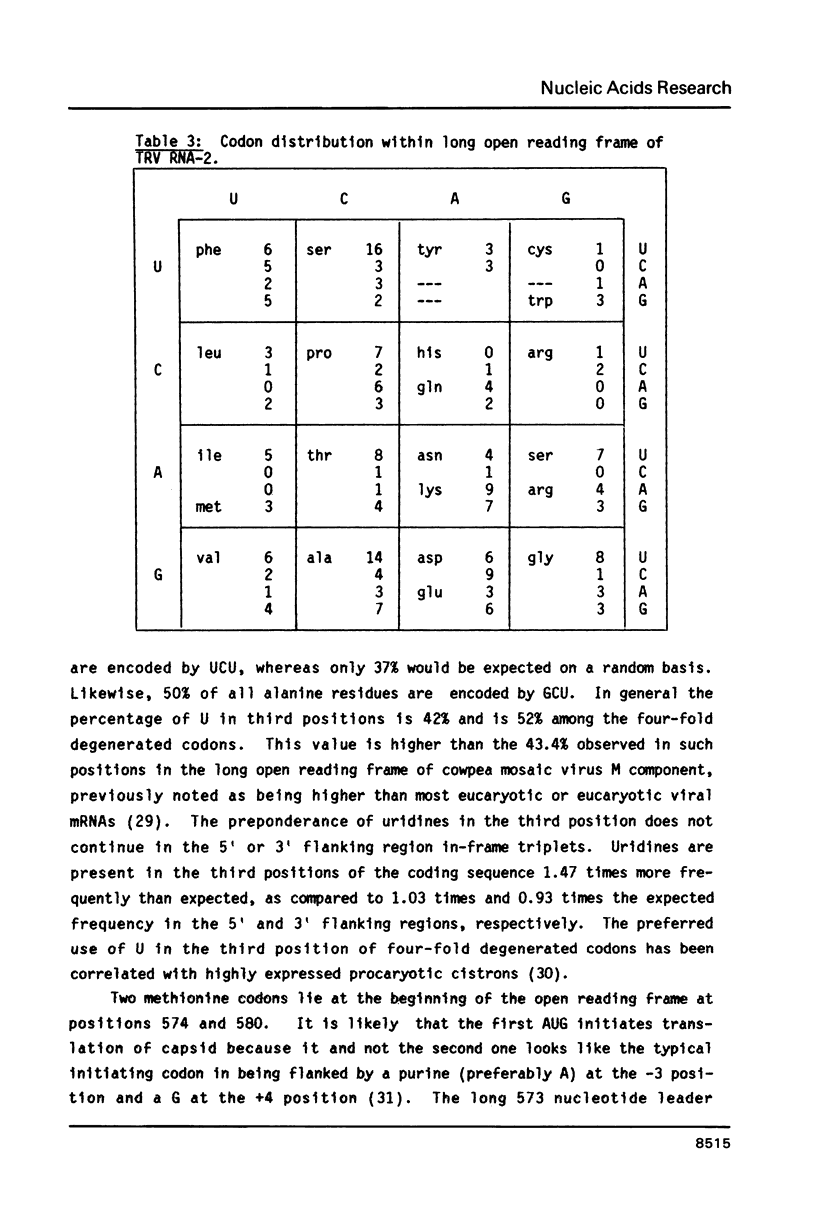
The nucleotide sequence of the smaller genomic strand (RNA-2) of the bipartite tobacco rattle virus (CAM strain) has been determined. RNA-2 is capped at the 5' terminus and contains 1799 nucleotide residues. There is a single 223 codon long open reading frame extending from nucleotide 574 to 1242 which designates a protein of Mr 23,654. The derived amino acid composition, in percent, matches that previously determined for the virus capsid protein. The long open reading frame is flanked by 5' and 3' untranslated regions of 573 and 554 nucleotides, respectively. The 5' leader sequence contains two different sets of direct repeats, one of 119 nucleotides and the other of 76. It also contains 13 apparently unused AUG codons, four of which lie in the same frame as the capsid protein cistron. The 3' terminal sequence of RNA-2 is identical to that of the larger genomic strand (RNA-1) for 459 nucleotides.
