Abstract
Light-induced fluorescence (LIF) was evaluated as a process analytical technology to monitor blend homogeneity and establish a relationship with high-performance liquid chromatography (HPLC). Secondary aims for this study included a determination of blend steady-state, acceptable mixing time interval, and mixing end point. Also, identification of potential “dead spots” in the 124 L intermediate bulk container mixing tote was explored. Individual samples from 13 sample locations were collected at 0.25, 0.5, 0.75, 1, 2, 5, 10, and 20 min and analyzed using LIF and HPLC. LIF and HPLC methods showed similar mixing profiles. A coefficient of determination (R2) of 0.86 (p value < 0.0001) was obtained for a second-degree polynomial bivariate fit of LIF counts by HPLC percent label claim (%LC). A significant linear relationship was determined between LIF percent relative standard (%RSD) and HPLC %RSD (R2 = 0.97, p < 0.0001). The LIF steady-state, acceptable mixing time interval, and mixing end point were determined to be 1–20, 2–20, and 2 min, respectively. The steady-state, acceptable mixing time interval, and mixing end point determined by HPLC were 1–20, 5–10, and 5 min, respectively. The Tukey–Kramer honestly significant difference analysis of HPLC %LC by sample location at 5 and 10 min mixing times showed that there was a statistical difference between the HPLC %LC group means at two blender locations.
KEY WORDS: blend homogeneity, light-induced fluorescence, powder mixing, powder mixing end point, process analytical technology
INTRODUCTION
Powder mixing is a common operation in the manufacture of pharmaceuticals. The homogeneity of the final blend depends on many parameters such as the principle operating design, active pharmaceutical ingredient (API) concentration, blender charging sequence, fill level, mixing time, mixing speed, and scale of blender. The tendency of a powder mixture to form a uniform blend or to segregate is dependent on its physical properties such as cohesiveness, particle size, shape, density, and other surface properties.
Most substances that absorb ultraviolet or visible light energy dissipate the excess energy as heat through collisions with neighboring atoms or molecules. However, some substances lose only part of the excess energy as heat and emit the remaining energy as electromagnetic radiation. This process is called fluorescence. Fluorescent analytical methods can have significant advantages over adsorption spectroscopy. Fluorescence has the advantage of using two wavelengths, an excitation and emission wavelength, which increases the specificity of the method. Also, it is reported that the sensitivity of fluorescence is approximately 100–1,000 times greater than absorption spectrophotometric methods (1). These advantages are further enhanced by patented technology within the sensor used in this study, where the emitted photon is measured at right angles to the excitation wavelength (internal dichroic mirror) which results in a high signal-to-noise ratio.
A major disadvantage of fluorescence is the sensitivity of fluorescence intensity to fluctuations in pH and temperature (2). Fluorometry also exhibits self-quenching at high concentrations. The emission intensity actually decreases with increasing fluorophore concentration because the absorption increases more rapidly than the emission (3). Lastly, as the concentration of the fluorophore increases, saturation of the detector can occur, limiting the linear response range of the detector.
Currently, there is a great deal of interest in applying process analytical technology (PAT) for improving the understanding of pharmaceutical operations as well as for monitoring and controlling them. Inline monitoring of blending requires fast, accurate, reliable, precise and sensitive techniques that require no sample preparation. The definition of PAT according to the Food and Drug Administration (FDA) is: “A system for designing, analyzing, and controlling manufacturing through timely measurements of critical quality and performance attributes of raw and in-process materials and processes with the goal of ensuring final product quality” (4). LIF has been evaluated as a potential PAT tool for the evaluation of content blend uniformity, monitoring the blending of powders, and measuring the content uniformity of tablets (5–7).
The primary aim of this study was to evaluate LIF as a PAT tool to assess blend homogeneity as a function of time for a validated commercial product and establish a relationship between LIF and high-pressure liquid chromatography (HPLC) as the standard analytical method. Secondary aims for this study included a determination of blend steady-state, acceptable mixing time interval, and mixing end point. Also, identification of potential “dead-spots” in the 124 L intermediate bulk container (IBC) mixing tote was explored. Earlier blend studies for this product had suggested that blend homogeneity could be achieved within the first 2 min of the validated 20-min blending process. Therefore, this research focused primarily on the first 10 min of the blending process with a possible opportunity to identify an earlier mixing end point and thereby significantly reduce blend times, decrease production cycle times, and increase material throughputs.
