This retrospective analysis of the US Oncology network's electronic medical records evaluated the association between progression-free survival and overall survival times and bevacizumab treatment to progression among patients with advanced non-small cell lung cancer.
Keywords: Bevacizumab, Non-small cell lung cancer, Chemotherapy, Progression-free survival
Abstract
Bevacizumab significantly extends progression-free survival (PFS) and overall survival (OS) times when combined with initial chemotherapy and continued as monotherapy until disease progression or unacceptable toxicity in patients with nonsquamous non-small cell lung cancer (NSCLC). In clinical practice, bevacizumab is sometimes discontinued after completion of chemotherapy. This retrospective analysis of the US Oncology network's electronic medical records evaluated the association between PFS and OS times and bevacizumab monotherapy to progression (BTP) among patients with advanced NSCLC.
Patients treated from July 2006 through June 2008 were analyzed as two cohorts based on whether or not they received BTP after completion of first-line chemotherapy plus bevacizumab. Hazard ratios for PFS and OS were estimated using Cox proportional hazards, adjusting for relevant treatment and patient characteristics. To account for survivorship bias, landmark analyses were conducted at 18, 21, and 26 weeks from initial therapy to examine residual PFS and OS times, defined as the time from the landmark to disease progression or death.
From the total 498 nonsquamous NSCLC patients, 403 received first-line chemotherapy plus bevacizumab: 154 received BTP, 249 did not. Longer PFS and OS times were observed in patients who received BTP than in those who received no BTP (median OS, 20.9 months versus 10.2 months; median PFS, 10.3 months versus 6.5 months). BTP was associated with a longer residual OS time at all specified landmarks and longer residual PFS time at week 18 than with no BTP.
In conclusion, this retrospective analysis provides supportive evidence that continued vascular endothelial growth factor suppression in advanced nonsquamous NSCLC patients is associated with favorable clinical outcomes.
Introduction
Lung cancer is the second most common cancer diagnosed in the U.S. and is the leading cause of cancer-related deaths. In 2009, an estimated 219,440 new patients were diagnosed and 159,390 patients succumbed to the disease [1]. Non-small cell lung cancer (NSCLC) accounts for >80% of all lung cancer cases. The large majority (70%) of advanced NSCLC patients are diagnosed with advanced stage (stage III/IV) disease. For these patients, the prognosis tends to be poor, with 5-year survival rates estimated at 9%–25% for patients with stage III disease and only 2% for patients with stage IV disease [2]. Traditional chemotherapy regimens include platinum-based doublets, such as carboplatin plus paclitaxel and cisplatin plus gemcitabine, and more recent regimens such as cisplatin plus pemetrexed. Although there is some evidence [3] that newer treatment options have led to incremental improvements in overall survival (OS) and progression-free survival (PFS) times, there is currently no universally accepted standard regimen for the first-line treatment of advanced NSCLC patients.
Inhibition of vascular endothelial growth factor (VEGF), an epithelial cell–specific mitogen, has become an important modality in cancer therapy because of the mechanistic role of angiogenesis in tumor progression [4]. Bevacizumab (Avastin®; Genentech, Inc., South San Francisco, CA), a humanized monoclonal antibody targeting VEGF, was approved by the U.S. Food and Drug Administration (FDA) in 2006 for the first-line treatment of advanced NSCLC patients in combination with carboplatin and paclitaxel. Numerous clinical trials have demonstrated the safety and efficacy of bevacizumab (BV) when combined with chemotherapy (CT) for the treatment of advanced NSCLC patients. A phase II trial [5] provided initial evidence of efficacy, as demonstrated by a longer PFS interval. In 2006, the results of the phase III Eastern Cooperative Oncology Group (ECOG) 4599 study demonstrated longer OS and PFS times in patients treated with carboplatin, paclitaxel, and BV [3]. A second phase III study [6] demonstrated a longer PFS interval in the BV plus CT arm. In the ECOG 4599 and Avastin in Lung trial, BV was administered through all cycles of CT and then continued as monotherapy until disease progression or unacceptable toxicity, supported by preclinical evidence that sustained VEGF inhibition maintains tumor regression [7, 8]. In clinical practice, however, BV is often discontinued after completion of CT.
With the increasing use of electronic medical records (EMRs) in the oncology setting, there will be new opportunities to explore real-world clinical outcomes and to generate hypotheses on the comparative effectiveness of therapies, including the incremental benefit of maintenance therapy. The goal of this retrospective EMR-based study was to evaluate the potential association between BTP and OS and PFS among advanced nonsquamous NSCLC patients receiving first-line CT plus BV in the outpatient community setting.
