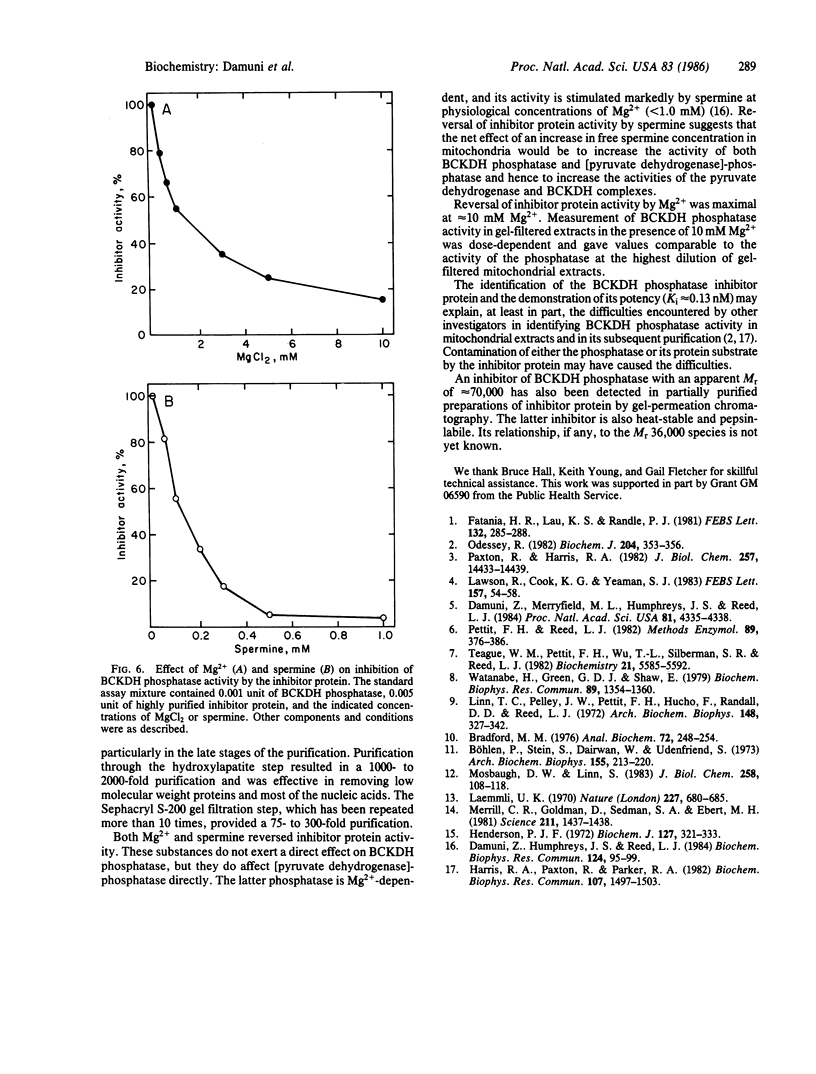
Abstract
A heat- and acid-stable protein inhibitor of the [branched-chain alpha-keto acid dehydrogenase]-phosphatase was purified over 100,000-fold from extracts of bovine kidney mitochondria. The nearly homogeneous protein was recovered with a yield of 4-8%. The apparent molecular weight of the inhibitor is about 36,000. This protein is a noncompetitive inhibitor of the phosphatase, and the inhibitor constant (Ki) is about 0.13 nM. The inhibition was reversed 50% by about 1.3 mM Mg2+ and about 0.1 mM spermine. This protein inhibitor is different from the cytosolic protein phosphatase inhibitors 1 and 2.
