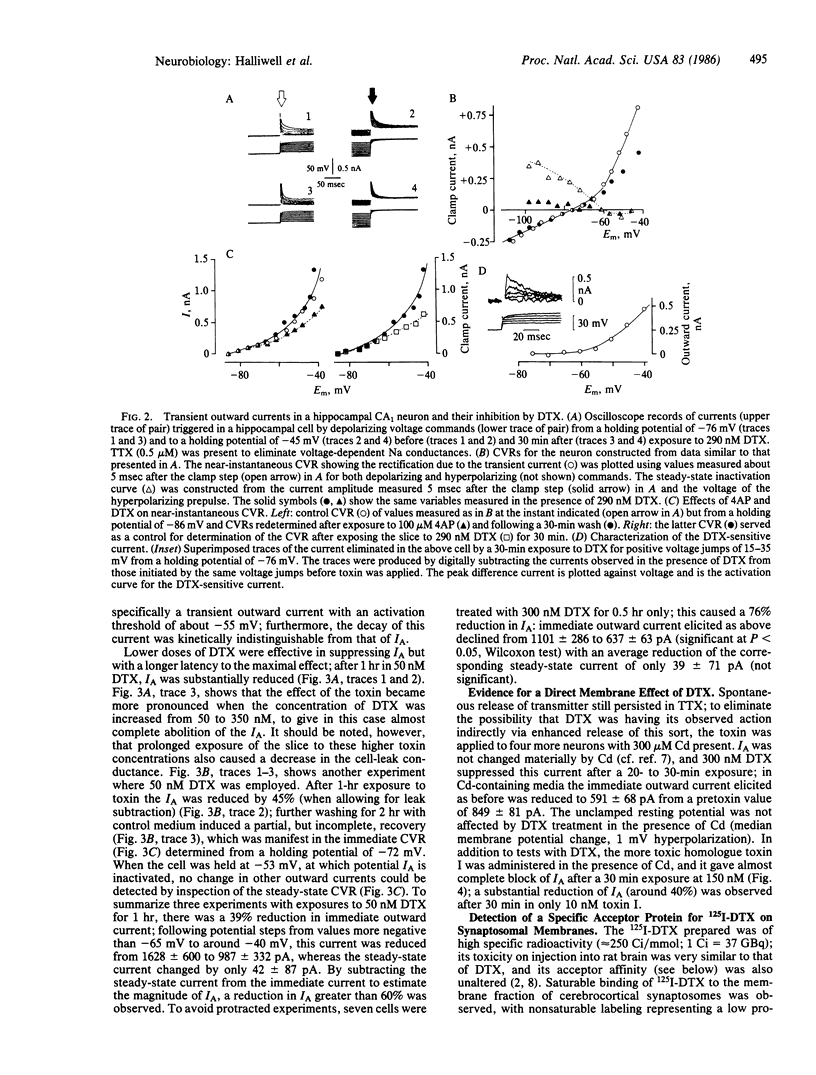
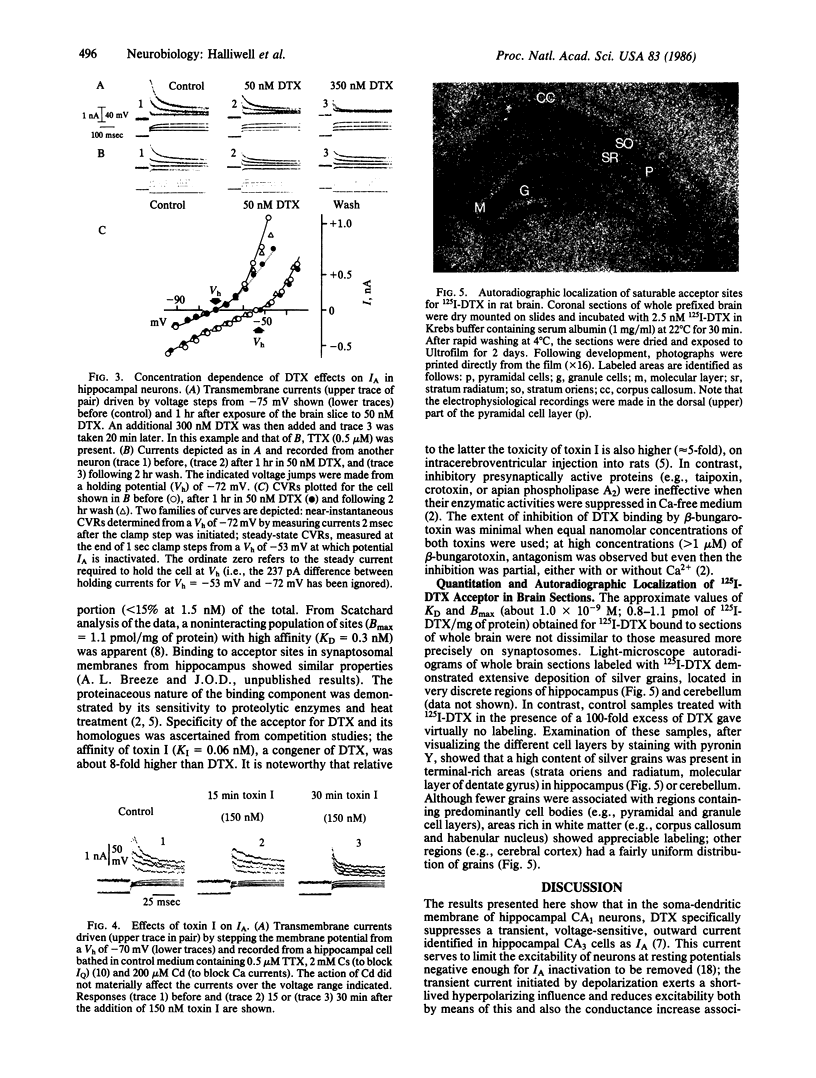
Abstract
Dendrotoxin, a small single-chain protein from the venom of Dendroaspis angusticeps, is highly toxic following intracerebroventricular injection into rats. Voltage-clamp analysis of CA1 neurons in hippocampal slices, treated with tetrodotoxin, revealed that nanomolar concentrations of dendrotoxin reduce selectively a transient, voltage-dependent K conductance. Epileptiform activity known to be induced by dendrotoxin can be attributed to such an action. Membrane currents not affected directly by the toxin include (i) Ca-activated K conductance; (ii) noninactivating voltage-dependent K conductance; (iii) inactivating and noninactivating Ca conductances; (iv) persistent inward (anomalous) rectifier current. Persistence of the effects of the toxin when Cd was included to suppress spontaneous transmitter release indicates a direct action on the neuronal membrane. Using biologically active, 125I-labeled dendrotoxin, protein acceptor sites of high affinity were detected on cerebrocortical synaptosomal membranes and sections of rat brain. In hippocampus, toxin binding was shown autoradiographically to reside in synapse-rich and white matter regions, with lower levels in cell body layers. This acceptor is implicated in the action of toxin because its affinities for dendrotoxin congeners are proportional to their central neurotoxicities and potencies in reducing the transient, voltage-dependent K conductance.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bostock H., Sears T. A., Sherratt R. M. The effects of 4-aminopyridine and tetraethylammonium ions on normal and demyelinated mammalian nerve fibres. J Physiol. 1981;313:301–315. doi: 10.1113/jphysiol.1981.sp013666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown D. A., Griffith W. H. Calcium-activated outward current in voltage-clamped hippocampal neurones of the guinea-pig. J Physiol. 1983 Apr;337:287–301. doi: 10.1113/jphysiol.1983.sp014624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown D. A., Griffith W. H. Persistent slow inward calcium current in voltage-clamped hippocampal neurones of the guinea-pig. J Physiol. 1983 Apr;337:303–320. doi: 10.1113/jphysiol.1983.sp014625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buckle P. J., Haas H. L. Enhancement of synaptic transmission by 4-aminopyridine in hippocampal slices of the rat. J Physiol. 1982 May;326:109–122. doi: 10.1113/jphysiol.1982.sp014180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Connor J. A., Stevens C. F. Voltage clamp studies of a transient outward membrane current in gastropod neural somata. J Physiol. 