Materials and Methods
Data Source
Clinical data were abstracted from iKnowMed (iKM), US Oncology Inc.'s (The Woodlands, TX) oncology-specific EMR system, which contains data from 884 community-based oncologists across US Oncology practices or clinics in 20 states. Patient characteristics abstracted from iKM included age, gender, region of practice, payor status, clinical stage, histologic type, Karnofsky performance status (KPS) score, body mass index (BMI), hemoglobin (Hgb) level, and location of metastases. The KPS score, Hgb level, and BMI were measured prior to initiation and after completion of CT. Treatment characteristics included dose and duration of CT and BV. Documented vital status in iKM was supplemented with data from the Social Security Death Master File to identify additional decedents. The institutional review board at US Oncology approved the use of institutional patient data for this study.
Study Population
All patients with advanced nonsquamous NSCLC who were initiated on a first-line CT regimen plus BV were retrospectively identified within the US Oncology network between July 1, 2006 and June 30, 2008. Patients were followed to June 30, 2009 or the date of death/last follow-up date for the purpose of measuring clinical outcomes. Identified patients had a potential follow-up duration of 12–36 months. Patients who were enrolled in a clinical study or received care for any cancer other than nonsquamous NSCLC during the study period were also excluded.
Patients were analyzed in two treatment cohorts based on whether or not they continued to receive BV monotherapy to disease progression (BTP) following the completion of first-line combination therapy with CT and BV. In order to be included in the BTP cohort, patients had to have been initiated on BV monotherapy within 30 days following their final CT administration. Using an intent-to-treat approach, patients who received BV monotherapy within 30 days following CT completion, but did not continue until disease progression, were assigned to the BTP cohort. Patients who progressed within 30 days following completion of CT were excluded from the study because they would not have had the opportunity to receive BV monotherapy following completion of CT and as such could not be appropriately assigned to either cohort.
Statistical Analyses
Descriptive summaries of patient characteristics were conducted overall and by treatment cohort to describe the study population and to identify potential underlying differences between treatment cohorts. Outcomes included the OS time measured from the initiation of CT until death or loss to follow-up and the PFS interval measured from the initiation of CT to progression, which was defined by an escalation in line of therapy or death. Following the completion of first-line therapy, the date of progression was identified as the date that a patient was initiated on second-line therapy. This escalation in line of therapy is documented in a standardized fashion in iKM. Switches in regimens resulting from toxicity or other nonprogression events do not lead to an escalation in line of therapy. Patients who were lost to follow-up or who had not experienced disease progression or death were censored at their last clinic visit date or the end of the study period (June 30, 2009), respectively.
Standard survival analysis techniques were used for estimating and comparing OS and PFS times (Kaplan–Meier and log-rank tests, Cox proportional hazard regression). The proportionality assumption was confirmed using the goodness-of-fit test developed by Harrell and Lee [9]. Covariates and stratification variables that were considered in the primary modeling included age, gender, clinical stage at diagnosis, KPS score, BMI, Hgb level, and dose of BV during CT. Backwards elimination was used to identify significant covariates for inclusion in the Cox regression models. Statistical significance was determined at an α = 0.05 level and all tests of significance were two-sided.
Landmark analyses were performed to address the possibility of survivorship and selection bias that results from patients surviving long enough to have the opportunity to receive BTP [10–12]. Three landmark timepoints were chosen for this study—18, 21, and 26 weeks. Only patients who were alive and progression free at the time of the landmarks were included. Residual OS and PFS times were measured with the index date assigned as the landmark timepoint with follow-up to death or progressive disease, respectively. Cox proportional hazard models were run for each of the landmarks.
Sensitivity Analyses
Sensitivity analyses were performed to evaluate the robustness of the associations observed in this study. Because patients in the BTP cohort received a greater number of CT cycles than those in the no BTP cohort, this covariate was included in a secondary Cox model in order to better understand the differences in OS and PFS between the BTP and no BTP cohorts. Furthermore, whereas the primary analysis used an intent-to-treat approach in which patients were included in the BTP cohort if they received BV monotherapy that was discontinued >21 days prior to disease progression, we conducted secondary analyses in which these patients were assigned to the no BTP cohort to assess whether or not this alternate assignment to BTP cohorts would impact the results. Finally, to assess whether differences in second-line therapies might have contributed to differences in OS between the BTP and no BTP cohorts, second-line therapies that patients received following their first disease progression were described.