Full text
PDF
Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Böhlen P., Stein S., Dairman W., Udenfriend S. Fluorometric assay of proteins in the nanogram range. Arch Biochem Biophys. 1973 Mar;155(1):213–220. doi: 10.1016/s0003-9861(73)80023-2. [DOI] [PubMed] [Google Scholar]
- Damuni Z., Humphreys J. S., Reed L. J. Stimulation of pyruvate dehydrogenase phosphatase activity by polyamines. Biochem Biophys Res Commun. 1984 Oct 15;124(1):95–99. doi: 10.1016/0006-291x(84)90921-5. [DOI] [PubMed] [Google Scholar]
- Damuni Z., Merryfield M. L., Humphreys J. S., Reed L. J. Purification and properties of branched-chain alpha-keto acid dehydrogenase phosphatase from bovine kidney. Proc Natl Acad Sci U S A. 1984 Jul;81(14):4335–4338. doi: 10.1073/pnas.81.14.4335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fatania H. R., Lau K. S., Randle P. J. Inactivation of purified ox kidney branched-chain 2-oxoacid dehydrogenase complex by phosphorylation. FEBS Lett. 1981 Sep 28;132(2):285–288. doi: 10.1016/0014-5793(81)81180-5. [DOI] [PubMed] [Google Scholar]
- Harris R. A., Paxton R., Parker R. A. Activation of the branched-chain alpha-ketoacid dehydrogenase complex by a broad specificity protein phosphatase. Biochem Biophys Res Commun. 1982 Aug 31;107(4):1497–1503. doi: 10.1016/s0006-291x(82)80168-x. [DOI] [PubMed] [Google Scholar]
- Henderson P. J. A linear equation that describes the steady-state kinetics of enzymes and subcellular particles interacting with tightly bound inhibitors. Biochem J. 1972 Apr;127(2):321–333. doi: 10.1042/bj1270321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lawson R., Cook K. G., Yeaman S. J. Rapid purification of bovine kidney branched-chain 2-oxoacid dehydrogenase complex containing endogenous kinase activity. FEBS Lett. 1983 Jun 27;157(1):54–58. doi: 10.1016/0014-5793(83)81115-6. [DOI] [PubMed] [Google Scholar]
- Linn T. C., Pelley J. W., Pettit F. H., Hucho F., Randall D. D., Reed L. J. -Keto acid dehydrogenase complexes. XV. Purification and properties of the component enzymes of the pyruvate dehydrogenase complexes from bovine kidney and heart. Arch Biochem Biophys. 1972 Feb;148(2):327–342. doi: 10.1016/0003-9861(72)90151-8. [DOI] [PubMed] [Google Scholar]
- Merril C. R., Goldman D., Sedman S. A., Ebert M. H. Ultrasensitive stain for proteins in polyacrylamide gels shows regional variation in cerebrospinal fluid proteins. Science. 1981 Mar 27;211(4489):1437–1438. doi: 10.1126/science.6162199. [DOI] [PubMed] [Google Scholar]
- Mosbaugh D. W., Linn S. Excision repair and DNA synthesis with a combination of HeLa DNA polymerase beta and DNase V. J Biol Chem. 1983 Jan 10;258(1):108–118. [PubMed] [Google Scholar]
- Odessey R. Purification of rat kidney branched-chain oxo acid dehydrogenase complex with endogenous kinase activity. Biochem J. 1982 Apr 15;204(1):353–356. doi: 10.1042/bj2040353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paxton R., Harris R. A. Isolation of rabbit liver branched chain alpha-ketoacid dehydrogenase and regulation by phosphorylation. J Biol Chem. 1982 Dec 10;257(23):14433–14439. [PubMed] [Google Scholar]
- Pettit F. H., Reed L. J. Pyruvate dehydrogenase complex from bovine kidney and heart. Methods Enzymol. 1982;89(Pt 500):376–386. doi: 10.1016/s0076-6879(82)89067-8. [DOI] [PubMed] [Google Scholar]
- Teague W. M., Pettit F. H., Wu T. L., Silberman S. R., Reed L. J. Purification and properties of pyruvate dehydrogenase phosphatase from bovine heart and kidney. Biochemistry. 1982 Oct 26;21(22):5585–5592. doi: 10.1021/bi00265a031. [DOI] [PubMed] [Google Scholar]
- Usa T., Morimoto Y., Fukuda T., Anami K., Setoguchi M., Maruyama Y. [Pharmacological studies on Y-8894. (III). Its effect on the abnormal electrocorticogram induced by destruction of the internal capsule]. Nihon Yakurigaku Zasshi. 1986 Oct;88(4):289–297. doi: 10.1254/fpj.88.289. [DOI] [PubMed] [Google Scholar]
- Watanabe H., Green G. D., Shaw E. A comparison of the behavior of chymotrypsin and cathepsin B towards peptidyl diazomethyl ketones. Biochem Biophys Res Commun. 1979 Aug 28;89(4):1354–1360. doi: 10.1016/0006-291x(79)92158-2. [DOI] [PubMed] [Google Scholar]