Abstract
Inhibition of the HSP90 chaperone leads to degradation of the HER2 receptor. The HSP90 inhibitor tanespimycin in combination with trastuzumab is active in patients with HER2-overexpressing metastatic breast cancer. This combination is one of several HER2-targeted therapies that will significantly improve the outcome of patients with this subtype of breast cancer.
Keywords: HER2 (ERBB2), HSP90, tanespimycin, trastuzumab, breast cancer
In this issue of Clinical Cancer Research, Modi et al. (1) report the clinical activity of tanespimycin with trastuzumab in patients with HER2-overexpressing metastatic breast cancer (MBC).
Approximately a quarter of breast cancers overexpress the HER2 (ErbB2) proto-oncogene. HER2 is a member of the ErbB family of transmembrane receptor tyrosine kinases (RTKs), which also includes the EGF receptor, HER3, and HER4. Binding of ligands to EGFR, HER3, and HER4 induces the formation of receptor homodimers and heterodimers that contain HER2, activation of the receptors’ kinase activity, and engagement of signal transducers associated with enhanced tumor cell proliferation and survival. The antibody trastuzumab is currently approved or the treatment of early and metastatic HER2-overexpressing breast cancer. Trastuzumab binds to an epitope in the juxtamembrane region of the HER2 receptor. This binding induces uncoupling of ligand-independent HER2-HER3 heterodimers and inhibition of downstream signaling as well as antibody-dependent, cell-mediated cytotoxicity. Although many breast cancers with HER2 gene amplification respond to trastuzumab, a signification fraction of these eventually progress. Several mechanisms of resistance to the antibody have been reported. These include enhanced signaling by RTKs in the ErbB family or in other receptor networks, amplification of phosphatidylinositol-3 kinase (PI3K) signaling as result of mutations in this pathway, and the presence of truncated forms of HER2 devoid of the antibody binding epitope in the receptor’s ectodomain [reviewed in (2)].
The HER2 tyrosine kinase inhibitors (TKIs) lapatinib and neratinib have shown clinical activity as single agents or in combination with chemotherapy in patients who have progressed on trastuzumab (2). These data suggest that trastuzumab-resistant tumors continue to depend on the HER2 tyrosine kinase. These patients may still need trastuzumab beyond progression as suggested by a recent study where the combination of lapatinib and trastuzumab was superior to lapatinib alone at improving progression-free survival, clinical response, and overall survival in patients with HER2+ MBC who had progressed on trastuzumab (3). These data imply that even in advanced stages, HER2+ breast cancers remain dependent on HER2 and that combinations of drugs targeted to the HER2 receptor network will be required for better inhibition of this pathway and hence, improved clinical activity.
Recent clinical evidence supports the efficacy of combinations of anti-HER2 therapies that improve on the inhibition of HER2 by trastuzumab. For example, the combination of trastuzumab and pertuzumab, an antibody that blocks ligand-induced HER2 heterodimerization by binding to an epitope in HER2 different to that of trastuzumab, induces clinical responses in trastuzumab-resistant patients (4). The Neo-ALTTO study compared trastuzumab, lapatinib, or the combination, each arm together with paclitaxel in patients with HER2+ tumors >2 cm in the preoperative setting. A pathological complete response, defined as no invasive cancer in the breast or only DCIS in the breast specimen, was significantly higher in the combination arm (51.3%) vs. 29.5% and 24.7% in the trastuzumab and lapatinib arms, respectively (5)
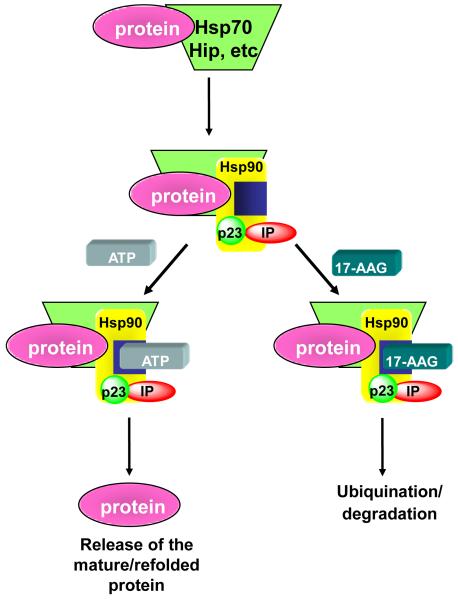
Inhibitors of heat shock protein (HSP) 90 are another rational approach against HER2+ breast cancers that progress on primary anti-HER2 therapy. HSP90 is an abundant molecular chaperone that is required for the refolding of proteins under conditions of environmental stress and for the conformational maturation a several proteins involved in signal transduction such as steroid receptors, RAF-1, CDK4, AKT, MET, HIF-1α, etc. (Fig. 1)(6). The antitumor antibiotic geldanamyicin binds to the ATP pocket of HSP90, thus inhibiting its function. This results in ubiquitination and proteasomal degradation of the HSP90 client proteins. HER2 is among the most sensitive HSP90 clients (7). Indeed, treatment of HER2-overexpressing cancer cells and xenografts causes potent and rapid degradation of HER2, concomitant inhibition of PI3K/AKT, and inhibition of tumor growth (6). Similar activity of HSP90 inhibitors has been observed against HER2+ tumors with acquired resistance to trastuzumab following prolonged adaptation to the antibody, coexpression of mutant PI3KH1047R (p110α), expression of truncated p95-HER2, and upregulation of membrane-associated mucin 4 (8-10).