Results
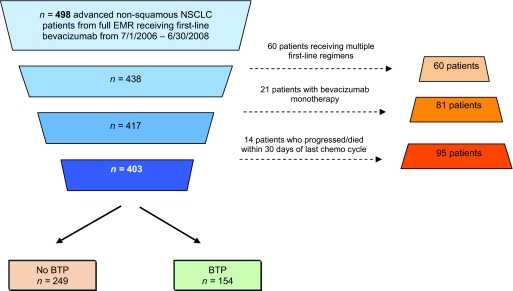
We identified 498 advanced nonsquamous NSCLC patients who received first-line CT with BV from July 1, 2006 to June 30, 2008 (Fig. 1). Of these 498 patients, 95 were excluded because their regimens were modified or changed during the course of their first-line therapy (n = 60), they received only BV monotherapy without CT (n = 21), or they progressed/died within 30 days of completion of CT (n = 14). The final study population included 403 patients: 249 (62%) did not continue receiving BV monotherapy following the completion of CT (no BTP cohort) and 154 (38%) continued to receive BV monotherapy following the completion of CT (BTP cohort). In the BTP cohort, 19 patients received BV after CT but it was discontinued >21 days prior to disease progression.
Figure 1.
Nonsquamous NSCLC patients identified for the BTP and no BTP cohorts.
Abbreviations: BTP, bevacizumab monotherapy to progression; EMR, electronic medical record; NSCLC, non-small cell lung cancer.
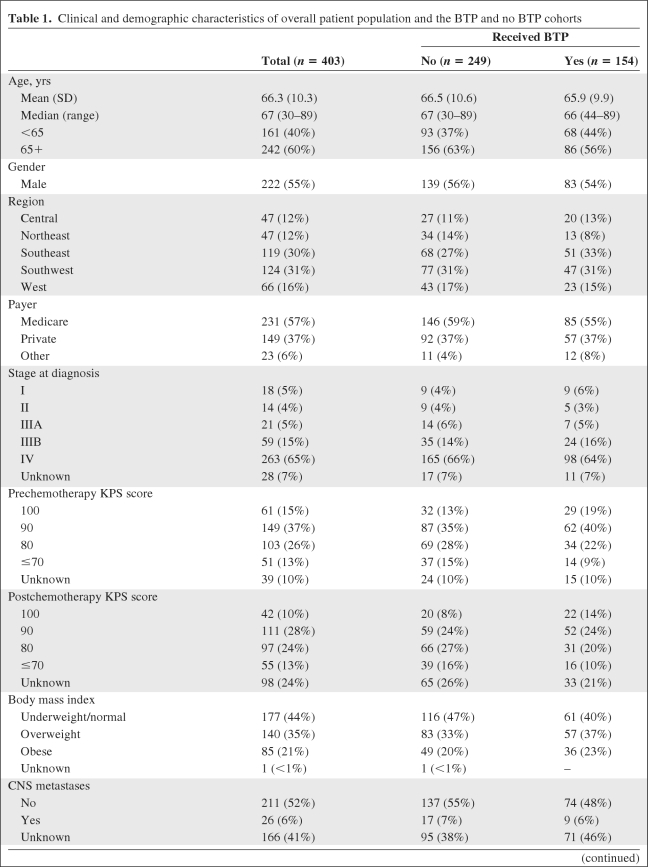
Table 1 presents clinical/demographic characteristics overall and by treatment cohort. For the entire cohort, the median age was 67 years (range, 30–89 years), 55% (n = 222) were male, 80% (n = 322) were diagnosed with advanced stage disease (stage IIIB and stage IV) and the remainder were diagnosed with earlier disease stage but later progressed to metastatic disease, and 83% (n = 336) were treated with combination platinum- and taxane-based therapy. The majority of patients (61%) were treated at practices in the southeast (30%) and southwest (31%) regions of the U.S., whereas the remainder were treated in the west (16%), central (12%), and northeast (12%) regions. Given the age of this population, most (57%) were Medicare patients. Baseline and treatment characteristics for the BTP and no BTP cohorts were similar except that the BTP cohort tended to have better KPS scores prior to CT and a greater number of completed CT cycles.
Table 1.
Clinical and demographic characteristics of overall patient population and the BTP and no BTP cohorts
Table 1a.
(Continued)
Abbreviations: BTP, bevacizumab monotherapy to progression; CNS, central nervous system; KPS, Karnofsy performance status.
