Abstract
A 45-year-old man with bipolar disease type I and post-traumatic frontal lobe lesions following a previous psychotic episode was hospitalised after having stopped taking aripiprazole (15 mg/day) and lithium (126 mg twice/daily). He presented with hypomania, psychosis, verbal unresponsiveness and disorientation. He engaged in compulsive onanism in public which resulted in the restriction of his freedom, suggesting impulse control disorder (ICD). Electroencephalogram showed no epileptiform activity. Brain CT-scan showed post-traumatic bifrontal and left occipital lesions. Viral and bacterial serologies were normal. Lithium was initially reinstated (same dose), but it was aripiprazole reinstatement at a lower dose (10 mg) that made his condition rapidly improve within 24–36 h. Aripiprazole is a partial dopamine D2-receptor antagonist that blocks dopamine at higher dopamine concentrations and augments/releases prefrontal cortex dopamine at lower concentrations. The rapid recovery suggests stabilisation of dopamine levels in the frontal lobes and that ICD may be modified by aripiprazole treatment.
Background
The case is important since our patient presented with an atypical form of hypomania and the clinical picture was suggestive of a rare form of psychomotor epilepsy (compulsive onanism). The behavioural abnormalities were difficult to differentiate from non-convulsive epilepsy owing to the absence of psychomotor agitation. This atypical form was probably potentiated by pre-existing neurological damage in the frontal lobes. The loss of impulse control was resistant to lithium reinstatement but quickly responded to aripiprazole within 24–36 h, which may suggest hypofunction of the dopamine system in the frontal lobes as responsible for impulse control loss.
This observation, together with preliminary reports of an improvement of impulse control with aripiprazole in the setting of drug addiction, for example, may promote debate and raise general interest for larger studies of pharmacological control of impulsivity in affective disorders.
Case presentation
This is a case report of a 45-year-old divorced man, with known bipolar disease type I since the age of 28, presenting with psychotic episodes during both manic as well as depressive episodes and with anxiety comorbidity. At the age of 30, he suffered from a severe manic psychosis which drove him to jump off a window from a 15 metre height. He was in coma for 2 weeks requiring intensive care and neurosurgical reconstruction of the skull, orbita and frontal sinuses (right-sided craniotomy). At present, he sustains sequellar peripheral facial paresis on the left side, anosmia and mild symptoms of right frontal lobe dysfunction but no other neurological deficits. He works with handcraft. He is a former smoker who now uses nicotine substitution. Family history was unremarkable. Neuropsychological tests performed about 10 months ago showed decreased executive functioning and planning, attentional deficits and reduced task-related speed. Visuospatial learning and memory performance were decreased to minus 3 SD in the Rey Complex Figure Test and Recognition Trial and Wisconsin Card Sorting test. His verbal capacities, abstraction and working memory were normal.
Our patient had been relatively stable for the last 3 years with oral lithium (126 mg twice daily) and aripiprazole (15 mg/day), a partial dopamine D2-receptor antagonist, known to have partial D2-agonism at low dopamine concentrations, as well as serotonin 5-HT1A receptor agonism and a 5-HT2A receptor antagonism. Aripiprazole is a licensed drug for schizophrenia,1 2 with good efficacy in bipolar disease.3 He stopped taking aripiprazole in the spring with the consent of his psychiatrist, because of side-effects (drowsiness, increased appetite and weight gain). According to relatives, he broke contact just a few weeks after he stopped taking aripiprazole. He was hospitalised 3 months later displaying aggressive behaviour, disinhibition, lability, auditory hallucinosis and psychotic delusions of grandiosity with a hypersexual theme. He had stopped taking lithium 5 days prior to admission and refused to take any medicine other than lithium. Serum lithium levels on admission were undetectable. About 24 h after lithium reinstatement (126 mg twice/daily), he presented no signs of psychomotor agitation but was found lying on the floor at night, confused and unresponsive. He could only vaguely remember the events from the day before. The following days he then engaged in compulsive onanism in public several times per day. His freedom was therefore restricted to extra observation and room confinement (every 15 min a nurse or care giver would visit the patient in his room, meals were delivered to him in his room) to minimise exhibitionism. Despite restrictions, this behaviour was not reduced. When confronted, he looked confused and embarrassed. He also talked to himself in his room in a splittered fashion. The symtoms lasted for 1 week and did not respond to lithium therapy. In the absence of psychomotor agitation, the proposed diagnosis was one of hypomania but the behavioural unresponsiveness with disinhibition and mnesic symptoms suggested an ictal cause.
