Abstract
Several ETS transcription factors, including MEF/ELF4 and ERG, can function as oncogenes and are overexpressed in human cancer. MEF cooperates in tumorigenesis in retroviral insertional mutagenesis-based mouse models of cancer and MEF is overexpressed in human lymphoma and ovarian cancer tissues via unknown mechanisms. ERG (Ets-related gene) overexpression or increased activity has been found in various human cancers, including sarcomas, acute myeloid leukemia and prostate cancer, where the ERG gene is rearranged due to chromosomal translocations. We have been examining how MEF functions as an oncogene and recently showed that MEF can cooperate with H-RasG12V and can inhibit both p53 and p16 expression thereby promoting transformation. In fact, in cells lacking p53, the absence of MEF abrogates H-RasG12V-induced transformation of mouse embryonic fibroblasts, at least in part due to increased p16 expression. We discuss the known mechanisms by which the ETS transcription factors MEF and ERG contribute to the malignant transformation of cells.
Key words: Ras, AML1-ETO, p53, MDM2, prostate cancer, AML
ELF4 (also known as myeloid Elf-1 like factor, MEF) belongs to the ETS family of transcription factors, which contains over 30 family members. These have an evolutionarily conserved ETS domain that binds to a consensus “GGAA” DNA sequence via a winged helix-turn-helix motif. While some ETS proteins are transcriptional repressors, most are transcriptional activators (including MEF and ERG). ETS proteins are regulated by mitogenic signaling transduction pathways (e.g., Ras/MAPK), and play important roles in cellular differentiation, proliferation, apoptosis and tissue remodeling. ETS proteins implicated in hematopoietic cell differentiation include PU.1, FLI-1, ETS-1, ETS-2 and TEL. Aberrant expression of ETS proteins have been observed in prostate cancer, breast cancer, sarcoma, glioma and hematological malignancies.
The MEF gene is located on the X chromosome (Xq26) and MEF is normally expressed in many tissues, especially in ovary, placenta, colon and hematopoietic cells.1 MEF activates the expression of a diverse set of target genes (e.g., IL-3, GM-CSF, IL-8, Perforin and MDM2), and also plays a critical role in innate immunity affecting NK cell development and perforin gene expression.2,3
While the MEF gene has been reported to be fused to the ERG gene in a single patient with AML,4 and is overexpressed as result of retroviral insertional mutagenesis in mice,5–7 ERG overexpression is more commonly associated with human cancer. In a remarkable and paradigm changing discovery, a fusion involving the prostate-specific gene transmembrane protease serine 2 gene (TMPRSS2) and the ERG gene was identified in 80% of prostate cancer specimens.8 This gene fusion allows the androgen responsive 5′ regulatory element in TMPRSS2 to control ERG expression, which promotes prostate cancer. TMPRSS2 is also rarely fused to other ETS members such as ETV1, ETV4 and ETV5 in prostate cancer.9 ERG overexpression induces murine epithelial hyperplasia, but it requires collaboration with either PI3K signaling or androgen receptor overexpression to promote the development of prostate cancer in mice.10 The ERG gene is also overexpressed in Ewing's sarcoma (EWS/ERG), peripheral primitive neuro-ectodermal tumors (FUS/ERG), and in AML (also FUS/ERG) via chromosomal translocations. Furthermore, in AML cases with normal cytogenetics, ERG overexpression predicts for a worse outcome than “normal” ERG expression.11 To address the physiological role of ERG in blood cell proliferation, the ERG gene was targeted in mice: a loss of function Erg mutant was shown to impair definitive hematopoiesis and HSC function, demonstrating that Erg is essential for normal hematopoiesis.12 Erg also plays an important role in megakaryocytic differentiation and maturation.13 ERG overexpression promotes the proliferative and self-renewal capacity of HSCs, however overexpression of the FUS/TLS-ERG fusion gene is not sufficient to promote the development of AML.14 These studies imply that ERG overexpression plays a critical role in promoting the growth of various human tumors, however, what ERG target genes contribute to the development of cancer remain largely unknown.
MEF was first isolated approximately 15 years ago. MEF regulates the cell cycle transition from G1 to S phase, thereby promoting cell proliferation. Mef-null mice show greater number of quiescent hematopoietic stem cells (HSCs), indicating that Mef also promotes the transition of HSCs from G0 to G1 phase. By generating Mef-null/p53-null mice we determined that the enhanced quiescence of Mef null HSCs depends on p53 (Fig. 1A).15 To further determine the relevance of MEF to human tumors, we examined the level of MEF expression in ovarian cancer and B-cell lymphoma. MEF is overexpressed in ovarian cancer, with protein expression being seen in 48% of serous cystadenocarcinomas, 43% of endometrioid tumors, and 21% of clear cell tumors (where p53 mutations are more common).16 In 26 B-cell lymphoma patient samples with presumably wild type in p53, 23% (6 of 26) showed higher expression of MEF (and enhanced expression of MDM2, see below).17 MEF is also expressed in human AML, with higher expression in poor prognosis subtypes.18 Although the MEF gene is involved in a case of AML with a t(X;21), the main consequence of this translocation, which generates a fusion between MEF and ERG, may be the regulation of ERG expression by the MEF promoter.4 Thus, MEF may contribute to the development of ovarian and hematologic malignancies, consistent with its expression profile in normal human tissues.
