Abstract
Background
β-amyloid protein (Aβ) accumulation and caspase activation have been shown to contribute to Alzheimer’s disease neuropathogenesis. Aβ is produced from amyloid precursor protein through proteolytic processing by aspartyl protease β-site APP-cleaving enzyme (BACE). The inhaled anesthetic isoflurane has been shown to induce caspase activation and increase levels of BACE and Aβ. However, the underlying mechanisms and interventions of the isoflurane-induced neurotoxicity remain largely to be determined. The glucose analog 2-deoxy-d-glucose (2-DG) has neuroprotective effects. Therefore, we sought to determine whether 2-DG can reduce caspase-3 activation and the increase in the levels of BACE and reactive oxygen species (ROS) induced by isoflurane.
Methods
H4 human neuroglioma cells were treated with saline or 2-DG (5 mM) for one hour followed by a control condition or 2% isoflurane for six hours. The levels of caspase-3 cleavage (activation), BACE, cytosolic calcium, and ROS were determined. Two-way ANOVA analysis was used to assess the interactions of 2-DG and isoflurane on caspase-3 activation, and levels of BACE and ROS.
Results
In H4 human neuroglioma cells 2-DG reduced the caspase-3 activation (477% versus 186%, F = 8.68; # P = 0.019) and the increase in BACE levels (345% versus 123%, F = 42.24; ** P = 0.0002) induced by isoflurane. 2-DG decreased the levels of cytosolic calcium and ROS (100% versus 66%, F = 1.94; * P = 0.014).
Conclusions
These results suggest that 2-DG may decrease oxidative stress and increase of cytosolic calcium levels, thus attenuating isoflurane-induced neurotoxicity.
Introduction
Accumulation of β-amyloid protein (Aβ) plays a fundamental role in the neuropathogenesis of Alzheimer’s disease (AD) (1,2). Aβ is produced from amyloid precursor protein (APP) through proteolytic processing by the aspartyl protease β-site APP-cleaving enzyme (BACE) and γ-secretase (3). Increasing evidence suggests a role for caspase activation and apoptosis in AD neuropathogenesis (4,5,6).
Several studies have suggested that isoflurane, a commonly used inhaled anesthetic, can induce caspase activation and apoptosis, affect APP processing (e.g., increases in BACE levels), and enhance levels and aggregation of Aβ (7–12,13,14). In addition, mechanistic studies have shown that isoflurane may induce caspase activation and apoptosis via increasing cytosolic calcium levels (12,15,16) and causing mitochondrial dysfunction [e.g., increases in reactive oxygen species (ROS) levels] (17). We have found that isoflurane increases ROS levels, which causes mitochondrial damage, leading to caspase-3 activation and apoptosis (17). The caspase activation and apoptosis then induce an increase in BACE levels (10). Finally, isoflurane may induce caspase-3 activation and apoptosis by increasing cytosolic calcium levels (12). However, the targeted prevention and treatment of the isoflurane-induced neurotoxicity have not been investigated.
2-deoxy-d-glucose (2-DG), an analog of glucose, has been shown to have neuroprotective effects via decreasing levels of cellular oxidative stress and enhancing calcium homeostasis (18,19). Specifically, 2-DG attenuates glutamate- or Fe2+-induced ROS generation and the glutamate-induced increase of intracellular calcium levels (18). We sought to determine whether 2-DG can reduce caspase-3 activation and attenuate the increase in BACE, ROS, and cytosolic calcium levels induced by isoflurane. We used H4 human neuroglioma cells (H4 naïve cells) in the studies because these cells are more physiologically relevant without the artificial over-expression of human APP.
Materials and Methods
Cell lines
We used H4 human neuroglioma cells (H4 naive cells) in the current experiments. The cells were cultured in Dulbecco’s modified eagle medium (high glucose) containing 9% heat-inactivated fetal calf serum, 100 units/ml penicillin, 100 μg/ml streptomycin, and 2 mM L-glutamine.
Cell treatment
The cell treatments were similar to those in our previous studies (9,10,20). Twenty-one percent O2, 5% CO2 and 2% isoflurane (air component: 2% isoflurane, 5% CO2, 21% O2 and balanced nitrogen) were delivered from an anesthesia machine to a sealed plastic box in a 37° Celsius incubator containing six-well plates seeded with one million cells in 1.5 ml of cell culture media as described in our previous studies (9,10,20). The control condition included 5% CO2 plus 21% O2 (air component: 5% CO2, 21% O2 and balanced nitrogen), which did not affect caspase-3 activation or BACE levels (9). The Datex infrared gas analyzer (Puritan-Bennett, Tewksbury, MA) was used to continuously monitor the delivered CO2, O2, and isoflurane concentration. We treated the cells with 2% isoflurane for six hours, during which time the cells were incubated in serum-free media. In the interaction studies, the cells were treated with 2-DG (5 mM) (18) one hour before the treatment of 2% isoflurane and throughout the time of the isoflurane treatment, i.e., a total of 7 hours of treatment with 2-DG. The pretreatment time of 2-DG was consistent with that of L-685,458, Congo red, Z-VAD, Aβ, iAβ5, clioquinol, 2-aminoethoxydiphenyl borate, and memantine in our previous studies (10,12,21–24). The treatment of 2-DG was not a preconditioning stimulus. In the studies to determine the effects of serum-free cell culture media on caspase-3 activation, and levels of BACE and ROS, the H4 naïve cells were treated with either cell culture media containing serum or serum-free cell culture media for six hours.
Cell lysis and protein amount quantification
Cell pellets were detergent-extracted on ice using immunoprecipitation buffer (10 mM Tris-HCl, pH 7.4, 150 mM NaCl, 2 mM EDTA, 0.5% Nonidet P-40) plus protease inhibitors (one −g/ml aprotinin, one −g/ml leupeptin, one −g/ml pepstatin A). The lysates were collected, centrifuged at 12,000 rpm for 10 min, and quantified for total proteins by a bicinchoninic acid protein assay kit (Pierce Technology Corporation, Iselin, NJ).
Western blot analysis
The cells were harvested at the end of the experiments and were subjected to Western blot analyses as described by Xie et al. (10). A caspase-3 antibody (1:1,000 dilution; Cell Signaling Technology, Inc. Beverly, MA) was used to recognize the caspase-3 fragment (17–20 kDa), which results from cleavage at the asparate position 175, and caspase-3 FL (35–40 kDa). BACE antibody (1:1,000 dilution; Abcam, Cambridge, MA) was used to recognize BACE (65 kDa). Antibody to the nontargeted protein β-Actin (42 kDa, 1:5,000, Sigma, St. Louis, MO) was used to control for loading differences in total protein amounts. Each band in the Western blot represents an independent experiment. We have averaged the results from three to six independent experiments. The intensity of signals in each Western blot was analyzed using the National Institute of Health image program (National Institute of Health Image 1.62, Bethesda, MD). We quantified the Western blots using two steps. First, we used levels of β-Actin to normalize (e.g., determine the ratio of the amount of FL-caspase-3 to the amount of β-Actin) the levels of BACE and caspase-3 to control for any loading differences in total protein amounts. Second, we presented changes in the levels of BACE and caspase-3 in the treated cells as percentages of those in cells from the control condition.
