This review presents the evidence base and discusses novel experimental approaches for the combination of biomarker modalities to allow optimization of an individualized treatment approach in GEJ adenocarcinoma patients that may be relevant to other tumor types as well.
Keywords: Gastroesophageal junction, Adenocarcinoma, Response, Positron emission tomography, Biomarkers
Learning Objectives
After completing this course, the reader will be able to:
Contrast the subtypes of gastroesophageal adenocarcinoma in order to select optimal therapeutic approaches for given subtypes.
Compare the various tools (CT, MRI, PET, PET-CT, etc.) for evaluating response to therapy in order to determine whether to initiate new therapy.
Evaluate response to neoadjuvant therapy, utilizing imaging, histopathogy of resected specimens, and biomarkers, to plan postoperative treatment.
This article is available for continuing medical education credit at CME.TheOncologist.com
Abstract
The incidence of adenocarcinomas of the gastroesophageal junction (GEJ) is rapidly rising, and even in early-stage locoregional confined disease the 5-year survival rate rarely exceeds 25%–35%. Randomized trials and meta-analyses have demonstrated a benefit with neoadjuvant or perioperative chemotherapy and with neoadjuvant chemoradiotherapy. However, the optimal approach in individual patients is not clear and remains controversial. A consistent finding is that patients who have a histopathological response to neoadjuvant therapy are more likely to receive a survival benefit. These clinical data provide a strong argument for the urgent development of methods to predict histopathological response to neoadjuvant therapies for GEJ adenocarcinoma. Published data demonstrate that clinicopathological features (tumor location), imaging (fluorodeoxyglucose-positron emission tomography “metabolic response”), and tissue/molecular biomarkers may all have a predictive value for neoadjuvant therapies. However, it is uncertain from published data whether or not they will be useful for clinical decision making in individual patients. Existing candidate biomarkers need to be properly qualified and validated and novel biomarkers are required; and an optimal approach should involve the combination and integration of clinical, imaging, and molecular biomarkers. This review presents the evidence base and discusses novel experimental approaches for the combination of biomarker modalities to allow optimization of an individualized treatment approach in GEJ adenocarcinoma patients that may be relevant to other tumor types as well.
Introduction
The clinical management of early operable adenocarcinomas of the gastroesophageal junction (GEJ) provides a clear paradigm for the application of predictive biomarkers [1]. International consensus is that chemotherapy and/or radiotherapy in addition to surgery improves outcome; however, there is disagreement regarding the optimal approach. Randomized clinical trial data support the use of either preoperative (neoadjuvant) chemotherapy, perioperative (neoadjuvant and adjuvant) chemotherapy, or preoperative concurrent chemoradiotherapy [2–7]. Accordingly, a central translational research objective for GEJ adenocarcinomas is to define subgroups of patients who optimally benefit from particular combined-modality approaches [1]. Unfortunately, interpretation of clinical trials is complicated by heterogeneity in trial data, both with regard to patient populations—for example, a mix of histology in older studies (squamous and adenocarcinoma), a mix of tumor types (esophageal, GEJ, and gastric), and different staging protocols (multidetector [or not] computed tomography [CT], endoscopic ultrasound [EUS], laparoscopy, endobronchial ultrasound, fluorodeoxyglucose-positron emission tomography [FDG-PET], or FDG-PET/CT)—and also with regard to all aspects of treatment protocols (surgery, chemotherapy, and radiotherapy). New approaches to clinical investigation to identify predictive biomarkers are needed and discussed in this review.
Clinical Epidemiology of Gastroesophageal Adenocarcinomas and Disease Subtypes
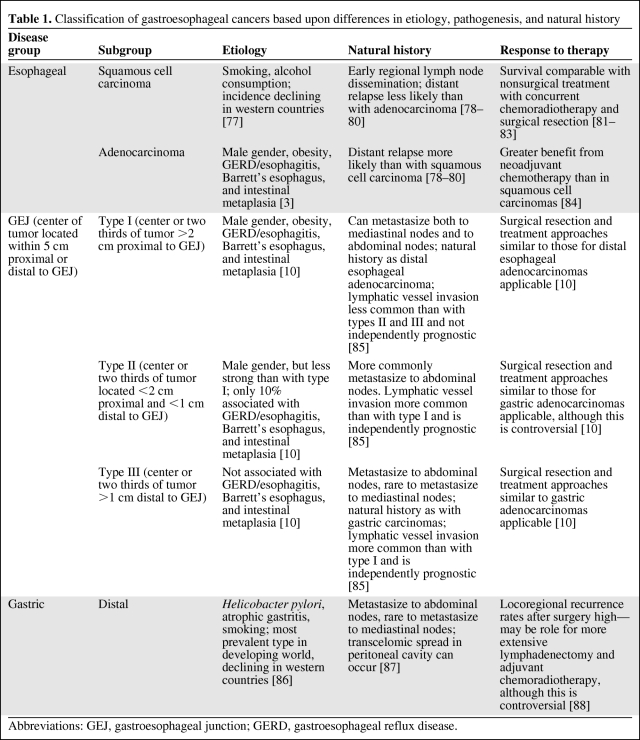
Gastroesophageal carcinomas have undergone distinct changes in their epidemiology over recent years [8]. The incidence of gastric adenocarcinoma is declining, but in western countries (but not Asian countries) there is an ongoing rise in adenocarcinomas of the distal esophagus and GEJ of more than sixfold in the past three decades [9]. This rising incidence has led to the recognition of adenocarcinoma of the GEJ as a distinct disease entity and the need to reclassify esophageal and gastric adenocarcinomas to reflect this [10]. Data support a classification with disease subtypes with differences in etiology, pathogenesis, and natural history (Table 1). According to U.S. cancer registry statistics, the 5-year survival rate is only 10%–15% overall, and in early-stage, locoregional, confined disease the 5-year survival rate is 25%–30% [11].
Table 1.
Classification of gastroesophageal cancers based upon differences in etiology, pathogenesis, and natural history
Abbreviations: GEJ, gastroesophageal junction; GERD, gastroesophageal reflux disease.
This review is concerned with the emerging predominant group, adenocarcinomas of the GEJ, defined as tumors occurring within 5 cm proximal or distal to the GEJ [10]. The recognition of subtypes of gastroesophageal adenocarcinomas is important for locoregional management [3, 12]. Although it is of less relevance for systemic treatment with cytotoxic drugs [13], emerging data with targeted therapy suggest differential responses for these clinicopathological subtypes (Table 1) with novel targeted agents (e.g., the epidermal growth factor receptor [EGFR] tyrosine kinase inhibitor erlotinib [14]), and this is supported by data showing differential expression of targets and biomarkers in these disease subgroups (e.g., human epidermal growth factor receptor [HER]-2) [15]. Recognition of these known disease subgroups is important for predictive biomarker investigation, especially discovery, particularly if a differential benefit of the intervention is seen in different subgroups. Here, unless disease subgroups are acknowledged, the biomarker discovered or being investigated may ultimately prove to be a surrogate for disease subtype rather than clinical benefit or response and therefore of no clinical utility as a predictive biomarker.
Response Assessment to Neoadjuvant Therapy in Early GEJ Adenocarcinomas as a Surrogate for Clinical Efficacy
There has been considerable interest in response to neoadjuvant therapy as a surrogate for survival benefit both in terms of the therapy modality being used and also as a prognostic factor for outcome following surgery [1]. A number of methods of response assessment have been evaluated in GEJ adenocarcinomas as surrogates for survival.
Clinical Response
Clinical response to therapy is often inconsistently defined, varies according to study, and correlates poorly with survival [1].
Radiological Response
Radiological response assessed by CT, and as defined by the Response Evaluation Criteria in Solid Tumors, is often used in current routine clinical practice and clinical trials [16]. However, a decline in tumor size is a relatively late event in response to chemotherapy or radiotherapy, limiting its use as a predictive biomarker. In addition, the presence of fibrotic or necrotic tissue may not accurately reflect the viable tumor cell fraction in a residual mass. Thus, esophageal thickness and tumor response measured by CT does not correlate with survival, meaning that it is not a useful surrogate for survival and has limited clinical utility for assessing benefit from neoadjuvant therapy and as a prognostic factor to facilitate subsequent clinical treatment decisions [1, 17].
