Abstract
Antigens bind to immunoreceptors on the surface of T-cells and B-cells, activating signal transduction pathways that allow them to proliferate, differentiate, and synthesize effector molecules involved in the immune response.
T cells and B cells are stimulated when antigens bind to T-cell receptors (TCRs) and B-cell receptors (BCRs) in their respective plasma membranes (Fig. 1). The antigens presented to T cells are in the form of short peptides that have been processed in infected cells and are displayed on the surface bound to major histocompatibility complex (MHC) class I or class II molecules. BCRs, in contrast, recognize free antigens in their native form present in the extracellular milieu or on cell surfaces.
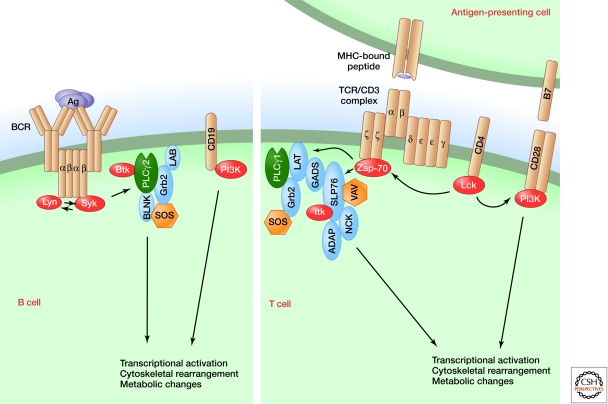
Figure 1.
Early events in T cell and B cell receptor signaling.
Despite superficial differences, the responses in the two cell types are remarkably similar. For both, signaling begins with engagement of a complex receptor composed of antigen receptor subunits with immunoglobulin or immunoglobulin-like domains. Both receptors also contain non-polymorphic subunits, the TCRζ chain and CD3 subunits (γ, δ, and ε) for the TCR, and the α and β chains of the BCR (Reth 1995; Wucherpfennig et al. 2010). Coreceptors are important in both T cells (CD4 for helper T cells; CD8 for cytotoxic T cells) and B cells (CD19). For both receptors, members of two protein tyrosine kinase (PTK) families, Src (Lck for T cells and Lyn for B cells) and Syk (Zap-70 for T cells and Syk for B cells) (Bradshaw 2010; Liu et al. 2010; Wang et al. 2010), are critical signaling molecules closely associated with the receptors and coreceptors. Activation of tyrosine kinases is the first biochemical change that follows receptor engagement.
Following antigen engagement, activated tyrosine kinases phosphorylate several adapter proteins and signaling enzymes, which then interact. For T cells, this is exemplified by the phosphorylation of the adapters LAT and SLP-76 (Fig. 2), the formation of multiprotein complexes nucleated at these adapters, and the inclusion of such enzymes as phospholipase Cγ (PLCγ1) and VAV in the protein assemblies (Balagopalan et al. 2010; Jordan and Koretzky 2010). Activation of an additional protein tyrosine kinase, Itk in T cells and Btk in B cells, occurs in these complexes (Andreotti et al. 2010). Similarly in B-cells, phosphorylation of BLNK and CD19 leads to recruitment of adapters and signaling enzymes. In both cell types these events occur at the plasma membrane, where critical lipid substrates of enzymes such as PLCγ1 and PI3 kinase (PI3K) are located.
Figure 2.
T cell receptor signaling.
Downstream from these initial events, a number of other protein complexes are formed, and various signaling pathways are activated. Many of these involve activation of protein serine kinases (Finlay and Cantrell 2010). In T cells and B cells, activation of protein kinase C (PKC) leads to formation and activation of the CARMA1–Bcl10–MALT1 complex (Thome et al. 2010). Stimulation of PI3K leads to activation of the kinase Akt (Huang and Sauer 2010). Downstream from the TCR and BCR, one also observes activation of small G proteins such as Ras, which, in turn, leads to activation of the Raf/MEK and ERK kinases. Finally, in both cell types, these various events lead to cytoskeletal changes and gene expression induced by transcriptional activators such as NF-AT and NF-κB. T cells and B cells thus activated can proliferate, differentiate, and synthesize the cytokines and effector molecules that allow them to fulfill their roles in the adaptive immune response.
(Figures adapted with kind permission of Cell Signaling Technology (http://cellsignal.com.)
Editors: Lewis Cantley, Tony Hunter, Richard Sever, and Jeremy Thorner
Additional Perspectives on Signal Transduction available at www.cshperspectives.org
REFERENCES
- Andreotti AH, Schwartzberg PL, Joseph RE, Berg LJ 2010. T-cell signaling regulated by the Tec family kinase, Itk. Cold Spring Harb Perspect Biol 2: a002287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balagopalan L, Coussens NP, Sherman E, Samelson LE, Sommers CL 2010. The LAT story: A tale of cooperativity, coordination, and choreography. Cold Spring Harb Perspect Biol 2: a005512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradshaw JM 2010. The Src, Syk, and Tec family kinases: Distinct types of molecular switches. Cell Signal 22: 1175–1184 [DOI] [PubMed] [Google Scholar]
- Finlay D, Cantrell D 2010. The coordination of T-cell function by serine/threonine kinases. Cold Spring Harb Perspect Biol 3: a002261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang YH, Sauer K 2010. Lipid signaling in T-cell development and function. Cold Spring Harb Perspect Biol 2: a002428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jordan MS, Koretzky GA 2010. Coordination of receptor signaling in multiple hematopoietic cell lineages by the adaptor protein SLP-76. Cold Spring Harb Perspect Biol 2: a002501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu W, Sohn HW, Tolar P, Pierce SK 2010. It’s all about change: The antigen-driven initiation of B-cell receptor signaling. Cold Spring Harb Perspect Biol 2: a002295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reth M 1995. The B-cell antigen receptor complex and co-receptors. Immunol Today 16: 310–313 [DOI] [PubMed] [Google Scholar]
- Thome M, Charton JE, Pelzer C, Hailfinger S 2010. Antigen receptor signaling to NF-κB via CARMA1, BCL10, and MALT1. Cold Spring Harb Perspect Biol 2: a003044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang H, Kadlecek TA, Au-Yeung BB, Goodfellow HE, Hsu LY, Freedman TS, Weiss A 2010. ZAP-70: An essential kinase in T-cell signaling. Cold Spring Harb Perspect Biol 2: a002279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wucherpfennig KW, Gagnon E, Call MJ, Huseby ES, Call ME 2010. Structural biology of the T-cell receptor: Insights into receptor assembly, ligand recognition, and initiation of signaling. Cold Spring Harb Perspect Biol 2: a005140. [DOI] [PMC free article] [PubMed] [Google Scholar]