Abstract
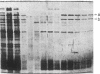
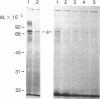
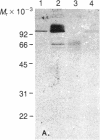
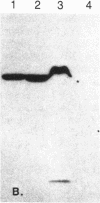
The H+-translocating ATPase located on vacuolar membranes of Neurospora crassa was partially purified by solubilization in two detergents, Triton X-100 and N-hexadecyl-N,N-dimethyl-3-ammonio-1-propanesulfonate, followed by centrifugation on sucrose density gradients. Two polypeptides of Mr approximately equal to 70,000 and approximately equal to 62,000 consistently migrated with activity, along with several minor bands of lower molecular weight. Radioactively labeled inhibitors of ATPase activity, N-[14C]ethylmaleimide and 7-chloro-4-nitro[14C]benzo-2-oxa-1,3-diazole, labeled the Mr approximately equal to 70,000 polypeptide; this labeling was reduced in the presence of ATP. N,N'-[14C]dicyclohexylcarbodiimide labeled a polypeptide of Mr approximately equal to 15,000. Estimation of the functional size of the vacuolar membrane ATPase by radiation inactivation gave a value of Mr 5.2 X 10(5), 10-15% larger than the mitochondrial ATPase. The Neurospora vacuolar ATPase showed no crossreactivity with antiserum to plasma membrane or mitochondrial ATPase but strongly crossreacted with antiserum against a polypeptide of Mr approximately equal to 70,000 associated with the tonoplast ATPase of corn coleoptiles. These results suggest that fungal and plant vacuolar ATPases may be large multisubunit complexes, somewhat similar to, but immunologically distinct from, known F0F1 ATPases.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bowman B. J., Berenski C. J., Jung C. Y. Size of the plasma membrane H+-ATPase from Neurospora crassa determined by radiation inactivation and comparison with the sarcoplasmic reticulum Ca2+-ATPase from skeletal muscle. J Biol Chem. 1985 Jul 25;260(15):8726–8730. [PubMed] [Google Scholar]
- Bowman B. J., Blasco F., Slayman C. W. Purification and characterization of the plasma membrane ATPase of Neurospora crassa. J Biol Chem. 1981 Dec 10;256(23):12343–12349. [PubMed] [Google Scholar]
- Bowman E. J., Bowman B. J., Slayman C. W. Isolation and characterization of plasma membranes from wild type Neurospora crassa. J Biol Chem. 1981 Dec 10;256(23):12336–12342. [PubMed] [Google Scholar]
- Bowman E. J. Comparison of the vacuolar membrane ATPase of Neurospora crassa with the mitochondrial and plasma membrane ATPases. J Biol Chem. 1983 Dec 25;258(24):15238–15244. [PubMed] [Google Scholar]
- Brooker R. J., Slayman C. W. [14C]N-ethylmaleimide labeling of the plasma membrane [H+]-ATPase of Neurospora crassa. J Biol Chem. 1983 Jan 10;258(1):222–226. [PubMed] [Google Scholar]
- Clarke D. J., Fuller F. M., Morris J. G. The proton-translocating adenosine triphosphatase of the obligately anaerobic bacterium Clostridium pasteurianum. 1. ATP phosphohydrolase activity. Eur J Biochem. 1979 Aug 1;98(2):597–612. doi: 10.1111/j.1432-1033.1979.tb13222.x. [DOI] [PubMed] [Google Scholar]
- Esch F. S., Allison W. S. On the subunit stoichiometry of the F1-ATPase and the sites in it that react specifically with p-fluorosulfonylbenzoyl-5'-adenosine. J Biol Chem. 1979 Nov 10;254(21):10740–10746. [PubMed] [Google Scholar]
- Ferguson S. J., Lloyd W. J., Radda G. K. The mitochondrial ATPase. Selective modification of a nitrogen residue in the beta subunit. Eur J Biochem. 1975 May;54(1):127–133. doi: 10.1111/j.1432-1033.1975.tb04121.x. [DOI] [PubMed] [Google Scholar]
- Forgac M., Cantley L., Wiedenmann B., Altstiel L., Branton D. Clathrin-coated vesicles contain an ATP-dependent proton pump. Proc Natl Acad Sci U S A. 1983 Mar;80(5):1300–1303. doi: 10.1073/pnas.80.5.1300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goffeau A., Slayman C. W. The proton-translocating ATPase of the fungal plasma membrane. Biochim Biophys Acta. 1981 Dec 30;639(3-4):197–223. doi: 10.1016/0304-4173(81)90010-0. [DOI] [PubMed] [Google Scholar]
- Kakinuma Y., Ohsumi Y., Anraku Y. Properties of H+-translocating adenosine triphosphatase in vacuolar membranes of SAccharomyces cerevisiae. J Biol Chem. 1981 Nov 10;256(21):10859–10863. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Ludwig B., Prochaska L., Capaldi R. A. Arrangement of oligomycin-sensitive adenosine triphosphatase in the mitochondrial inner membrane. Biochemistry. 1980 Apr 1;19(7):1516–1523. doi: 10.1021/bi00548a039. [DOI] [PubMed] [Google Scholar]
- Mandala S., Taiz L. Partial purification of a tonoplast ATPase from corn coleoptiles. Plant Physiol. 1985 Jun;78(2):327–333. doi: 10.1104/pp.78.2.327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rott R., Nelson N. Purification and immunological properties of proton-ATPase complexes from yeast and rat liver mitochondria. J Biol Chem. 1981 Sep 10;256(17):9224–9228. [PubMed] [Google Scholar]
- Schneider D. L. Membranous localization and properties of ATPase of rat liver lysosomes. J Membr Biol. 1977 Jun 6;34(2-3):247–261. doi: 10.1007/BF01870302. [DOI] [PubMed] [Google Scholar]
- Uchida E., Ohsumi Y., Anraku Y. Purification and properties of H+-translocating, Mg2+-adenosine triphosphatase from vacuolar membranes of Saccharomyces cerevisiae. J Biol Chem. 1985 Jan 25;260(2):1090–1095. [PubMed] [Google Scholar]
- Vaitukaitis J., Robbins J. B., Nieschlag E., Ross G. T. A method for producing specific antisera with small doses of immunogen. J Clin Endocrinol Metab. 1971 Dec;33(6):988–991. doi: 10.1210/jcem-33-6-988. [DOI] [PubMed] [Google Scholar]
- Vaughn L. E., Davis R. H. Purification of vacuoles from Neurospora crassa. Mol Cell Biol. 1981 Sep;1(9):797–806. doi: 10.1128/mcb.1.9.797. [DOI] [PMC free article] [PubMed] [Google Scholar]