Abstract
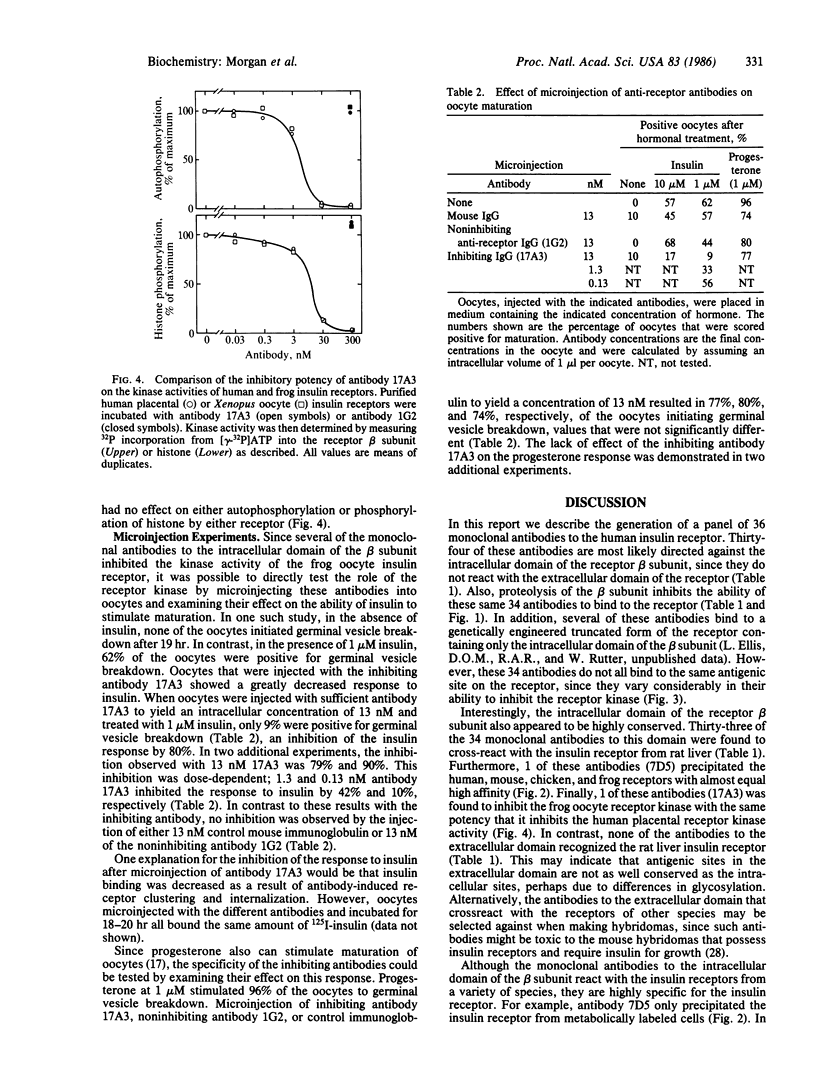
Thirty-six monoclonal antibodies to the human insulin receptor were produced. Thirty-four bound the intracellular domain of the receptor beta subunit, the domain containing the tyrosine-specific kinase activity. Of these 34 antibodies, 33 recognized the rat receptor and 1 was shown to precipitate the receptors from mice, chickens, and frogs with high affinity. Another of the antibodies inhibited the kinase activities of the human and frog receptors with equal potencies. This antibody inhibited the kinase activities of these receptors by more than 90%, whereas others had no effect on either kinase activity. Microinjection of the inhibiting antibody into Xenopus oocytes blocked the ability of insulin to stimulate oocyte maturation. In contrast, this inhibiting antibody did not block the ability of progesterone to stimulate the same response. Furthermore, control immunoglobulin and a noninhibiting antibody to the receptor beta subunit did not block this response to insulin. These results strongly support a role for the tyrosine-specific kinase activity of the insulin receptor in mediating this biological effect of insulin.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Avruch J., Alexander M. C., Palmer J. L., Pierce M. W., Nemenoff R. A., Blackshear P. J., Tipper J. P., Witters L. A. Role of insulin-stimulated protein phosphorylation in insulin action. Fed Proc. 1982 Aug;41(10):2629–2633. [PubMed] [Google Scholar]
- Chang T. H., Steplewski Z., Koprowski H. Production of monoclonal antibodies in serum free medium. J Immunol Methods. 1980;39(4):369–375. doi: 10.1016/0022-1759(80)90237-9. [DOI] [PubMed] [Google Scholar]
- Dumont J. N. Oogenesis in Xenopus laevis (Daudin). I. Stages of oocyte development in laboratory maintained animals. J Morphol. 1972 Feb;136(2):153–179. doi: 10.1002/jmor.1051360203. [DOI] [PubMed] [Google Scholar]
- Ebina Y., Ellis L., Jarnagin K., Edery M., Graf L., Clauser E., Ou J. H., Masiarz F., Kan Y. W., Goldfine I. D. The human insulin receptor cDNA: the structural basis for hormone-activated transmembrane signalling. Cell. 1985 Apr;40(4):747–758. doi: 10.1016/0092-8674(85)90334-4. [DOI] [PubMed] [Google Scholar]
- El-Etr M., Schorderet-Slatkine S., Baulieu E. E. Meiotic maturation in Xenopus laevis oocytes initiated by insulin. Science. 1979 Sep 28;205(4413):1397–1399. doi: 10.1126/science.472755. [DOI] [PubMed] [Google Scholar]
- Feldman R. A., Wang L. H., Hanafusa H., Balduzzi P. C. Avian sarcoma virus UR2 encodes a transforming protein which is associated with a unique protein kinase activity. J Virol. 1982 Apr;42(1):228–236. doi: 10.1128/jvi.42.1.228-236.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grigorescu F., Flier J. S., Kahn C. R. Defect in insulin receptor phosphorylation in erythrocytes and fibroblasts associated with severe insulin resistance. J Biol Chem. 1984 Dec 25;259(24):15003–15006. [PubMed] [Google Scholar]