Cytosolic calcium measurement
Cytosolic calcium levels were determined as described by Zhang et al. (24). Specifically, H4 naive cells were loaded with Fura-2 (Invitrogen), perfused with Tyrode buffer, and [Ca2+]i transients were recorded as changes in Fura-2 ratio (340/380 nm) using a spectrofluoroscope system (Ionoptix, Milton, MA). The cells were exposed to isoflurane (0.57 mM or 2%) or inomycin (10 uM) with or without 2-DG (5 mM) pretreatment.
Reactive oxygen species (ROS) measurement
ROS levels were measured as described by Zhang et al. (17). OxiSelectTM ROS assay kit (Cell Biolabs, Inc. San Diego, CA) was used in the experiments. The experiment was performed according to the protocol provided by the company. Briefly, cultured H4 naive cells were placed in a clear 96-well cell culture plate (5 × 104 cells each well) overnight in the incubator. We then added the 2′,7′-dichlorofluorescein diacetate/media solution to the cells for 30 minutes, then washed the cells twice with Dulbecco’s phosphate buffered saline. The H4 naive cells loaded with 2′,7′-dichlorofluorescein diacetate were then exposed to 2% isoflurane for six hours. The treated cells were lysed by adding 100 −l of cell lysis buffer and were mixed thoroughly and incubated for five minutes at room temperature. We then transferred 150 −l of the mixture to each well of a 96-well plate that was suitable for fluorescence measurement. Finally, the fluorescence was read with a fluorometric plate reader at 480/530 nm.
Statistics
Given the presence of background caspase-3 activation, BACE, and ROS levels in the cells cultured in serum-free media, we did not use absolute values to describe changes in caspase-3 activation, BACE, and ROS levels. Instead, these changes were presented as a percentage of those of the control group. One hundred percent of caspase-3 activation, BACE or ROS refers to control levels for the purpose of comparison to experimental conditions. Data were expressed as mean ± S.D.. The number of samples varied from three to six, and the samples were normally distributed (tested by normality test, data not shown). We used two-way ANOVA and post hoc (Tukey) test to determine the difference among groups and the interaction of 2-DG and isoflurane on caspase-3 activation, levels of BACE and ROS. This was a type III sum of squares. We used a two-tailed t-test (assuming equal variances) to determine the effects of serum-free cell culture media on ROS levels in the H4 naïve cells. * or ** indicated the difference between the control group and treatment group(s); # or ## indicated the interaction of 2-DG and isoflurane.
Results
2-DG attenuates the isoflurane-induced caspase-3 activation
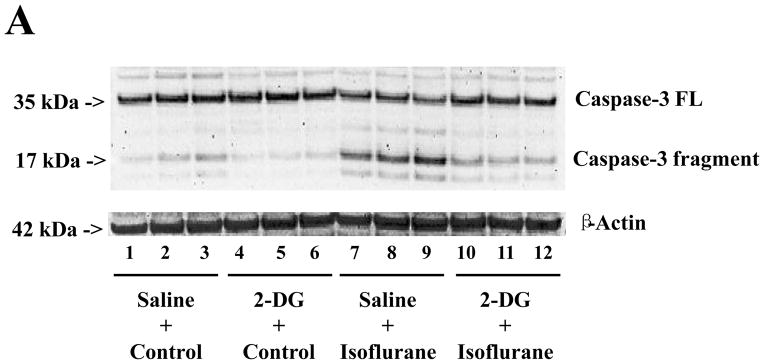
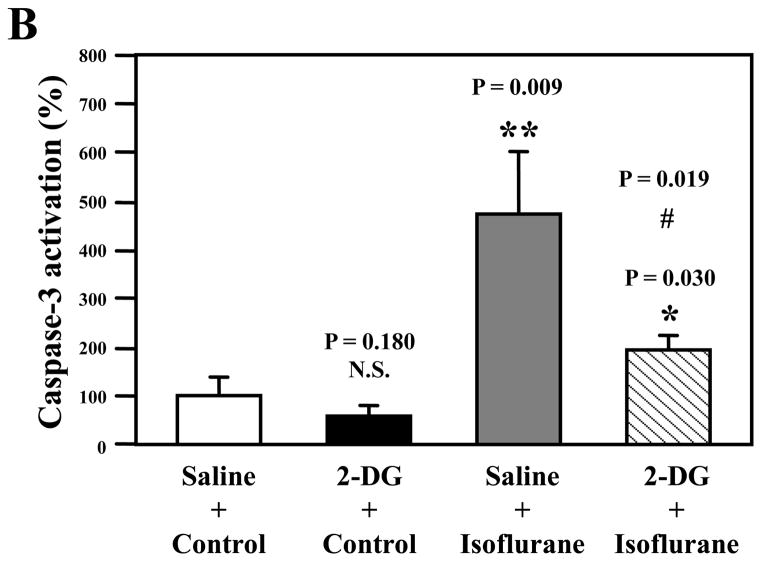
Caspase-3 immunoblotting revealed that isoflurane treatment induced caspase-3 activation (Figure 1A) as evidenced by increased ratios of cleaved (activated) caspase-3 fragment (17 kDa) to full-length (FL) (35 – 40 kDa) caspase-3. Treatment with 2-DG for six hours alone did not induce caspase-3 activation, but the 2-DG treatment attenuated the isoflurane-induced caspase-3 activation (Figure 1A). Quantification of the Western blots, based on the ratio of caspase-3 fragment to FL caspase-3, revealed that the isoflurane treatment led to caspase-3 activation as compared to the control condition: 100% versus 477%, F = 3.94; P = 0.0090. Two-way ANOVA showed that there was an interaction of 2-DG and isoflurane on caspase-3 activation and that 2-DG attenuated the isoflurane-induced caspase-3 activation: 477% versus 186%, F = 8.68; # P = 0.019. These findings suggest that 2-DG mitigates isoflurane-induced caspase-3 activation.
Figure 1. 2-DG attenuates the isoflurane-induced caspase-3 activation in H4 naïve cells.
A. Isoflurane (lanes 7 to 9) induces caspase-3 cleavage (activation) by increasing caspase-3 fragment levels and decreasing full length (FL)-caspase-3 levels as compared to control conditions (lanes 1 to 3) in Western blot analysis. Treatment with 2-deoxy-d-glucose (2-DG) (lanes 4 to 6) does not induce caspase-3 activation as compared to the control condition (lanes 1 to 3), but 2-DG treatment attenuates isoflurane-induced caspase-3 activation (lanes 10 to 12) as compared to isoflurane treatment alone (lanes 7 to 9). B. Quantification of the Western blots shows that isoflurane treatment (gray bar, F = 3.94; ** P = 0.009), but not 2-DG (black bar, F= 0.68; P = 0.180, N.S.), induces caspase-3 activation, assessed by quantifying the ratio of caspase-3 fragment to FL-caspase-3, as compared to that of control conditions (white bar). Two-way ANOVA shows that there is an interaction of 2-DG and isoflurane on caspase-3 activation (striped bar, F = 8.68; # P = 0.019) and that 2-DG attenuates isoflurane-induced caspase-3 activation. N = 3.