Metabolic Response
Several workers have evaluated the potential of serial FDG-PET scans during the course of neoadjuvant therapy to define a metabolic response or nonresponse and to use this as a predictive biomarker or surrogate for benefit from neoadjuvant therapy and allow therapy to be changed early if it is likely to be ineffective [18–27]. These studies are discussed in detail below; however, in summary, preliminary evidence supports a role for FDG-PET–defined metabolic response as a potentially useful surrogate for survival benefit from neoadjuvant chemotherapy in particular [21, 23–26]. Confirmatory retrospective and prospective studies are needed, and the greater value of metabolic response may be to predict histopathological response, which has been shown to be a stronger surrogate for survival benefit following neoadjuvant therapy than is metabolic response [26].
Histopathological Response
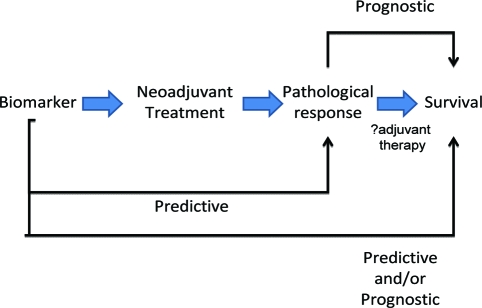
Histopathological response to treatment appears to have value as a surrogate for survival in GEJ adenocarcinomas. A consistent finding is that histopathological response identifies patients who benefit from both neoadjuvant chemotherapy and chemoradiotherapy, and patients who do not respond to neoadjuvant therapy have survival rates that may not be significantly different from those who are treated with surgery alone [1, 5, 28–30]. Therefore, the ability to predict histopathological response to neoadjuvant therapy would allow optimal combined-modality treatment selection preoperatively. In addition, the subsequent confirmation (or otherwise) of histopathological response may facilitate subsequent postoperative (adjuvant) therapy (Fig. 1). Implicit in this statement is the hypothesis that histopathological response is indicative of the underlying disease biology, although the molecular mechanisms behind this are not known.
Figure 1.
Predictive biomarkers in gastroesophageal junction adenocarcinomas. A biomarker predictive of histopathological response, and hence survival benefit from a neoadjuvant therapy, may also be predictive for adjuvant use of the same therapy and may also have therapy-independent prognostic value. Histopathological response has prognostic value and may confirm or modify the relevance of a predictive biomarker, and hence influence adjuvant therapy decisions.
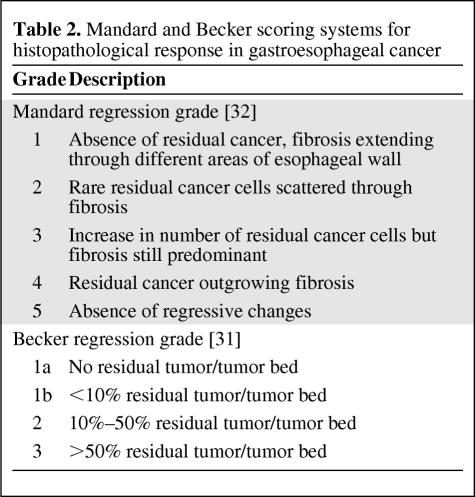
Definition of Histopathological Response
Histopathological response has been defined by a variety of methods commonly involving estimation of the percentage of viable tumor relative to therapy-induced fibrosis [30–34]. The validated Becker [31] and Mandard [32] scoring systems are used in gastroesophageal cancer (Table 2). A major histopathological response is often defined as “<10% residual tumor cells” (Mandard grade 1 and grade 2, Becker grade 1a and grade 1b). Histopathological response evaluation requires resection of the tumor and can be determined only at the end of treatment, and cannot be used to modify preoperative treatment. Ideally, other measures of response that can predict histopathological response are needed.
Table 2.
Mandard and Becker scoring systems for histopathological response in gastroesophageal cancer
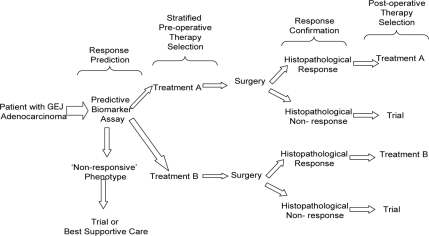
In the remainder of this review we discuss the potential use of biomarkers to predict histopathological response to preoperative therapies in GEJ adenocarcinoma. It is envisaged that the clinical use of such predictive biomarkers will provide the means to stratify patients for combined-modality therapy in a clinical management paradigm similar to that outlined in Figure 2.
Figure 2.
Paradigm for the use of predictive biomarkers to stratify preoperative therapy selection in gastroesophageal junction (GEJ) adenocarcinomas. Histopathological response can be subsequently used for confirmation or modification of adjuvant therapy. This requires prospective qualification and validation of predictive biomarkers before implementation. Randomization between alternative treatments is also possible in this paradigm.
Clinicopathological Predictive Biomarkers
Anatomical Disease Classification
The classification of GEJ adenocarcinomas outlined in Table 1 reveals etiological and histological differences and represents a practical and pragmatic disease classification based upon the anatomical location of the tumor, which can be applied in clinical practice with the use of routine staging procedures preoperatively (endoscopy, EUS, CT, laparoscopy) and postoperatively (histopathology). Distinct patterns of lymph node spread have been defined for type I, type II, and type III GEJ adenocarcinomas, and proposals for optimized treatment volumes for preoperative and definitive chemoradiotherapy have been made using this [12]. Distinct pathogenesis and biological features are implied by the observations of distinct etiologies and histological features, and recent and current therapeutic trials have recognized this by the definition of inclusion criteria based upon this disease classification [6, 35]. These clinical trials will provide data for hypothesis generation regarding the biological basis and therapeutic relevance of anatomical GEJ adenocarcinoma classification, which at present are not defined. It is important for the use of biomarker protocols to recognize these disease subtypes, because clinical response phenotypes may be different in each, and accordingly biomarkers must be able to delineate between being predictive for clinical benefit and being diagnostic or surrogates/markers for disease subtype. This is well illustrated by early clinical trial data of the oral tyrosine kinase inhibitor erlotinib, whereby differential sensitivity between GEJ adenocarcinomas and gastric adenocarcinomas was demonstrated [14].
Clinical Symptoms
Measurable clinical symptoms such as dysphagia (dysphagia scores) and weight gain have been evaluated as predictive biomarkers. However, although improvements in dysphagia or weight gain following neoadjuvant chemotherapy correlate with radiological response, they do not correlate with histopathological response [36].
Rebiopsy of Tumors
Endoscopic rebiopsy of tumors after completion of neoadjuvant therapy has been evaluated as a means to predict histopathological response, but this approach is unreliable in the prediction of response [31, 37].
Imaging Predictive Biomarkers
Anatomical Imaging
Endoscopy
Macroscopic appearance after treatment is associated with histopathological response, but with a limited accuracy of 50% (sensitivity, 57%; specificity, 36%) [37].
CT and EUS
CT scans are routinely performed following the completion of neoadjuvant therapy. However, they are of limited sensitivity and specificity in histopathological response assessment [38]. Progressive disease on CT after therapy is a well-acknowledged surrogate for the absence of clinical benefit from therapy in malignancy in general [16]. In early GEJ adenocarcinomas, CT progression identifies primary chemotherapy- or chemoradiotherapy-resistant disease associated with a very poor prognosis and provides a strong argument for avoidance of surgery in such patients. In a systematic review on assessment of histopathological response to therapy using CT, EUS, and FDG-PET, EUS was superior to CT, with sensitivities of 50%–100% and specificities of 36%–100%, compared with 33%–55% and 50%–71%, respectively, for CT [39]. However, EUS is an invasive procedure, and in 20%–50% of cases the probe cannot be passed through the narrowed esophagus. Moreover, the EUS technique is not as widespread as CT scanning, mainly because of the limited availability of operators with sufficient expertise. In addition, the need to ensure the lack of distant progression at least by CT is considered mandatory by all clinicians before radical treatment such as surgical resection is planned.
Functional Imaging
Biochemical and molecular imaging potentially offer significant advantages in evaluating histopathological response over anatomical imaging by detecting changes, for example, the decrease in cell proliferation, increase in cell death, and decline in the number of viable tumor cells that precede a decline in tumor size [40].