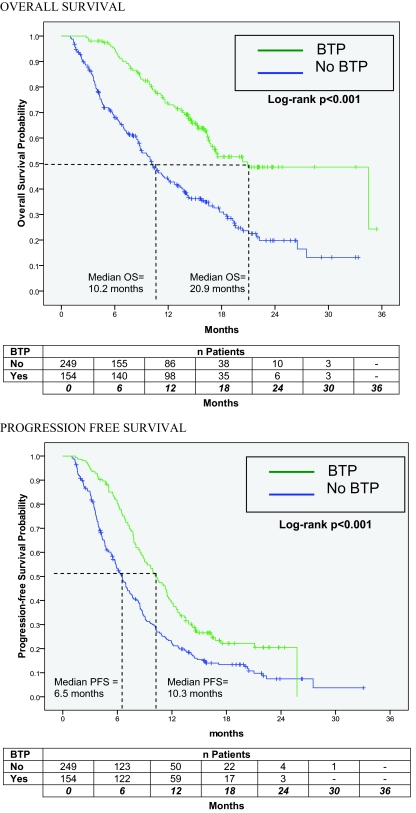
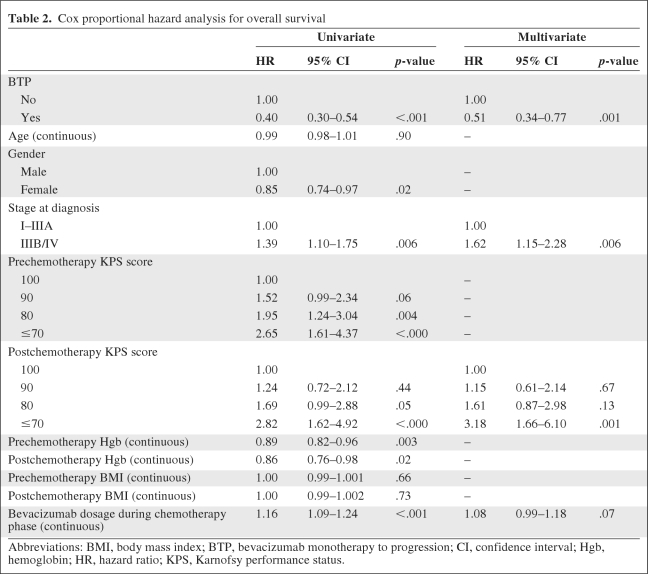
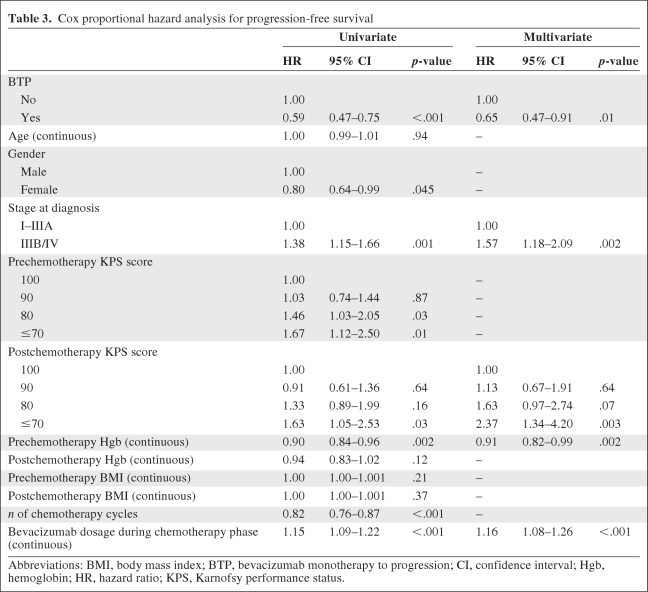
Unadjusted Kaplan–Meier estimates of the median OS and PFS times for the BTP and no BTP cohorts are shown in Figure 2. The median OS time for the BTP cohort was significantly longer than that for the no BTP cohort (20.9 months versus 10.2 months, respectively). The PFS duration was also significantly longer in the BTP cohort than in the no BTP cohort (10.3 months versus 6.5 months, respectively). In a Cox regression analysis using a backwards elimination approach (Table 2), the BTP cohort was found to have a 49% lower overall mortality risk than the no BTP cohort (hazard ratio [HR], 0.51; 95% confidence interval [CI], 0.34–0.77) after adjusting for stage at diagnosis, post-CT KPS score, and BV dose during the CT phase. Patients in the BTP cohort had a 35% lower risk for disease progression relative to those in the no BTP cohort (HR, 0.65; 95% CI, 0.47–0.91) after controlling for stage at diagnosis, post-CT KPS score, post-CT Hgb, and BV dose during the CT phase (Table 3).