Figure 1.
Ansamycins (17-AAG) bind the ADP/ATP switch site in HSP90. HSP90 (in orange) is a molecular chaperone involved in the maturation and refolding of several oncoproteins. It has three domains: an amino terminal region that binds ATP (blue box) and drugs and interacts with co-chaperones (i.e., HSP70, Hip, etc.); a middle domain with docking sites for client proteins and co-chaperones, which participates in generating the ATPase activity; and a carboxy-terminal domain that contains a dimerization motif, a second drug-binding region, and interaction sites for other co-chaperones (i.e. p23, IP). Binding of ATP to the amino-terminal domain and its subsequent hydrolysis by HSP90 result in a conformation that is essential for chaperone activity. All HSP90 inhibitors in clinical development interact with the amino-terminal ATP-binding pocket, prevent ATP binding, and disrupt the chaperone cycle, resulting in ubiquitination and degradation of client proteins such as HER2, EGFR, nuclear steroid receptors, RAF-1, CDK4, AKT, MET, HIF-1α, etc.
Tanespimycin [17-allylamino-17-demethoxy-geldanamycin (17-AAG)] is a geldanamycin derivative that potently inhibits HSP90 in preclinical cancer models. Its early clinical development was limited by poor aqueous solubility and pharmacokinetic properties. A formulation of 17-AAG, KOS-953, that contains Cremophor, was tested in a phase I trial in patients with advanced cancer (11). Inhibition of the drug target was supported by induction of HSP70, a marker of inhibition of HSP90, in patients’ lymphocytes at all dose levels. Tanespimycin was overall well tolerated and, in combination with trastuzumab, exhibited antitumor activity only in patients with HER2-overexpressing MBC who had progressed on prior trastuzumab.
In this issue of the Journal and building on their phase I experience, Modi et al. (1) report the results of a phase II trial of tanespimycin plus trastuzumab in 31 patients with HER2+ MBC progressing on trastuzumab. Tanespimycin (KOS-953) was administered weekly at a dose of 450 mg/m2 intravenously and trastuzumab at a conventional weekly dose. After 21 patients had been enrolled, a second formulation of tanespimycin without Cremophor was substituted in order to avoid hypersensitivity reactions. Therapy was continued until disease progression with clinical response being assessed by RECIST criteria. The most common toxicities were diarrhea, nausea, headaches, fatigue, and neuropathy mostly grade 1/2. The overall response rate was 22% with a clinical benefit rate of 59%. Median progression-free survival was 6 months and the overall survival was 17 months. Of note, patients responding to the Cremophor-based formulation maintained this response after cross-over to the second formulation and there was no significant cardiac toxicity observed. Drug-induced downregulation of HER2 in tumors was not reported.
This level of clinical activity is not inconsistent and in some cases may appear superior to that seen with lapatinib plus trastuzumab, pertuzumab plus trastuzumab, trastuzumab-DM1, and neratinib in patients with similar characteristics [reviewed in (12)]. Whether trastuzumab is necessary for the antitumor activity of the combination cannot be ascertained without a randomized study. However, the clinical data supporting trastuzumab’s activity beyond progression (3, 13), its tolerability, the likely possibility that single-agent HSP90 inhibitors will be unable to completely downregulate HER2 (and other client proteins) in sustained fashion, and the preclinical data supporting synergy of both types of drugs (8, 9), would together argue for HSP90 inhibitors to be used in combination with trastuzumab or another HER2 (pathway) inhibitor in patients with HER2+ breast cancer. Thus, this rational combination is one of several HER2-targeted therapies that, pending further development and approval, should improve the outcome of patients with this subtype of breast cancer.
With the positive outcome of this trial, it is then disappointing to read in the report by Modi et al. that ‘the development of tanespimycin as a cancer therapy has been suspended by the sponsor for non-clinical reasons.’ The usual road blocks to drug development, i.e., lack of efficacy against its molecular target(s), undue toxicity, lack of clinical activity, production issues, etc., do not seem to be at play here (http://www.myelomabeacon.com/news/2010/07/22/tanespimycin-development-halted/). Hence, I am afraid that given the potential of this drug to benefit cancer patients, this cryptic explanation might not be acceptable. Will other HSP90 inhibitors, for which to my knowledge there are yet no clinical efficacy data in HER2+ breast cancers, fill this niche? Through federal grants to academia and/or tax breaks to industry to support the entrepreneurial pursuit of projects that should help society like the one discussed herein, (tax-paying) patients, advocates, and cancer care providers contribute to this research and, therefore, deserve a better explanation as to why the development of this useful therapy is now truncated.