Investigations
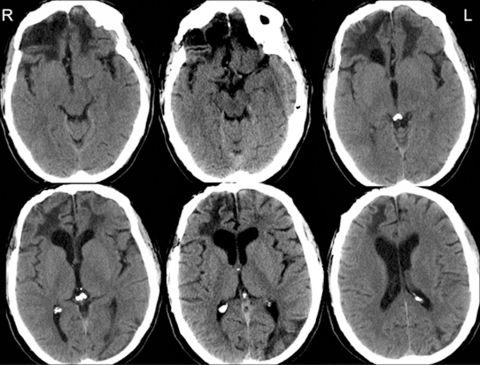
All investigations were undertaken with the patient’s consent. Brain CT-scan with angiography revealed old bifrontal and left occipital post-contusional lesions with predominant substance loss in the right hemisphere (figure 1). The cerebral CT-angiography was normal. Owing to metal artifacts from previous neurosurgery, MRI could not be performed. The electroencephalograms (EEG) only revealed low frontal lobe activity within the 4–6 Hz range, amplitude 50 microV, predominantly in the right frontal lobe. The right hemisphere featured low activity particularly in the right frontal region on one occasion after sleep deprivation. No signs of epileptiform activity were detected. Blood count, electrolytes, creatinine, liver function tests, folate, B12, cholesterol panel, thyroid function were unremarkable. Viral and bacterial serologies were negative. The patient refused to undergo a lumbar punction.
Figure 1.
Non-enhanced and contrast cerebral CT-scan revealed only post-traumatic substance loss in the orbitofrontal and ventromedial cortex bilaterally, predominant on the right side and to a lesser degree, a contre-coup left occipital lesion.
Differential diagnosis
Differential diagnosis included postlesional non-convulsive frontotemporal epilepsy, stroke, cerebral haemorrhage, space-occupying lesion (brain tumour, aneurysm, arteriovenous malformation) and infectious disease (herpes encephalitis, neuroborreliosis, syphilis).
Outcome and follow-up
After 1 week, and showing no signs of improvement despite therapeutic serum lithium levels (0.87 mmol/l), the patient accepted to be reinstated on a lower aripiprazole dose (10 mg/day). This was an agreement owing to the patient’s reluctance in taking a dose that previously induced side-effects. Despite the lower dose, aripiprazole reinstatement resulted in a cessation of compulsive onanism within 24–36 h and a decrease in confusional, mnesic and psychotic symtoms.
Discussion
This is an unusual course of a hypomanic episode in the context of type I bipolar disease, rapidly changing from a psychotic agitated state on admission to a more compulsive sexual behaviour in the absence of clear psychomotor agitation. He engaged in such behaviour despite adverse consequences to his freedom, suggesting impulse control disorder (ICD). The core symtoms of ICD include repetitive, compulsive engagement in a behaviour despite adverse consequences, reduced control over the behaviour, an urge or craving for the behaviour before it is initiated and a hedonic characteristic during the performance of the behavior.4 Due to unresponsiveness while awake, the initial proposed investigations aimed at uncovering non-convulsive status epilepticus owing to substantial post-traumatic lesional load in both frontal cortices. Indeed post-ictal or peri-ictal true onanism has been reported to occur at the end of the ictal discharge beginning either in the perisylvian region or in the anterior part of the cingular gyrus.5 The EEG only showed low frontal cortex activity, predominating in the right side. This was line with a more extensive damage to the right frontobasal cortex found in the CT-scan. The absence of epileptiform activity following sleep deprivation potentially excluded psychomotor epilepsy.
Loss of impulse control is thought to result from dysregulation of the dopaminergic system in the mesocorticolimbic pathways, including, among other, the ventromedial and orbitofrontal cortex, involved in planning and judgment, and it has been reported in bipolar disease.6 On the other hand, orbitofrontal activity measured with positron emission tomography (PET) and single-photon emission CT studies has been reported to be decreased in manic patients.7 In our patient, with an already compromised orbitofrontal lobe by previous trauma, a cumulative disinhibitory effect caused by hypomania may explain the ‘wiping out’ of impulse control accompanied by hyperactivity in the subcortical limbic system. There are two lines of evidence in support of this notion, one is coming from fMRI and PET studies showing hyperactive subcortical limbic activity in the ventral striatum and amygdala during mania. Both the ventral striatum and amygdala are interconnected with the cingulate, medial and orbitofrontal cortex. A second line of evidence comes from a few rare stroke patients with lesions in the right hemisphere (frontal or temporal lobe, thalamus and caudate nucleus) who can develop mania with impulsive features due to a dysfunction in the fronto-striato-capsulo-thalamo-cortical loop.8 Furthermore it has been found that there is a reduction in the activation and functioning of the frontal lobe in several conditions where impulse control is compromised such as attention deficit disorder, Parkinson’s disease, depression and schizophrenia.6 9–11
Our patient can thus be described to be in a vulnerable state predisposing to ICD. Dopamine levels have been proposed to mediate gating of impulses within the frontal lobe.12 Aripiprazole, a partial dopamine D2-receptor antagonist appears to block dopamine at higher dopamine concentrations and augments/releases prefrontal cortex dopamine at lower dopamine concentrations thus acting as a dopamine stabiliser. The rapid reversal of the symptomatology with this drug suggests that there is a hypofunction of the dopamine system in the frontal lobe responsible for ICD, which is rescued by partial dopamin agonism. Similar effects on impulse control have been reported with aripiprazole in other conditions. Decreased alcohol consumption was observed in patients with lower self-control as a measure of impulsivity following aripiprazole titrated up to 15 mg/day over a 8 day-period in a double-blind, placebo-controlled randomised study.13 Interestingly, aripiprazole 10 mg/daily (combined with memantine 10 mg and sertraline 100 mg/daily) given to a patient suffering from frontotemporal dementia was shown to improve a severe apathy syndrome and to increase glucose metabolism in the frontal and anterior temporal cortices, as measured with PET-scan.14
Learning points.