MATERIALS AND METHODS
Materials and Equipment
Fluorescent API granules were obtained from GlaxoSmithKline. Spectralon fluorescence reference standards were used to assess LIF instrument performance. Acetonitrile (HPLC grade), acetic acid (HPLC grade), and water (reagent grade or equivalent) were used for HPLC testing.
All blending was completed at GSK facilities using a Metolift post blender and a 124 L intermediate bulk container (IBC). Additional laboratory test equipment included Waters HPLC, Waters 600 controller, Waters 717 auto sampler, Waters 486 tunable absorbance detector, centrifuge, and ultrasonic bath.
Fluorescence detection was accomplished using a Honeywell purpose built LIF sensor with a xenon strobe light source and photomultiplier tube style detector. The strobe had an adjustable energy range of 100–180 mJ at 1–20 Hz. The wavelength range was 190–650 nm and was configured with excitation and emission filters appropriate to the API.
Methods
LIF Method
A preblend of two separate API granulations were mixed in a ratio of 25:75 in a 124 L IBC tote blender. The nonfluorescent API granulation was charged into the IBC first and the fluorescent API granulation was layered on top. The excitation wavelength of the fluorescent API was 330–360 nm and the emission wavelength was 325–440 nm. The LIF probe was placed on the surface of the collected samples to get the fluorescent readings. The LIF sensitivity was set at 42. Data from the LIF sensor was analyzed and compared with HPLC assay data. The LIF unit emitted a spot size of 10 mm. Penetration depth is variable dependent on the excitation wavelength used, particle size, and packing density and was nominally determined to be approximately 1,500 μm (5). The spot size and penetration gives a sample volume of approximately 0.117 cm3 which for a nominal bulk density of 0.6 g/cm3 yields a sample weight of 70.6 mg when using LIF.
Sampling
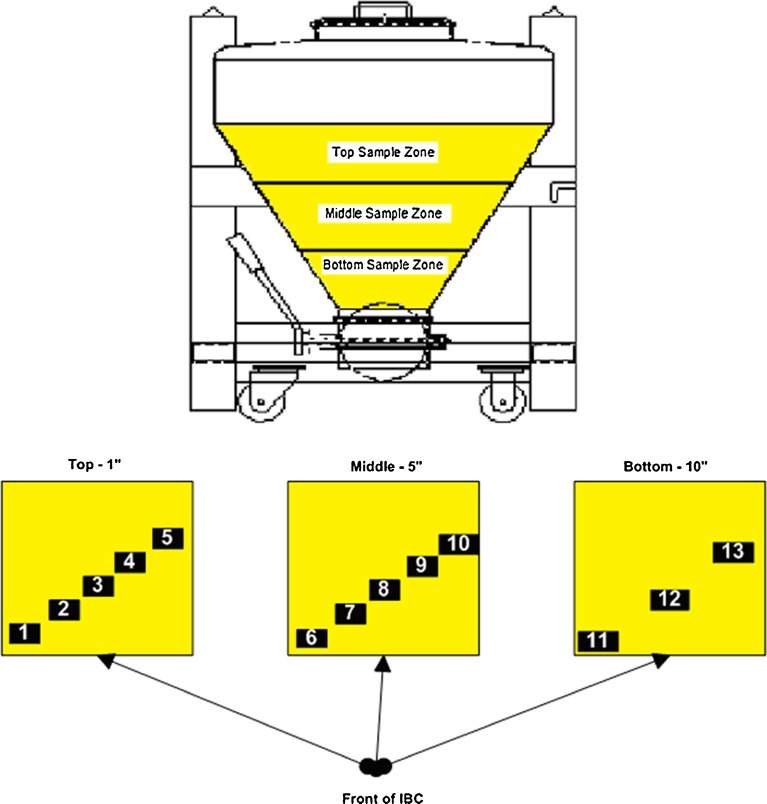
The preblend granulation was blended at 16 rpm and samples were collected at rotations 4, 8, 12, and 16 (0.25, 0.5, 0.75, and 1 min) for the first minute and then at 2, 5, 10, and 20 min. Samples were collected from 13 sample locations of the 124 L IBC as indicated in Fig. 1. Each sample weighed approximately 80–240 mg which is one to three times the target weight of the intended API content of the final tablet. The samples were collected using a stainless steel grain type sampling thief and stored in appropriate labeled containers. Five LIF measurements were made on each sample. Once the LIF measurements were made, the total contents of the sample container were used for HLPC analysis.
