Abstract
Transplantation of hematopoietic stem cells from healthy donors can cure patients with many diseases. Donor T cells can protect against recurrence of infection and disease, but some of these (alloreactive) T cells recognize patient tissues as foreign, causing graft-versus-host disease. Removing T cells from donor grafts before transplantation reduces graft-versus-host disease but increases infection and disease recurrence. Inactivation of alloreactive T cells by inducing tolerance to patient cells (anergization) before transplantation preserves beneficial donor T cell effects while reducing graft-versus-host disease. We show that this approach also results in expansion of regulatory T cells that specifically suppress alloreactive donor T cell responses in the recipient. In addition to reducing graft-versus-host disease, antigen-specific regulatory T cells generated with this strategy could suppress unwanted T cell responses that cause rejection of solid organ transplants and tissue damage in autoimmune disorders.
Introduction
Allogeneic hematopoietic stem cell transplantation is the process of administering chemotherapy or radiation therapy to patients to eliminate their own bone marrow followed by the infusion of normal hematopoietic stem cells from a donor, a healthy individual. This treatment can cure patients with a broad range of diseases. Most allogeneic transplants are performed for malignant disorders such as leukemia and lymphoma, although this treatment is also curative for a broad range of inherited or acquired disorders of hematopoiesis and the immune system, including thalassemia and severe congenital immunodeficiencies. The use of sibling donors who are fully matched at human leukocyte antigen (HLA) histocompatibility loci produces the best results after allogeneic transplantation. As a result of reduced family size, however, the likelihood of a patient having such a donor is no more than 25% (1). Although there are alternate sources of hematopoietic stem cells for individuals without an appropriate sibling donor, failure to locate well-matched donors remains an impediment to successful transplantation from unrelated donors (2). This is a particularly significant problem for patients belonging to ethnic groups that are underrepresented in volunteer donor bone marrow registries and umbilical cord blood banks (3–5).
Most patients, however, will have family donors who match at half the HLA loci. The use of hematopoietic stem cells from these half-matched (haploidentical) donors would increase the availability of allogeneic transplantation as a treatment (6). Increased HLA mismatch between donor and recipient results in greater frequency and severity of acute graft-versus-host disease (GVHD) (7). Acute GVHD, in which some donor T cells recognize, attack, and damage recipient tissues, is a significant and potentially fatal complication after allogeneic transplantation. Severe acute GVHD is effectively prevented by depleting T cells from donor grafts before transplantation. This non-selective process removes alloreactive donor T cells that recognize recipient tissues and cause GVHD but also removes donor T cells that provide pathogen- and tumor-specific immunity, markedly delaying reconstitution of the immune system, significantly increasing infection, and contributing to disease relapse (8–12).
Strategies have therefore been developed to selectively remove or de- stroy only alloreactive cells within T cell pools from HLA-mismatched donors before transplantation (13–16). In these allodepletion approaches, the donor T cells are stimulated by alloantigens on recipient cells. The alloreactive T cells are removed or destroyed after collection from the donor but before transplantation into the recipient. These depletion techniques, which identify alloreactive T cells by cell surface markers or metabolic activity, lack specificity and may lead to undesirable loss of T cells that would otherwise provide anti-pathogen or anti-tumor immunity. An alternative to these depletion strategies involves altering the function of the alloreactive T cells. One approach takes advantage of the requirement of T cells for both an HLA-restricted, antigen-specific signal and a second nonspecific costimulatory signal to become activated and proliferate. The dominant costimulatory signal for human T cell activation is delivered by the B7.1 and B7.2 molecules on antigen- presenting cells to the CD28 receptor on CD4+ T cells (17). Blockade of B7-CD28 interaction during antigen presentation induces antigen- specific hyporesponsiveness (anergy) in T cells (18, 19). We have successfully used this strategy to induce alloantigen-specific anergy in two pilot studies in which large doses of alloanergized haploidentical donor T cells were infused together with donor bone marrow into the recipients, resulting in rapid immune reconstitution and less severe GVHD than that of historical control recipients of non–T cell depleted haploidentical bone marrow transplantation (20–22).
One benefit of this alloanergization strategy is preservation of viral- and tumor-associated antigen-specific T cell responses in the transplanted cells, thus enhancing protection from infections and providing anti-tumor immunity despite the induction of hyporesponsiveness in alloreactive effector T cells (23, 24). In addition, alloanergization is a way to generate cell-mediated allosuppression in vitro (25–27). Human allosuppressive cells generated after alloanergization, which have not been well characterized, could be used to prevent or suppress alloreactive T cell responses after allogeneic transplantation.
Previous studies suggest that CD4+ regulatory T (Treg) cells play a role in abrogation of alloresponses after alloanergization (28, 29). Only starting cell populations containing CD4+ Treg cells showed optimal abrogation of murine T cell alloresponses after alloanergization with costimulatory blockade (28). Furthermore, depletion of CD4+ Treg cells from human CD4+ T cell populations before alloanergization with costimulatory blockade reduced the acquisition of allosuppressive capacity, suggesting that such cells may mediate allosuppression in this context (29). We therefore measured the frequency, suppressive function, and specificity of CD4+ Treg cells in peripheral blood from patients who had received alloanergized haploidentical bone marrow transplantation in two previously reported phase I studies (21). To further assess the mechanisms and the potential utility of this strategy for use in HLA-mismatched transplantation, we also examined in vitro the effect of alloanergization and subsequent allorestimulation of donor peripheral blood mononuclear cells (PBMCs) on CD4+ Treg cell frequency, phenotype, and allosuppressive function.
Materials and Methods
Alloanergized bone marrow transplantation
Patients received haploidentical alloanergized bone marrow transplants in an institutional review board (IRB)–approved phase I clinical trial. Four patients with high-risk acute lymphoblastic leukemia and one with amegakaryocytic thrombocytopenia were evaluable for 100 days after transplant. Conditioning regimen, ex vivo alloanergization technique, and supportive care were previously described (21) (Supplementary Methods and fig. S1).
Patient samples
Cryopreserved PBMCs from patients and marrow donors were thawed and resuspended in culture medium with ribonuclease-free deoxyribonuclease (15 U/ml; Roche Diagnostics) for 1 hour. Cells were washed and stained with surface antibodies (Supplementary Methods), fixed, permeabilized, and stained with fluorescein isothiocyanate (FITC)–conjugated rat antibody against human FOXP3 (PCH101; Ebioscience) (44). Cells were acquired on an FC500 flow cytometer (Beckman Coulter) and data were analyzed with FlowJo version 4 software. Hematopoietic chimerism was determined as previously described (20). Donor origin of CD4+ Treg cells was shown by measurement of surface expression of HLA-A2 in patient 2. PBMCs were stained with HLA-A2–FITC (BB7.2; BD Biosciences), CD127-phycoerythrin (PE), CD3–ECD (energy-coupled dye), CD25-PE-Cy5, and CD4-PE-Cy7 antibodies (Beckman Coulter). For flow cytometric cell sorting of patient samples, PBMCs were labeled with CD25-FITC (B1.49.9), CD4-PE-Cy5 (13B8.2), CD8-PE-Cy7 (SFCI21Thy2D3), CD127-PE (HIL-7R- M21) (all Beckman Coulter), and CD3-APC (allophycocyanin) (HIT3a; Becton Dickinson) and sorted into CD3+CD4+CD8–CD25hiCD127lo and CD3+CD4+CD8–CD25–CD127+ cell fractions on a FACSAria high-speed cell sorter (Becton Dickinson).
