Abstract
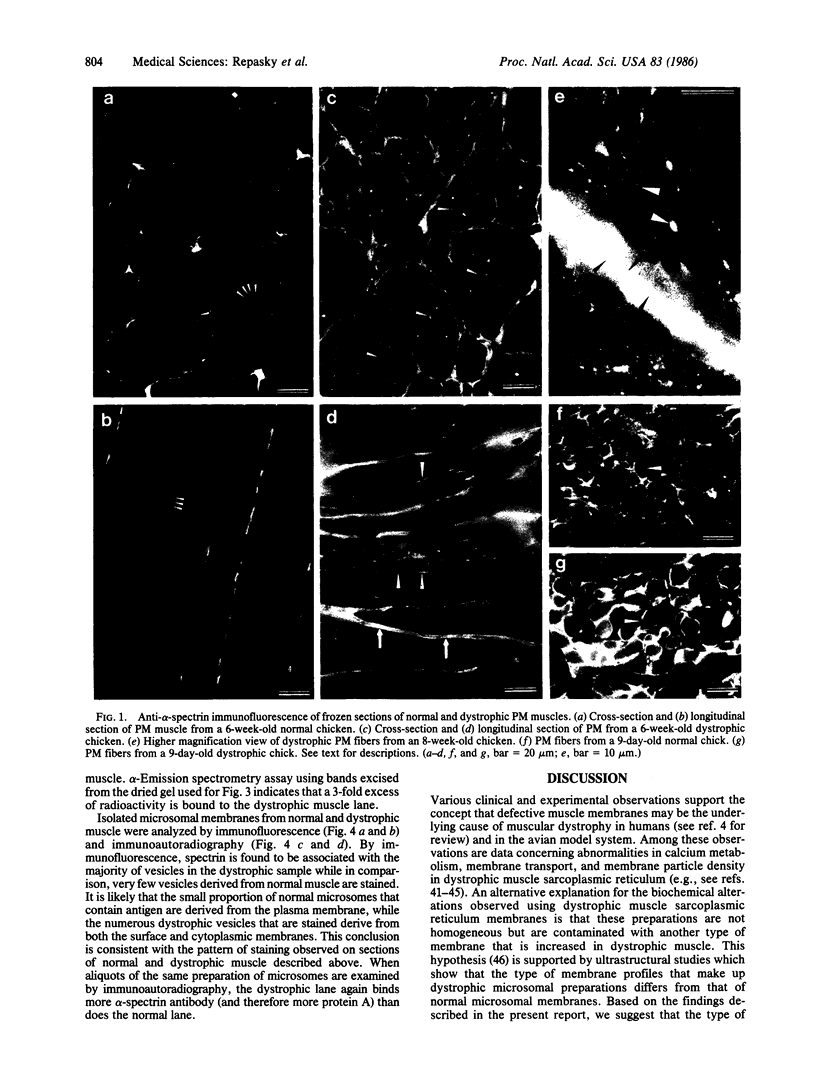
A significant increase in the concentration of spectrin has been observed in dystrophic chicken pectoralis major muscle when compared to normal fast-twitch muscle. In normal muscle, alpha-spectrin-specific immunofluorescence delineates each myofiber with a network pattern of staining at the sarcolemma with little staining within the cytoplasm. In dystrophic fibers, numerous intensely stained areas occur within the cytoplasm and staining at the sarcolemma is increased, thereby obscuring or eliminating the highly regular network arrangement of spectrin usually seen in this region. When immunofluorescence experiments are performed on microsomal vesicles isolated from normal and dystrophic tissues, only a small fraction of normal vesicles are stained, whereas most of the dystrophic vesicles are associated with spectrin. An increase in spectrin concentration is observed using immunoautoradiography of whole muscle and isolated microsomes, thus supporting the immunofluorescent observations described above. The early-age post-hatching when increases in spectrin concentration can be detected and the simplicity of the immunofluorescent technique make this observation useful as a new diagnostic parameter. This observation also shows that the distribution of spectrin and its concentration within nonerythroid cells can be modified by abnormal physiological states; this modification may contribute to subsequent symptoms, such as increased rigidity and abnormal calcium metabolism, that are observed in dystrophy.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Apgar J. R., Herrmann S. H., Robinson J. M., Mescher M. F. Triton X-100 extraction of P815 tumor cells: evidence for a plasma membrane skeleton structure. J Cell Biol. 1985 May;100(5):1369–1378. doi: 10.1083/jcb.100.5.1369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Appleyard S. T., Dunn M. J., Dubowitz V., Scott M. L., Pittman S. J., Shotton D. M. Monoclonal antibodies detect a spectrin-like protein in normal and dystrophic human skeletal muscle. Proc Natl Acad Sci U S A. 1984 Feb;81(3):776–780. doi: 10.1073/pnas.81.3.776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baudry M., Bundman M. C., Smith E. K., Lynch G. S. Micromolar calcium stimulates proteolysis and glutamate binding in rat brain synaptic membranes. Science. 1981 May 22;212(4497):937–938. doi: 10.1126/science.7015504. [DOI] [PubMed] [Google Scholar]
- Ben-Ze'ev A., Duerr A., Solomon F., Penman S. The outer boundary of the cytoskeleton: a lamina derived from plasma membrane proteins. Cell. 1979 Aug;17(4):859–865. doi: 10.1016/0092-8674(79)90326-x. [DOI] [PubMed] [Google Scholar]
- Bennett V. The membrane skeleton of human erythrocytes and its implications for more complex cells. Annu Rev Biochem. 1985;54:273–304. doi: 10.1146/annurev.bi.54.070185.001421. [DOI] [PubMed] [Google Scholar]