Fig. 1.
Preblend sampling zones inside IBC
Calibration of the LIF Sensor
The LIF sensor was calibrated using fluorescence reference standard (8,9). The LIF response for the reflectance standard, baseline, and seven fluorescence standards were noted at different arbitrary gain sensitivity percents (42, 44, 46, 48, and 50). The sensitivity is the amount of gain applied to the photomultiplier tube and helps to improve the sensitivity of the measurement. However, as one increases the gain, both the signal and noise are increased, so optimization of the gain for these experiments was necessary to acquire optimal data. The LIF response was plotted against fluorescence standards at a sensitivity setting of 42. This sensitivity setting had an R2 = 0.98 and was chosen for analysis of all samples.
HPLC Blend Content Assay
The total contents of the sample bottle, approximately 80–240 mg of granules were weighed and transferred into the 100 mL amber volumetric flask. Approximately 75 mL of mobile phase was added to the flask and sonicated for 15 min. The solution was equilibrated to room temperature, diluted to 100 mL, and mixed thoroughly. An aliquot of the sample solution was centrifuged for 5 min at about 13,000 rpm. A portion of the supernatant was transferred to vials and analyzed by HPLC. The final results of the HPLC analysis were normalized per the individual weights of each sample.
Data Analysis
Blending data obtained from LIF and HPLC results were analyzed using John Sall’s Mac Program by SAS (10). LIF counts were compared to HPLC percent label claim (%LC). Blend homogeneity was further investigated using Bartlett’s test (11) to assess the sample location variability.
Blend uniformity was assessed using tiered criteria similar to that suggested in the FDA guidance for process unit dose batch homogeneity (12). For purposes of this study, blend homogeneity for a given blend time was classified as Readily Pass when the percent relative standard (%RSD) for all 13 locations was ≤4%. Similarly, blending times having a %RSD ≤ 6% were classified as Marginally Pass. Steady-state was defined as the mixing interval where the group means by time were statistically equivalent. An acceptable mixing time interval was the time interval when steady-state mixing existed and the %RSD was classified as Readily Pass (%RSD ≤ 4%). The mixing end point was chosen as the first time point that met the acceptable mixing time interval criteria.
RESULTS AND DISCUSSION
In accordance with the aims of the study, the establishment of a relationship between LIF counts and HPLC %LC was addressed qualitatively with a comparison of data from the blend mixing profiles. Statistical comparisons of LIF and HPLC data provided quantitative relationships between the two analytical technologies. Blend homogeneity as a function of mixing time was explored and followed by the identification of potential mixing tote dead spots.
Relationship of LIF and HPLC Technologies
LIF Count Results for Mixing Profiles
Five LIF readings were recorded for each sample and sample means, standard deviation (SD), and %RSD were calculated and are summarized in Table I. LIF count results from Table I demonstrate that from 2 to 20 min the blend meets the %RSD ≤ 4% criterion and suggests that the blend is homogeneous during that blending interval. The 1 min time point meets the Marginally Pass criterion. The earlier time points do not meet the Marginally Pass criterion because they were taken during the mixing phase which is discussed in the “Similarity of Mixing Profiles” section.
Table I.