Alloanergization of PBMCs in vitro
PBMCs from healthy volunteer donors were isolated by Ficoll-Paque density gradient centrifugation (Amersham Biosciences) on an IRB- approved protocol. PBMCs were alloanergized by co-culture of responder PBMCs with γ-irradiated (3.5 Gy) stimulator PBMCs from fully HLA-mismatched unrelated or haploidentical related donors for 72 hours in 70ml flasks (Corning) with 10 mg of humanized B7.1 and B7.2 antibodies (Wyeth) per 106 cells, as described (24). Untreated (control) and alloanergized responder PBMCs were washed and stained with surface antibodies (Supplementary Methods), fixed, permeabilized, and stained with FOXP3-FITC (PCH101; Ebioscience) or FOXP3-PE (259D; Biolegend).
Immunomagnetic cell sorting
For immunomagnetic purification of CD4+ Treg cells, a modification of the strategy described by Godfrey et al. (45) was used, described further in Supplementary Methods. Depletion strategies using immunomagnetic cell sorting are also described in Supplementary Methods.
Suppression assays
A flow diagram of suppression assays used is shown in fig. S2. Washed alloanergized responder PBMCs (or purified CD4+ Treg cells) were added to 105 untreated autologous responder PBMCs in U-bottomed 96-well plates (Nunc) in culture medium (RPMI, penicillin-streptomycin, and 10% human AB serum; Sigma-Aldrich) in a volume of 100 ml and stimulated with equal numbers of first-party (those used in alloanergization) or third-party γ-irradiated PBMCs or with cytomegalovirus-infected human foreskin fibroblast cell lysate (Colorado Health Sciences) and incubated for 5 days. [3H] Thymidine (0.037 MBq; 1 µCi per well; Amersham) was added 18 hours before harvesting with a Tomtec cell harvester (Wallac). Thymidine incorporation was measured by Microbeta liquid scintillation counting (Perkin-Elmer). Triplicate wells were set up for each condition. Percentage suppression was calculated as; 1 0 0 × [ 1 – (proliferation of untreated responders + Treg cells)/proliferation (untreated responders alone)]. Because of the limited numbers of available cells, suppression assays with patient samples were performed with only 5 × 103 donor PBMCs and an equivalent number of γ-irradiated patient pre-transplant PBMCs or HLA-mismatched third-party PBMCs as stimulators in U-bottomed 96-well plates (Nunc). Sorted CD3+CD4+CD8–CD25hiCD127lo Treg cells (0.5 × 103 to 5 × 103) from patients after transplant were added and proliferation was measured as above. The effect of removal of donor-derived CD4+ Treg cells on alloresponses of patient PBMCs after transplant was assessed by coculture of 5 × 103 Treg-depleted CD4+ cells (CD3+CD4+CD8–CD25-CD127+ or 5 × 103 Treg-replete CD4+ cells (obtained by recombining CD3+CD4+CD8–CD25hiCD127lo cells and CD3+CD4+CD8–CD25-CD127+ cells) from patient samples after transplant with 5 × 103 γ-irradiated patient pre-transplant or third-party stimulator PBMCs. Proliferation was measured by [3H] thymidine incorporation at 5 days.
Statistics
Statistical analysis was performed with GraphPad Prism 4 software. A P value of <0.05 was used to reject the null hypothesis.
Results
Expansion of donor CD4+ Treg cells occurs in vivo after alloanergized haploidentical transplantation
A transient increase in the percentage of CD4+ cells expressing intracellular FOXP3 (the product of the forkhead box P3 gene, present in functional Treg cells) occurred in all five evaluable patients, with 10-fold increases above the levels seen in donors, peaking by day 60 after transplantation, with elevated levels sustained beyond day 300 (Fig. 1A). In patients 2 through 5, who reconstituted CD4+ T cells rapidly, absolute numbers of CD4+FOXP3+ cells reached levels above 100 cells/μl of peripheral blood (Fig. 1B). We further characterized the phenotype of CD4+FOXP3+ cells in patients by five-color flow cytometry. CD4+FOXP3+ cells had a CD25hiHLADRdimCD45RO+CTLA4+ phenotype characteristic of antigen-experienced (memory) Treg cells. In addition, ~50% of FOXP3+CD4+ cells accumulated intracellular interleukin-10 (IL-10) as measured by intracellular cytokine flow cytometry (Fig. 1C). More than 90% of FOXP3+CD4+ cells had high expression of CD25 and low expression of the IL-7 receptor subunit α (CD127; Fig. 1D). All recipients of alloanergized transplants had achieved 100% donor chimerism in unsorted PBMCs by day 28. To exclude the possibility that expanded populations of CD4+ Treg cells were of recipient origin, we measured surface expression of HLA class I in HLA-A2– patient 2 transplanted with cells from an HLA-A2+ donor. At day 29 after alloanergized transplantation, 98% of patient peripheral blood CD4+ Treg cells expressed HLA-A2 (Fig. 1E).
Fig. 1.
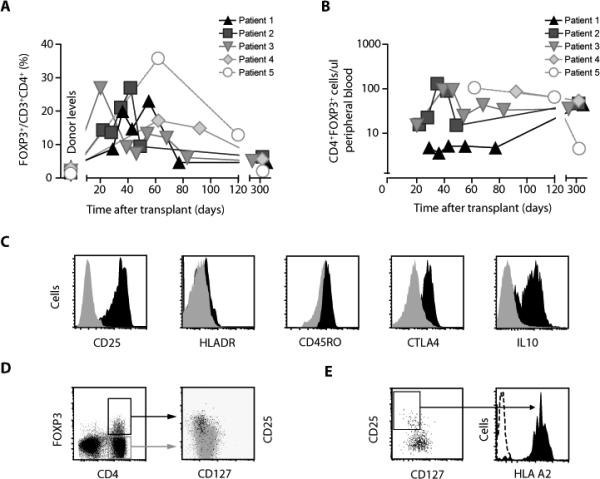
Expansion of donor-derived CD4+ Treg cells after transplantation of bone marrow containing alloanergized haploidentical donor T cells. (A) Frequency of FOXP3+ cells (expressed as percentage of CD3+CD4+ cells) in peripheral blood from patients after alloanergized haploidentical bone marrow transplantation and from their donors. (B) Absolute number of CD4+FOXP3+ cells per microliter of peripheral blood from patients after alloanergized haploidentical transplantation. (C) Cell surface and intracellular markers on FOXP3+and FOXP3–CD4+ cells from peripheral blood of patients after alloanergized haploidentical transplantation. Illustrative histograms gated on CD3+CD4+ cells from patient 2 at day 36 are shown. Black, FOXP3+ cells; gray, FOXP3– cells. (D) Cell surface expression of CD25 and CD127 on CD4+ cells in peripheral blood after alloanergized haploidentical transplantation. Left, dot plot of CD4 and intracellular FOXP3 expression (gated on CD3+ cells); right, dot plot showing expression of CD25 and CD127 on cells from the boxed regions in the left panel (black, FOXP3+ cells; gray, FOXP3– cells) from patient 2 at day 36. (E) Left, dot plot of CD25 and CD127 expression on CD3+CD4+ cells from peripheral blood of an HLA-A2– patient on day 30 after receiving an alloanergized haploidentical transplant from an HLA-A2+ donor; right, histogram showing HLA-A2 expression (black) on cells from the boxed region in the left panel and an isotype control (dotted line).