-
▶
The loss of impulse control in our patient was likely the manifestation of disinhibition owing to decreased frontal lobe metabolism induced by hypomania and low reserve for inhibition owing to considerable post-traumatic frontal lobe damage.
-
▶
Reinstatement of lithium therapy likely improved hypomania but aripiprazole seemed to play a more determining role in frontal lobe stabilisation.
-
▶
Further, larger studies are warranted to access the efficacy and safety profile in impulse control disorders but also in conditions where frontal lobe functioning is compromised.
Acknowledgments
The authors would like to thank Professor Torbjörn Tomson, Department of Neurology, Karolinska Solna and the nursing staff of avdelning 53, NSP, Stockholm.
Footnotes
Competing interests None.
Patient consent Obtained.
References
- 1.DeLeon A, Patel NC, Crismon ML. Aripiprazole: a comprehensive review of its pharmacology, clinical efficacy, and tolerability. Clin Ther 2004;26:649–66 [DOI] [PubMed] [Google Scholar]
- 2.Marder SR, McQuade RD, Stock E, et al. Aripiprazole in the treatment of schizophrenia: safety and tolerability in short-term, placebo-controlled trials. Schizophr Res 2003;61:123–36 [DOI] [PubMed] [Google Scholar]
- 3.Keck PE, Jr, Calabrese JR, McQuade RD, et al. A randomized, double-blind, placebo-controlled 26-week trial of aripiprazole in recently manic patients with bipolar I disorder. J Clin Psychiatry 2006;67:626–37 [DOI] [PubMed] [Google Scholar]
- 4.Brewer JA, Potenza MN. The neurobiology and genetics of impulse control disorders: relationships to drug addictions. Biochem Pharmacol 2008;75:63–75 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Stoffels C, Munari C, Bonis A, et al. Genital and sexual manifestations occurring in the course of partial seizures in man. Rev Electroencephalogr Neurophysiol Clin 1980;10:386–92 [DOI] [PubMed] [Google Scholar]
- 6.Ceravolo R, Frosini D, Rossi C, et al. Impulse control disorders in Parkinson’s disease: definition, epidemiology, risk factors, neurobiology and management. Parkinsonism Relat Disord 2009;15 Suppl 4:S111–5 [DOI] [PubMed] [Google Scholar]
- 7.Gonul AS, Coburn K, Kula M. Cerebral blood flow, metabolic, receptor, and transporter changes in bipolar disorder: the role of PET and SPECT studies. Int Rev Psychiatry 2009;21:323–35 [DOI] [PubMed] [Google Scholar]
- 8.Carota A, Dieguez S, Bogousslavsky J. [Psychopathology of stroke]. Psychol Neuropsychiatr Vieil 2005;3:235–49 [PubMed] [Google Scholar]
- 9.Seidman LJ, Valera EM, Makris N. Structural brain imaging of attention-deficit/hyperactivity disorder. Biol Psychiatry 2005;57:1263–72 [DOI] [PubMed] [Google Scholar]
- 10.Rogers MA, Kasai K, Koji M, et al. Executive and prefrontal dysfunction in unipolar depression: a review of neuropsychological and imaging evidence. Neurosci Res 2004;50:1–11 [DOI] [PubMed] [Google Scholar]
- 11.Pantelis C, Yücel M, Wood SJ, et al. Structural brain imaging evidence for multiple pathological processes at different stages of brain development in schizophrenia. Schizophr Bull 2005;31:672–96 [DOI] [PubMed] [Google Scholar]
- 12.Bechara A. Decision making, impulse control and loss of willpower to resist drugs: a neurocognitive perspective. Nat Neurosci 2005;8:1458–63 [DOI] [PubMed] [Google Scholar]
- 13.Voronin K, Randall P, Myrick H, et al. Aripiprazole effects on alcohol consumption and subjective reports in a clinical laboratory paradigm–possible influence of self-control. Alcohol Clin Exp Res 2008;32:1954–61 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fellgiebel A, Müller MJ, Hiemke C, et al. Clinical improvement in a case of frontotemporal dementia under aripiprazole treatment corresponds to partial recovery of disturbed frontal glucose metabolism. World J Biol Psychiatry 2007;8:123–6 [DOI] [PubMed] [Google Scholar]