2-DG attenuates the isoflurane-induced increases in BACE levels
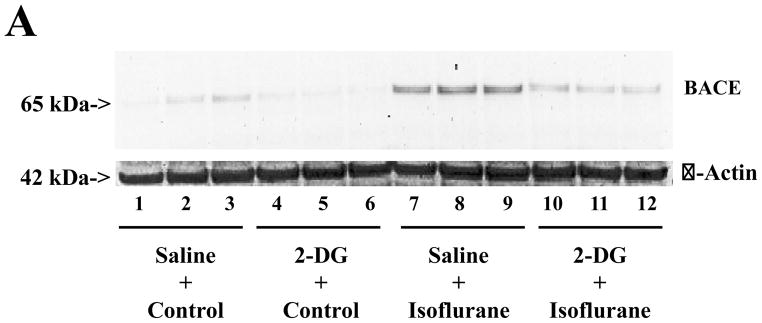
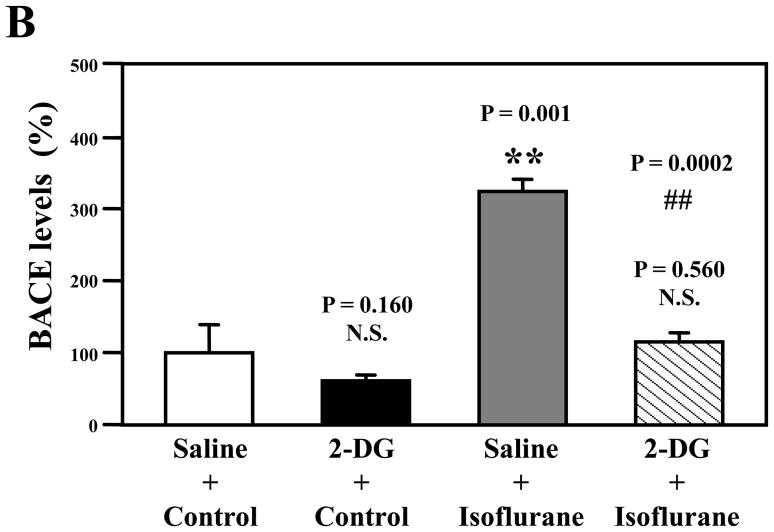
As can be seen in Figure 2A, BACE immunoblotting illustrated that isoflurane increased BACE levels as compared to the control. 2-DG alone did not change BACE levels as compared to controls. However, 2-DG attenuated the isoflurane-induced increases in the BACE levels (Figure 2A). Western blot quantification showed that isoflurane increased BACE levels as compared to controls: 100% versus 345%, F = 1.013; P = 0.001. Two-way ANOVA showed that there was an interaction of 2-DG and isoflurane on BACE, and that 2-DG attenuated the isoflurane-induced increase in BACE levels: 345% versus 123%, F = 42.24; ## P = 0.0002. These findings suggest that 2-DG mitigates isoflurane-induced increases in BACE.
Figure 2. 2-DG attenuates the isoflurane-induced increases in BACE levels in H4 naïve cells.
A. Isoflurane treatment (lanes 7 to 9) increases the levels of β-site APP-cleaving enzyme (BACE) as compared to control conditions (lanes 1 to 3) in Western blot analysis. Treatment with 2-deoxy-d-glucose (2-DG) (lanes 4 to 6) does not change the BACE levels as compared to control conditions (lanes 1 to 3), but 2-DG treatment attenuates isoflurane-induced increases in BACE levels (lanes 10 to 12) as compared to isoflurane treatment alone (lanes 7 to 9). B. Quantification of Western blots shows that isoflurane treatment (gray bar, F = 1.013; ** P = 0.001), but neither 2-DG (black bar, F = 2.44; P = 0.160, N.S.) nor 2-DG plus isoflurane (net bar, F = 1.67, P = 0.560, N.S.), increases levels of BACE as compared to that of control conditions (white bar). Two-Way ANOVA shows that there is an interaction of 2-DG with isoflurane on BACE levels (striped bar, F = 42.24; ## P = 0.0002) and that 2-DG attenuates isoflurane-induced increases in BACE levels. N = 3.
2-DG decreases cytosolic calcium levels
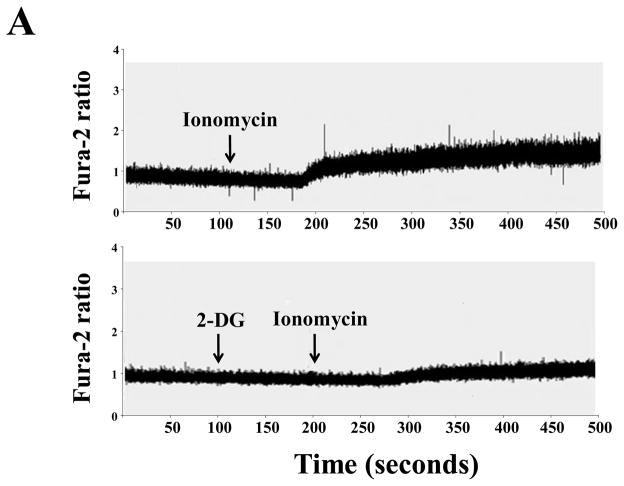
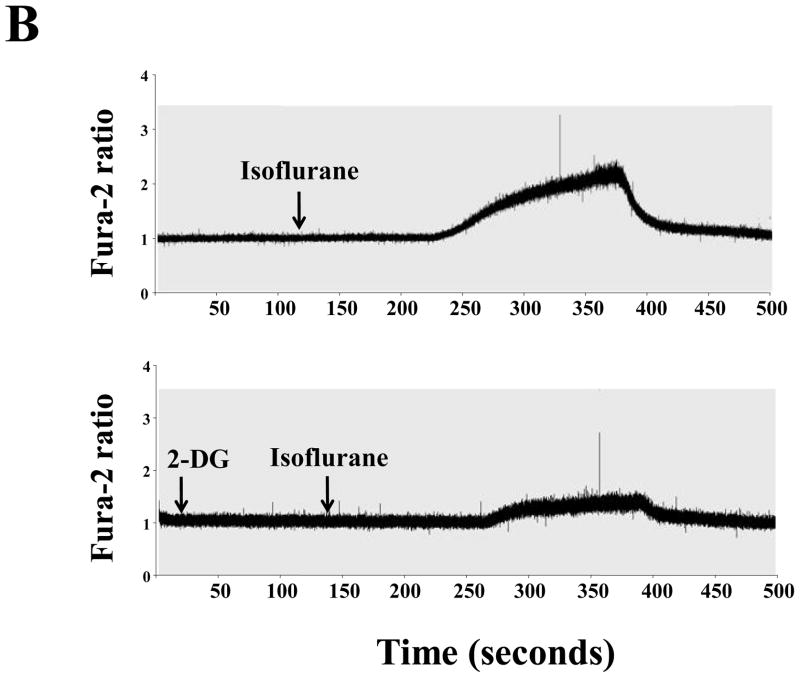
2% isoflurane increased cytosolic calcium (Figure 3B upper panel) in the H4 naïve cells. 2-DG treatment inhibited isoflurane-induced increase in cytosolic calcium (Figure 3B, lower panel). 2-DG attenuated the increase in cytosolic calcium induced by inomycin, which can increase intracellular calcium (25) (Figure 3A). Thus, H4 naive cells released calcium, and 2-DG inhibited both inomycin and isoflurane-induced increase in cytosolic calcium. Finally, 2-DG alone did not increase cytosolic calcium (data not shown). Taken together, these findings suggest that 2-DG may attenuate an isoflurane-induced increase in cytosolic calcium.