LIF Counts for the Preblend Samples
| Time (min) | 0.25 | 0.5 | 0.75 | 1 | 2 | 5 | 10 | 20 |
|---|---|---|---|---|---|---|---|---|
| Mean (LIF counts) | 61.7 | 43.6 | 45.5 | 50.5 | 49.4 | 49.9 | 49.8 | 50.9 |
| SD | 2.82 | 5.99 | 3.24 | 2.15 | 0.99 | 0.80 | 1.32 | 1.03 |
| %RSD | 4.57 | 13.7 | 7.12 | 4.26 | 2.00 | 1.60 | 2.65 | 2.02 |
HPLC Results for Mixing Profiles
HPLC analysis was performed from the same sample that LIF measurements were taken. Percent label claim was calculated and HPLC results at the various locations and time stamps are summarized in Table II. The results support that the preblend is homogeneous after 5–10 min of blending and passes the Readily Pass blend homogeneity criteria. The 2-min time point meets the Marginally Pass criteria. The 0.25, 0.5, 0.75, and 20 min blend samples were outside the Marginally Pass classification. The commercial product blend time is 20 min so the observed high %RSD result was unexpected. After thorough investigation, no assignable cause could be attributed to the high %RSD HPLC result. The observed variation may be due to demixing, thief sampling technique, analytical error, or a combination of errors. It is important to note that the 20-min blending process has been validated and used in the production of hundreds of commercial batches with no content uniformity issues noted in the product history. As such, it is highly unlikely that demixing is the source of variability. Powder blend sampling techniques are widely recognized as a potential source of error in assessments of blend homogeneity. The correct choice of blend thief and associated sampling technique is critical to acquiring representative blend uniformity data (13,14). Compared to LIF data acquisition, the more complex HPLC sample preparation, analysis methodology, and lack of replicates could have also contributed to observed high %RSD. As it was not possible to identify the exact root cause, the data point was included in the data set.
Table II.
HPLC %LC for the Preblend Samples
| Time (min) | 0.25 | 0.5 | 0.75 | 1 | 2 | 5 | 10 | 20 |
|---|---|---|---|---|---|---|---|---|
| Mean | 242.7 | 56.2 | 69.4 | 118.2 | 93.1 | 93.4 | 94.4 | 104.1 |
| SD | 44.52 | 40.0 | 22.0 | 16.90 | 4.05 | 2.46 | 3.44 | 14.76 |
| %RSD | 18.34 | 71.2 | 31.7 | 14.30 | 4.35 | 2.63 | 3.64 | 14.18 |
Similarity of Mixing Profiles
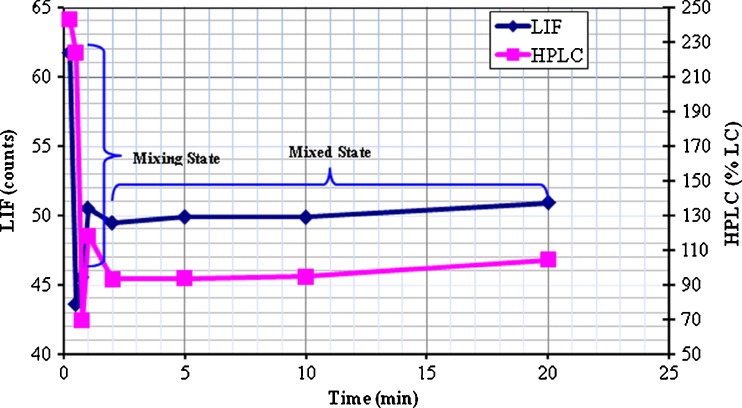
Figure 2 is a graph of LIF counts and HPLC %LC data versus mixing time. Both analytical methods show similar mixing profiles. The profiles can be divided into two general states or phases of blending involving a rapidly changing mixing state and a mixed state. The visual similarity between the LIF and HPLC mixing profiles suggests that a qualitative relationship exists between LIF count data and HPLC %LC. A quantitative relationship is established in the next two sections.
Fig. 2.
Mixing profiles of LIF means (diamond) and HPLC means (square) for the preblend samples
Relationship Between LIF Count and HPLC %LC Using a Bivariate Fit
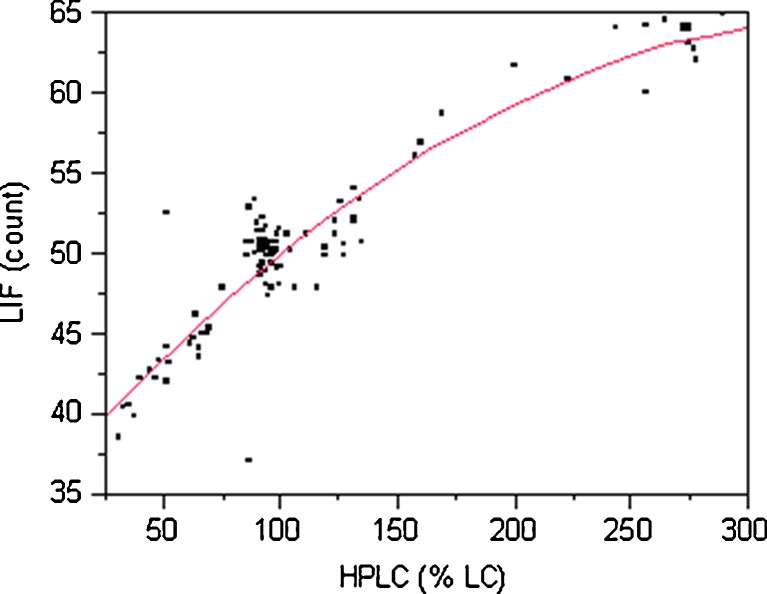
A bivariate fit between LIF count and HPLC %LC is depicted in Fig. 3. A second-order polynomial fit gave an R2 of 0.86 (p value < 0.0001) which indicates that 86% of the variability can be explained by this model. The analysis also shows that there is a statistically significant second order polynomial relationship between LIF and HPLC data. Lai et al. (6,7) have reported a similar nonlinear fit for LIF response versus percent w/w blend. The nonlinearity is thought to be due to saturation of the photomultiplier tube.