After transplantation, CD4+ Treg cells suppress recipient-specific alloresponses in vivo
In view of the marked expansion of donor-derived CD4+ Treg cells after alloanergized haploidentical transplantation, we next investigated their allosuppressive function and specificity. Sufficient cells were available for functional analysis from two of the five evaluable patients. The CD25hiCD127lo surface phenotype, which identifies functional human CD4+ Treg cells (30), was used to purify CD4+ Treg cells. Flow cytometric sorting of recipient peripheral blood samples after alloanergized transplantation yielded ~98% pure populations of CD25hiCD127loCD4+ Treg cells and Treg-depleted CD4+ cells with minimal (<0.2%) residual Treg cell content (Fig. 2A). Purified CD4+ Treg cells from recipient blood samples (patient 2 at day 43 and patient 3 at day 42) suppressed recipient-stimulated proliferation of untreated donor PBMCs in a dose-dependent manner but did not reduce proliferation after stimulation with cells obtained from a different unrelated (third-party) individual, showing that the CD4+ Treg cells that expanded in vivo after alloanergized transplantation had acquired a degree of specificity for recipient alloantigens (Fig. 2B). Additionally, we measured alloproliferative responses of cells from these patient samples before and after depletion of CD4+ Treg cells. Depletion of CD4+ Treg cells increased recipient alloantigen-stimulated proliferation when compared to Treg-replete CD4+ cells but had no effect on proliferation stimulated by third-party cells. These data are consistent with suppression of proliferation of T cells responsive to recipient but not third-party antigens by expanded populations of donor-derived CD4+ Treg cells after alloanergized transplantation (Fig. 2C).
Fig. 2.
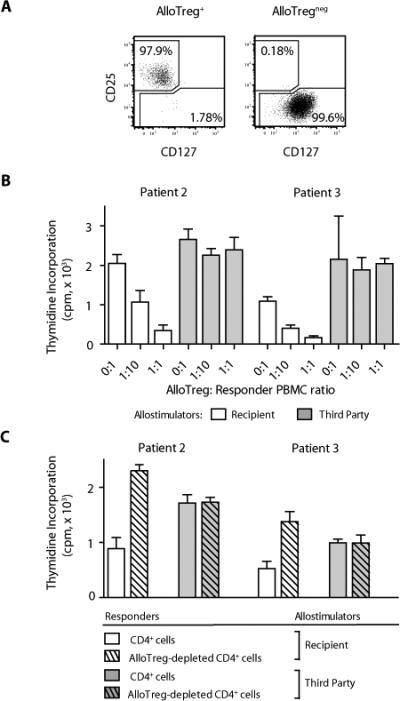
CD4+ Treg cells from recipients of alloanergized transplants suppressed recipient-specific alloresponses. (A) Flow cytometric sorting of patient PBMCs after transplant yielded ~98% pure populations of CD4+CD25hiCD127lo Treg cells (left) and Treg-depleted CD4+ cells with minimal (<0.2%) residual Treg cell content (right). Dot plots are gated on viable CD3+CD4+ cells. (B) Effect of addition of purified Treg cells from patient peripheral blood after transplant on mean (± SD) proliferation (thymidine incorporation) of fresh donor PBMCs stimulated with recipient (patient) or third-party allostimulators. Results are shown for patient 2 (day 43) and patient 3 (day 42). cpm, counts per minute. (C) Mean proliferation (± SD) of Treg-replete and Treg-depleted CD4+ cells from peripheral blood of patients 2 and 3 after transplant stimulated with recipient or third- party allostimulators. Results are shown for patient 2 (day 43) and patient 3 (day 42).
Alloanergization of PBMCs increases the frequency of CD4+ Treg cells in vitro
To more fully understand the in vivo expansion of functional and specific allosuppressive CD4+ Treg cells after alloanergized transplantation, we examined the generation of allosuppressive CD4+ Treg cells from donor PBMCs in vitro using the same strategy employed to alloanergize haploidentical bone marrow. The proportion of CD4+ cells expressing FOXP3 in alloanergized PBMCs (median, 2.7%; range, 1.8 to 4.5%) was significantly higher than the proportion of CD4+ cells expressing FOXP3 in untreated PBMCs cultured for the same period (median, 0.5%; range, 0.3 to 2.2%) (Fig. 3, A and B). Using the PCH101 FOXP3 clone or the alternate FOXP3 clone 259D, we identified similar frequencies of FOXP3+CD4+ cells within alloanergized PBMCs. FOXP3+CD4+ cells in alloanergized PBMCs had a phenotype similar to that of the expanded populations of FOXP3+CD4+ cells we had seen in vivo after alloanergized transplantation (CD25hiHLADRdimCD45RO+CTLA4+), although they did not secrete IL-10. To assess the potential of these cells for migration from peripheral blood to secondary lymphoid organs and tissue targets of GVHD, we also measured the expression of adhesion molecules and chemokine receptors on FOXP3+CD4+ cells. FOXP3+CD4+ cells were CD45RO+CD62L+CCR4+ but were CCR7– (Fig. 3C). FOXP3+CD4+ cells in alloanergized PBMCs also co-expressed high levels of CD25 and low levels of CD127 (Fig. 3D). The proportion of CD4+ cells with CD25hiCD127lo expression was also significantly higher in alloanergized PBMCs (median, 7.6%; range, 3.9 to 13%) than in control cells (median, 4.5%; range, 1.7 to 8.5%; Fig. 3D). Finally, we examined the frequency of CD4+ Treg cells in healthy donor PBMCs before and after alloanergization with haploidentical allostimulators to more closely represent the degree of HLA mismatch in our clinical samples. The proportion of CD4+ cells with CD25hiCD127lo expression was also significantly higher in PBMCs alloanergized with haploidentical allostimulators (median, 6.0%; range, 2.4 to 8.5%) than in control cells (median, 3.4%; range, 1.5 to 4.5%; Fig. 3E).
Fig. 3.
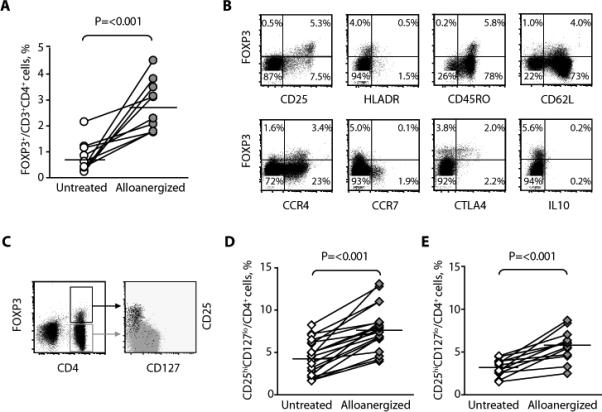
Alloanergization increased the frequency of CD4+ cells with a Treg phenotype in vitro. (A) Frequency of FOXP3+ cells, expressed as percentage of CD3+CD4+ cells in untreated and alloanergized responder PBMCs in 10 HLA-mismatched responder- stimulator pairs. Horizontal lines represent median values. P value is for a two-tailed paired t test. (B) Phenotype of CD4+ Treg cells after alloanergization of PBMCs. Dot plots of CD3+CD4+ cells are from a representative experiment (of four). (C) Cell surface expression of CD25 and CD127 on CD4+ cells in alloanergized responder PBMCs. Left, dot plot of CD4 and intracellular FOXP3 expression (gated on CD3+ cells); right, dot plot showing expression of CD25 and CD127 on cells from the boxed regions in the left panel (black, FOXP3+ cells; gray, FOXP3– cells). One representative experiment (of four) is shown. (D) Frequency of CD25hiCD127lo cells, expressed as percentage of CD3+CD4+ cells in untreated and alloanergized responder PBMCs in 20 HLA- mismatched responder-stimulator pairs. Horizontal lines represent median values. P value is for a two-tailed paired t test. (E) Frequency of CD25hiCD127lo cells, expressed as percentage of CD3+CD4+ cells in untreated and alloanergized responder PBMCs in 12 haploidentical responder-stimulator pairs. Horizontal lines represent median values. P value is for a two-tailed paired t test.