PET
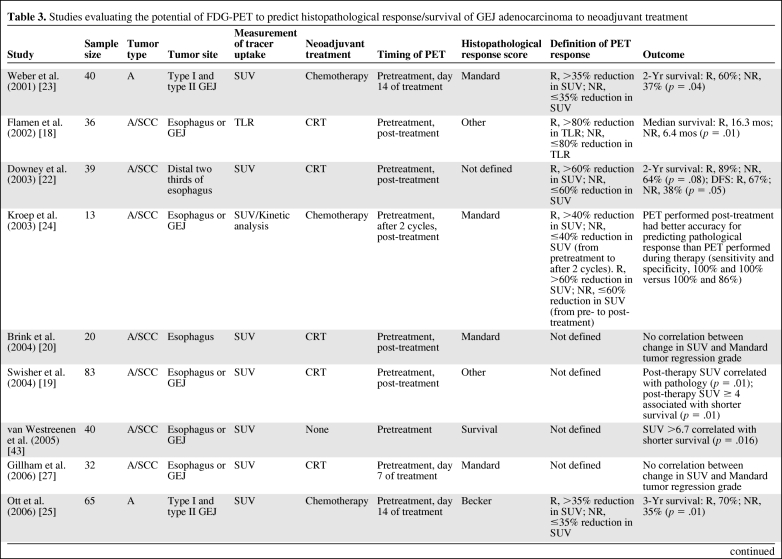
PET allows noninvasive visualization and quantitative assessment of physiological and biochemical processes within tumors [41]. [18F]-2-fluoro-2-deoxy-d-glucose (18FDG) is the glucose analog 2-deoxy-d-glucose labeled with 18F and is by far the most commonly used radiopharmaceutical for oncological PET studies. The use of 18FDG is based on differences in aerobic glycolysis between nonmalignant and malignant cells, although the molecular mechanisms that determine greater 18FDG uptake in tumors are inadequately understood [42]. Several studies have evaluated the potential of FDG-PET to predict histopathological response/survival with neoadjuvant treatment in GEJ cancers (Table 3).
Table 3.
Studies evaluating the potential of FDG-PET to predict histopathological response/survival of GEJ adenocarcinoma to neoadjuvant treatment
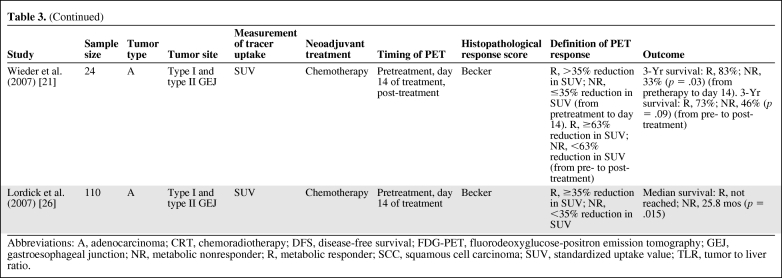
Table 3.
(Continued)
Abbreviations: A, adenocarcinoma; CRT, chemoradiotherapy; DFS, disease-free survival; FDG-PET, fluorodeoxyglucose-positron emission tomography; GEJ, gastroesophageal junction; NR, metabolic nonresponder; R, metabolic responder; SCC, squamous cell carcinoma; SUV, standardized uptake value; TLR, tumor to liver ratio.
Pretreatment FDG-PET
Conflicting results have been reported in the use of pretreatment FDG-PET to predict histopathological response [18–27, 43]. Several authors have demonstrated that the predictive value of changes in tumor 18FDG uptake with serial imaging during therapy is superior to measurements of absolute pretherapy 18FDG uptake (Table 3).
Serial FDG-PET
A number of studies have demonstrated the ability of serial FDG-PET imaging early in the course of neoadjuvant treatment to predict histopathological response (Table 3) [21, 23–26]. There are significant issues yet to be resolved with regard to the standardization of PET imaging protocols [44], and the optimal method and time point for repeat imaging is not fully resolved. Ideally, this would be as soon as possible after the start of treatment, thereby allowing potentially toxic therapy to be discontinued in those identified as nonresponders. Most studies performing FDG-PET imaging during therapy have performed them on day 14 of treatment [21, 23, 25, 26]. In contrast, Gillham et al. [27] performed them on day 7 of treatment. The optimal time point is likely to be different for chemotherapy than for chemoradiotherapy, and may be different again for novel targeted therapies. This most likely reflects the different therapeutic mechanisms of these different treatment modalities, for example, the acute and chronic inflammatory response to radiation and the contribution of host stromal reaction to therapy [45].
The strongest data supporting the use of FDG-PET as a predictive biomarker are derived from prospective studies in GEJ adenocarcinomas [21, 23, 25, 26]. The data are unicentric from a specialized, high-volume center performed using a PET, not PET-CT, scanner and clearly need multicentric evaluation. Initially, Weber et al. [23] demonstrated, in a prospective study of patients with locally advanced GEJ adenocarcinomas, that a reduction in metabolic activity after 2 weeks of chemotherapy was correlated with a subsequent decrease in tumor size, a higher rate of curative resections (p = .01), histopathological tumor regression (p = .001), and patient survival (p = .04). These prospectively collected data allowed the authors to define an optimal cutoff value of a 35% reduction in standard uptake value (SUV) (day 14 from baseline) to distinguish responding and nonresponding tumors with a sensitivity of 93% (95% confidence interval [CI], 68%–100%) and specificity of 95% (95% CI, 77%–100%) [23]. This definition was then validated prospectively in a larger population with longer follow-up. Metabolic responders (i.e., those with a reduction in 18FDG uptake of 35% 14 days after the initiation of therapy) showed a histopathological response rate of 44%, with a 3-year survival rate of 70%. In contrast, prognosis was poor for metabolic nonresponders, with a histopathological response rate of 5% (p = .001) and a 3-year survival rate of 35% (p = .01). A multivariate analysis demonstrated that metabolic response was the only factor that predicted recurrence (p = .018) in patients whose tumors were completely resected [25]. Early metabolic response (14 days after the start of therapy) provided at least the same accuracy for prediction of treatment outcome as with late 18FDG changes (3 months after the start of therapy) [21], and FDG-PET after completion of chemotherapy did not result in a higher accuracy for the prediction of histopathological response.
Subsequently, the Metabolic response evalUatioN for Individualisation of neoadjuvant Chemotherapy in esOphageal and esophagogastric adeNocarcinoma trial assessed the feasibility of a PET response–guided treatment algorithm. FDG-PET scans were performed at baseline and 14 days after the start of chemotherapy (i.e., after one cycle). Patients whose tumor SUV had decreased by ≥35% were defined as metabolic responders and went on to receive further chemotherapy before undergoing surgery. Metabolic nonresponders discontinued chemotherapy and proceeded to surgery. Metabolic responders were found to have a good long-term prognosis, with a median overall survival duration not yet reached, whereas nonresponders had a median overall survival time of 25.8 months (hazard ratio [HR] 2.13; 95% CI, 1.14–3.99; p = .015) [26]. Together with previous investigations, that study suggested that FDG-PET may provide an effective predictive biomarker to identify nonresponders to neoadjuvant chemotherapy, with a major histopathological response rate of <5% in FDG-PET early metabolic nonresponders, and a definitive randomized trial is needed and planned to determine clinical utility [25, 26]. However, whereas FDG-PET early metabolic responders had a higher histopathological response rate, approximately 50% of those predicted to have a response did not, and therefore do not receive clinical benefit from neoadjuvant therapy. This problem is clearly illustrated by the HR of 4.55 (95% CI, 1.37–15.04; p = .004) for survival between those who have an FDG-PET metabolic response and a major histopathological response and those who have an FDG-PET early metabolic response but no histopathological response [26]. Therefore, histopathological response after neoadjuvant chemotherapy remains the strongest indicator of long-term clinical outcome, and so has value as a prognostic indicator (assessed after therapy) but no predictive value to assist in planning of optimized neoadjuvant therapy (Fig. 1).
Accordingly, improvement in the accuracy of early prediction of response remains a key aim for research. A better understanding of the biological basis of FDG-PET metabolic response and subsequent histopathological response or nonresponse would be valuable and also provide insights into tumor biology that would be of therapeutic relevance. Although a change in 18FDG uptake has been demonstrated to be indicative of a lower viable cell number and lower rate of glucose metabolism per cell [45], the molecular pathways and mechanisms of a decrease in 18FDG uptake following cytotoxic chemotherapy are unknown and may be treatment and tumor type specific [46]. Caution is necessary in making unvalidated generalizations. In particular, studies based on examination of specific pathways and approaches have so far failed to provide a molecular basis for the greater uptake of 18FDG in tumors and the decrease that characterizes early metabolic response to therapy.