Fig. 3.
Polynomial bivariate fit of LIF counts by HPLC %LC for all samples
Relationship Between LIF %RSD and HPLC %RSD Using Bivariate Fit
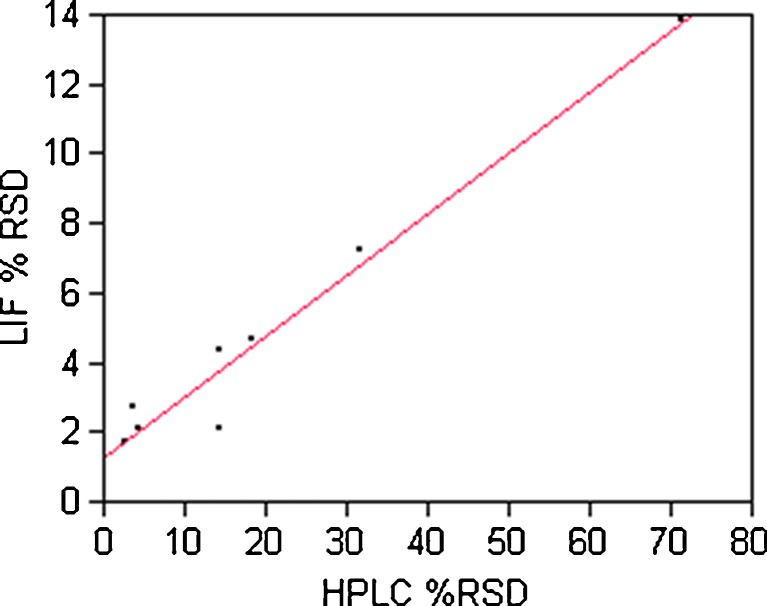
A bivariate fit provided in Fig. 4 was plotted between LIF %RSD and HPLC %RSD. The linear fit has an R2 of 0.97 and is statistically significant at a p value of <0.0001. This means that 97% of the experimental variation can be explained by the linear model and the relationship between LIF %RSD and HPLC %RSD is statistically significant. Since %RSD is a key measure of blend uniformity, this is an important finding and suggests that LIF can be used to qualitatively document blend homogeneity. The most disparate point on the graph (the fourth point to the right of the origin) is the 20-min blend sample. The high HPLC %RSD value resulted in the deviation from the statistically significant best-fit line.
Fig. 4.
Bivariate fit of LIF %RSD by HPLC %RSD
The LIF and HPLC mixing profiles are qualitatively similar and the bivariate fit analyses show that there are statistically significant relationships between LIF count and HLPC %LC and LIF %RSD and HPLC %RSD.
Determination of Acceptable Mixing Time Interval and End Point
For purposes of this study, an acceptable mixing time interval was the time interval when steady-state mixing existed and the %RSD was classified as Readily Pass (%RSD ≤ 4%). Steady-state was defined as the mixing interval where the group means by time were statistically equivalent. The mixing end point was chosen as the first time point that met the acceptable mixing time interval criteria.