In vitro allorestimulation of alloanergized PBMCs increases CD4+ Treg cells
To determine whether the expansion of CD4+ Treg cells that we had observed in vivo after transplant resulted from re-exposure of alloanergized donor T cells to recipient alloantigens in the absence of costimulatory blockade, we measured the effect of a 7-day in vitro allorestimulation of alloanergized PBMCs. As we and others have reported, PBMCs allostimulated with concomitant costimulatory blockade were hyporesponsive to subsequent allorestimulation (19, 31) with a media 89% reduction in alloproliferation (range, 60 to 97%) in 12 unique HLA-mismatched stimulator-responder pairs. However, allorestimulation of alloanergized PBMCs resulted in a further two- to threefold increase in the proportion of CD4+ cells expressing intracellular FOXP3 to a median frequency of almost 5% (Fig. 4A). A similar incremental increase in the proportion of CD4+ cells with a CD25hiCD127lo phenotype was seen (Fig. 4B). Repeat rounds of in vitro allostimulation led to further increases in both the frequency and the absolute numbers of CD4+ Treg cells (Fig. S3).
Fig. 4.
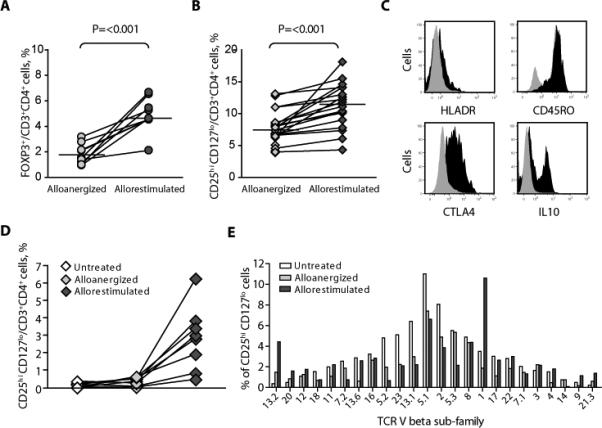
The effect of in vitro allorestimulation of alloanergized PBMCs on the frequency and phenotype of CD4+ Treg cells. (A) Frequency of FOXP3+ cells, expressed as percentage of CD4+ cells, in alloanergized and allorestimulated alloanergized responder PBMCs in nine HLA-mismatched responder-stimulator pairs. Horizontal lines represent median values. P value is for a two-tailed paired t test. (B) Frequency of CD25hiCD127lo cells, expressed as percentage of CD4+ cells in alloanergized and allorestimulated alloanergized responder PBMCs in 22 different HLA- mismatched responder-stimulator pairs. Horizontal lines represent median values. P value is for a two-tailed paired t test. (C) Cell surface and intracellular markers on CD4+ Treg cells after in vitro allorestimulation of alloanergized responder PBMCs. Histograms are from one representative experiment (of six). Black, FOXP3+CD4+ cells; gray, FOXP3–CD4+ cells. (D) Frequency of CD25hiCD127lo cells, expressed as percentage of CD4+ cells in untreated, alloanergized, and allorestimulated alloanergized responder PBMCs in eight HLA-mismatched responder-stimulator pairs after depletion of CD4+ Treg cells from starting populations of untreated PBMCs. (E) Histogram showing TCR Vb subfamily distribution in CD4+CD25hiCD127lo Treg cells before and after alloanergization and allorestimulation with HLA-mismatched stimulators. One representative experiment (of three) is shown.
The phenotype of CD4+ Treg cells that we had observed in vitro in alloanergized PBMCs was maintained after allorestimulation of alloanergized PBMCs, except that ~30% of CD4+ Treg cells in allorestimulated alloanergized PBMCs secreted IL-10, mirroring the phenotype of CD4+ Treg cells we had observed in vivo after alloanergized transplantation (Fig. 4C).
We next sought to determine whether the increased frequencies of CD4+ Treg cells we had observed after alloanergization and allorestimulation were due to expansion of pre-existing CD4+ Treg cells or to the generation of induced CD4+ Treg cells originating from non-Treg cells. Depletion of pre-existing CD4+ Treg cells from starting PBMC populations prevented the increase in frequency of CD4+ Treg cells after alloanergization, consistent with an expansion of pre-existing CD4+ Treg cells. However, allorestimulation of these cells resulted in a significant increase in the proportion of CD4+ Treg cells, suggesting that allorestimulation may also lead to conversion of non-Treg CD4+ cells to Treg cells (Fig. 4D). T cell receptor (TCR) Vβ subfamily distribution was similar in CD4+CD25hiCD127lo Treg cells within untreated and alloanergized PBMCs. However, expanded proportions of individual subfamilies were present in CD4+CD25hiCD127lo Treg cells within allorestimulated alloanergized PBMCs, consistent with alloantigen-driven expansion of these cells (Fig. 4E).
CD4+ Treg cells from alloanergized PBMCs selectively suppress proliferation responses
Using immunomagnetic cell sorting, we isolated CD4+ Treg cells from untreated, alloanergized, or allorestimulated alloanergized PBMCs and investigated their ability to suppress proliferation responses of autologous PBMCs. Isolated populations contained a median of 90% (range, 80 to 94%) CD25hiCD127lo Treg cells and a median of 38% (range, 30 to 48%) CD25hiFOXP3+ cells. Treg cells from each PBMC source suppressed alloresponses of autologous untreated PBMCs. The mean (± SD) proliferation of responder PBMCs to first-party allostimulation without the addition of any isolated Treg cells was 14,085 cpm (± 5895 cpm). This proliferation was reduced by a mean of 71% when equal numbers of CD4+ Treg cells isolated from untreated PBMCs were added to responder PBMCs. However, the addition of CD4+ Treg cells isolated from alloanergized PBMCs or from allorestimulated PBMCs suppressed these proliferative responses more than the addition of Treg cells isolated from untreated PBMCs (Fig. 5A), particularly at lower Treg-to-untreated PBMC ratios.
Fig. 5.
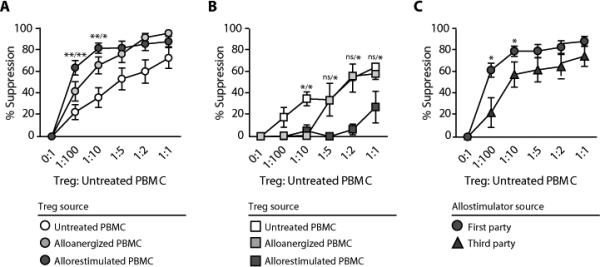
Suppression by CD4+ Treg cells after in vitro alloanergization and allorestimulation of PBMCs. (A) Mean percentage suppression (± SD) of first-party stimulated alloproliferation of untreated autologous responder PBMCs by CD4+ Treg cells from untreated, alloanergized, and allorestimulated alloanergized PBMCs. Data are for 12 HLA-mismatched stimulator- responder pairs. *P < 0.05; **P <0.01 [two-tailed paired t test com- paring suppression of first-party stimulated alloproliferation by CD4+ Treg cells from allorestimulated alloanergized PBMCs with CD4+ Treg cells from untreated (left) or alloanergized (right) PBMCs]. (B) Mean percentage suppression (± SD) of cytomegalovirus- specific proliferation of untreated autologous responder PBMCs by CD4+ Treg cells purified from untreated, alloanergized, and allorestimulated alloanergized PBMCs. Data are for three different HLA-mismatched stimulator- responder pairs. *P < 0.05 [two-tailed paired t test comparing suppression of cytomegalovirus proliferation by CD4+ Treg cells from untreated PBMCs with CD4+ Treg cells from alloanergized (left) or alloanergized allorestimulated (right) PBMCs]. ns, not significant. (C) Mean percentage suppression (± SD) of first- and third-party stimulated alloproliferation of untreated autologous responder PBMCs by CD4+ Treg cells purified from allorestimulated alloanergized PBMCs. Data are for 12 HLA-mismatched stimulator-responder pairs. *P < 0.05 (two-tailed paired t test comparing suppression of first-party stimulated alloproliferation with third-party stimulated alloproliferation).