Figure 2.
Kaplan–Meier estimates of OS and PFS for the BTP and no BTP cohorts (n = 403 patients).
Abbreviations: BTP, bevacizumab monotherapy to progression; NSCLC, non-small cell lung cancer; OS, overall survival; PFS, progression-free survival.
Table 2.
Cox proportional hazard analysis for overall survival
Abbreviations: BMI, body mass index; BTP, bevacizumab monotherapy to progression; CI, confidence interval; Hgb, hemoglobin; HR, hazard ratio; KPS, Karnofsy performance status.
Table 3.
Cox proportional hazard analysis for progression-free survival
Abbreviations: BMI, body mass index; BTP, bevacizumab monotherapy to progression; CI, confidence interval; Hgb, hemoglobin; HR, hazard ratio; KPS, Karnofsy performance status.
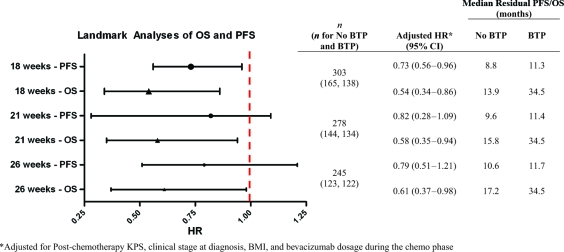
Landmark Analyses
To control for survivorship bias between the BTP and no BTP cohorts, landmark analyses were conducted at 18, 21, and 26 weeks from treatment initiation to evaluate the association between BTP and the residual OS and PFS times from these landmarks (Fig. 3). BTP remained associated with a longer residual OS time at each landmark. Among those who were alive and progression free at 18 weeks (no BTP, n = 165; BTP, n = 138), BTP was associated with a 46% lower risk for death (HR, 0.54; 95% CI, 0.34–0.86). This association between BTP and a longer residual OS time persisted among patients who remained progression free and alive at 21 weeks (HR, 0.58; 95% CI, 0.35–0.94) and at 26 weeks (HR, 0.61; 95% CI, 0.37–0.98).
Figure 3.
Landmark analyses for residual overall and progression-free survival at 18, 21, and 26 weeks.
Abbreviations: BMI, body mass index; BTP, bevacizumab monotherapy to progression; CI, confidence interval; HR, hazard ratio; KPS, Karnofsky performance status; OS, overall survival; PFS, progression-free survival.
In landmark analyses of PFS, BTP was associated with a longer PFS time at 18 weeks (HR, 0.73; 95% CI, 0.56–0.96), but the association was no longer observed at 21 weeks (HR, 0.82; 95% CI, 0.28–1.09) and 26 weeks (HR, 0.79; 95% CI, 0.51–1.21).
Sensitivity Analyses
Sensitivity analyses were conducted to evaluate the robustness of our observed associations. Because the BTP and no BTP cohorts differed in the number of CT cycles they received, we conducted secondary analyses wherein the number of CT cycles was included as a covariate in the Cox models. Point estimates for OS remained similar when the number of CT cycles was added to the model (HR, 0.55; 95% CI, 0.37–0.84). However, the association between BTP and PFS was attenuated when the number of CT cycles was added to the model (HR, 0.75; 95% CI, 0.53–1.06).
Sensitivity analyses were also carried out to determine how cohort assignment affected our results. Although the primary strategy for cohort assignment included an intent-to-treat approach in which patients (n = 19) with BV monotherapy that was discontinued prior to disease progression were included in the BTP cohort, secondary analyses were conducted in which those patients were reassigned to the no BTP cohort. When these patients were assigned to the no BTP cohort, the median OS times (20.9 months versus 10.9 months, respectively) and median PFS times (10.1 months versus 6.9 months, respectively) for the BTP and no BTP cohorts were similar to those observed in the original analyses. In the landmark analyses, BTP remained significantly associated with OS at 18 weeks (HR, 0.56; 95% CI, 0.35–0.88) and 21 weeks (HR, 0.61; 95% CI, 0.38–0.98), but was only marginally associated with OS at 26 weeks (HR, 0.61; 95% CI, 0.36–1.02). BTP remained associated with PFS at 18 weeks (HR, 0.68; 95% CI, 0.47–0.98) but not at 21 weeks (HR, 0.78; 95% CI, 0.53–1.14) or 26 weeks (HR, 0.77; 95% CI, 0.52–1.14).