Molecular Predictive Biomarkers
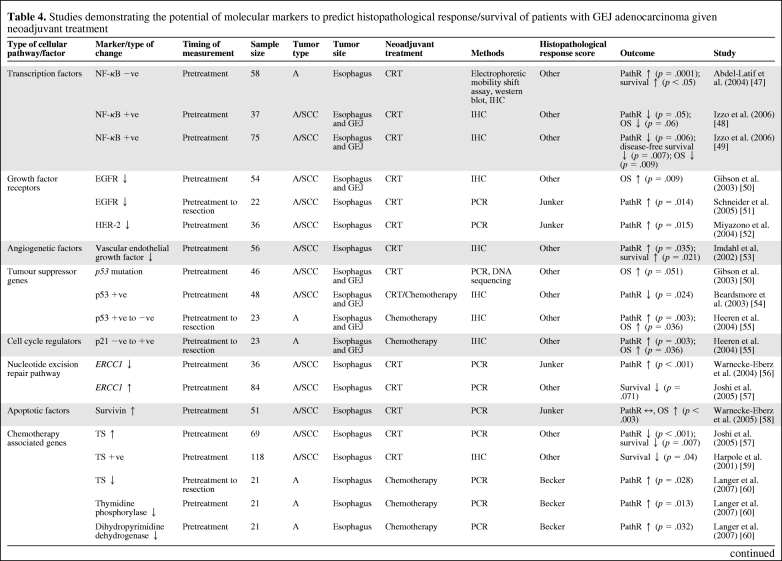
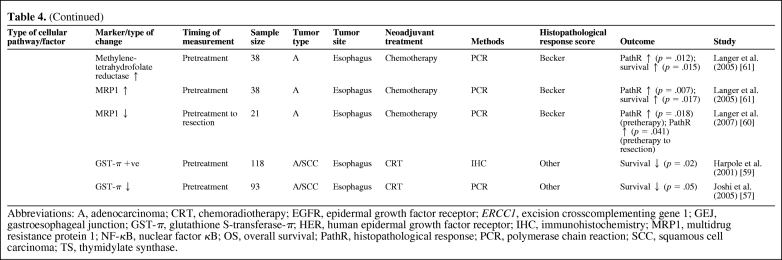
Table 4 summarizes the studies that have demonstrated the predictive value of a number of molecular biomarkers in assessing histopathological response/survival in GEJ cancer patients with neoadjuvant therapy [47–61]. None of the biomarkers available have been prospectively tested, and most studies are on small patient populations. Those molecular biomarkers that are likely to be relevant to future targeted therapies or have shown consistently positive results for histopathological response prediction are discussed below.
Table 4.
Studies demonstrating the potential of molecular markers to predict histopathological response/survival of patients with GEJ adenocarcinoma given neoadjuvant treatment
Table 4.
(Continued)
Abbreviations: A, adenocarcinoma; CRT, chemoradiotherapy; EGFR, epidermal growth factor receptor; ERCC1, excision crosscomplementing gene 1; GEJ, gastroesophageal junction; GST-π, glutathione S-transferase-π; HER, human epidermal growth factor receptor; IHC, immunohistochemistry; MRP1, multidrug resistance protein 1; NF-κB, nuclear factor κB; OS, overall survival; PathR, histopathological response; PCR, polymerase chain reaction; SCC, squamous cell carcinoma; TS, thymidylate synthase.
Nuclear Factor-κB
Nuclear factor-κB appears to hold promise as a predictive biomarker following neoadjuvant chemoradiotherapy [47–49, 62].
EGFR Family
Expression of EGFR is found in 29%–92% of patients with esophageal cancer [63]. Amplification of HER-2 has been seen in 15%–19% of esophageal adenocarcinomas [15, 64]. Although results for EGFR and histopathological response/survival prediction with chemoradiotherapy have generally been positive [50, 51], the method of assay of EGFR (immunohistochemistry [IHC], mutation) may determine the predictive impact, and optimal methods of assessment in this predictive context are yet to be resolved. The same applies to the use of novel EGFR-targeted agents, and both monoclonal antibodies and small molecules are in early-phase clinical trials in the neoadjuvant setting [65, 66]. Although limited, data so far suggest that EGFR mutation appears to be relatively infrequent in GEJ adenocarcinomas, and therefore clinically effective translation of EGFR mutation as a predictive biomarker for EGFR-targeted agents, as has been proven to be useful in non-small cell lung cancer (NSCLC) patients, seems unlikely. Similarly, although K-ras has clinical utility as a predictive biomarker for EGFR-targeted agents in colorectal adenocarcinomas and NSCLC, K-ras is wild type in 70%–80% of gastroesophageal cancer patients (much higher than in NSCLC and colorectal cancer patients), so its impact and use as a biomarker may be limited in gastroesophageal cancer.
Updated results of a preplanned subgroup analysis of the Trastuzumab in Gastric Cancer (ToGA) trial (a phase III randomized trial of cisplatin and 5-fluorouracil or capecitabine with or without trastuzumab in advanced gastric and GEJ adenocarcinomas) have suggested that HER-2 IHC (score 3+) with the use of HER-2 fluorescence in situ hybridization in borderline IHC (score 2+) cases predicts those patients who will benefit from the addition of trastuzumab [15], and these data are potentially transferable to predictive biomarker use in the neoadjuvant setting. Trastuzumab is being assessed in the neoadjuvant setting; however, results may have to be readdressed in light of the HER-2 predictive biomarker data from the ToGA trial [15].
Vascular Endothelial Growth Factor
A potential predictive role for vascular endothelial growth factor (VEGF) was reported by Imdahl et al. [53], who found that, for patients undergoing neoadjuvant chemoradiotherapy, a higher VEGF index in pretreatment biopsies correlated with a poorer histopathological response (p = .035) [53]. Several early-phase trials are investigating anti-VEGF or anti-VEGF receptor agents in GEJ patients in the advanced setting [67–70], and the U.K. STO3 phase II/III trial is evaluating standard neoadjuvant chemotherapy with or without bevacizumab in patients with gastric and type III GEJ adenocarcinomas.
Gene Expression Profiling
Luthra et al. [71] performed oligonucleotide microarrays on pretreatment endoscopic biopsies from 19 patients (16 with adenocarcinoma, two with squamous cell carcinoma, and one with adenosquamous carcinoma) prior to neoadjuvant chemoradiotherapy. Unsupervised hierarchical cluster analysis segregated the cancers into two molecular subtypes, consisting of 10 and nine specimens, respectively. Most cancers (five of six) that had a complete pathological response clustered in molecular subtype I. Subtype II, with one exception, consisted of cancers with less than a complete pathological response. Levels of p53 effector related to peripheral myelin protein 22, S100A2 (which encodes a calcium binding protein), and SPRR3 (a small proline-rich protein) allowed discrimination of complete from less than complete pathological response with high sensitivity and specificity (85%). Pathway analysis identified the apoptotic pathway as one of the key functions downregulated in molecular subtype II, in comparison with molecular subtype I [71].
Using cDNA microarrays to compare gene expression profiles, Greenawalt et al. [72] identified 3,516 genes that were differentially expressed among normal esophageal tissue, esophageal adenocarcinoma, squamous cell carcinoma, and Barrett's esophagus, and allowed separation in unsupervised hierarchical cluster analysis consistent with histology. These data may be useful to identify the molecular mechanisms underlying the different behavior and responsiveness to treatment of these histological disease subtypes [72].
Discussion
Mechanisms and Paradigms for Stratified Clinical Decision Making in Early GEJ Adenocarcinomas
A key translational research objective for GEJ adenocarcinomas is to define subgroups of patients with optimal survival from particular combined-modality treatment approaches. Clinical and radiological responses are not reliable as surrogates for survival. However, a consistent finding is that histopathological response identifies patients with longer survival times both after neoadjuvant chemotherapy and after chemoradiotherapy [5, 28, 29]. Histopathological response can only be determined at the time of resection and so the ability to predict histopathological response earlier in the course of neoadjuvant therapy would be beneficial. Prediction of histopathological response to neoadjuvant therapy would allow optimal combined-modality treatment selection, and the subsequent determination of histopathological response or nonresponse may facilitate subsequent clinical treatment decisions for postoperative (adjuvant) therapy. This provides a useful model for clinical investigation and a paradigm for stratified clinical decision making in early gastroesophageal adenocarcinoma that is potentially readily transferable to clinical practice (Figs. 1 and 2).