1971 Feb;213(1):21–30. doi: 10.1113/jphysiol.1971.sp009365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dolly J. O., Halliwell J. V., Black J. D., Williams R. S., Pelchen-Matthews A., Breeze A. L., Mehraban F., Othman I. B., Black A. R. Botulinum neurotoxin and dendrotoxin as probes for studies on transmitter release. J Physiol (Paris) 1984;79(4):280–303. [PubMed] [Google Scholar]
- Dubois J. M. Evidence for the existence of three types of potassium channels in the frog Ranvier node membrane. J Physiol. 1981 Sep;318:297–316. doi: 10.1113/jphysiol.1981.sp013865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galvan M. A transient outward current in rat sympathetic neurones. Neurosci Lett. 1982 Aug 31;31(3):295–300. doi: 10.1016/0304-3940(82)90036-2. [DOI] [PubMed] [Google Scholar]
- Galvan M., Franz P., Vogel-Wiens C. Actions of potassium channel blockers on guinea-pig lateral olfactory tract axons. Naunyn Schmiedebergs Arch Pharmacol. 1984 Jan;325(1):8–11. doi: 10.1007/BF00507047. [DOI] [PubMed] [Google Scholar]
- Gustafsson B., Galvan M., Grafe P., Wigström H. A transient outward current in a mammalian central neurone blocked by 4-aminopyridine. Nature. 1982 Sep 16;299(5880):252–254. doi: 10.1038/299252a0. [DOI] [PubMed] [Google Scholar]
- Haas H. L., Wieser H. G., Yaşargil M. G. 4-Aminopyridine and fiber potentials in rat and human hippocampal slices. Experientia. 1983 Jan 15;39(1):114–115. doi: 10.1007/BF01960661. [DOI] [PubMed] [Google Scholar]
- Halliwell J. V., Adams P. R. Voltage-clamp analysis of muscarinic excitation in hippocampal neurons. Brain Res. 1982 Oct 28;250(1):71–92. doi: 10.1016/0006-8993(82)90954-4. [DOI] [PubMed] [Google Scholar]
- Halliwell J. V., Dolly J. O. Preferential action of beta-bungarotoxin at nerve terminal regions in the hippocampus. Neurosci Lett. 1982 Jun 30;30(3):321–327. doi: 10.1016/0304-3940(82)90420-7. [DOI] [PubMed] [Google Scholar]
- Harvey A. L., Karlsson E. Protease inhibitor homologues from mamba venoms: facilitation of acetylcholine release and interactions with prejunctional blocking toxins. Br J Pharmacol. 1982 Sep;77(1):153–161. doi: 10.1111/j.1476-5381.1982.tb09281.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacDermott A. B., Weight F. F. Action potential repolarization may involve a transient, Ca2+-sensitive outward current in a vertebrate neurone. Nature. 1982 Nov 11;300(5888):185–188. doi: 10.1038/300185a0. [DOI] [PubMed] [Google Scholar]