- Birkenmeier C. S., Bodine D. M., Repasky E. A., Helfman D. M., Hughes S. H., Barker J. E. Remarkable homology among the internal repeats of erythroid and nonerythroid spectrin. Proc Natl Acad Sci U S A. 1985 Sep;82(17):5671–5675. doi: 10.1073/pnas.82.17.5671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brain M. C., Kohn I., McComas A. J., Missirlis Y. F., Rathbone M. P., Vickers J. Red-cell stability in Duchenne's syndrome. N Engl J Med. 1978 Feb 16;298(7):403–403. doi: 10.1056/NEJM197802162980716. [DOI] [PubMed] [Google Scholar]
- Branton D., Cohen C. M., Tyler J. Interaction of cytoskeletal proteins on the human erythrocyte membrane. Cell. 1981 Apr;24(1):24–32. doi: 10.1016/0092-8674(81)90497-9. [DOI] [PubMed] [Google Scholar]
- Brown H. D., Chattopadhyay S. K., Patel A. B. Erythrocyte abnormality in human myopathy. Science. 1967 Sep 29;157(3796):1577–1578. doi: 10.1126/science.157.3796.1577. [DOI] [PubMed] [Google Scholar]
- Burridge K. Direct identification of specific glycoproteins and antigens in sodium dodecyl sulfate gels. Methods Enzymol. 1978;50:54–64. doi: 10.1016/0076-6879(78)50007-4. [DOI] [PubMed] [Google Scholar]
- Butterfield D. A., Leung P. K. Erythrocyte membrane fluidity in chicken muscular dystrophy. Life Sci. 1978 May 22;22(20):1783–1788. doi: 10.1016/0024-3205(78)90593-3. [DOI] [PubMed] [Google Scholar]
- COSMOS E. INTRACELLULAR DISTRIBUTION OF CALCIUM IN DEVELOPING BREAST MUSCLE OF NORMAL AND DYSTROPHIC CHICKENS. J Cell Biol. 1964 Nov;23:241–252. doi: 10.1083/jcb.23.2.241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen C. M. The molecular organization of the red cell membrane skeleton. Semin Hematol. 1983 Jul;20(3):141–158. [PubMed] [Google Scholar]
- Cosmos E. Enzymatic activity of differentiating muscle fibers. I. Development of phosphorylase in muscles of the domestic fowl. Dev Biol. 1966 Apr;13(2):163–181. doi: 10.1016/0012-1606(66)90062-5. [DOI] [PubMed] [Google Scholar]
- ENGEL W. K., CUNNINGHAM G. G. RAPID EXAMINATION OF MUSCLE TISSUE. AN IMPROVED TRICHROME METHOD FOR FRESH-FROZEN BIOPSY SECTIONS. Neurology. 1963 Nov;13:919–923. doi: 10.1212/wnl.13.11.919. [DOI] [PubMed] [Google Scholar]
- Ettienne E. M., Singer R. H. Ca2+ binding, ATP-dependent Ca2+ transport, and total tissue Ca2+ in embryonic and adult avian dystrophic pectoralis. J Membr Biol. 1978 Dec 29;44(3-4):195–210. doi: 10.1007/BF01944221. [DOI] [PubMed] [Google Scholar]
- Geiger B. Membrane-cytoskeleton interaction. Biochim Biophys Acta. 1983 Aug 11;737(3-4):305–341. doi: 10.1016/0304-4157(83)90005-9. [DOI] [PubMed] [Google Scholar]
- Glenney J. R., Jr, Glenney P., Osborn M., Weber K. An F-actin- and calmodulin-binding protein from isolated intestinal brush borders has a morphology related to spectrin. Cell. 1982 Apr;28(4):843–854. doi: 10.1016/0092-8674(82)90063-0. [DOI] [PubMed] [Google Scholar]
- Goodman S. R., Shiffer K. The spectrin membrane skeleton of normal and abnormal human erythrocytes: a review. Am J Physiol. 1983 Mar;244(3):C121–C141. doi: 10.1152/ajpcell.1983.244.3.C121. [DOI] [PubMed] [Google Scholar]
- Haest C. W. Interactions between membrane skeleton proteins and the intrinsic domain of the erythrocyte membrane. Biochim Biophys Acta. 1982 Dec;694(4):331–352. doi: 10.1016/0304-4157(82)90001-6. [DOI] [PubMed] [Google Scholar]