Moreover, CD4+ Treg cells from allorestimulated alloanergized PBMCs showed the greatest potency in suppressing alloresponses at Treg-to-untreated PBMC ratios below 1:5. The allospecificity of CD4+ Treg cells was examined by evaluating their effect on cytomegalovirus-specific proliferation. CD4+ Treg cells isolated from alloanergized PBMCs suppressed cytomegalovirus-specific responses less than CD4+ Treg cells purified from untreated PBMCs at low but not high ratios, whereas CD4+ Treg cells purified from allorestimulated alloanergized PBMCs suppressed cytomegalovirus-specific responses less than CD4+ Treg cells purified from untreated PBMCs at all ratios (Fig. 5B). This allospecificity was further examined by evaluating the effects of CD4+ Treg cells on proliferative response to the stimulator cells used in the anergization step (first-party) or third-party allostimulators. CD4+ Treg cells from allorestimulated alloanergized PBMCs suppressed alloproliferation to the first-party stimulator significantly more than to a third-party stimulator at Treg-to-responder PBMC ratios lower than 1:5 (Fig. 5C).
Unsorted alloanergized PBMCs also suppress alloresponses
Unsorted alloanergized PBMCs generated in vitro also suppressed alloproliferative responses of untreated autologous responder PBMCs. This suppression was dose-dependent and only significant at high alloanergized-to-untreated PBMC ratios. Unsorted alloanergized PBMCs suppressed first-party stimulated alloresponses more than they suppressed third-party stimulated alloresponses, indicating a degree of specificity for first-party alloantigens. Unsorted alloanergized PBMCs also suppressed first-party stimulated alloresponses of autologous PBMCs previously primed for 3 days with first-party allostimulators (Fig. 6A), indicating that unsorted alloanergized PBMCs could suppress ongoing alloresponses. In contrast, unsorted alloanergized PBMCs did not suppress cytomegalovirus-specific proliferation of untreated autologous responder PBMCs, even at high alloanergized-to-untreated PBMC ratios (Fig. 6B). We then depleted CD4+ or CD25+ cells from PBMCs after alloanergization to determine whether these depleted alloanergized PBMCs had residual suppressive activity. CD4 depletion resulted in a median 100-fold reduction in CD4+ cells. CD25 depletion resulted in a median 300- and 220-fold reduction in CD4+CD25+ and CD4+CD25+FOXP3+ cells to levels below 0.02% and 0.01% of CD4+ cells, respectively. Depletion of either CD4+ or CD25+ cells abrogated suppression except at the highest alloanergized PBMC- to-untreated responder PBMC ratio (Fig. 6C), showing that the majority (but not all) of the suppressive activity resided in the CD4+ and CD25+ cell populations.
Fig. 6.
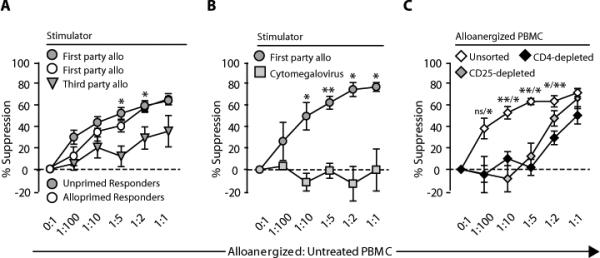
Specific suppression of alloresponses by unsorted alloanergized PBMCs. (A) Mean percentage suppression (± SD) of first- and third-party stimulated alloproliferation of untreated or first-party alloprimed autologous responder PBMCs by alloanergized PBMCs. Data are for 16 HLA-mismatched stimulator-responder pairs. *P <0.05 (two-tailed paired t test com- paring suppression of first- and third-party stimulated alloproliferation). (B) Mean percentage suppression (± SD) of cytomegalovirus lysate–stimulated proliferation and first-party stimulated alloproliferation of untreated autologous responder PBMCs by alloanergized PBMCs. Data are for three HLA-mismatched stimulator-responder pairs. *P < 0.05; **P < 0.01 (two-tailed paired t test comparing suppression of cytomegalovirus- stimulated proliferation and alloproliferation). (C) Mean percentage suppression (± SD) of first-party stimulated alloproliferation of untreated autologous responder PBMCs by unsorted alloanergized PBMC and alloanergized PBMCs depleted of CD4+ or CD25+ cells. Data are for six HLA-mismatched responder- stimulator pairs. *P < 0.05; **P < 0.01 [two-tailed paired t test comparing suppression by alloanergized PBMCs depleted of CD4+ (left) or CD25+ (right) cells with suppression by unsorted alloanergized PBMCs.
Discussion
Alloanergization is a strategy to induce alloantigen-specific tolerance in T cells by exposing them to alloantigens in the presence of costimulatory blockade. This strategy has been successfully used to induce tolerance in human donor T cells within hematopoietic stem cell grafts to reduce GVHD after HLA-mismatched allogeneic bone marrow transplantation. Alloanergization induces tolerance in donor T cells by triggering cellular pathways within alloreactive donor T cells, making them anergic (unable to proliferate in response to restimulation by alloantigens). We show that a second mechanism, the expansion of CD4+ Treg cells that specifically suppress the proliferation of alloreactive donor T cells, also contributes to control of donor T cell alloresponses after alloanergization.
A persistent expansion of donor CD4+ Treg cells occurred in the peripheral blood of patients after alloanergized haploidentical bone marrow transplantation. This expansion occurred in patients whose donor bone marrow had been anergized with either CTLA4- immunoglobulin or the combination of B7.1 and B7.2 antibodies to block costimulation. These cells specifically suppressed donor T cell responses to recipient alloantigens. To determine whether the in vivo expansion of these allospecific CD4+ Treg cells resulted from the re-exposure of alloanergized donor T cells to recipient alloantigens after transplantation, we modeled this re-exposure in vitro. We characterized CD4+ Treg cells within blood cells from normal volunteers before and after alloanergization and again after re-exposure to alloantigen. Alloanergization doubled the frequency of CD4+ Treg cells. Re-exposure to first-party alloantigen resulted in a further increase in the frequency of CD4+ Treg cells and also increased their potency and specificity. The selective suppression of alloreactive T cell responses, mediating both GVHD after hematopoietic stem cell transplantation and graft rejection after solid organ transplantation, by alloantigen-specific CD4+ Treg cells generated after in vitro alloanergization could avoid the off-target effects of less specific approaches to immunosuppression.