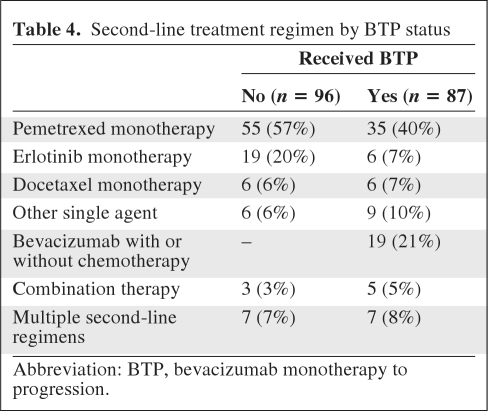
To assess whether differences in second-line therapies might have contributed to differences in OS between the BTP and no BTP cohorts, second-line therapies that patients received following their first disease progression were analyzed. Thirty-nine percent of patients in the no BTP cohort (n = 96) versus 56% (n = 87) of patients in the BTP cohort received second-line therapy. A large proportion of patients received second-line monotherapy with pemetrexed, erlotinib, docetaxel, or another agent (90% in the no BTP cohort, 64% in the BTP cohort). The most notable difference in second-line use between the BTP and no BTP cohorts was that 21% of patients (n = 19) in the BTP cohort continued to receive BV in the second-line setting, whereas no patient in the no BTP cohort received second-line BV (Table 4).
Table 4.
Second-line treatment regimen by BTP status
Abbreviation: BTP, bevacizumab monotherapy to progression.
Discussion
In this retrospective study, we observed longer OS and PFS times among patients who received BTP. We found that patients who received BTP had favorable OS (49% lower risk) and PFS (35% lower risk) risks relative to patients who discontinued BV at the same time CT was discontinued. The association between OS and BTP persisted in all landmark analyses of residual survival, whereas BTP remained significantly associated with PFS in the overall population and among patients who were progression free at 18 weeks. Among patients who were progression free at 21 and 26 weeks, the association between BTP and PFS was not statistically significant. Although the nonrandomized nature of this study precludes us from making strong conclusions related to causality and does not completely account for inherent selection bias related to treatment selection, some important inferences can be made based on the observed associations.
Results of this study estimating the population-based effectiveness of BTP demonstrated that the median OS times for the BTP and no BTP cohorts were 21.0 months and 10.1 months, respectively, with a median OS time for the overall study population of 13.8 months. The demonstrated OS duration of 21 months in the BTP arm is significantly longer than the median OS of 12.3 months that was previously published from the ECOG 4599 clinical trial of first-line BV plus CT treatment followed by BV monotherapy as maintenance treatment [3]. Because the key difference between our two cohorts was whether or not BV was continued as maintenance monotherapy, we postulated that this large difference in OS could also be attributed to other clinical variables, such as best response to therapy or tolerability of therapy. Our study is unable to distinguish between these possibilities. In addition, this difference in OS might be partly attributed to the fact that patients in this nonrandomized study were preselected and may have had more favorable prognostic factors than patients who were included in the original pivotal trials. Furthermore, because the data for this study are very recent, it is possible that the availability of more treatment choices today may have impacted the observed outcomes relative to what was observed in past trials.
To better understand what factors may be influencing the decision to discontinue BV treatment after CT, we assessed adverse events, including BV-related toxicities, in the two cohorts and whether there were differences by practice. From the initiation of BV plus CT treatment to 30 days following the patients' last treatment, the overall rate of grade ≥3 toxicities in the BTP cohort (1.9%) was similar to that in the no BTP cohort (1.6%). The rate of all toxicity grades was greater in the BTP cohort (11.7%) than in the no BTP cohort (6.8%). Although we acknowledge that toxicities may be underreported in EMRs, because physicians may not systematically complete the toxicity fields for all patients, these rates suggest that tolerability to treatment did not appear to be a driving factor in the decision to discontinue BV after CT. We also did not observe any notable differences in physician practice of BTP versus no BTP. Of the 45 clinics that contributed patient data to this study, 36 (80%) prescribed both BTP and no BTP, six clinics (13%) prescribed exclusively no BTP, and three clinics (7%) treated exclusively with BTP for their patients.