Figure 3. Isoflurane-induced elevation of cytosolic calcium levels can be inhibited by 2-DG in H4 naïve cells.
A. The upper panel shows that 10 μM inomycin treatment induces an increase in the fura-2 ratio (340 nm/380 nm) in H4 naïve cells. Treatment with 2-deoxy-d-glucose (2-DG) (lower panel) attenuates the inomycin-induced increase of cytosolic calcium levels in H4 naïve cells. These are representative tracings of two independent experiments. B. Upper panel shows that 2% isoflurane treatment induces a robust increase in the fura-2 ratio (340 nm/380 nm) in H4 naïve cells, which suggests that isoflurane treatment can increase cytosolic calcium levels. Treatment with 2-DG (lower panel) attenuates the isoflurane-induced elevation of cytosolic calcium in H4 naïve cells. These are representative tracings of two independent experiments.
2-DG decreases ROS levels
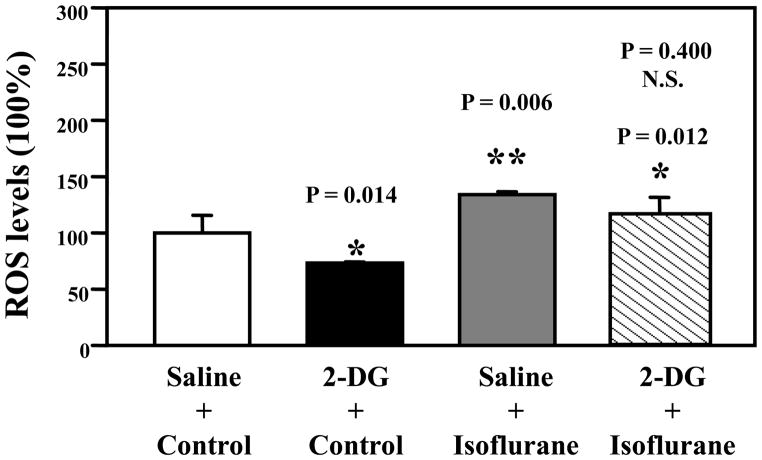
Isoflurane increased the levels of ROS (gray bar, F = 1.78; P = 0.006) and 2-DG treatment attenuated ROS levels (black bar, F = 1.94; P = 0.014) (Figure 4). Two-way ANOVA showed that there was no interaction of isoflurane and 2-DG on ROS (F = 0.69; P = 0.400, N.S.). These results suggest that 2-DG may not specifically attenuate an isoflurane-induced increase in ROS, but 2-DG decreased ROS, mitigating the isoflurane-induced increase in ROS.
Figure 4. 2-DG attenuates ROS levels.
Fluorescence assay shows that 2-deoxy-d-glucose (2-DG) (black bar, F = 1.94, * P = 0.014) decreases ROS levels; isoflurane (gray bar, F = 1.78; ** P = 0.006) increases reactive oxygen species (ROS) levels, and 2-DG plus isoflurane (net bar, F = 2.04; * P = 0.012) increase ROS levels as compared to control conditions (white bar). Two-way ANOVA shows that there is no interaction of isoflurane and 2-DG on ROS levels (F = 0.69; P = 0.400, N.S.). N = 6.
Effects of serum-free cell culture media on caspase-3 activation, BACE levels and ROS levels
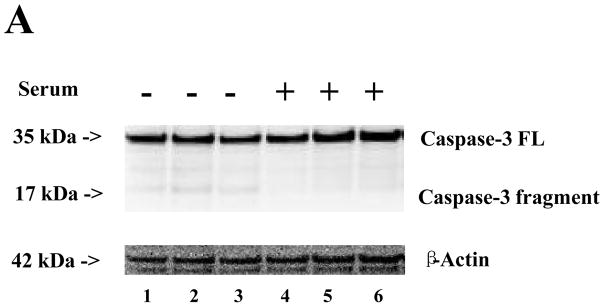
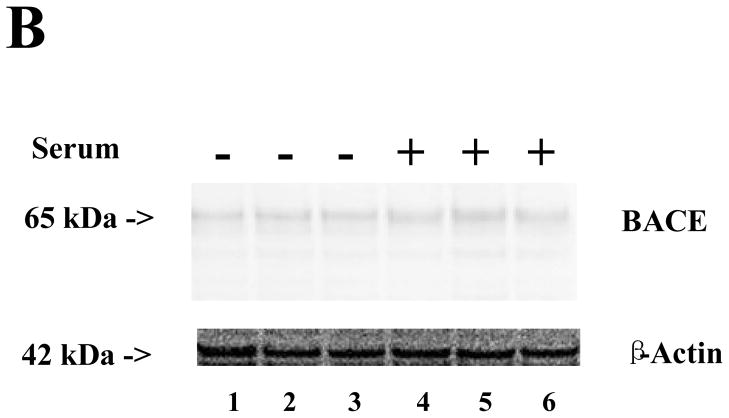
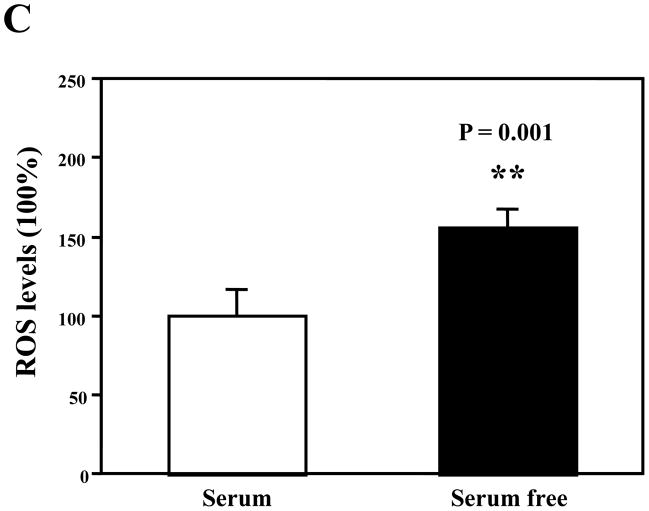
Treatment with serum-free cell culture media for six hours induced a slight caspase-3 activation (Figure 5A), slight increases in BACE (Figure 5B), and significant increases in ROS (Figure 5C) as compared to the cell culture media containing serum in the H4 naïve cells. These results suggest that the serum deprivation could induce caspase-3 activation and increase BACE and ROS, and that these effects can be attenuated by 2-DG (Figures 1, 2, and 4, black bars).