In our previous studies of alloanergized haploidentical transplantation, we observed a lower than expected incidence of both acute and chronic GVHD even though the patients received large doses of donor T cells. Moreover, these patients showed a low incidence of viral infection (21), consistent with our previous work showing that alloanergization did not significantly reduce pathogen-specific T cell immune responses (24). These results suggested to us that alloanergization functionally inactivated alloreactive T cells while sparing responses in non-alloreactive T cells, making this approach to control alloreactivity after transplant comparable to other methods that selectively destroy or deplete alloreactive T cells within the donor T cell pool. Our in vitro data showing that alloanergization and re-exposure to alloantigen generate alloantigen-specific Treg cells suggest that this strategy also provides an additional active mechanism to limit alloreactivity after transplantation of alloanergized donor T cells. Previous murine and human in vitro studies have shown that alloanergization can generate suppressive cells (25, 26); our data show that these cells can persist, expand, and functionally suppress alloresponses in vivo after transplantation of alloanergized donor T cells in humans.
Our in vitro studies provided further insight into the phenotype and function of CD4+ Treg cells after alloanergization. First, their CD25hiCD127lo phenotype showed that these cells are CD4+ Treg cells rather than recently activated effector T cells that have increased expression of FOXP3 (32–34). Further, in addition to a CD45RO+ memory cell phenotype, these CD4+ Treg cells preferentially expressed CD62L, which is associated with homing to lymph nodes and suppression of lethal murine GVHD (35, 36), and the chemokine receptor CCR4, which is expressed on human CD4+ Treg cells primed for effective suppression of CD8+ T cell responses in vitro (37). Allorestimulation resulted in two important functional changes. Whereas IL-10 was not secreted by CD4+ Treg cells after in vitro alloanergization, one-third of these cells secreted IL-10 after allorestimulation, as observed in CD4+ Treg cells isolated from our patients. Activated or allostimulated human CD4+FOXP3+ Treg cells can secrete IL-10 (38, 39). IL-10 secretion by CD4+ Treg cells, which protects against GVHD in a xenogeneic murine model (40), could also contribute to limiting alloreactivity in vivo. Allorestimulation in vitro also increased both the potency and the specificity of allosuppression mediated by CD4+ Treg cells. These cells had no suppressive effects on proliferation after stimulation with cytomegalovirus, a common and clinically important viral pathogen after transplantation. This suggests that the allosuppressive potency and specificity of CD4+ Treg cells within alloanergized donor T cells increase after re-exposure to alloantigen.
CD4+ Treg cell infusion can reduce the rate of GVHD after murine stem cell transplantation (41). The role of CD4+ Treg cells after clinical transplantation has not been clearly defined, although several studies have reported increased peripheral blood CD4+ Treg cells after allogeneic hematopoietic stem cell transplantation in patients who do not develop severe acute or chronic GVHD (42, 43). By 3 months after transplant, all the patients we studied had CD4+FOXP3+ cell numbers at least double the number (four cells per microliter of peripheral blood) associated with the absence of severe acute GVHD after T cell–depleted, HLA-matched allogeneic transplantation (42). Four patients reached and sustained levels of 80 or more CD4+FOXP3+ cells per microliter of peripheral blood. In the patients we describe, acute GVHD was limited to the gut (patients 1, 3, and 4) and resolved completely with brief courses of corticosteroids, although these patients received large doses of haploidentical donor T cells. None of these patients developed chronic GVHD, consistent with the low incidence we have reported in a larger cohort of patients receiving alloanergized haploidentical transplants (21). Although no firm conclusions can be drawn from such a small number of patients, the ability to confer protection against GVHD may depend critically on Treg cell antigen specificity as well as on Treg cell frequency.
In aggregate, these data suggest that alloanergization of donor T cells serves to limit alloresponses after transplantation by suppression of recipient-specific immune responses in addition to directly rendering donor T cells hyporesponsive to the recipient's alloantigens. This Treg cell–mediated suppression process, which may be both amplified and sustained by continuous allorestimulation of donor T cells by recipient alloantigens in vivo, may serve as a fail-safe mechanism preventing expansion of alloreactive donor cells that escape alloanergization or undergo cytokine-mediated reversal of anergy. Furthermore, the high degree of antigen specificity of CD4+ Treg cells generated by this process suggests that this approach could be used to generate non-alloreactive donor T cells enriched with allospecific Treg cells for use as targeted cell therapy for the prophylaxis or treatment of acute GVHD. In addition, the in vivo generation of antigen-specific CD4+ Treg cells after in vitro anergization of patient T cells has therapeutic potential in a wide range of settings outside allogeneic hematopoietic stem cell transplantation. These include the prevention or treatment of rejection after solid organ transplantation and suppression of autoantigen-specific immune responses in patients with autoimmune diseases.
Supplementary Material
Acknowledgments
We thank L. Brennan for collating patient information and D. Yuk for performing suppression assays.
Funding: Supported by NIH grants U19 CA100265 and R21 CA137645, Leukemia & Lymphoma Society (J.K.D.), and the American Society of Blood and Marrow Transplantation–Otsuka New Investigator Award (J.K.D.).
Footnotes
Author contributions: All authors analyzed data and wrote the manuscript. J.K.D. and E.C.G. contributed to the experimental design. J.K.D. conceived and performed laboratory experiments. E.C.G. conceived and conducted the clinical trial. L.M.N. developed and provided critical reagents.
Competing interests: The authors declare no financial conflicts of interest.
References and Notes
- 1.Oudshoorn M, Lie J, Bakker J, Van Der Zanden H, Claas F. In: Clinical Bone Marrow and Blood Stem Cell Transplantation. Atkinson K, editor. Cambridge University Press; Cambridge: 2004. pp. 369–382. [Google Scholar]
- 2.Kernan NA, Bartsch G, Ash RC, Beatty PG, Champlin R, Filipovich A, Gajewski J, Hansen JA, Henslee-Downey J, McCullough J, McGlave P, Perkins HA, Phillips GL, Sanders J, Stroncek D, Thomas ED, Blume KG. Analysis of 462 transplantations from unrelated donors facilitated by the National Marrow Donor Program. N. Engl. J. Med. 1993;328:593–602. doi: 10.1056/NEJM199303043280901. [DOI] [PubMed] [Google Scholar]