- Hanna S. D., Baskin R. J. Calcium transport and phosphoenzyme formation in sarcoplasmic reticulum isolated from normal and dystrophic chickens. Biochem Med. 1977 Jun;17(3):300–309. doi: 10.1016/0006-2944(77)90036-9. [DOI] [PubMed] [Google Scholar]
- Hsu Q. S., Kaldor G. Studies on the sarcoplasmic reticulum of normal and dystrophic animals. Proc Soc Exp Biol Med. 1969 Sep;131(4):1398–1402. doi: 10.3181/00379727-131-34116. [DOI] [PubMed] [Google Scholar]
- Hudecki M. S., Pollina C. M., Heffner R. R., Bhargava A. K. Enhanced functional ability in drug-treated dystrophic chickens: trial results with indomethacin, diphenylhydantoin, and prednisolone. Exp Neurol. 1981 Jul;73(1):173–185. doi: 10.1016/0014-4886(81)90053-4. [DOI] [PubMed] [Google Scholar]
- Kuettner J. F., Dreher K. L., Rao G. H., Eaton J. W., Blackshear P. L., Jr, White J. G. Influence of the ionophore A23187 on the plastic behavior of normal erythrocytes. Am J Pathol. 1977 Jul;88(1):81–94. [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lazarides E., Granger B. L. Preparation and assay of the intermediate filament proteins desmin and vimentin. Methods Enzymol. 1982;85(Pt B):488–508. doi: 10.1016/0076-6879(82)85048-9. [DOI] [PubMed] [Google Scholar]
- Levine J., Willard M. Fodrin: axonally transported polypeptides associated with the internal periphery of many cells. J Cell Biol. 1981 Sep;90(3):631–642. doi: 10.1083/jcb.90.3.631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lumb E. M., Emery A. E. Erythrocyte deformation in duchenne muscular dystrophy. Br Med J. 1975 Aug 23;3(5981):467–468. doi: 10.1136/bmj.3.5981.467-a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lux S. E. Spectrin-actin membrane skeleton of normal and abnormal red blood cells. Semin Hematol. 1979 Jan;16(1):21–51. [PubMed] [Google Scholar]
- Malouf N. N., Sommer J. R. Chicken dystrophy. The geometry of the transverse tubules. Am J Pathol. 1976 Aug;84(2):299–316. [PMC free article] [PubMed] [Google Scholar]
- Martonosi A. Sarocoplasmic reticulum. VI. Microsomal Ca2+ transport in genetic muscular dystrophy of mice. Proc Soc Exp Biol Med. 1968 Mar;127(3):824–828. doi: 10.3181/00379727-127-32813. [DOI] [PubMed] [Google Scholar]
- Menold M. M., Repasky E. A. Heterogeneity of spectrin distribution among avian muscle fiber types. Muscle Nerve. 1984 Jun;7(5):408–414. doi: 10.1002/mus.880070511. [DOI] [PubMed] [Google Scholar]
- Mescher M. F., Jose M. J., Balk S. P. Actin-containing matrix associated with the plasma membrane of murine tumour and lymphoid cells. Nature. 1981 Jan 15;289(5794):139–144. doi: 10.1038/289139a0. [DOI] [PubMed] [Google Scholar]
- Nelson W. J., Lazarides E. Expression of the beta subunit of spectrin in nonerythroid cells. Proc Natl Acad Sci U S A. 1983 Jan;80(2):363–367. doi: 10.1073/pnas.80.2.363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson W. J., Lazarides E. Posttranslational control of membrane-skeleton (ankyrin and alpha beta-spectrin) assembly in early myogenesis. J Cell Biol. 1985 May;100(5):1726–1735. doi: 10.1083/jcb.100.5.1726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson W. J., Lazarides E. Switching of subunit composition of muscle spectrin during myogenesis in vitro. 1983 Jul 28-Aug 3Nature. 304(5924):364–368. doi: 10.1038/304364a0. [DOI] [PubMed] [Google Scholar]