Acknowledgments
Supported by NCI grant R01 CA80195, a grant from the Breast Cancer Research Foundation; ACS Clinical Research Professorship Grant CRP-07-234; the Lee Jeans Translational Breast Cancer Research Program; Breast Cancer Specialized Program of Research Excellence (SPORE) grant P50 CA98131, and Vanderbilt-Ingram Cancer Center Support Grant P30 CA68485
References
- 1.Modi S, Stopeck AT, Linden HM, et al. HSP90 Inhibition is Effective in Breast Cancer: A Phase 2 Trial of Tanespimycin (17AAG) plus Trastuzumab in Patients with HER2-Positive Metastatic Breast Cancer Progressing on Trastuzumab. Clin Cancer Res. 2011 doi: 10.1158/1078-0432.CCR-11-0072. [DOI] [PubMed] [Google Scholar]
- 2.Garrett JT, Arteaga CL. Resistance to HER2-directed antibodies and tyrosine kinase inhibitors: Mechanisms and clinical implications. Cancer Biol Ther. 2011;11:793–800. doi: 10.4161/cbt.11.9.15045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Blackwell KL, Burstein HJ, Storniolo AM, et al. Randomized study of Lapatinib alone or in combination with trastuzumab in women with ErbB2-positive, trastuzumab-refractory metastatic breast cancer. J Clin Oncol. 2010;28:1124–30. doi: 10.1200/JCO.2008.21.4437. [DOI] [PubMed] [Google Scholar]
- 4.Baselga J, Gelmon KA, Verma S, et al. Phase II trial of pertuzumab and trastuzumab in patients with human epidermal growth factor receptor 2-positive metastatic breast cancer that progressed during prior trastuzumab therapy. J Clin Oncol. 2010;28:1138–44. doi: 10.1200/JCO.2009.24.2024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Baselga J, Bradbury I, Eidtmann H, et al. First results of the NeoALTTO trial (BIG 01-06/EGF 106903): A phase III, randomized, open label, neoadjuvant study of lapatinib, trastuzumab, and their combination plust paclitaxel in women with HER2-positive primary breast cancer. Cancer Res. 2010;70:82s. [Google Scholar]
- 6.Workman P, Burrows F, Neckers L, Rosen N. Drugging the cancer chaperone HSP90: combinatorial therapeutic exploitation of oncogene addiction and tumor stress. Ann N Y Acad Sci. 2007;1113:202–16. doi: 10.1196/annals.1391.012. [DOI] [PubMed] [Google Scholar]
- 7.Xu W, Mimnaugh E, Rosser MF, et al. Sensitivity of mature Erbb2 to geldanamycin is conferred by its kinase domain and is mediated by the chaperone protein Hsp90. J Biol Chem. 2001;276:3702–8. doi: 10.1074/jbc.M006864200. [DOI] [PubMed] [Google Scholar]
- 8.Chandarlapaty S, Scaltriti M, Angelini P, et al. Inhibitors of HSP90 block p95-HER2 signaling in Trastuzumab-resistant tumors and suppress their growth. Oncogene. 2010;29:325–34. doi: 10.1038/onc.2009.337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Scaltriti M, Serra V, Normant E, et al. Antitumor Activity of the Hsp90 Inhibitor IPI-504 in HER2-Positive Trastuzumab-Resistant Breast Cancer. Mol Cancer Ther. 2011;10:817–24. doi: 10.1158/1535-7163.MCT-10-0966. [DOI] [PubMed] [Google Scholar]
- 10.Leow CC, Chesebrough J, Coffman KT, et al. Antitumor efficacy of IPI-504, a selective heat shock protein 90 inhibitor against human epidermal growth factor receptor 2-positive human xenograft models as a single agent and in combination with trastuzumab or lapatinib. Mol Cancer Ther. 2009;8:2131–41. doi: 10.1158/1535-7163.MCT-08-1038. [DOI] [PubMed] [Google Scholar]
- 11.Modi S, Stopeck AT, Gordon MS, et al. Combination of trastuzumab and tanespimycin (17-AAG, KOS-953) is safe and active in trastuzumab-refractory HER-2 overexpressing breast cancer: a phase I dose-escalation study. J Clin Oncol. 2007;25:5410–7. doi: 10.1200/JCO.2007.11.7960. [DOI] [PubMed] [Google Scholar]
- 12.Abramson V, Arteaga CL. New strategies in HER2-overexpressing breast cancer: many combinations of targeted drugs available. Clin Cancer Res. 2011;17:952–8. doi: 10.1158/1078-0432.CCR-09-1947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.von Minckwitz G, du Bois A, Schmidt M, et al. Trastuzumab beyond progression in human epidermal growth factor receptor 2-positive advanced breast cancer: a german breast group 26/breast international group 03-05 study. J Clin Oncol. 2009;27:1999–2006. doi: 10.1200/JCO.2008.19.6618. [DOI] [PubMed] [Google Scholar]