Figure 5. Comparison of serum and serum-free cell culture media on caspase-3 activation, BACE levels, and ROS levels in H4 naïve cells.
A. There are slight increases in caspase-3 activation in the cells after 6-hour treatment in serum-free cell culture media (lanes 1 to 3) as compared to those after 6-hour treatment in cell culture media containing serum. N = 3. B. There is no significant difference in β-site APP-cleaving enzyme (BACE) levels between the cells after 6-hour treatment with serum-free cell culture media (lanes 1 to 3) and cells after treatment with cell culture media containing serum (lanes 4 to 6). N = 3. C. There is a significant increase in reactive oxygen species (ROS) levels in the cells after 6-hour treatment in serum-free cell culture media (black bar, ** P = 0.001) as compared to cells after treatment with cell culture media containing serum (white bar). N = 6.
Discussion
AD is one of the most common forms of dementia, affects 5.4 million Americans and costs $183 billion a year in the United States. a An estimated 200 million patients worldwide undergo anesthesia and surgery each year. Even though anesthesia and surgery may not increase the incidence of AD (26), it has been reported that age of onset of AD is inversely related to cumulative exposure to anesthesia and surgery before age 50 years (27). Several studies have suggested an association between anesthesia exposure and AD risk (28,29). However, other studies have suggested that there is little or no relationship between anesthesia and AD (30–32). More population studies, defining the role of anesthesia and surgery in AD, are necessary (14,33). Because we cannot clinically prove or disprove the relationship of anesthesia and AD and performing such studies may take many years, it is equally important to perform animal studies and in vitro studies, which will establish mechanistic hypotheses, vulnerable windows, less provocative anesthetics, and potential interventions. The results from these in vitro and animal studies would help to design more targeted and focused clinical research (14).
We found that 2-DG attenuated isoflurane-induced caspase-3 activation in H4 naïve cells. Our previous studies have shown that isoflurane-induced caspase-3 activation can increase the levels of BACE, which will promote APP processing to generate Aβ (10). Thus, we determined the effects of 2-DG on the BACE levels and have found that 2-DG can also attenuate isoflurane-induced increase in BACE levels. Given that many studies have shown that isoflurane can promote AD neuropathogenesis (13,14,17), the results from the current studies may suggest that 2-DG could mitigate isoflurane-induced promotion of AD neuropathogenesis.
We chose to use H4 naïve cells because these cells are more physiologically relevant without the artificial over-expression of human APP. However, it is technically difficult to detect APP processing and Aβ levels in these nonhuman APP over-expression cells, as described in our previous studies (10). Therefore, one limitation in the current studies is that we were unable to directly assess the effects of 2-DG on isoflurane-induced APP processing and Aβ generation. However, increases in BACE levels strongly suggest the promotion of APP processing and Aβ generation (10,34). Our previous studies have shown that isoflurane can promote APP processing and increase Aβ generation by enhancing BACE levels (10). Thus, the measurement of BACE could serve as an indirect assay for Aβ in the current studies. Taken together, our findings that 2-DG mitigates the effects of isoflurane on increasing BACE levels highly suggest that 2-DG is able to mitigate the effects of isoflurane on promoting APP processing and Aβ generation. Our future experiments will include the in vivo relevance studies in primary (cortex/hippocampus) cultured neurons, wild type mice, and AD transgenic mice (which have higher Aβ levels for a relatively easy measurement of Aβ) to further assess the potential protective effects of 2-DG on isoflurane or other anesthetics (e.g., sevoflurane) induced neurotoxicity.
The current studies are also limited by the use of serum-free cell culture media, which suggest that the changes in caspase-3 activation, levels of BACE and ROS could result from the combination of isoflurane treatment and serum deprivation. We used serum-free cell culture media in order to more easily demonstrate the effects of isoflurane on caspase-3 activation, increases in BACE and ROS levels, because serum could protect the cells from injuries, which may cover the potential cytotoxic effects of isoflurane. The findings that the anesthetics isoflurane and sevoflurane induce caspase activation, apoptosis, cell death, increase in BACE and Aβ levels in the brain tissues of mice (11,12,21,22) and in the cells incubated in the cell culture media containing serum (7,8,15,35) suggest that the isoflurane-induced cytotoxic effects may not be dependent on serum deprivation. Moreover, we have found that serum deprivation increases the levels of ROS but not caspase-3 activation and levels of BACE in the H4 naïve cells (Figure 5). Nevertheless, given the fact that serum-free cell culture media was used in the current experiments, it is conceivable that 2-DG may attenuate caspase-3 activation, increases in the levels of BACE and ROS induced by the combination of isoflurane and serum deprivation. As such, conclusions about the effects of 2-DG on isoflurane-induced cytotoxicity must be made cautiously and will require independent confirmation both in vitro and in vivo.
Several studies have shown that 2-DG has cytoprotective effects through decreasing ROS generation and cytosolic calcium increase (18,19). Isoflurane has been shown to induce caspase-3 activation by increasing cytosolic calcium levels (24) and increasing ROS generation (17). We therefore investigated the underlying mechanism by which 2-DG inhibits the isoflurane-induced caspase-3 activation by assessing the effects of 2-DG on the isoflurane-induced increase of cytosolic calcium levels and ROS accumulation in H4 naïve cells. The findings that 2-DG can attenuate the inomycin-induced increase of cytosolic calcium (Figure 3A) have validated the calcium study system in the H4 naïve cells used in our experiments. We then found that the effects of 2-DG on attenuating the levels of cytosolic calcium and ROS mitigate the effects of isoflurane on increasing the levels of cytosolic calcium and ROS. Collectively, these studies have further demonstrated that isoflurane may induce neurotoxicity via the mitochondria pathway of apoptosis (17) and have suggested that 2-DG may ameliorate isoflurane-induced neurotoxicity by inhibiting the mitochondrial pathway of apoptosis. Specifically, we have postulated that 2-DG inhibits the isoflurane-induced increase of cytosolic calcium levels, leading to attenuation of the isoflurane-induced increase of ROS. The reduced ROS levels will prohibit isoflurane-induced caspase activation and apoptosis as suggested by Zhang et al. (17). Finally, the inhibition of caspase activation and apoptosis will lead to attenuation of isoflurane-induced increases in BACE levels (10). Alternatively, 2-DG may attenuate the isoflurane-induced increase of BACE levels via affecting oxidative stress and hypoxia-inducible factor 1 (HIF1)-α because BACE levels can be enhanced by oxidative stress and HIF1-α (36). Finally, p75 neurotrophin receptor (p75NTR) can regulate BACE levels and this regulation can be prevented by caloric restriction (e.g., 2-DG treatments) (37). Thus, 2-DG may mitigate isoflurane-induced increases in BACE levels through affecting p75NTR. Future studies should include an assessment of the interaction of 2-DG and isoflurane on the levels of HIF1-α and p75NTR to further understand the underlying mechanisms by which 2-DG attenuates isoflurane-induced cytotoxicity.