- 3.Kollman C, Abella E, Baitty RL, Beatty PG, Chakraborty R, Christiansen CL, Hartzman RJ, Hurley CK, Milford E, Nyman JA, Smith TJ, Switzer GE, Wada RK, Setterholm M. Assessment of optimal size and composition of the U.S. National Registry of hematopoietic stem cell donors. Transplantation. 2004;78:89–95. doi: 10.1097/01.tp.0000132327.40702.97. [DOI] [PubMed] [Google Scholar]
- 4.Confer DL. The National Marrow Donor Program. Meeting the needs of the medically underserved. Cancer. 2001;91:274–278. doi: 10.1002/1097-0142(20010101)91:1+<274::aid-cncr18>3.0.co;2-e. [DOI] [PubMed] [Google Scholar]
- 5.Ballen KK, Hicks J, Dharan B, Ambruso D, Anderson K, Bianco C, Bemiller L, Dickey W, Lottenberg R, O'Neill M, Popovsky M, Skerrett D, Sniecinski I, Wingard JR. Racial and ethnic composition of volunteer cord blood donors: Comparison with volunteer unrelated marrow donors. Transfusion. 2002;42:1279–1284. doi: 10.1046/j.1537-2995.2002.00191.x. [DOI] [PubMed] [Google Scholar]
- 6.Spitzer TR. Haploidentical stem cell transplantation: The always present but overlooked donor. Hematology Am. Soc. Hematol. Educ. Program. 2005:390–395. doi: 10.1182/asheducation-2005.1.390. [DOI] [PubMed] [Google Scholar]
- 7.Szydlo R, Goldman JM, Klein JP, Gale RP, Ash RC, Bach FH, Bradley BA, Casper JT, Flomenberg N, Gajewski JL, Gluckman E, Henslee-Downey PJ, Hows JM, Jacobsen N, Kolb HJ, Lowenberg B, Masaoka T, Rowlings PA, Sondel PM, van Bekkum DW, van Rood JJ, Vowels MR, Zhang MJ, Horowitz MM. Results of allogeneic bone marrow transplants for leukemia using donors other than HLA-identical siblings. J. Clin. Oncol. 1997;15:1767–1777. doi: 10.1200/JCO.1997.15.5.1767. [DOI] [PubMed] [Google Scholar]
- 8.Munn RK, Henslee-Downey PJ, Romond EH, Marciniak EJ, Fleming DR, Messino MJ, Macdonald JS, Rayens MK, Harder EJ, Phillips GL, Thompson JS. Treatment of leukemia with partially matched related bone marrow transplantation. Bone Marrow Transplant. 1997;19:421–427. doi: 10.1038/sj.bmt.1700681. [DOI] [PubMed] [Google Scholar]
- 9.Aversa F, Tabilio A, Velardi A, Cunningham I, Terenzi A, Falzetti F, Ruggeri L, Barbabietola G, Aristei C, Latini P, Reisner Y, Martelli MF, Felicini R, Falcinelli F, Carotti A, Perruccio K, Ballanti S, Santucci A, Gambelunghe C. Treatment of high-risk acute leukemia with T-cell- depleted stem cells from related donors with one fully mismatched HLA haplotype. N. Engl. J. Med. 1998;339:1186–1193. doi: 10.1056/NEJM199810223391702. [DOI] [PubMed] [Google Scholar]
- 10.Keever CA, Small TN, Flomenberg N, Heller G, Pekle K, Black P, Pecora A, Gillio A, Kernan NA, O'Reilly RJ. Immune reconstitution following bone marrow transplantation: Comparison of recipients of T-cell depleted marrow with recipients of conventional marrow grafts. Blood. 1989;73:1340–1350. [PubMed] [Google Scholar]
- 11.Horowitz MM, Gale RP, Sondel PM, Goldman JM, Kersey J, Kolb HJ, Rimm AA, Ringdén O, Rozman C, Speck B, Truitt RL, Zwaan FE, Bortin MM. Graft-versus-leukemia reactions after bone marrow transplantation. Blood. 1990;75:555–562. [PubMed] [Google Scholar]
- 12.Henslee PJ, Thompson JS, Romond EH, Doukas MA, Metcalfe M, Marshall ME, MacDonald JS. T cell depletion of HLA and haploidentical marrow reduces graft-versus-host disease but it may impair a graft-versus-leukemia effect. Transplant. Proc. 1987;19:2701–2706. [PubMed] [Google Scholar]
- 13.Cavazzana-Calvo M, Fromont C, Le Deist F, Lusardi M, Coulombel L, Derocq JM, Gerota I, Griscelli C, Fischer A. Specific elimination of alloreactive T cells by an anti-interleukin-2 receptor B chain-specific immunotoxin. Transplantation. 1990;50:1–7. doi: 10.1097/00007890-199007000-00001. [DOI] [PubMed] [Google Scholar]
- 14.Davies JK, Koh MB, Lowdell MW. Antiviral immunity and T-regulatory cell function are retained after selective alloreactive T-cell depletion in both the HLA-identical and HLA- mismatched settings. Biol. Blood Marrow Transplant. 2004;10:259–268. doi: 10.1016/j.bbmt.2003.12.001. [DOI] [PubMed] [Google Scholar]
- 15.Amrolia PJ, Muccioli-Casadei G, Yvon E, Huls H, Sili U, Wieder ED, Bollard C, Michalek J, Ghetie V, Heslop HE, Molldrem JJ, Rooney CM, Schlinder J, Vitetta E, Brenner MK. Selective depletion of donor alloreactive T cells without loss of antiviral or antileukemic responses. Blood. 2003;102:2292–2299. doi: 10.1182/blood-2002-11-3516. [DOI] [PubMed] [Google Scholar]
- 16.Mielke S, Nunes R, Rezvani K, Fellowes VS, Venne A, Solomon SR, Fan Y, Gostick E, Price DA, Scotto C, Read EJ, Barrett AJ. A clinical-scale selective allodepletion approach for the treatment of HLA-mismatched and matched donor-recipient pairs using expanded T lymphocytes as antigen-presenting cells and a TH9402-based photodepletion technique. Blood. 2008;111:4392–4402. doi: 10.1182/blood-2007-08-104471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.June CH, Bluestone JA, Nadler LM, Thompson CB. The B7 and CD28 receptor families. Immunol. Today. 1994;15:321–331. doi: 10.1016/0167-5699(94)90080-9. [DOI] [PubMed] [Google Scholar]
- 18.Gimmi CD, Freeman GJ, Gribben JG, Gray G, Nadler LM. Human T-cell clonal anergy is induced by antigen presentation in the absence of B7 costimulation. Proc. Natl. Acad. Sci. U.S.A. 1993;90:6586–6590. doi: 10.1073/pnas.90.14.6586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tan P, Anasetti C, Hansen JA, Melrose J, Brunvand M, Bradshaw J, Ledbetter JA, Linsley PS. Induction of alloantigen-specific hyporesponsiveness in human T lymphocytes by blocking interaction of CD28 with its natural ligand B7/BB1. J. Exp. Med. 1993;177:165–173. doi: 10.1084/jem.177.1.165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Guinan EC, Boussiotis VA, Neuberg D, Brennan LL, Hirano N, Nadler LM, Gribben JG. Transplantation of anergic histoincompatible bone marrow allografts. N. Engl. J. Med. 1999;340:1704–1714. doi: 10.1056/NEJM199906033402202. [DOI] [PubMed] [Google Scholar]
- 21.Davies JK, Gribben JG, Brennan LL, Yuk D, Nadler LM, Guinan EC. Outcome of alloanergized haploidentical bone marrow transplantation after ex vivo costimulatory blockade: Results of 2 phase 1 studies. Blood. 2008;112:2232–2241. doi: 10.1182/blood-2008-03-143636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Powles RL, Morgenstern GR, Kay HE, McElwain TJ, Clink HM, Dady PJ, Barrett A, Jameson B, Depledge MH, Watson JG, Sloane J, Leigh M, Lumley H, Hedley D, Lawler SD, Filshie J, Robinson B. Mismatched family donors for bone-marrow transplantation as treat- ment for acute leukaemia. Lancet. 1983;1:612–615. doi: 10.1016/s0140-6736(83)91793-2. [DOI] [PubMed] [Google Scholar]