Current data do not provide definitive answers regarding the optimal methods to predict histopathological response. Recent guidance on biomarker development is useful and should be applied to subsequent investigations with predictive biomarkers in GEJ adenocarcinomas for both existing candidate biomarkers discussed here and novel markers emerging from discovery investigations [73–76]. However, in GEJ adenocarcinomas (and other solid tumor types), there is a clinical need to use biomarker development methodology to assess combinations of biomarkers, which necessarily complicates clinical protocols. Thus, in GEJ adenocarcinomas, current data suggest that FDG-PET may have utility as a predictive biomarker that has a high negative predictive value (95%) but limited positive predictive value (45%–50%) for histopathological response. These data support investigation in a prospective, randomized, clinical trial in which FDG-PET is used as a predictive biomarker to alter the clinical treatment decision for neoadjuvant therapy (and this study is planned—the European Organization for Research and Treatment of Cancer IMAGE trial). If confirmed, the use of FDG-PET as a predictive biomarker in this way would surpass the current use of therapy based on clinicopathological grounds; there is a clear clinical need to improve the positive predictive value. This implies the need to combine biomarker modalities, for example, with a tissue biomarker (from tumor, serum, or other biospecimen), providing a subclassification of FDG-PET–defined metabolic response to identify which metabolic responders will or will not have a histopathological response. There are several possible permutations of clinical application involving the combined use of a tissue biomarker and/or FDG-PET metabolic response as an imaging predictive biomarker that need to be investigated. One example is the utility of initial subclassification on the basis of a tumor-measured predictive tissue biomarker and then refinement/modification according to serial FDG-PET–measured metabolic response. Alternatively, the utility of serial tissue biomarker analysis alongside serial FDG-PET would be feasible with a serum biomarker, and there are other combinations that may also be useful and worthy of investigation. Furthermore, ongoing investigation must be set in the context of emerging novel agents and targets for gastroesophageal adenocarcinoma; however, clinical trial design should be able to accommodate this. Nevertheless, data with FDG-PET in chemotherapy versus chemoradiotherapy provide a potential warning regarding generalization to different treatment modalities and the need for optimization of each, which may be a matter of timing of assessment and/or PET technique (tracer or quantification).
It is not yet clear whether the subclassification of GEJ adenocarcinomas into type I, type II, and type III, which have different etiologies, will be relevant to systemic therapy. Nevertheless, emerging data suggest that it will be important for targeted therapies with potentially differential responses in these subtypes, and current clinical protocols have recognized this with subtype-specific inclusion criteria.
Predictive Biomarker Discovery in Early GEJ Adenocarcinomas
This review illustrates the need for ongoing discovery of predictive biomarkers of histopathological response and clinical benefit from distinct combined-modality treatment approaches in early GEJ adenocarcinomas. Molecular biomarker discovery must, to a certain extent, be driven by the requirements of emerging therapeutic targets and novel agents in preclinical and early-phase clinical development. So far, in molecular biomarker discovery in GEJ adenocarcinoma, the emphasis has been on pathway-specific approaches. More recently, hypothesis-generating approaches to discover biomarkers using “omic” platforms were used by Luthra et al. [71] and Greenawalt et al. [72]. These data are promising, and larger scale retrospective evaluation using archived and clinical trial–derived material is needed for hypothesis generation.
Aside from such “blue sky” omic discovery approaches, there are several aspects of current understanding and practice in GEJ adenocarcinomas that would benefit from discovery investigations that have the potential for rapid clinical translation with the application of a hypothesis-testing (as opposed to hypothesis-generating) approach:
The identification of biological and pathogenic differences among type I, type II, and type III GEJ adenocarcinomas
The molecular pathways and mechanisms responsible for metabolic and/or histopathological response
The extent of overlap between molecular mechanisms that define GEJ adenocarcinoma subtypes and metabolic and histopathological response.
Investigational approaches that select patients based on clinicopathological subtypes and that use tissue and imaging biomarkers in the same cohort of patients will be required to address these questions. Serial endoscopic tumor biopsies during therapy are also likely to be important. Data from such investigations should be “back translated” for mechanistic studies in preclinical models to confirm potential leads for predictive biomarkers for prospective evaluation in a combined clinicopathological, imaging, and tissue predictive biomarker strategy for stratified optimal treatment selection in early GEJ adenocarcinomas.
Acknowledgments
R.D.P. and G.B. are engaged in research in the area of gastroesophageal biomarkers funded by NHS Grampian, Chief Scientists Office, Scotland and the Friends of Aberdeen and the North Centre for Oncology Haematology and Radiotherapy.
Author Contributions
Conception/Design: Russell D. Petty
Manuscript writing: Russell D. Petty, Gillian H. Bain
Final approval of manuscript: Russell D. Petty, Gillian H. Bain
References
- 1.Brücher BL, Swisher SG, Königsrainer A, et al. Response to preoperative therapy in upper gastrointestinal cancers. Ann Surg Oncol. 2009;16:878–886. doi: 10.1245/s10434-009-0315-x. [DOI] [PubMed] [Google Scholar]
- 2.Medical Research Council Oesophageal Cancer Working Group. Surgical resection with or without preoperative chemotherapy in oesophageal cancer: A randomised controlled trial. Lancet. 2002;359:1727–1733. doi: 10.1016/S0140-6736(02)08651-8. [DOI] [PubMed] [Google Scholar]
- 3.Gebski V, Burmeister B, Smithers BM, et al. Survival benefits from neoadjuvant chemoradiotherapy or chemotherapy in oesophageal carcinoma: A meta-analysis. Lancet Oncol. 2007;8:226–234. doi: 10.1016/S1470-2045(07)70039-6. [DOI] [PubMed] [Google Scholar]
- 4.Cunningham D, Allum WH, Stenning SP, et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med. 2006;355:11–20. doi: 10.1056/NEJMoa055531. [DOI] [PubMed] [Google Scholar]
- 5.Kelsen DP, Winter KA, Gunderson LL, et al. Long-term results of RTOG trial 8911 (USA intergroup 113): A random assignment trial comparison of chemotherapy followed by surgery compared with surgery alone for esophageal cancer. J Clin Oncol. 2007;25:3719–3725. doi: 10.1200/JCO.2006.10.4760. [DOI] [PubMed] [Google Scholar]
- 6.Stahl M, Walz MK, Stuschke M, et al. Phase III comparison of preoperative chemotherapy compared with chemoradiotherapy in patients with locally advanced adenocarcinoma of the esophagogastric junction. J Clin Oncol. 2009;27:851–856. doi: 10.1200/JCO.2008.17.0506. [DOI] [PubMed] [Google Scholar]