To the best of our knowledge, this is the first large, nationwide and community-based study evaluating the association between the use of BTP and clinical outcomes in NSCLC patients. The strengths of this study include the use of clinically detailed EMR data that allowed for a very detailed characterization of the treatment, clinical, and demographic characteristics of the patient population. As opposed to claims-based data sources, the use of oncology-specific EMR data, as is available through iKM, provides the opportunity to more closely mimic the data that are typically incorporated into the analysis of randomized clinical trials (RCTs). Also, the use of EMR data allows for the evaluation of all age ranges, as opposed to claims-based studies that may be limited to a younger managed care population or older Medicare population. Because community-based clinical practice may differ from the clinical trial setting, it is critical to conduct observational research to evaluate how clinical trial treatment protocols are translated to the real-world setting and to measure resultant outcomes. While RCTs provide the highest level of causal evidence in a setting in which physician and patient compliance is closely monitored and subjects are strictly selected (i.e., strong internal validity), observational research provides an opportunity to complement the current literature and to generate hypotheses for future clinical studies. Relative to RCTs, the application of sound study design and analytic methods to geographically dispersed population-based data has the potential to yield results that are more generalizable to community-based care, may be more reflective of real-world outcomes, and provide an opportunity to bridge the gap between clinical efficacy and real-world effectiveness.
This study had its limitations. Although the study population was representative of the general age distribution of advanced NSCLC patients, there was an underrepresentation of the Medicaid and indigent care population, which may limit generalizability of the results to this patient population. An inherent limitation and challenge in conducting any retrospective observational research stems from the fact that patients are not randomly assigned to comparison groups. As such, it is impossible to rule out the possibility of underlying selection bias with regard to cohort assignment. We attempted to address this concern to the greatest extent possible by characterizing patients according to several clinical and treatment factors that may have confounded the observed associations and then by controlling for these factors in the statistical analyses. Furthermore, comparisons of therapies that differ in duration introduce a risk for survivorship bias because assignment to comparison groups is dependent on therapy duration, which is to some degree correlated with survival. To minimize and evaluate the potential impact of this survivorship bias on our study results, patients who died or progressed in their disease within 30 days of completion of CT were excluded and landmark analyses were conducted to evaluate the association between BTP and residual PFS among subsets of the study population who remained progression free at predetermined times. Most importantly, it is impossible to fully determine from this data source and study design whether the longer OS and PFS times observed in the BTP cohort were secondary to therapy or simply a reflection of an inherent selection bias whereby patients receiving BTP were those who were observed to be responding better to the therapy. Physicians may have been more conscientious in their continuation of BV maintenance in those patients who were having an excellent response, exhibiting disease control, or tolerating therapy particularly well. In these cases, the survival advantage we observed could be viewed as a proxy for these favorable clinical factors.
Despite the potential biases in an observational study, these results have an important role in clinical decision making. Treatment options have expanded with the recent FDA approvals of erlotinib and pemetrexed as first-line maintenance treatments for advanced NSCLC patients. Erlotinib and pemetrexed have resulted in longer OS times (1 month and 2.8 months, respectively) and PFS times (0.2 and 1.7 months, respectively) than with placebo [13, 14]. Since 2005, when ECOG 4599 trial data were first presented to the American Society of Clinical Oncology [15], we have attempted to understand the benefits of BV given with CT versus its benefits as a single agent in the maintenance setting. In the absence of published RCTs that address this very specific question, these results demonstrate that continuing BV in certain populations may provide clinically meaningful outcomes. Whether or not these subpopulations can be characterized as patients who are responding to or tolerating CT well will remain questions for future studies.
In conclusion, this study provides supportive evidence that advanced nonsquamous NSCLC patients receiving continued VEGF suppression with BV had more favorable clinical outcomes than patients who discontinued BV when CT was completed. Although this study was unable to substantiate a causal link between BTP and better clinical outcomes, it highlights the utility of using EMR data in observational research. Further, the study findings provide significant insight into real-world patterns of care and associated outcomes and provide important evidence on which to base future comparative effectiveness research.
Author Contributions
Conception/Design: Eric Nadler, Steve Gruschkus, Amy Pratt Sing, Michael Forsyth, Elaine Yu, Arliene Ravelo
Financial support: Elaine Yu, Arliene Ravelo
Administrative support: Elaine Yu, Arliene Ravelo
Provision of study material or patients: Steve Gruschkus
Collection and/or assembly of data: Steve Gruschkus
Data analysis and interpretation: Eric Nadler, Steve Gruschkus, Elaine Yu, Arliene Ravelo
Manuscript writing: Eric Nadler, Steve Gruschkus, Amy Pratt Sing, Michael Forsyth, Elaine Yu, Arliene Ravelo
Final approval of manuscript: Eric Nadler, Steve Gruschkus, Amy Pratt Sing, Michael Forsyth, Elaine Yu, Arliene Ravelo
References
- 1.American Cancer Society. Statistics for 2009: Cancer Facts & Figures, 2009. [accessed March 11, 2011]. Available at http://www.cancer.org/Research/CancerFactsFigures/CancerFactsFigures/cancer-facts-figures-2009.