- Grunberger G., Zick Y., Gorden P. Defect in phosphorylation of insulin receptors in cells from an insulin-resistant patient with normal insulin binding. Science. 1984 Mar 2;223(4639):932–934. doi: 10.1126/science.6141638. [DOI] [PubMed] [Google Scholar]
- Hedo J. A., Simpson I. A. Internalization of insulin receptors in the isolated rat adipose cell. Demonstration of the vectorial disposition of receptor subunits. J Biol Chem. 1984 Sep 10;259(17):11083–11089. [PubMed] [Google Scholar]
- Heldin C. H., Westermark B. Growth factors: mechanism of action and relation to oncogenes. Cell. 1984 May;37(1):9–20. doi: 10.1016/0092-8674(84)90296-4. [DOI] [PubMed] [Google Scholar]
- Kull F. C., Jr, Jacobs S., Su Y. F., Svoboda M. E., Van Wyk J. J., Cuatrecasas P. Monoclonal antibodies to receptors for insulin and somatomedin-C. J Biol Chem. 1983 May 25;258(10):6561–6566. [PubMed] [Google Scholar]
- Köhler G., Milstein C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature. 1975 Aug 7;256(5517):495–497. doi: 10.1038/256495a0. [DOI] [PubMed] [Google Scholar]
- Maller J. L., Koontz J. W. A study of the induction of cell division in amphibian oocytes by insulin. Dev Biol. 1981 Jul 30;85(2):309–316. doi: 10.1016/0012-1606(81)90262-1. [DOI] [PubMed] [Google Scholar]
- Morgan D. O., Roth R. A. Plate binding assay for monoclonal anti-receptor antibodies. Endocrinology. 1985 Mar;116(3):1224–1226. doi: 10.1210/endo-116-3-1224. [DOI] [PubMed] [Google Scholar]
- Reynhout J. K., Taddei C., Smith L. D., LaMarca M. J. Response of large oocytes of Xenopus laevis to progesterone in vitro in relation to oocyte size and time after previous HCG-induced ovulation. Dev Biol. 1975 Jun;44(2):375–379. doi: 10.1016/0012-1606(75)90408-x. [DOI] [PubMed] [Google Scholar]
- Roth R. A., Cassell D. J. Insulin receptor: evidence that it is a protein kinase. Science. 1983 Jan 21;219(4582):299–301. doi: 10.1126/science.6849137. [DOI] [PubMed] [Google Scholar]
- Roth R. A., Cassell D. J., Maddux B. A., Goldfine I. D. Regulation of insulin receptor kinase activity by insulin mimickers and an insulin antagonist. Biochem Biophys Res Commun. 1983 Aug 30;115(1):245–252. doi: 10.1016/0006-291x(83)90996-8. [DOI] [PubMed] [Google Scholar]