- 7.Boige V, Pignon J, Saint-Aubert B, et al. Final results of a randomized trial comparing preoperative 5-fluorouracil (F)/cisplatin (P) to surgery alone in adenocarcinoma of stomach and lower esophagus (ASLE): FNLCC ACCORD07-FFCD 9703 trial. J Clin Oncol. 2007;25(18 suppl):4510. [Google Scholar]
- 8.Koshy M, Esiashvilli N, Landry JC, et al. Multiple management modalities in esophageal cancer: Epidemiology, presentation and progression, work-up, and surgical approaches. The Oncologist. 2004;9:137–146. doi: 10.1634/theoncologist.9-2-137. [DOI] [PubMed] [Google Scholar]
- 9.Pohl H, Welch HG. The role of overdiagnosis and reclassification in the marked increase of esophageal adenocarcinoma incidence. J Natl Cancer Inst. 2005;97:142–146. doi: 10.1093/jnci/dji024. [DOI] [PubMed] [Google Scholar]
- 10.Rüdiger Siewert J, Feith M, Werner M, et al. Adenocarcinoma of the esophagogastric junction: Results of surgical therapy based on anatomical/topographic classification in 1,002 consecutive patients. Ann Surg. 2000;232:353–361. doi: 10.1097/00000658-200009000-00007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008;58:71–96. doi: 10.3322/CA.2007.0010. [DOI] [PubMed] [Google Scholar]
- 12.Meier I, Merkel S, Papadopoulos T, et al. Adenocarcinoma of the esophagogastric junction: The pattern of metastatic lymph node dissemination as a rationale for elective lymphatic target volume definition. Int J Radiat Oncol Biol Phys. 2008;70:1408–1417. doi: 10.1016/j.ijrobp.2007.08.053. [DOI] [PubMed] [Google Scholar]
- 13.Chau I, Norman AR, Cunningham D, et al. The impact of primary tumour origins in patients with advanced oesophageal, oesophago-gastric junction and gastric adenocarcinoma—individual patient data from 1775 patients in four randomised controlled trials. Ann Oncol. 2009;20:885–891. doi: 10.1093/annonc/mdn716. [DOI] [PubMed] [Google Scholar]
- 14.Dragovich T, McCoy S, Fenoglio-Preiser CM, et al. Phase II trial of erlotinib in gastroesophageal junction and gastric adenocarcinomas: SWOG 0127. J Clin Oncol. 2006;24:4922–4927. doi: 10.1200/JCO.2006.07.1316. [DOI] [PubMed] [Google Scholar]
- 15.Chung H, Bang YJ, Xu JM, et al. Human epidermal growth factor receptor 2 (HER2) in gastric cancer (GC): Results of the ToGA trial screening programme and recommendations for HER2 testing. Eur J Cancer Suppl. 2009;7:6511. [Google Scholar]
- 16.Therasse P, Arbuck SG, Eisenhauer EA, et al. New guidelines to evaluate the response to treatment in solid tumours. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst. 2000;92:205–216. doi: 10.1093/jnci/92.3.205. [DOI] [PubMed] [Google Scholar]
- 17.Swisher SG, Maish M, Erasmus JJ, et al. Utility of PET, CT, and EUS to identify pathologic responders in esophageal cancer. Ann Thorac Surg. 2004;78:1152–1160. doi: 10.1016/j.athoracsur.2004.04.046. [DOI] [PubMed] [Google Scholar]
- 18.Flamen P, Van Cutsem E, Lerut A, et al. Positron emission tomography for assessment of the response to induction radiochemotherapy in locally advanced oesophageal cancer. Ann Oncol. 2002;13:361–368. doi: 10.1093/annonc/mdf081. [DOI] [PubMed] [Google Scholar]
- 19.Swisher SG, Erasmus J, Maish M, et al. 2-Fluoro-2-deoxy-D-glucose positron emission tomography imaging is predictive of pathologic response and survival after preoperative chemoradiation in patients with esophageal carcinoma. Cancer. 2004;101:1776–1785. doi: 10.1002/cncr.20585. [DOI] [PubMed] [Google Scholar]
- 20.Brink I, Hentschel M, Bley TA, et al. Effects of neoadjuvant radio-chemotherapy on 18F-FDG-PET in esophageal carcinoma. Eur J Surg Oncol. 2004;30:544–550. doi: 10.1016/j.ejso.2004.03.007. [DOI] [PubMed] [Google Scholar]
- 21.Wieder HA, Ott K, Lordick F, et al. Prediction of tumor response by FDG-PET: Comparison of the accuracy of single and sequential studies in patients with adenocarcinomas of the esophagogastric junction. Eur J Nucl Med Mol Imaging. 2007;34:1925–1932. doi: 10.1007/s00259-007-0521-3. [DOI] [PubMed] [Google Scholar]
- 22.Downey RJ, Akhurst T, Ilson D, et al. Whole body 18FDG-PET and the response of esophageal cancer to induction therapy: Results of a prospective trial. J Clin Oncol. 2003;21:428–432. doi: 10.1200/JCO.2003.04.013. [DOI] [PubMed] [Google Scholar]
- 23.Weber WA, Ott K, Becker K, et al. Prediction of response to preoperative chemotherapy in adenocarcinomas of the esophagogastric junction by metabolic imaging. J Clin Oncol. 2001;19:3058–3065. doi: 10.1200/JCO.2001.19.12.3058. [DOI] [PubMed] [Google Scholar]
- 24.Kroep JR, Van Groeningen CJ, Cuesta MA, et al. Positron emission tomography using 2-deoxy-2-[18F]-fluoro-D-glucose for response monitoring in locally advanced gastroesophageal cancer; a comparison of different analytical methods. Mol Imaging Biol. 2003;5:337–346. doi: 10.1016/j.mibio.2003.09.007. [DOI] [PubMed] [Google Scholar]
- 25.Ott K, Weber WA, Lordick F, et al. Metabolic imaging predicts response, survival, and recurrence in adenocarcinomas of the esophagogastric junction. J Clin Oncol. 2006;24:4692–4698. doi: 10.1200/JCO.2006.06.7801. [DOI] [PubMed] [Google Scholar]
- 26.Lordick F, Ott K, Krause BJ, et al. PET to assess early metabolic response and to guide treatment of adenocarcinoma of the oesophagogastric junction: The MUNICON phase II trial. Lancet Oncol. 2007;8:797–805. doi: 10.1016/S1470-2045(07)70244-9. [DOI] [PubMed] [Google Scholar]
- 27.Gillham CM, Lucey JA, Keogan M, et al. (18)FDG uptake during induction chemoradiation for oesophageal cancer fails to predict histomorphological tumour response. Br J Cancer. 2006;95:1174–1179. doi: 10.1038/sj.bjc.6603412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Berger AC, Farma J, Scott WJ, et al. Complete response to neoadjuvant chemoradiotherapy in esophageal carcinoma is associated with significantly improved survival. J Clin Oncol. 2005;23:4330–4337. doi: 10.1200/JCO.2005.05.017. [DOI] [PubMed] [Google Scholar]
- 29.Wu TT, Chirieac LR, Abraham SC, et al. Excellent interobserver agreement on grading the extent of residual carcinoma after preoperative chemoradiation in esophageal and esophagogastric junction carcinoma: A reliable predictor for patient outcome. Am J Surg Pathol. 2007;31:58–64. doi: 10.1097/01.pas.0000213312.36306.cc. [DOI] [PubMed] [Google Scholar]
- 30.Brücher BL, Becker K, Lordick F, et al. The clinical impact of histopathologic response assessment by residual tumor cell quantification in esophageal squamous cell carcinomas. Cancer. 2006;106:2119–2127. doi: 10.1002/cncr.21850. [DOI] [PubMed] [Google Scholar]
- 31.Becker K, Mueller JD, Schulmacher C, et al. Histomorphology and grading of regression in gastric carcinoma treated with neoadjuvant chemotherapy. Cancer. 2003;98:1521–1530. doi: 10.1002/cncr.11660. [DOI] [PubMed] [Google Scholar]
- 32.Mandard AM, Dalibard F, Mandard JC, et al. Pathologic assessment of tumor regression after preoperative chemoradiotherapy of esophageal carcinoma. Clinicopathologic correlations. Cancer. 1994;73:2680–2686. doi: 10.1002/1097-0142(19940601)73:11<2680::aid-cncr2820731105>3.0.co;2-c. [DOI] [PubMed] [Google Scholar]
- 33.Salzer-Kuntschik M, Delling G, Beron G, et al. Morphological grades of regression in osteosarcoma after polychemotherapy—study COSS 80. J Cancer Res Clin Oncol. 1983;106(suppl):21–24. doi: 10.1007/BF00625047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Junker K, Langner K, Klinke F, et al. Grading of tumor regression in non-small cell lung cancer: Morphology and prognosis. Chest. 2001;120:1584–1591. doi: 10.1378/chest.120.5.1584. [DOI] [PubMed] [Google Scholar]