- 2.Goldstraw P, Crowley J, Chansky K, et al. The IASLC Lung Cancer Staging Project: Proposals for the revision of the TNM stage groupings in the forthcoming (seventh) edition of the TNM classification of malignant tumours. J Thorac Oncol. 2007;2:706–714. doi: 10.1097/JTO.0b013e31812f3c1a. [DOI] [PubMed] [Google Scholar]
- 3.Sandler A, Gray R, Perry MC, et al. Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell lung cancer. N Engl J Med. 2006;355:2542–2550. doi: 10.1056/NEJMoa061884. [DOI] [PubMed] [Google Scholar]
- 4.Folkman J. Tumor angiogenesis: Therapeutic implications. N Engl J Med. 1971;285:1182–1186. doi: 10.1056/NEJM197111182852108. [DOI] [PubMed] [Google Scholar]
- 5.Johnson DH, Fehrenbacher L, Novotny WF, et al. Randomized phase II trial comparing bevacizumab plus carboplatin and paclitaxel with carboplatin and paclitaxel alone in previously untreated locally advanced or metastatic non-small-cell lung cancer. J Clin Oncol. 2004;22:2184–2191. doi: 10.1200/JCO.2004.11.022. [DOI] [PubMed] [Google Scholar]
- 6.Hirsh V, Ramlau R, von Pawel J, et al. Final safety results of BO17704 (AVAiL): A phase III randomized study of first-line bevacizumab combined with cisplatin/gemcitabine (CG) in patients with advanced or recurrent non-squamous, non-small cell lung cancer (NSCLC) [abstract 8039] J Clin Oncol. 2009;27(15 suppl):416s. [Google Scholar]
- 7.Klement G, Baruchel S, Rak J, et al. Continuous low-dose therapy with vinblastine and VEGF receptor-2 antibody induces sustained tumor regression without overt toxicity. J Clin Invest. 2000;105:R15–R24. doi: 10.1172/JCI8829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Klement G, Huang P, Mayer B, et al. Differences in therapeutic indexes of combination metronomic chemotherapy and an anti-VEGFR-2 antibody in multidrug-resistant human breast cancer xenografts. Clin Cancer Res. 2002;8:221–232. [PubMed] [Google Scholar]
- 9.Harrel F, Lee K. Verifying assumptions of the proportional hazards model. Proc 11th Annual SAS Users Group Int. 1986;11:823–828. [Google Scholar]
- 10.Anderson JR, Cain KC, Gelber RD. Analysis of survival by tumor response. J Clin Oncol. 1983;1:710–719. doi: 10.1200/JCO.1983.1.11.710. [DOI] [PubMed] [Google Scholar]
- 11.Anderson JR, Cain KC, Gelber RD. Analysis of survival by tumor response and other comparisons of time-to-event by outcome variables. J Clin Oncol. 2008;26:3913–3915. doi: 10.1200/JCO.2008.16.1000. [DOI] [PubMed] [Google Scholar]
- 12.Lara PN, Jr, Redman MW, Kelly K, et al. Disease control rate at 8 weeks predicts clinical benefit in advanced non-small-cell lung cancer: Results from Southwest Oncology Group randomized trials. J Clin Oncol. 2008;26:463–467. doi: 10.1200/JCO.2007.13.0344. [DOI] [PubMed] [Google Scholar]
- 13.Cappuzzo F, Ciuleanu T, Stelmakh L, et al. Erlotinib as maintenance treatment in advanced non-small-cell lung cancer: A multicentre, randomised, placebo-controlled phase 3 study. Lancet Oncol. 2010;11:521–529. doi: 10.1016/S1470-2045(10)70112-1. [DOI] [PubMed] [Google Scholar]
- 14.Ciuleanu T, Brodowicz T, Zielinski C, et al. Maintenance pemetrexed plus best supportive care versus placebo plus best supportive care for non-small-cell lung cancer: A randomised, double-blind, phase 3 study. Lancet. 2009;24:1432–1440. doi: 10.1016/S0140-6736(09)61497-5. [DOI] [PubMed] [Google Scholar]
- 15.Sandler AB, Gray R, Brahmer J, et al. Randomized phase II/III trial of paclitaxel (P) plus carboplatin (C) with or without bevacizumab (NSC # 704865) in patients with advanced non-squamous non-small cell lung cancer (NSCLC): An Eastern Cooperative Oncology Group (ECOG) trial - E4599. J Clin Oncol. 2005;23(suppl 16):4. [Google Scholar]