It is interesting that 2-DG can reduce an increase of cytosolic calcium levels induced by both isoflurane and inomycin. Given that isoflurane and inomycin may increase the levels of cytosolic calcium via different mechanisms, it is likely that the mitigation effects of 2-DG on isoflurane-induced increase of cytosolic calcium are not specific to isoflurane. Moreover, these findings may suggest that the effects of isoflurane and 2-DG on cytosolic calcium levels, ROS generation, caspase-3 activation, and BACE could be through independent mechanistic pathways.
2-DG, a glucose analog, is a competitive inhibitor of hexokinase, which can decrease glucose availability and energy of cells. 2-DG has been shown to significantly reduce seizure-induced spatial memory deficits and hippocampus neuron loss in rats (18). High concentrations of 2-DG (e.g., 10 or 20 mM) may induce ROS release from the mitochondria, leading to cytotoxic effects (18). However, low concentrations of 2-DG (e.g., 5 mM) can protect oxidative injury in primary neurons, neural progenitor cells, or synaptic terminals (38,39). We therefore used a low concentration of 2-DG (5 mM) in our current experiments to determine whether 2-DG can mitigate the isoflurane-induced cytotoxicity. It is unclear as to why 2-DG decreased the ROS levels as presented in Figure 4. We used serum-free cell culture media in the studies and serum deprivation can increase ROS levels (Figure 5). It is therefore conceivable that 2-DG may also mitigate the serum deprivation-induced increases in ROS levels (Figure 4, black bar). It is also possible that 2-DG may affect the autophagic state of cells, however, the results from such studies would only provide additional evidence to demonstrate that 2-DG may mitigate isoflurane-induced cytotoxicity, which will only serve to further support the current conclusion. We therefore will systematically assess the interactive effects of 2-DG with isoflurane and other anesthetics (e.g., sevoflurane) on the autophagic state of cells in our future studies. Impaired glucose use and reduced energy in the brain are associated with AD neuropathogenesis (40,41), and the anesthetic isoflurane has been shown to promote AD neuropathogenesis (13,14). Furthermore, our current findings suggest that 2-DG can attenuate isoflurane-induced neurotoxicity through mitochondria-related mechanisms. Taken together, it is conceivable that isoflurane may induce mitochondrial dysfunction, impair generation of cell energy, leading to caspase activation, apoptosis, and Aβ accumulation. Our future studies should include systematically assessing the effects of isoflurane and other anesthetics on mitochondrial function and cellular energy levels.
In conclusion, we found that 2-DG might protect against isoflurane-induced cytotoxicity in cultured cells. Specifically, 2-DG decreased ROS and cytosolic calcium, mitigating caspase-3 activation and increase in BACE induced by isoflurane. These findings should lead to more studies investigating the neurotoxicity of anesthesia and AD neuropathgenesis, especially the underlying mechanisms and possible interventions.
Acknowledgments
Funding: This research was supported by K08NS048140, R21AG029856 and R01 GM088801 (National Institutes of Health), Jahnigen Career Development Award (American Geriatrics Society), Investigator Initiated Research Grant (Alzheimer’s Association) (to Z. X.), R01GM079360 (National Institutes of Health) (to FI), and China National Science Foundation Oversea young scholar collaboration research award NSF30928036 (to Y. Y. and Z. X.).
The authors would like to thank Dr. Hui Zheng, the Assistant Professor in Medicine and statistician in Massachusetts General Hospital and Harvard Medical School, for the advice regarding statistic analysis of the data in the studies.
Footnotes
2011 Alzheimer’s Facts and Figures, Alzheimer’s Association, http://www.alz.org/downloads/Facts_Figures_2011.pdf (Accessed July 15, 2011)
The experiments were performed at Massachusetts General Hospital and Harvard Medical School.
The authors declare no conflicts of interest.
Reprints will not be available from the authors.
DISCLOSURES:
Name: Jun Zhang, M.D., Ph.D.
Contribution: Data collection and data analysis. Attestation: Jun Zhang approved the final manuscript.
Name: Yuanlin Dong, M.D., M.S.
Contribution: Data collection and data analysis.
Attestation: Yuanlin Dong approved the final manuscript.
Name: Zhipeng Xu, M.D., Ph.D.
Contribution: Data collection and data analysis.
Attestation: Zhipeng Xu approved the final manuscript.
Name: Yiying Zhang, M.D., M.S.
Contribution: Data collection and data analysis.
Attestation: Yiying Zhang approved the final manuscript.
Name: Chuxiong Pan, M.D., M.S.
Contribution: Data collection and data analysis.
Attestation: Chuxiong Pan approved the final manuscript.
Name: Sayre McAuliffe, B.A.
Contribution: Data collection, data analysis and manuscript preparation. Attestation: Sayre McAuliffe approved the final manuscript.
Name: Fumito Ichinose, M.D.
Contribution: Data collection, data analysis and manuscript preparation Attestation: Fumito Ichinose approved the final manuscript.
Name: Yun Yue, M.D., M.S.
Contribution: Manuscript preparation.
Attestation: Yun Yue approved the final manuscript.
Name: Weimin Liang, M.D., Ph.D. Contribution: Manuscript preparation.
Attestation: Weimin Liang approved the final manuscript.
Name: Zhongcong Xie, M.D., Ph.D. *
Contribution: Study design and manuscript preparation. Attestation: Zhongcong Xie approved the final manuscript.