- Roth R. A., Cassell D. J., Wong K. Y., Maddux B. A., Goldfine I. D. Monoclonal antibodies to the human insulin receptor block insulin binding and inhibit insulin action. Proc Natl Acad Sci U S A. 1982 Dec;79(23):7312–7316. doi: 10.1073/pnas.79.23.7312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roth R. A., Mesirow M. L., Cassell D. J. Preferential degradation of the beta subunit of purified insulin receptor. Effect on insulin binding and protein kinase activities of the receptor. J Biol Chem. 1983 Dec 10;258(23):14456–14460. [PubMed] [Google Scholar]
- Sadler S. E., Maller J. L. Identification of a steroid receptor on the surface of Xenopus oocytes by photoaffinity labeling. J Biol Chem. 1982 Jan 10;257(1):355–361. [PubMed] [Google Scholar]
- Shulman M., Wilde C. D., Köhler G. A better cell line for making hybridomas secreting specific antibodies. Nature. 1978 Nov 16;276(5685):269–270. doi: 10.1038/276269a0. [DOI] [PubMed] [Google Scholar]
- Simpson I. A., Hedo J. A. Insulin receptor phosphorylation may not be a prerequisite for acute insulin action. Science. 1984 Mar 23;223(4642):1301–1304. doi: 10.1126/science.6367041. [DOI] [PubMed] [Google Scholar]
- Spivack J. G., Erikson R. L., Maller J. L. Microinjection of pp60v-src into Xenopus oocytes increases phosphorylation of ribosomal protein S6 and accelerates the rate of progesterone-induced meiotic maturation. Mol Cell Biol. 1984 Aug;4(8):1631–1634. doi: 10.1128/mcb.4.8.1631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takayama S., White M. F., Lauris V., Kahn C. R. Phorbol esters modulate insulin receptor phosphorylation and insulin action in cultured hepatoma cells. Proc Natl Acad Sci U S A. 1984 Dec;81(24):7797–7801. doi: 10.1073/pnas.81.24.7797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tamura S., Brown T. A., Whipple J. H., Fujita-Yamaguchi Y., Dubler R. E., Cheng K., Larner J. A novel mechanism for the insulin-like effect of vanadate on glycogen synthase in rat adipocytes. J Biol Chem. 1984 May 25;259(10):6650–6658. [PubMed] [Google Scholar]
- Tamura S., Fujita-Yamaguchi Y., Larner J. Insulin-like effect of trypsin on the phosphorylation of rat adipocyte insulin receptor. J Biol Chem. 1983 Dec 25;258(24):14749–14752. [PubMed] [Google Scholar]
- Ullrich A., Bell J. R., Chen E. Y., Herrera R., Petruzzelli L. M., Dull T. J., Gray A., Coussens L., Liao Y. C., Tsubokawa M. Human insulin receptor and its relationship to the tyrosine kinase family of oncogenes. 1985 Feb 28-Mar 6Nature. 313(6005):756–761. doi: 10.1038/313756a0. [DOI] [PubMed] [Google Scholar]
- Zick Y., Rees-Jones R. W., Taylor S. I., Gorden P., Roth J. The role of antireceptor antibodies in stimulating phosphorylation of the insulin receptor. J Biol Chem. 1984 Apr 10;259(7):4396–4400. [PubMed] [Google Scholar]
- de StGroth S. F., Scheidegger D. Production of monoclonal antibodies: strategy and tactics. J Immunol Methods. 1980;35(1-2):1–21. doi: 10.1016/0022-1759(80)90146-5. [DOI] [PubMed] [Google Scholar]