Full text
PDF









Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ahlquist P., Dasgupta R., Kaesberg P. Near identity of 3- RNA secondary structure in bromoviruses and cucumber mosaic virus. Cell. 1981 Jan;23(1):183–189. doi: 10.1016/0092-8674(81)90283-x. [DOI] [PubMed] [Google Scholar]
- Ahlquist P., Dasgupta R., Shih D. S., Zimmern D., Kaesberg P. Two-step binding of eukaryotic ribosomes to brome mosaic virus RNA3. Nature. 1979 Sep 27;281(5729):277–282. doi: 10.1038/281277a0. [DOI] [PubMed] [Google Scholar]
- Bailey J. M., Davidson N. Methylmercury as a reversible denaturing agent for agarose gel electrophoresis. Anal Biochem. 1976 Jan;70(1):75–85. doi: 10.1016/s0003-2697(76)80049-8. [DOI] [PubMed] [Google Scholar]
- Biggin M. D., Gibson T. J., Hong G. F. Buffer gradient gels and 35S label as an aid to rapid DNA sequence determination. Proc Natl Acad Sci U S A. 1983 Jul;80(13):3963–3965. doi: 10.1073/pnas.80.13.3963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Callahan P. L., Mizutani S., Colonno R. J. Molecular cloning and complete sequence determination of RNA genome of human rhinovirus type 14. Proc Natl Acad Sci U S A. 1985 Feb;82(3):732–736. doi: 10.1073/pnas.82.3.732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crouse G. F., Frischauf A., Lehrach H. An integrated and simplified approach to cloning into plasmids and single-stranded phages. Methods Enzymol. 1983;101:78–89. doi: 10.1016/0076-6879(83)01006-x. [DOI] [PubMed] [Google Scholar]
- Davies J. W., Stanley J., Van Kammen A. Sequence homology adjacent to the 3' terminal poly(A) of cowpea mosaic virus RNAs. Nucleic Acids Res. 1979 Sep 25;7(2):493–500. doi: 10.1093/nar/7.2.493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- England T. E., Bruce A. G., Uhlenbeck O. C. Specific labeling of 3' termini of RNA with T4 RNA ligase. Methods Enzymol. 1980;65(1):65–74. doi: 10.1016/s0076-6879(80)65011-3. [DOI] [PubMed] [Google Scholar]
- Fritsch C., Mayo M. A., Hirth L. Further studies on the translation products of tobacco rattle virus RNA in vitro. Virology. 1977 Apr;77(2):722–732. doi: 10.1016/0042-6822(77)90494-9. [DOI] [PubMed] [Google Scholar]
- Gergen J. P., Stern R. H., Wensink P. C. Filter replicas and permanent collections of recombinant DNA plasmids. Nucleic Acids Res. 1979 Dec 20;7(8):2115–2136. doi: 10.1093/nar/7.8.2115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghabrial S. A., Lister R. M. Coat protein and symptom specification in tobacco rattle virus. Virology. 1973 Mar;52(1):1–12. doi: 10.1016/0042-6822(73)90392-9. [DOI] [PubMed] [Google Scholar]
- Grantham R., Gautier C., Gouy M., Jacobzone M., Mercier R. Codon catalog usage is a genome strategy modulated for gene expressivity. Nucleic Acids Res. 1981 Jan 10;9(1):r43–r74. doi: 10.1093/nar/9.1.213-b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haidar M. A., Hirth L. 5'-terminal structure of tobacco rattle virus RNA: evidence for polarity of reconstitution. Virology. 1977 Jan;76(1):173–185. doi: 10.1016/0042-6822(77)90294-x. [DOI] [PubMed] [Google Scholar]
- Harrison B. D., Robinson D. J. The tobraviruses. Adv Virus Res. 1978;23:25–77. doi: 10.1016/s0065-3527(08)60097-4. [DOI] [PubMed] [Google Scholar]