- Mehraban F., Breeze A. L., Dolly J. O. Identification by cross-linking of a neuronal acceptor protein for dendrotoxin, a convulsant polypeptide. FEBS Lett. 1984 Aug 20;174(1):116–122. doi: 10.1016/0014-5793(84)81088-1. [DOI] [PubMed] [Google Scholar]
- Meves H., Pichon Y. The effect of internal and external 4-aminopyridine on the potassium currents in intracellularly perfused squid giant axons. J Physiol. 1977 Jun;268(2):511–532. doi: 10.1113/jphysiol.1977.sp011869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Othman I. B., Spokes J. W., Dolly J. O. Preparation of neurotoxic 3H-beta-bungarotoxin: demonstration of saturable binding to brain synapses and its inhibition by toxin I. Eur J Biochem. 1982 Nov;128(1):267–276. doi: 10.1111/j.1432-1033.1982.tb06961.x. [DOI] [PubMed] [Google Scholar]
- Pelhate M., Pichon Y. Proceedings: Selective inhibition of potassium current in the giant axon of the cockroach. J Physiol. 1974 Oct;242(2):90P–91P. [PubMed] [Google Scholar]
- Schauf C. L., Colton C. A., Colton J. S., Davis F. A. Aminopyridines and sparteine as inhibitors of membrane potassium conductance: effects on Myxicola giant axons and the lobster neuromuscular junction. J Pharmacol Exp Ther. 1976 May;197(2):414–425. [PubMed] [Google Scholar]
- Segal M., Rogawski M. A., Barker J. L. A transient potassium conductance regulates the excitability of cultured hippocampal and spinal neurons. J Neurosci. 1984 Feb;4(2):604–609. doi: 10.1523/JNEUROSCI.04-02-00604.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimahara T. Presynaptic modulation of transmitter release by the early outward potassium current in Aplysia. Brain Res. 1983 Mar 14;263(1):51–56. doi: 10.1016/0006-8993(83)91199-x. [DOI] [PubMed] [Google Scholar]
- Strydom D. J., Joubert F. J. The amino acid sequence of a weak trypsin inhibitor B from Dendroaspis Polylepis polylepis (black mamba) venom. Hoppe Seylers Z Physiol Chem. 1981 Oct;362(10):1377–1384. doi: 10.1515/bchm2.1981.362.2.1377. [DOI] [PubMed] [Google Scholar]
- Thompson S. H. Three pharmacologically distinct potassium channels in molluscan neurones. J Physiol. 1977 Feb;265(2):465–488. doi: 10.1113/jphysiol.1977.sp011725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tillotson D. Inactivation of Ca conductance dependent on entry of Ca ions in molluscan neurons. Proc Natl Acad Sci U S A. 1979 Mar;76(3):1497–1500. doi: 10.1073/pnas.76.3.1497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Unnerstall J. R., Niehoff D. L., Kuhar M. J., Palacios J. M. Quantitative receptor autoradiography using [3H]ultrofilm: application to multiple benzodiazepine receptors. J Neurosci Methods. 1982 Jul;6(1-2):59–73. doi: 10.1016/0165-0270(82)90016-4. [DOI] [PubMed] [Google Scholar]