- 35.Tepper JE, O'Neil B. Transition in biology and philosophy in the treatment of gastroesophageal junction adenocarcinoma. J Clin Oncol. 2009;27:836–837. doi: 10.1200/JCO.2008.19.5982. [DOI] [PubMed] [Google Scholar]
- 36.Forshaw MJ, Gossage JA, Chrystal K, et al. Symptomatic responses to neoadjuvant chemotherapy for carcinoma of the oesophagus and oesophagogastric junction: Are they worth measuring? Clin Oncol (R Coll Radiol) 2006;18:345–350. doi: 10.1016/j.clon.2006.02.008. [DOI] [PubMed] [Google Scholar]
- 37.Brown WA, Thomas J, Gotley D, et al. Use of oesophagogastroscopy to assess the response of oesophageal carcinoma to neoadjuvant therapy. Br J Surg. 2004;91:199–204. doi: 10.1002/bjs.4411. [DOI] [PubMed] [Google Scholar]
- 38.Jones DR, Parker LA, Jr, Detterbeck FC, et al. Inadequacy of computed tomography in assessing patients with esophageal carcinoma after induction chemoradiotherapy. Cancer. 1999;85:1026–1032. doi: 10.1002/(sici)1097-0142(19990301)85:5<1026::aid-cncr3>3.0.co;2-n. [DOI] [PubMed] [Google Scholar]
- 39.Westerterp M, van Westreenen HL, Reitsma JB, et al. Esophageal cancer: CT, endoscopic US, and FDG PET for assessment of response to neoadjuvant therapy—systematic review. Radiology. 2005;236:841–851. doi: 10.1148/radiol.2363041042. [DOI] [PubMed] [Google Scholar]
- 40.Mankoff DA, Eary JF, Link JM, et al. Tumor-specific positron emission tomography imaging in patients: [18F] fluorodeoxyglucose and beyond. Clin Cancer Res. 2007;13:3460–3469. doi: 10.1158/1078-0432.CCR-07-0074. [DOI] [PubMed] [Google Scholar]
- 41.Weber WA. Positron emission tomography as an imaging biomarker. J Clin Oncol. 2006;24:3282–3292. doi: 10.1200/JCO.2006.06.6068. [DOI] [PubMed] [Google Scholar]
- 42.Pauwels EK, Sturm EJ, Bombardieri E, et al. Positron-emission tomography with [18F]fluorodeoxyglucose. Part I. Biochemical uptake mechanism and its implication for clinical studies. J Cancer Res Clin Oncol. 2000;126:549–559. doi: 10.1007/PL00008465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.van Westreenen HL, Plukker JT, Cobben DC, et al. Prognostic value of the standardized uptake value in esophageal cancer. AJR Am J Roentgenol. 2005;185:436–440. doi: 10.2214/ajr.185.2.01850436. [DOI] [PubMed] [Google Scholar]
- 44.Lordick F, Ruers T, Aust DE, et al. European Organisation of Research and Treatment of Cancer (EORTC) Gastrointestinal Group: Workshop on the role of metabolic imaging in the neoadjuvant treatment of gastrointestinal cancer. Eur J Cancer. 2008;44:1807–1819. doi: 10.1016/j.ejca.2008.06.005. [DOI] [PubMed] [Google Scholar]
- 45.Spaepen K, Stroobants S, Dupont P, et al. [18F]FDG PET monitoring of tumour response to chemotherapy: Does [18F]FDG uptake correlate with the viable tumour cell fraction? Eur J Nucl Med Mol Imaging. 2003;30:682–688. doi: 10.1007/s00259-003-1120-6. [DOI] [PubMed] [Google Scholar]
- 46.Weber WA, Petersen V, Schmidt B, et al. Positron emission tomography in non-small-cell lung cancer: Prediction of response to chemotherapy by quantitative assessment of glucose use. J Clin Oncol. 2003;21:2651–2657. doi: 10.1200/JCO.2003.12.004. [DOI] [PubMed] [Google Scholar]
- 47.Abdel-Latif MM, O'Riordan J, Windle HJ, et al. NF-κB activation in esophageal adenocarcinoma: Relationship to Barrett's metaplasia, survival, and response to neoadjuvant chemoradiotherapy. Ann Surg. 2004;239:491–500. doi: 10.1097/01.sla.0000118751.95179.c6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Izzo JG, Malhotra U, Wu TT, et al. Association of activated transcription factor nuclear factor κB with chemoradiation resistance and poor outcome in esophageal carcinoma. J Clin Oncol. 2006;24:748–754. doi: 10.1200/JCO.2005.03.8810. [DOI] [PubMed] [Google Scholar]
- 49.Izzo JG, Correa AM, Wu TT, et al. Pretherapy nuclear factor-κB status, chemoradiation resistance, and metastatic progression in esophageal carcinoma. Mol Cancer Ther. 2006;5:2844–2850. doi: 10.1158/1535-7163.MCT-06-0351. [DOI] [PubMed] [Google Scholar]
- 50.Gibson MK, Abraham SC, Wu TT, et al. Epidermal growth factor receptor, p53 mutation, and pathological response predict survival in patients with locally advanced esophageal cancer treated with preoperative chemoradiotherapy. Clin Cancer Res. 2003;9:6461–6468. [PubMed] [Google Scholar]
- 51.Schneider S, Uchida K, Brabender J, et al. Downregulation of TS, DPD, ERCC1, GST-Pi, EGFR, and HER2 gene expression after neoadjuvant three-modality treatment in patients with esophageal cancer. J Am Coll Surg. 2005;200:336–344. doi: 10.1016/j.jamcollsurg.2004.10.035. [DOI] [PubMed] [Google Scholar]
- 52.Miyazono F, Metzger R, Warnecke-Eberz U, et al. Quantitative c-erbB-2 but not c-erbB-1 mRNA expression is a promising marker to predict minor histopathologic response to neoadjuvant radiochemotherapy in oesophageal cancer. Br J Cancer. 2004;91:666–672. doi: 10.1038/sj.bjc.6601976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Imdahl A, Bognar G, Schulte-Mönting J, et al. Predictive factors for response to neoadjuvant therapy in patients with oesophageal cancer. Eur J Cardiothorac Surg. 2002;21:657–663. doi: 10.1016/s1010-7940(02)00044-1. [DOI] [PubMed] [Google Scholar]
- 54.Beardsmore DM, Verbeke CS, Davies CL, et al. Apoptotic and proliferative indexes in esophageal cancer: Predictors of response to neoadjuvant therapy [corrected] J Gastrointest Surg. 2003;7:77–86. discussion 86–87. [PubMed] [Google Scholar]
- 55.Heeren PA, Kloppenberg FW, Hollema H, et al. Predictive effect of p53 and p21 alteration on chemotherapy response and survival in locally advanced adenocarcinoma of the esophagus. Anticancer Res. 2004;24:2579–2583. [PubMed] [Google Scholar]
- 56.Warnecke-Eberz U, Metzger R, Miyazono F, et al. High specificity of quantitative excision repair cross-complementing 1 messenger RNA expression for prediction of minor histopathological response to neoadjuvant radiochemotherapy in esophageal cancer. Clin Cancer Res. 2004;10:3794–3799. doi: 10.1158/1078-0432.CCR-03-0079. [DOI] [PubMed] [Google Scholar]
- 57.Joshi MB, Shirota Y, Danenberg KD, et al. High gene expression of TS1, GSTP1, and ERCC1 are risk factors for survival in patients treated with trimodality therapy for esophageal cancer. Clin Cancer Res. 2005;11:2215–2221. doi: 10.1158/1078-0432.CCR-04-1387. [DOI] [PubMed] [Google Scholar]
- 58.Warnecke-Eberz U, Hokita S, Xi H, et al. Overexpression of survivin mRNA is associated with a favorable prognosis following neoadjuvant radiochemotherapy in esophageal cancer. Oncol Rep. 2005;13:1241–1246. [PubMed] [Google Scholar]
- 59.Harpole DH, Jr, Moore MB, Herndon JE, 2nd, et al. The prognostic value of molecular marker analysis in patients treated with trimodality therapy for esophageal cancer. Clin Cancer Res. 2001;7:562–569. [PubMed] [Google Scholar]
- 60.Langer R, Specht K, Becker K, et al. Comparison of pretherapeutic and posttherapeutic expression levels of chemotherapy-associated genes in adenocarcinomas of the esophagus treated by 5-fluorouracil- and cisplatin-based neoadjuvant chemotherapy. Am J Clin Pathol. 2007;128:191–197. doi: 10.1309/1U6X4L9XFJLJV940. [DOI] [PubMed] [Google Scholar]