This manuscript was handled by: Steven L. Shafer, MD
Contributor Information
Jun Zhang, Geriatric Anesthesia Research Unit, Department of Anesthesia, Critical Care and Pain Medicine, Massachusetts General Hospital and Harvard Medical School, Charlestown, MA. (Current Affiliation: Department of Anesthesiology, Huashan Hospital, Fudan University, Shanghai, P. R. China)
Yuanlin Dong, Geriatric Anesthesia Research Unit, Department of Anesthesia, Critical Care and Pain Medicine, Massachusetts General Hospital and Harvard Medical School, Charlestown, MA
Zhipeng Xu, Geriatric Anesthesia Research Unit, Department of Anesthesia, Critical Care and Pain Medicine, Massachusetts General Hospital and Harvard Medical School, Charlestown, MA
Yiying Zhang, Geriatric Anesthesia Research Unit, Department of Anesthesia, Critical Care and Pain Medicine, Massachusetts General Hospital and Harvard Medical School, Charlestown, MA
Chuxiong Pan, Geriatric Anesthesia Research Unit, Department of Anesthesia, Critical Care and Pain Medicine, Massachusetts General Hospital and Harvard Medical School, Charlestown, MA. (Current Affiliation: Department of Anesthesia, Beijing Tongren Hospital, Capital Medical University, Beijing, P. R. China)
Sayre McAuliffe, Geriatric Anesthesia Research Unit, Department of Anesthesia, Critical Care and Pain Medicine, Massachusetts General Hospital and Harvard Medical School, Charlestown, MA
Fumito Ichinose, Department of Anesthesia, Critical Care and Pain Medicine, Massachusetts General Hospital and Harvard Medical School, Charlestown, MA
Yun Yue, Department of Anesthesia, Beijing Chaoyang Hospital, Capital Medical University, Beijing, P. R. China
Weimin Liang, Department of Anesthesiology, Huashan Hospital, Fudan University, Shanghai, P. R. China
Zhongcong Xie, Geriatric Anesthesia Research Unit, Department of Anesthesia, Critical Care and Pain Medicine, Massachusetts General Hospital and Harvard Medical School, Charlestown, MA
References
- 1.Tanzi RE, Bertram L. Twenty years of the Alzheimer’s disease amyloid hypothesis: a genetic perspective. Cell. 2005;120:545–55. doi: 10.1016/j.cell.2005.02.008. [DOI] [PubMed] [Google Scholar]
- 2.Selkoe DJ. Alzheimer’s disease: genes, proteins, and therapy. Physiol Rev. 2001;81:741–66. doi: 10.1152/physrev.2001.81.2.741. [DOI] [PubMed] [Google Scholar]
- 3.Xie Z, Tanzi RE. Alzheimer’s disease and post-operative cognitive dysfunction. Exp Gerontol. 2006;41:346–59. doi: 10.1016/j.exger.2006.01.014. [DOI] [PubMed] [Google Scholar]
- 4.Burguillos MA, Deierborg T, Kavanagh E, Persson A, Hajji N, Garcia-Quintanilla A, Cano J, Brundin P, Englund E, Venero JL, Joseph B. Caspase signalling controls microglia activation and neurotoxicity. Nature. 2011 doi: 10.1038/nature09788. [DOI] [PubMed] [Google Scholar]
- 5.Mattson MP. Contributions of mitochondrial alterations, resulting from bad genes and a hostile environment, to the pathogenesis of Alzheimer’s disease. Int Rev Neurobiol. 2002;53:387–409. doi: 10.1016/s0074-7742(02)53014-2. [DOI] [PubMed] [Google Scholar]
- 6.Raina AK, Hochman A, Ickes H, Zhu X, Ogawa O, Cash AD, Shimohama S, Perry G, Smith MA. Apoptotic promoters and inhibitors in Alzheimer’s disease: Who wins out? Prog Neuropsychopharmacol Biol Psychiatry. 2003;27:251–4. doi: 10.1016/S0278-5846(03)00020-4. [DOI] [PubMed] [Google Scholar]
- 7.Eckenhoff RG, Johansson JS, Wei H, Carnini A, Kang B, Wei W, Pidikiti R, Keller JM, Eckenhoff MF. Inhaled anesthetic enhancement of amyloid-beta oligomerization and cytotoxicity. Anesthesiology. 2004;101:703–9. doi: 10.1097/00000542-200409000-00019. [DOI] [PubMed] [Google Scholar]
- 8.Wei H, Kang B, Wei W, Liang G, Meng QC, Li Y, Eckenhoff RG. Isoflurane and sevoflurane affect cell survival and BCL-2/BAX ratio differently. Brain Res. 2005;1037:139–47. doi: 10.1016/j.brainres.2005.01.009. [DOI] [PubMed] [Google Scholar]
- 9.Xie Z, Dong Y, Maeda U, Alfille P, Culley DJ, Crosby G, Tanzi RE. The common inhalation anesthetic isoflurane induces apoptosis and increases amyloid beta protein levels. Anesthesiology. 2006;104:988–94. doi: 10.1097/00000542-200605000-00015. [DOI] [PubMed] [Google Scholar]
- 10.Xie Z, Dong Y, Maeda U, Moir RD, Xia W, Culley DJ, Crosby G, Tanzi RE. The inhalation anesthetic isoflurane induces a vicious cycle of apoptosis and amyloid beta-protein accumulation. J Neurosci. 2007;27:1247–54. doi: 10.1523/JNEUROSCI.5320-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Xie Z, Culley DJ, Dong Y, Zhang G, Zhang B, Moir RD, Frosch MP, Crosby G, Tanzi RE. The common inhalation anesthetic isoflurane induces caspase activation and increases amyloid beta-protein level in vivo. Ann Neurol. 2008;64:618–27. doi: 10.1002/ana.21548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zhang G, Dong Y, Zhang B, Ichinose F, Wu X, Culley DJ, Crosby G, Tanzi RE, Xie Z. Isoflurane-induced caspase-3 activation is dependent on cytosolic calcium and can be attenuated by memantine. J Neurosci. 2008;28:4551–60. doi: 10.1523/JNEUROSCI.5694-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tang J, Eckenhoff MF, Eckenhoff RG. Anesthesia and the old brain. Anesth Analg. 2010;110:421–6. doi: 10.1213/ANE.0b013e3181b80939. [DOI] [PubMed] [Google Scholar]
- 14.Bittner EA, Yue Y, Xie Z. Brief review: anesthetic neurotoxicity in the elderly, cognitive dysfunction and Alzheimer’s disease. Can J Anaesth. 2011;58:216–23. doi: 10.1007/s12630-010-9418-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wei H, Liang G, Yang H, Wang Q, Hawkins B, Madesh M, Wang S, Eckenhoff RG. The common inhalational anesthetic isoflurane induces apoptosis via activation of inositol 1,4,5-trisphosphate receptors. Anesthesiology. 2008;108:251–60. doi: 10.1097/01.anes.0000299435.59242.0e. [DOI] [PubMed] [Google Scholar]
- 16.Wei H, Xie Z. Anesthesia, calcium homeostasis and Alzheimer’s disease. Curr Alzheimer Res. 2009;6:30–5. doi: 10.2174/156720509787313934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhang Y, Dong Y, Wu X, Lu Y, Xu Z, Knapp A, Yue Y, Xu T, Xie Z. The mitochondrial pathway of anesthetic isoflurane-induced apoptosis. J Biol Chem. 2010;285:4025–37. doi: 10.1074/jbc.M109.065664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lee J, Bruce-Keller AJ, Kruman Y, Chan SL, Mattson MP. 2-Deoxy-D-glucose protects hippocampal neurons against excitotoxic and oxidative injury: evidence for the involvement of stress proteins. J Neurosci Res. 1999;57:48–61. doi: 10.1002/(SICI)1097-4547(19990701)57:1<48::AID-JNR6>3.0.CO;2-L. [DOI] [PubMed] [Google Scholar]