One-Way Analysis of Variance (ANOVA) of LIF Counts by Time
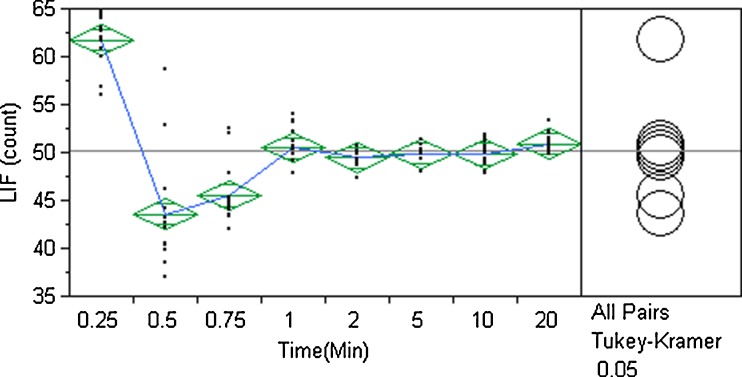
A one-way ANOVA was performed for LIF counts by time and is plotted in Fig. 5. At each time point, the LIF counts are provided for each of the 13 sample locations. In the first 0.75 min, there is a large variation in the number of counts for a given location. The one-way ANOVA “means diamonds” are shown in Fig. 5. The means diamonds are a graphical illustration of the t test. The line bisecting the diamond represents the group mean LIF counts. The vertical span represents the 95% confidence interval and horizontal lines near the tip of the diamonds are called “overlap” marks. If there is a vertical separation between the top overlap mark of one diamond and the bottom overlap mark of a second diamond, the group means are not considered statistically equivalent. The one-way ANOVA shows that the group means are statistically equivalent at 1 min mixing time and beyond. This is also supported by the Tukey–Kramer honestly significant difference (HSD) test (p < 0.05) which compares all group mean pairs. The Tukey–Kramer HSD test is considered a more conservative ANOVA test. The analysis is summarized in Table III. For time points at 1 min and beyond, the means are statistically equivalent and are associated with letter B. The blend mean at 0.25 min (represented by A) is statistically different from all other blends. Mixtures at 0.5 and 0.75 min (represented by C) are statistically different from all other mixtures. The 1–20 min blend means are statistically the same and represent a steady-state mixing interval. The acceptable mixing time interval is considered to be 2–20 min where the blend meets the Readily Pass classification and group means are statistically equivalent. The mixing end point for the LIF data is 2 min.
Fig. 5.
One-way analysis of variance of LIF counts by time
Table III.
Tukey–Kramer HSD Summary of Group Means Comparison for LIF Counts by Time
| Time (min) | Group mean | |||
|---|---|---|---|---|
| 0.25 | A | 61.7 | ||
| 20 | B | 50.9 | ||
| 1 | B | 50.5 | ||
| 5 | B | 49.9 | ||
| 10 | B | 49.8 | ||
| 2 | B | 49.4 | ||
| 0.75 | C | 45.0 | ||
| 0.5 | C | 43.5 |
Means not sharing a letter are statistically different
One-Way Analysis of Variance of HPLC %LC by Time
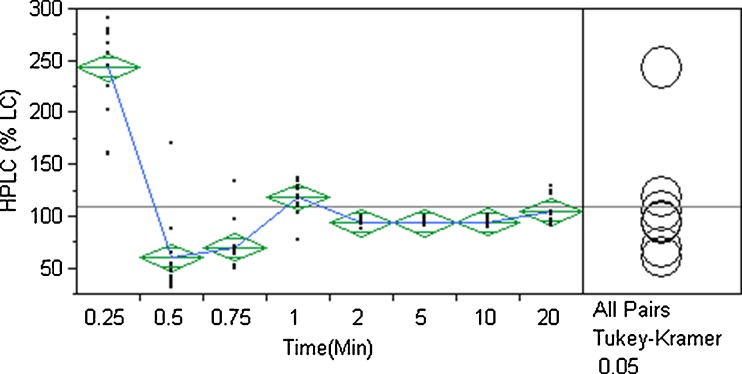
A one-way ANOVA was performed for HPLC %LC by time. The results are shown in Fig. 6. As seen with the LIF data, the first 0.75 min of blending exhibit a wide range of %LC values over the 13 sample locations. The Tukey–Kramer HSD test at p < 0.05 is provided in Table IV. HPLC one-way ANOVA shows that the 1, 2, 5, 10, and 20 min group means are statistically the same (associated with the letter B) and the steady-state is 1–20 min. This is the same steady-state interval determined by LIF analysis. Comparing all group mean pairs using the Tukey–Kramer HSD test, the 2, 5, and 10 min groups means are statistically the same. Using the more conservative ANOVA analysis, the blend steady-state is considered to be reached at the 2–10 min mixing interval. The acceptable mixing time interval is 5–10 min. At this time interval, the blend meets the Readily Pass criterion and group means are statistically the same. The mixing end point is 5 min. It should be noted that the %RSD at the 2 min time point was 4.35%; exceeding the Readily Pass criterion by 0.35%.