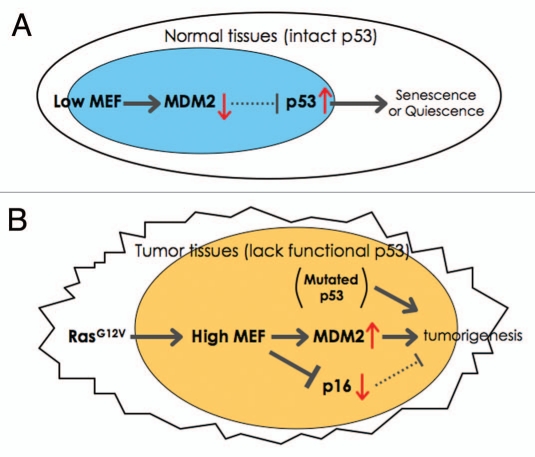
Figure 1.
A model of how MEF contributes to tumorigenesis. (A) This model shows how low (or null) MEF expression leads to senescence or quiescence due to enhanced p53 expression in normal tissues (e.g., HSCs). (B) The model for transformation in the absence of p53 and the presence of RasG12V (e.g., in ovarian cancer). Oncogenic-Ras can upregulate the expression of MEF. In the absence of wild type p53 (due to genetic deletion or mutations), MEF overexpression promotes transformation by activating MDM2 and suppressing Ras/Ets-1-induced p16 expression.
To understand the mechanisms by which MEF promotes tumorigenesis, we have recently shown that MEF can cooperate with oncogenic H-RasG12V in promoting transformation.17 In response to oncogenic stress, cells activate both the p53 and p16 tumor suppressor pathways, promoting apoptosis or cellular senescence in order to avoid transformation. In the absence of Mef, mouse embryonic fibroblasts show enhanced senescence with increased p53 expression, due to decreased Mdm2 expression. We determined that this is because Mef binds to and activates the Mdm2 promoter (Fig. 1A).17 MEF also has p53-independent effects on tumorigenesis inhibiting Ras/Ets-1 induced p16 expression; thus p53-null/Mef-null mouse embryonic fibroblasts are markedly resistant to H-RasG12V-induced transformation, at least in part due to the accumulated p16 expression (Fig. 1B). Thus, MEF promotes the tumorigenesis using both p53-dependent and p53-independent mechanisms.
In earlier studies, we showed that MEF also binds to wild type AML1 (also known as RUNX1) and to the AML1-ETO fusion protein, found in AML with t(8;21). AML1-ETO blocks the transcriptional activating function of MEF,19 and although MEF mRNA is detectable by northern blot in all AML cases studied, AML cases containing AML1-ETO do have lower MEF expression than other subtypes of AML.18 The role of MEF is likely context-dependent;18 however, given that ERG overexpression contributes to the more aggressive behavior of AML with normal cytogenetics, higher levels of Mef could function similarly. Yet, given the effects of MEF on HSC quiescence, the selection of low MEF-expressing cells could occur over time, as Mef-null HSCs are more resistant to chemotherapy or radiotherapy than normal HSCs. At this time, understanding how ETS proteins like MEF and ERG function in tumorigenesis, and how they are regulated by different signaling pathways, remains a priority and a fertile ground for exploration.