- 19.Duan W, Mattson MP. Dietary restriction and 2-deoxyglucose administration improve behavioral outcome and reduce degeneration of dopaminergic neurons in models of Parkinson’s disease. J Neurosci Res. 1999;57:195–206. doi: 10.1002/(SICI)1097-4547(19990715)57:2<195::AID-JNR5>3.0.CO;2-P. [DOI] [PubMed] [Google Scholar]
- 20.Xie Z, Dong Y, Maeda U, Moir R, Inouye SK, Culley DJ, Crosby G, Tanzi RE. Isoflurane-induced apoptosis: a potential pathogenic link between delirium and dementia. J Gerontol A Biol Sci Med Sci. 2006;61:1300–6. doi: 10.1093/gerona/61.12.1300. [DOI] [PubMed] [Google Scholar]
- 21.Lu Y, Wu X, Dong Y, Xu Z, Zhang Y, Xie Z. Anesthetic sevoflurane causes neurotoxicity differently in neonatal naive and Alzheimer disease transgenic mice. Anesthesiology. 2010;112:1404–16. doi: 10.1097/ALN.0b013e3181d94de1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Dong Y, Zhang G, Zhang B, Moir RD, Xia W, Marcantonio ER, Culley DJ, Crosby G, Tanzi RE, Xie Z. The common inhalational anesthetic sevoflurane induces apoptosis and increases beta-amyloid protein levels. Arch Neurol. 2009;66:620–31. doi: 10.1001/archneurol.2009.48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zhen Y, Dong Y, Wu X, Xu Z, Lu Y, Zhang Y, Norton D, Tian M, Li S, Xie Z. Nitrous oxide plus isoflurane induces apoptosis and increases beta-amyloid protein levels. Anesthesiology. 2009;111:741–52. doi: 10.1097/ALN.0b013e3181b27fd4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zhang B, Dong Y, Zhang G, Moir RD, Xia W, Yue Y, Tian M, Culley DJ, Crosby G, Tanzi RE, Xie Z. The inhalation anesthetic desflurane induces caspase activation and increases amyloid beta-protein levels under hypoxic conditions. J Biol Chem. 2008;283:11866–75. doi: 10.1074/jbc.M800199200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Morgan AJ, Jacob R. Ionomycin enhances Ca2+ influx by stimulating store-regulated cation entry and not by a direct action at the plasma membrane. Biochem J. 1994;300 ( Pt 3):665–72. doi: 10.1042/bj3000665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bohnen NI, Warner MA, Kokmen E, Beard CM, Kurland LT. Alzheimer’s disease and cumulative exposure to anesthesia: a case-control study. J Am Geriatr Soc. 1994;42:198–201. doi: 10.1111/j.1532-5415.1994.tb04952.x. [DOI] [PubMed] [Google Scholar]
- 27.Bohnen N, Warner MA, Kokmen E, Kurland LT. Early and midlife exposure to anesthesia and age of onset of Alzheimer’s disease. Int J Neurosci. 1994;77:181–5. doi: 10.3109/00207459408986029. [DOI] [PubMed] [Google Scholar]
- 28.Lee TA, Wolozin B, Weiss KB, Bednar MM. Assessment of the emergence of Alzheimer’s disease following coronary artery bypass graft surgery or percutaneous transluminal coronary angioplasty. J Alzheimers Dis. 2005;7:319–24. doi: 10.3233/jad-2005-7408. [DOI] [PubMed] [Google Scholar]
- 29.Bufill E, Bartes A, Moral A, Casadevall T, Codinachs M, Zapater E, Carles Rovira J, Roura P, Oliva R, Blesa R. Genetic and environmental factors that may influence in the senile form of Alzheimer’s disease: nested case control studies. Neurologia. 2009;24:108–12. [PubMed] [Google Scholar]
- 30.Knopman DS, Petersen RC, Cha RH, Edland SD, Rocca WA. Coronary artery bypass grafting is not a risk factor for dementia or Alzheimer disease. Neurology. 2005;65:986–90. doi: 10.1212/01.wnl.0000171954.92119.c7. [DOI] [PubMed] [Google Scholar]
- 31.Gasparini M, Vanacore N, Schiaffini C, Brusa L, Panella M, Talarico G, Bruno G, Meco G, Lenzi GL. A case-control study on Alzheimer’s disease and exposure to anesthesia. Neurol Sci. 2002;23:11–4. doi: 10.1007/s100720200017. [DOI] [PubMed] [Google Scholar]
- 32.Avidan MS, Searleman AC, Storandt M, Barnett K, Vannucci A, Saager L, Xiong C, Grant EA, Kaiser D, Morris JC, Evers AS. Long-term cognitive decline in older subjects was not attributable to noncardiac surgery or major illness. Anesthesiology. 2009;111:964–70. doi: 10.1097/ALN.0b013e3181bc9719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Harris RA, Eger EI., 2nd Alzheimer’s disease and anesthesia: out of body, out of mind… or not? Ann Neurol. 2008;64:595–7. doi: 10.1002/ana.21575. [DOI] [PubMed] [Google Scholar]
- 34.Vassar R, Bennett BD, Babu-Khan S, Kahn S, Mendiaz EA, Denis P, Teplow DB, Ross S, Amarante P, Loeloff R, Luo Y, Fisher S, Fuller J, Edenson S, Lile J, Jarosinski MA, Biere AL, Curran E, Burgess T, Louis JC, Collins F, Treanor J, Rogers G, Citron M. Beta-secretase cleavage of Alzheimer’s amyloid precursor protein by the transmembrane aspartic protease BACE. Science. 1999;286:735–41. doi: 10.1126/science.286.5440.735. [DOI] [PubMed] [Google Scholar]
- 35.Wei H, Liang G, Yang H. Isoflurane preconditioning inhibited isoflurane-induced neurotoxicity. Neurosci Lett. 2007;425:59–62. doi: 10.1016/j.neulet.2007.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Guglielmotto M, Aragno M, Autelli R, Giliberto L, Novo E, Colombatto S, Danni O, Parola M, Smith MA, Perry G, Tamagno E, Tabaton M. The up-regulation of BACE1 mediated by hypoxia and ischemic injury: role of oxidative stress and HIF1alpha. J Neurochem. 2009;108:1045–56. doi: 10.1111/j.1471-4159.2008.05858.x. [DOI] [PubMed] [Google Scholar]
- 37.Costantini C, Weindruch R, Della Valle G, Puglielli L. A TrkA-to-p75NTR molecular switch activates amyloid beta-peptide generation during aging. Biochem J. 2005;391:59–67. doi: 10.1042/BJ20050700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Guo ZH, Mattson MP. In vivo 2-deoxyglucose administration preserves glucose and glutamate transport and mitochondrial function in cortical synaptic terminals after exposure to amyloid beta-peptide and iron: evidence for a stress response. Exp Neurol. 2000;166:173–9. doi: 10.1006/exnr.2000.7497. [DOI] [PubMed] [Google Scholar]
- 39.Park M, Song KS, Kim HK, Park YJ, Kim HS, Bae MI, Lee J. 2-Deoxy-d-glucose protects neural progenitor cells against oxidative stress through the activation of AMP-activated protein kinase. Neurosci Lett. 2009;449:201–6. doi: 10.1016/j.neulet.2008.11.007. [DOI] [PubMed] [Google Scholar]
- 40.O’Connor T, Sadleir KR, Maus E, Velliquette RA, Zhao J, Cole SL, Eimer WA, Hitt B, Bembinster LA, Lammich S, Lichtenthaler SF, Hebert SS, De Strooper B, Haass C, Bennett DA, Vassar R. Phosphorylation of the translation initiation factor eIF2alpha increases BACE1 levels and promotes amyloidogenesis. Neuron. 2008;60:988–1009. doi: 10.1016/j.neuron.2008.10.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Velliquette RA, O’Connor T, Vassar R. Energy inhibition elevates beta-secretase levels and activity and is potentially amyloidogenic in APP transgenic mice: possible early events in Alzheimer’s disease pathogenesis. J Neurosci. 2005;25:10874–83. doi: 10.1523/JNEUROSCI.2350-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]