Fig. 6.
One-way analysis of variance of HPLC %LC by time
Table IV.
Tukey–Kramer HSD Summary of Group Means Comparison for HPLC %LC by Time
| Time (min) | Group mean | ||||
|---|---|---|---|---|---|
| 0.25 | A | 242.7 | |||
| 1 | B | 118.2 | |||
| 20 | B | 104.1 | |||
| 10 | B | C | 94.4 | ||
| 5 | B | C | 93.3 | ||
| 2 | B | C | 93.1 | ||
| 0.75 | C | D | 69.4 | ||
| 0.5 | D | 56.2 |
Means not sharing a letter are statistically different
Determination of Potential Blender Dead Spots
In order to evaluate the potential for blender dead spots, an appropriate mixing end point or mixing interval must be identified. The “acceptable mixing time interval” as defined above was chosen as a conservative mixing interval where the group means at each sample time are statistically equivalent and have a %RSD ≤ 4 (Readily Pass). The LIF sensor used in the study did not have sufficient sensitivity to reveal potential blender dead spots. The individual LIF mean counts ranged from 37 to 66 while the HPLC %LC results ranged from 22 to 290. Simply using the data ranges, a single LIF count is equivalent to 9.6 %LC. Therefore, only HPLC data was used to identify potential blender dead spots.
One-Way Analysis of Variance of HPLC %LC by Location over the Acceptable Mixing Time Interval
The 5- and 10-min sample times met the acceptable mixing time interval criteria and were used to evaluate the blender for potential dead spots. The Tukey–Kramer HSD analysis indicates that there is a difference in the blend means at locations 3 (top middle) and 13 (bottom right) in Fig. 1. These means are statistically different from the rest of the locations means. The Bartlett’s test was used to evaluate the assay variability among the sample locations. The test indicated that there was no statistical difference in assay variability. Therefore, the differences in the assay means at locations 3 and 13 cannot be attributed to differences in assay variability among sample locations (Table V).
Table V.
Tukey-Kramer HSD Summary of Group Means Comparison for HPLC %LC by Location at 5- and 10-Min Blend Times
| Sample Location | Mean | ||
|---|---|---|---|
| 3 | A | 98.8 | |
| 6 | A | B | 96.5 |
| 10 | A | B | 95.5 |
| 4 | A | B | 95.1 |
| 8 | A | B | 95.0 |
| 7 | A | B | 94.3 |
| 1 | A | B | 94.3 |
| 8 | A | B | 94.9 |
| 2 | A | B | 93.9 |
| 9 | A | B | 93.7 |
| 5 | A | B | 93.0 |
| 12 | A | B | 91.0 |
| 11 | A | B | 90.9 |
| 13 | B | 88.6 |
Means not sharing a letter are statistically different
CONCLUSIONS
This study met its primary aim of evaluating LIF as a PAT tool and establishing a relationship between LIF and HPLC analytical technologies. LIF and HPLC gave qualitatively similar mixing profiles. LIF counts and HPLC %LC data were fit to a second order polynomial that had an R2 of 0.86 (p < 0.0001) which covered a fourfold API concentration range. A linear relationship (R2 = 0.97, p < 0.0001) was observed between LIF %RSD and HPLC %RSD. Since %RSD is a key measure of blend uniformity, this is an important finding and suggests that LIF can be used to qualitatively document blend homogeneity.
A secondary aim of this study was to determine steady-state, acceptable mixing time interval and end point. The LIF steady-state, acceptable mixing time interval and end point were determined to be 1–20, 2–20, and 2 min, respectively. The steady-state, acceptable mixing time interval, and mixing end point determined by HPLC were 1–20, 5–10, and 5 min, respectively.