- Kemper B. Inactivation of parathyroid hormone mRNA by treatment with periodate and aniline. Nature. 1976 Jul 22;262(5566):321–323. doi: 10.1038/262321a0. [DOI] [PubMed] [Google Scholar]
- Kozak M. Possible role of flanking nucleotides in recognition of the AUG initiator codon by eukaryotic ribosomes. Nucleic Acids Res. 1981 Oct 24;9(20):5233–5252. doi: 10.1093/nar/9.20.5233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Land H., Grez M., Hauser H., Lindenmaier W., Schütz G. 5'-Terminal sequences of eucaryotic mRNA can be cloned with high efficiency. Nucleic Acids Res. 1981 May 25;9(10):2251–2266. doi: 10.1093/nar/9.10.2251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayo M. A., Fritsch C., Hirth L. Translation of tobacco rattle virus RNA in vitro using wheat germ extracts. Virology. 1976 Feb;69(2):408–415. doi: 10.1016/0042-6822(76)90472-4. [DOI] [PubMed] [Google Scholar]
- Mayo M. A., Robinson D. J. Revision of estimates of the molecular weights of tobravirus coat proteins. Intervirology. 1975;5(6):313–318. doi: 10.1159/000149929. [DOI] [PubMed] [Google Scholar]
- Messing J. New M13 vectors for cloning. Methods Enzymol. 1983;101:20–78. doi: 10.1016/0076-6879(83)01005-8. [DOI] [PubMed] [Google Scholar]
- Miki T., Okada Y. Comparative studies on some strains of tobacco rattle virus. Virology. 1970 Dec;42(4):993–998. doi: 10.1016/0042-6822(70)90347-8. [DOI] [PubMed] [Google Scholar]
- Sippel A. E. Purification and characterization of adenosine triphosphate: ribonucleic acid adenyltransferase from Escherichia coli. Eur J Biochem. 1973 Aug 1;37(1):31–40. doi: 10.1111/j.1432-1033.1973.tb02953.x. [DOI] [PubMed] [Google Scholar]
- Sänger H. L. Characteristics of tobacco rattle virus. I. Evidence that its two particles are functionally defective and mutually complementing. Mol Gen Genet. 1968 May 17;101(4):346–367. doi: 10.1007/BF00436232. [DOI] [PubMed] [Google Scholar]
- Sänger H. L. Functions of the two particles of tobacco rattle virus. J Virol. 1969 Mar;3(3):304–312. doi: 10.1128/jvi.3.3.304-312.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tautz D., Renz M. An optimized freeze-squeeze method for the recovery of DNA fragments from agarose gels. Anal Biochem. 1983 Jul 1;132(1):14–19. doi: 10.1016/0003-2697(83)90419-0. [DOI] [PubMed] [Google Scholar]
- Taylor J. M., Illmensee R., Summers J. Efficeint transcription of RNA into DNA by avian sarcoma virus polymerase. Biochim Biophys Acta. 1976 Sep 6;442(3):324–330. doi: 10.1016/0005-2787(76)90307-5. [DOI] [PubMed] [Google Scholar]
- Wickens M. P., Buell G. N., Schimke R. T. Synthesis of double-stranded DNA complementary to lysozyme, ovomucoid, and ovalbumin mRNAs. Optimization for full length second strand synthesis by Escherichia coli DNA polymerase I. J Biol Chem. 1978 Apr 10;253(7):2483–2495. [PubMed] [Google Scholar]
- van Wezenbeek P., Verver J., Harmsen J., Vos P., van Kammen A. Primary structure and gene organization of the middle-component RNA of cowpea mosaic virus. EMBO J. 1983;2(6):941–946. doi: 10.1002/j.1460-2075.1983.tb01525.x. [DOI] [PMC free article] [PubMed] [Google Scholar]