- 61.Langer R, Specht K, Becker K, et al. Association of pretherapeutic expression of chemotherapy-related genes with response to neoadjuvant chemotherapy in Barrett carcinoma. Clin Cancer Res. 2005;11:7462–7469. doi: 10.1158/1078-0432.CCR-05-0042. [DOI] [PubMed] [Google Scholar]
- 62.Abdel-Latif MM, O'Riordan JM, Ravi N, et al. Activated nuclear factor-kappa B and cytokine profiles in the esophagus parallel tumor regression following neoadjuvant chemoradiotherapy. Dis Esophagus. 2005;18:246–252. doi: 10.1111/j.1442-2050.2005.00497.x. [DOI] [PubMed] [Google Scholar]
- 63.Kuwano H, Kato H, Miyazaki T, et al. Genetic alterations in esophageal cancer. Surg Today. 2005;35:7–18. doi: 10.1007/s00595-004-2885-3. [DOI] [PubMed] [Google Scholar]
- 64.Reichelt U, Duesedau P, Tsourlakis MC, et al. Frequent homogeneous HER-2 amplification in primary and metastatic adenocarcinoma of the esophagus. Mod Pathol. 2007;20:120–129. doi: 10.1038/modpathol.3800712. [DOI] [PubMed] [Google Scholar]
- 65.Sgroi MM, Hanna NH, McCollum AD, et al. Preoperative cetuximab and radiation (XRT) for patients (pts) with surgically resectable esophageal and gastroesophageal (GE) junction carcinomas: A pilot study from the Hoosier Oncology Group and the University of Texas-Southwestern. J Clin Oncol. 2008;26(15 suppl):4564. [Google Scholar]
- 66.Safran H, DiPetrillo T, Akerman P, et al. Phase I/II study of trastuzumab, paclitaxel, cisplatin and radiation for locally advanced, HER2 overexpressing, esophageal adenocarcinoma. Int J Radiat Oncol Biol Phys. 2007;67:405–409. doi: 10.1016/j.ijrobp.2006.08.076. [DOI] [PubMed] [Google Scholar]
- 67.Shah MA, Ramanathan RK, Ilson DH, et al. Multicenter phase II study of irinotecan, cisplatin, and bevacizumab in patients with metastatic gastric or gastroesophageal junction adenocarcinoma. J Clin Oncol. 2006;24:5201–5206. doi: 10.1200/JCO.2006.08.0887. [DOI] [PubMed] [Google Scholar]
- 68.Enzinger PC, Ryan DP, Regan EM, et al. Phase II trial of docetaxel, cisplatin, irinotecan, and bevacizumab in metastatic esophagogastric cancer. J Clin Oncol. 2008;26(15 suppl):4552. [Google Scholar]
- 69.Jhawer MP, Ilson D, Robinson E, et al. Interim results of a phase II study of modified docetaxel, cisplatin, fluorouracil (mDCF), and bevacizumab (BEV) in patients with metastatic gastroesophageal (GR) adenocarcinoma [abstract 109]. Presented at the American Society of Clinical Oncology 2008 Gastrointestinal Cancers Symposium; January 25–27, 2008; Orlando, FL. [Google Scholar]
- 70.Sun W, Powell ME, O'Dwyer P, et al. A phase II study: Combination of sorafenib with docetaxel and cisplatin in the treatment of metastatic or advanced unresectable gastric and gastroesophageal junction (GEJ) adenocarcinoma (ECOG 5203) J Clin Oncol. 2008;26(15 suppl):4535. doi: 10.1200/JCO.2009.27.7988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Luthra R, Wu TT, Luthra MG, et al. Gene expression profiling of localized esophageal carcinomas: Association with pathologic response to preoperative chemoradiation. J Clin Oncol. 2006;24:259–267. doi: 10.1200/JCO.2005.03.3688. [DOI] [PubMed] [Google Scholar]
- 72.Greenawalt DM, Duong C, Smyth GK, et al. Gene expression profiling of esophageal cancer: Comparative analysis of Barrett's esophagus, adenocarcinoma, and squamous cell carcinoma. Int J Cancer. 2007;120:1914–1921. doi: 10.1002/ijc.22501. [DOI] [PubMed] [Google Scholar]
- 73.McShane LM, Altman DG, Sauerbrei W, et al. Reporting recommendations for tumor marker prognostic studies (REMARK) J Natl Cancer Inst. 2005;97:1180–1184. doi: 10.1093/jnci/dji237. [DOI] [PubMed] [Google Scholar]
- 74.Hayes DF, Bast RC, Desch CE, et al. Tumor marker utility grading system: A framework to evaluate clinical utility of tumor markers. J Natl Cancer Inst. 1996;88:1456–1466. doi: 10.1093/jnci/88.20.1456. [DOI] [PubMed] [Google Scholar]
- 75.Cummings J, Ward TH, Greystoke A, et al. Biomarker method validation in anticancer drug development. Br J Pharmacol. 2008;153:646–656. doi: 10.1038/sj.bjp.0707441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Cancer Research UK. Prognostic/Predictive Biomarker Roadmap- guide to applying for a Cancer Research UK Biomarker. [accessed October 9, 2009]. Available at http://science.cancerresearchuk.org/reps/pdfs/bidd_prognostic_roadmap.pdf.
- 77.Pandeya N, Williams G, Green AC, et al. Alcohol consumption and the risks of adenocarcinoma and squamous cell carcinoma of the esophagus. Gastroenterology. 2009;136:1215–1224. doi: 10.1053/j.gastro.2008.12.052. [DOI] [PubMed] [Google Scholar]
- 78.Siewert JR, Ott K. Are squamous and adenocarcinomas of the esophagus the same disease? Semin Radiat Oncol. 2006;17:38–44. doi: 10.1016/j.semradonc.2006.09.007. [DOI] [PubMed] [Google Scholar]
- 79.Siewert JR, Stein HJ, Feith M, et al. Histologic tumor type is an independent prognostic parameter in esophageal cancer: Lessons from more than 1,000 consecutive resections at a single center in the Western world. Ann Surg. 2001;234:360–367. doi: 10.1097/00000658-200109000-00010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Mariette C, Finzi L, Piessen G, et al. Esophageal carcinoma: Prognostic differences between squamous cell carcinoma and adenocarcinoma. World J Surg. 2005;29:39–45. doi: 10.1007/s00268-004-7542-x. [DOI] [PubMed] [Google Scholar]
- 81.Cooper JS, Guo MD, Herskovic A, et al. Chemoradiotherapy of locally advanced esophageal cancer: Long-term follow-up of a prospective randomized trial (RTOG 85–01). Radiation Therapy Oncology Group. JAMA. 1999;281:1623–1627. doi: 10.1001/jama.281.17.1623. [DOI] [PubMed] [Google Scholar]
- 82.Stahl M, Stuschke M, Lehmann N, et al. Chemoradiation with and without surgery in patients with locally advanced squamous cell carcinoma of the esophagus. J Clin Oncol. 2005;23:2310–2317. doi: 10.1200/JCO.2005.00.034. [DOI] [PubMed] [Google Scholar]
- 83.Bedenne L, Michel P, Bouché O, et al. Chemoradiation followed by surgery compared with chemoradiation alone in squamous cancer of the esophagus: FFCD 9102. J Clin Oncol. 2007;25:1160–1168. doi: 10.1200/JCO.2005.04.7118. [DOI] [PubMed] [Google Scholar]
- 84.Whiteman DC, Sadeghi S, Pandeya N, et al. Combined effects of obesity, acid reflux and smoking on the risk of adenocarcinomas of the oesophagus. Gut. 2008;57:173–180. doi: 10.1136/gut.2007.131375. [DOI] [PubMed] [Google Scholar]
- 85.von Rahden BH, Stein HJ, Feith M. Lymphatic vessel invasion as a prognostic factor in patients with primary resected adenocarcinomas of the esophagogastric junction. J Clin Oncol. 2005;23:874–879. doi: 10.1200/JCO.2005.12.151. [DOI] [PubMed] [Google Scholar]
- 86.Derakhshan MH, Malekzadeh R, Watabe H, et al. Combination of gastric atrophy, reflux symptoms and histological subtype indicates two distinct aetiologies of gastric cardia cancer. Gut. 2008;57:298–305. doi: 10.1136/gut.2007.137364. [DOI] [PubMed] [Google Scholar]
- 87.Aikou T, Shimazu H. Difference in main lymphatic pathways from the lower esophagus and gastric cardia. Jpn J Surg. 1989;19:290–295. doi: 10.1007/BF02471404. [DOI] [PubMed] [Google Scholar]
- 88.Van Cutsem E, Van de Velde C, Roth A, et al. Expert opinion on management of gastric and gastro-oesophageal junction adenocarcinoma on behalf of the European Organisation for Research and Treatment of Cancer (EORTC)-gastrointestinal cancer group. Eur J Cancer. 2008;44:182–194. doi: 10.1016/j.ejca.2007.11.001. [DOI] [PubMed] [Google Scholar]