Two locations in the 124 L IBC mixing tote were identified as potential dead spots. Tukey–Kramer HSD group mean analysis of HPLC %LC by sample location at 5- and 10-min mixing times showed that there was a statistical difference between the HPLC %LC group means at locations 3 and 13 compared to the rest of the sample locations.
The LIF sensor used in this study did not have sufficient sensitivity to reveal potential blender dead spots. This lack of sensitivity also probably accounts for qualitatively smaller %RSD values compared to the HPLC %RSDs.
FUTURE WORK
To use LIF as a quantitative PAT tool, LIF count data will need to be converted to %LC or % w/w API using solid state calibration standards. Preliminary investigations have shown that the fabrication of solid-state calibration standards is complex and dependent on the density of the standards. Additional work will be required to establish meaningful calibration standards.
A newer more sensitive LIF instrument has been developed based on the learnings from this study. This research project will be repeated with the more sensitive unit to further develop the relationships between LIF and HPLC technologies.
ACKNOWLEDGMENTS
The authors thank GlaxoSmithKline for their generous support of this work.
REFERENCES
- 1.Harris DC. Quantitative chemical analysis. 8. New York: Freeman; 2010. p. 612. [Google Scholar]
- 2.Tutorial on fluorescence and fluorescent instrumentation. In: FMRC Manual. http://fmrc.pulmcc.washington.edu/DOCUMENTS/FMRC299.pdf. Accessed 02 Nov 2010.
- 3.Harris DC. Quantitative chemical analysis. 84. New York: Freeman; 2010. pp. 406–412. [Google Scholar]
- 4.US Food and Drug Administration, Center for Drug Evaluation and Research. Office of Pharmaceutical Science (OPS), Process analytical technology (PAT) initiative. September 2004. http://www.fda.gov/cder/OPS/PAT.htm#Introduction. Accessed 02 Nov 2010.
- 5.Lai C-K, Cooney CL. Application of a fluorescence sensor for miniscale on-line monitoring of powder mixing kinetics. J Pharm Sci. 2004;93:60–70. doi: 10.1002/jps.10478. [DOI] [PubMed] [Google Scholar]
- 6.Lai C-K, Holt D, Leung JC, Raju GK, Hansen P, Cooney CL. Real time and noninvasive monitoring of dry powder blend homogeneity. AlChE J. 2001;47:2618–2622. doi: 10.1002/aic.690471124. [DOI] [Google Scholar]
- 7.Lai C-K, Zahari A, Miller B, Katstra WE, Cima MJ, Cooney CL. Nondestructive and on-line monitoring of tablets using light-induced fluorescence technology. AAPS PharmSciTech [serial online] 2004; 5(1): E3. [DOI] [PMC free article] [PubMed]
- 8.Corporation H. Honeywell blend homogeneity sensor user guide. Release 1. Fort Washington, PA: Honeywell Corporation; 2006. [Google Scholar]
- 9.Labsphere, Inc. Spectralon fluorescence standards guide. North Sutton, NH.
- 10.SAS Institute Inc . JMP® 8 Design of experiments software. Cary, NC: SAS Publishing; 2008. [Google Scholar]
- 11.Engineering Statistics Handbook. NIST/SEMATECH e-handbook of statistical methods. http://www.itl.nist.gov/div898/handbook/eda/section3/eda357.htm. Accessed 04 Nov 2010.
- 12.FDA Guidance for Industry. U.S. Department of Health and Human Services. Food and Drug Administration. Center for Drug Evaluation and Research (CDER). Powder blends and finished dosage units—stratified in-process dosage unit sampling and assessment. http://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/ucm070312.pdf. 2003. Accessed 05 Nov 2010.
- 13.Garcia TP, Wilkinson SJ, Scott JF. The development of a blend-sampling technique to assess the uniformity of a powder mixture. Drug Dev Ind Pharm. 2001;27:297–307. doi: 10.1081/DDC-100103729. [DOI] [PubMed] [Google Scholar]
- 14.Garcia TP, Taylor MK, Pande GS. Comparision of the performance of two sample thieves for the determination of the content uniformity of a powder blend. Pharm Dev Technol. 1998;3(1):7–12. doi: 10.3109/10837459809028474. [DOI] [PubMed] [Google Scholar]