- Palek J., Liu S. C. Dependence of spectrin organization in red blood cell membranes on cell metabolism: implications for control of red cell shape, deformability, and surface area. Semin Hematol. 1979 Jan;16(1):75–93. [PubMed] [Google Scholar]
- Percy A. K., Miller M. E. Reduced deformability of erythrocyte membranes from patients with Duchenne muscular dystrophy. Nature. 1975 Nov 13;258(5531):147–148. doi: 10.1038/258147a0. [DOI] [PubMed] [Google Scholar]
- Repasky E. A., Granger B. L., Lazarides E. Widespread occurrence of avian spectrin in nonerythroid cells. Cell. 1982 Jul;29(3):821–833. doi: 10.1016/0092-8674(82)90444-5. [DOI] [PubMed] [Google Scholar]
- Roses A. D., Appel S. H. Erythrocyte spectrin peak II phosphorylation in Duchenne muscular dystrophy. J Neurol Sci. 1976 Oct;29(2-4):185–193. doi: 10.1016/0022-510x(76)90170-2. [DOI] [PubMed] [Google Scholar]
- Rowland L. P. Biochemistry of muscle membranes in Duchenne muscular dystrophy. Muscle Nerve. 1980 Jan-Feb;3(1):3–20. doi: 10.1002/mus.880030103. [DOI] [PubMed] [Google Scholar]
- Siman R., Baudry M., Lynch G. Brain fodrin: substrate for calpain I, an endogenous calcium-activated protease. Proc Natl Acad Sci U S A. 1984 Jun;81(11):3572–3576. doi: 10.1073/pnas.81.11.3572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siman R., Baudry M., Lynch G. Purification from synaptosomal plasma membranes of calpain I, a thiol protease activated by micromolar calcium concentrations. J Neurochem. 1983 Oct;41(4):950–956. doi: 10.1111/j.1471-4159.1983.tb09039.x. [DOI] [PubMed] [Google Scholar]
- Sobue K., Kanda K., Adachi J., Kakiuchi S. Calmodulin-binding proteins that interact with actin filaments in a Ca2+-dependent flip-flop manner: survey in brain and secretory tissues. Proc Natl Acad Sci U S A. 1983 Nov;80(22):6868–6871. doi: 10.1073/pnas.80.22.6868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sobue K., Muramoto Y., Fujita M., Kakiuchi S. Calmodulin-binding protein of erythrocyte cytoskeleton. Biochem Biophys Res Commun. 1981 Jun 16;100(3):1063–1070. doi: 10.1016/0006-291x(81)91931-8. [DOI] [PubMed] [Google Scholar]
- Tsuchiya Y., Sugita H., Ishiura S., Imahori K. Spectrin extractability from erythrocyte in Duchenne muscular dystrophies and the effect of proteases on erythrocyte ghosts. Clin Chim Acta. 1981 Feb 5;109(3):285–293. doi: 10.1016/0009-8981(81)90314-4. [DOI] [PubMed] [Google Scholar]
- Warnick J. E., Lebeda F. J., Albuquerque E. X. Junctional and extrajunctional aspects of inherited muscular dystrophy in chickens: development and pharmacology. Ann N Y Acad Sci. 1979;317:263–284. doi: 10.1111/j.1749-6632.1979.tb56535.x. [DOI] [PubMed] [Google Scholar]
- Wilson B. W., Randall W. R., Patterson G. T., Entrikin R. K. Major physiologic and histochemical characteristics of inherited dystrophy of the chicken. Ann N Y Acad Sci. 1979;317:224–246. doi: 10.1111/j.1749-6632.1979.tb56531.x. [DOI] [PubMed] [Google Scholar]