Abbreviations
- MEF
myeloid Elf1-like factor
- ELF4
E74-like factor 4
- ERG
Ets-related gene
- TMPRSS2
transmembrane protease serine 2
- AML
acute myeloid leukemia
- HSC
hematopoietic stem cells
Footnotes
Previously published online: www.landesbioscience.com/journals/cc/article/13000
References
- 1.Miyazaki Y, Sun X, Uchida H, Zhang J, Nimer S. MEF, a novel transcription factor with an Elf-1 like DNA binding domain but distinct transcriptional activating properties. Oncogene. 1996;13:1721–1729. [PubMed] [Google Scholar]
- 2.Lacorazza HD, Miyazaki Y, Di Cristofano A, Deblasio A, Hedvat C, Zhang J, et al. The ETS protein MEF plays a critical role in perforin gene expression and the development of natural killer and NK-T cells. Immunity. 2002;17:437–449. doi: 10.1016/s1074-7613(02)00422-3. [DOI] [PubMed] [Google Scholar]
- 3.Lacorazza HD, Nimer SD. The emerging role of the myeloid Elf-1 like transcription factor in hematopoiesis. Blood Cells Mol Dis. 2003;31:342–350. doi: 10.1016/s1079-9796(03)00162-1. [DOI] [PubMed] [Google Scholar]
- 4.Moore SD, Offor O, Ferry JA, Amrein PC, Morton CC, Dal Cin P. ELF4 is fused to ERG in a case of acute myeloid leukemia with a t(X;21)(q25-26;q22) Leuk Res. 2006;30:1037–1042. doi: 10.1016/j.leukres.2005.10.014. [DOI] [PubMed] [Google Scholar]
- 5.Lund AH, Turner G, Trubetskoy A, Verhoeven E, Wientjens E, Hulsman D, et al. Genome-wide retroviral insertional tagging of genes involved in cancer in Cdkn2a-deficient mice. Nat Genet. 2002;32:160–165. doi: 10.1038/ng956. [DOI] [PubMed] [Google Scholar]
- 6.Mikkers H, Allen J, Knipscheer P, Romeijn L, Hart A, Vink E, et al. High-throughput retroviral tagging to identify components of specific signaling pathways in cancer. Nat Genet. 2002;32:153–159. doi: 10.1038/ng950. [DOI] [PubMed] [Google Scholar]
- 7.Du Y, Spence SE, Jenkins NA, Copeland NG. Cooperating cancer-gene identification through oncogenic-retrovirus-induced insertional mutagenesis. Blood. 2005;106:2498–2505. doi: 10.1182/blood-2004-12-4840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tomlins SA, Rhodes DR, Perner S, Dhanasekaran SM, Mehra R, Sun XW, et al. Recurrent fusion of TMPRSS2 and ETS transcription factor genes in prostate cancer. Science. 2005;310:644–648. doi: 10.1126/science.1117679. [DOI] [PubMed] [Google Scholar]
- 9.Kumar-Sinha C, Tomlins SA, Chinnaiyan AM. Recurrent gene fusions in prostate cancer. Nat Rev Cancer. 2008;8:497–511. doi: 10.1038/nrc2402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zong Y, Xin L, Goldstein AS, Lawson DA, Teitell MA, Witte ON. ETS family transcription factors collaborate with alternative signaling pathways to induce carcinoma from adult murine prostate cells. Proc Natl Acad Sci USA. 2009;106:12465–12470. doi: 10.1073/pnas.0905931106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Marcucci G, Baldus CD, Ruppert AS, Radmacher MD, Mrozek K, Whitman SP, et al. Overexpression of the ETS-related gene, ERG, predicts a worse outcome in acute myeloid leukemia with normal karyotype: a Cancer and Leukemia Group B study. J Clin Oncol. 2005;23:9234–9242. doi: 10.1200/JCO.2005.03.6137. [DOI] [PubMed] [Google Scholar]
- 12.Loughran SJ, Kruse EA, Hacking DF, de Graaf CA, Hyland CD, Willson TA, et al. The transcription factor Erg is essential for definitive hematopoiesis and the function of adult hematopoietic stem cells. Nat Immunol. 2008;9:810–819. doi: 10.1038/ni.1617. [DOI] [PubMed] [Google Scholar]
- 13.Stankiewicz MJ, Crispino JD. ETS2 and ERG promote megakaryopoiesis and synergize with alterations in GATA-1 to immortalize hematopoietic progenitor cells. Blood. 2009;113:3337–3347. doi: 10.1182/blood-2008-08-174813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pereira DS, Dorrell C, Ito CY, Gan OI, Murdoch B, Rao VN, et al. Retroviral transduction of TLS-ERG initiates a leukemogenic program in normal human hematopoietic cells. Proc Natl Acad Sci USA. 1998;95:8239–8244. doi: 10.1073/pnas.95.14.8239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Liu Y, Elf SE, Miyata Y, Sashida G, Liu Y, Huang G, et al. p53 regulates hematopoietic stem cell quiescence. Cell Stem Cell. 2009;4:37–48. doi: 10.1016/j.stem.2008.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yao JJ, Liu Y, Lacorazza HD, Soslow RA, Scandura JM, Nimer SD, et al. Tumor promoting properties of the ETS protein MEF in ovarian cancer. Oncogene. 2007;26:4032–4037. doi: 10.1038/sj.onc.1210170. [DOI] [PubMed] [Google Scholar]
- 17.Sashida G, Liu Y, Elf S, Miyata Y, Ohyashiki K, Izumi M, et al. ELF4/MEF activates MDM2 expression and blocks oncogene-induced p16 activation to promote transformation. Mol Cell Biol. 2009;29:3687–3699. doi: 10.1128/MCB.01551-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Fukushima T, Miyazaki Y, Tsushima H, Tsutsumi C, Taguchi J, Yoshida S, et al. The level of MEF but not ELF-1 correlates with FAB subtype of acute myeloid leukemia and is low in good prognosis cases. Leuk Res. 2003;27:387–392. doi: 10.1016/s0145-2126(02)00214-x. [DOI] [PubMed] [Google Scholar]
- 19.Mao S, Frank RC, Zhang J, Miyazaki Y, Nimer SD. Functional and physical interactions between AML1 proteins and an ETS protein, MEF: implications for the pathogenesis of t(8;21)-positive leukemias. Mol Cell Biol. 1999;19:3635–3644. doi: 10.1128/mcb.19.5.3635. [DOI] [PMC free article] [PubMed] [Google Scholar]