- 23.Comoli P, Locatelli F, Moretta A, Montagna D, Calcaterra V, Cometa A, Basso S, Zecca M, Maccario R. Human alloantigen-specific anergic cells induced by a combination of CTLA4- Ig and CsA maintain anti-leukemia and anti-viral cytotoxic responses. Bone Marrow Trans- plant. 2001;27:1263–1273. doi: 10.1038/sj.bmt.1703063. [DOI] [PubMed] [Google Scholar]
- 24.Davies JK, Yuk D, Nadler LM, Guinan EC. Induction of alloanergy in human donor T cells without loss of pathogen or tumor immunity. Transplantation. 2008;86:854–864. doi: 10.1097/TP.0b013e3181861b6c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Taylor PA, Friedman TM, Korngold R, Noelle RJ, Blazar BR. Tolerance induction of alloreactive T cells via ex vivo blockade of the CD40:CD40L costimulatory pathway results in the generation of a potent immune regulatory cell. Blood. 2002;99:4601–4609. doi: 10.1182/blood.v99.12.4601. [DOI] [PubMed] [Google Scholar]
- 26.Koenen HJ, Joosten I. Blockade of CD86 and CD40 induces alloantigen-specific immu- noregulatory T cells that remain anergic even after reversal of hyporesponsiveness. Blood. 2000;95:3153–3161. [PubMed] [Google Scholar]
- 27.Vermeiren J, Ceuppens JL, Van Ghelue M, Witters P, Bullens D, Mages HW, Kroczek RA, Van Gool SW. Human T cell activation by costimulatory signal-deficient allogeneic cells in- duces inducible costimulator-expressing anergic T cells with regulatory cell activity. J. Immunol. 2004;172:5371–5378. doi: 10.4049/jimmunol.172.9.5371. [DOI] [PubMed] [Google Scholar]
- 28.Taylor PA, Noelle RJ, Blazar BR. CD4+CD25+ immune regulatory cells are required for in- duction of tolerance to alloantigen via costimulatory blockade. J. Exp. Med. 2001;193:1311–1318. doi: 10.1084/jem.193.11.1311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Koenen HJ, Fasse E, Joosten I. IL-15 and cognate antigen successfully expand de novo- induced human antigen-specific regulatory CD4+ T cells that require antigen-specific ac- tivation for suppression. J. Immunol. 2003;171:6431–6441. doi: 10.4049/jimmunol.171.12.6431. [DOI] [PubMed] [Google Scholar]
- 30.Hartigan-O'Connor DJ, Poon C, Sinclair E, McCune JM. Human CD4+ regulatory T cells express lower levels of the IL-7 receptor alpha chain (CD127), allowing consistent identi- fication and sorting of live cells. J. Immunol. Methods. 2007;319:41–52. doi: 10.1016/j.jim.2006.10.008. [DOI] [PubMed] [Google Scholar]
- 31.Gribben JG, Guinan EC, Boussiotis VA, Ke XY, Linsley L, Sieff C, Gray GS, Freeman GJ, Nadler LM. Complete blockade of B7 family-mediated costimulation is necessary to induce human alloantigen-specific anergy: A method to ameliorate graft-versus-host disease and extend the donor pool. Blood. 1996;87:4887–4893. [PubMed] [Google Scholar]
- 32.Walker MR, Carson BD, Nepom GT, Ziegler SF, Buckner JH. De novo generation of antigen-specific CD4+CD25+ regulatory T cells from human CD4+CD25– cells. Proc. Natl. Acad. Sci. U.S.A. 2005;102:4103–4108. doi: 10.1073/pnas.0407691102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wang J, Ioan-Facsinay A, van der Voort EI, Huizinga TW, Toes RE. Transient expression of FOXP3 in human activated nonregulatory CD4+ T cells. Eur. J. Immunol. 2007;37:129–138. doi: 10.1002/eji.200636435. [DOI] [PubMed] [Google Scholar]
- 34.Seddiki N, Santner-Nanan B, Martinson J, Zaunders J, Sasson S, Landay A, Solomon M, Selby W, Alexander SI, Nanan R, Kelleher A, Fazekas de St Groth B. Expression of inter- leukin (IL)-2 and IL-7 receptors discriminates between human regulatory and activated T cells. J. Exp. Med. 2006;203:1693–1700. doi: 10.1084/jem.20060468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sallusto F, Lenig D, Förster R, Lipp M, Lanzavecchia A. Two subsets of memory T lympho- cytes with distinct homing potentials and effector functions. Nature. 1999;401:708–712. doi: 10.1038/44385. [DOI] [PubMed] [Google Scholar]
- 36.Ermann J, Hoffmann P, Edinger M, Dutt S, Blankenberg FG, Higgins JP, Negrin RS, Fathman CG, Strober S. Only the CD62L+ subpopulation of CD4+CD25+ regulatory T cells protects from lethal acute GVHD. Blood. 2005;105:2220–2226. doi: 10.1182/blood-2004-05-2044. [DOI] [PubMed] [Google Scholar]
- 37.Baatar D, Olkhanud P, Sumitomo K, Taub D, Gress R, Biragyn A. Human peripheral blood T regulatory cells (Tregs), functionally primed CCR4+ Tregs and unprimed CCR4– Tregs, regulate effector T cells using FasL. J. Immunol. 2007;178:4891–4900. doi: 10.4049/jimmunol.178.8.4891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tiemessen MM, Jagger AL, Evans HG, van Herwijnen MJ, John S, Taams LS. CD4+CD25+Foxp3+ regulatory T cells induce alternative activation of human monocytes/ macrophages. Proc. Natl. Acad. Sci. U.S.A. 2007;104:19446–19451. doi: 10.1073/pnas.0706832104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Dieckmann D, Plottner H, Berchtold S, Berger T, Schuler G. Ex vivo isolation and character- ization of CD4+CD25+ T cells with regulatory properties from human blood. J. Exp. Med. 2001;193:1303–1310. doi: 10.1084/jem.193.11.1303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Mutis T, van Rijn RS, Simonetti ER, Aarts-Riemens T, Emmelot ME, van Bloois L, Martens A, Verdonck LF, Ebeling SB. Human regulatory T cells control xenogeneic graft- versus-host disease induced by autologous T cells in RAG2–/–gc–/– immunodeficient mice. Clin. Cancer Res. 2006;12:5520–5525. doi: 10.1158/1078-0432.CCR-06-0035. [DOI] [PubMed] [Google Scholar]
- 41.Taylor PA, Lees CJ, Blazar BR. The infusion of ex vivo activated and expanded CD4+CD25+ immune regulatory cells inhibits graft-versus-host disease lethality. Blood. 2002;99:3493–3499. doi: 10.1182/blood.v99.10.3493. [DOI] [PubMed] [Google Scholar]
- 42.Rezvani K, Mielke S, Ahmadzadeh M, Kilical Y, Savani BN, Zeilah J, Keyvanfar K, Montero A, Hensel N, Kurlander R, Barrett AJ. High donor FOXP3-positive regulatory T-cell (Treg) con- tent is associated with a low risk of GVHD following HLA-matched allogeneic SCT. Blood. 2006;108:1291–1297. doi: 10.1182/blood-2006-02-003996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zorn E, Kim HT, Lee SJ, Floyd BH, Litsa D, Arumugarajah S, Bellucci R, Alyea EP, Antin JH, Soiffer RJ, Ritz J. Reduced frequency of FOXP3+ CD4+CD25+ regulatory T cells in patients with chronic graft-versus-host disease. Blood. 2005;106:2903–2911. doi: 10.1182/blood-2005-03-1257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Jung T, Schauer U, Heusser C, Neumann C, Rieger C. Detection of intracellular cytokines by flow cytometry. J. Immunol. Methods. 1993;159:197–207. doi: 10.1016/0022-1759(93)90158-4. [DOI] [PubMed] [Google Scholar]
- 45.Godfrey WR, Ge YG, Spoden DJ, Levine BL, June CH, Blazar BR, Porter SB. In vitro-expanded human CD4+CD25+ T-regulatory cells can markedly inhibit allogeneic dendritic cell-stimulated MLR cultures. Blood. 2004;104:453–461. doi: 10.1182/blood-2004-01-0151. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


