Abstract
Implantable medical devices are increasingly important in the practice of modern medicine. Unfortunately, almost all medical devices suffer to a different extent from adverse reactions, including inflammation, fibrosis, thrombosis and infection. To improve the safety and function of many types of medical implants, a major need exists for development of materials that evoked desired tissue responses. Because implant-associated protein adsorption and conformational changes thereafter have been shown to promote immune reactions, rigorous research efforts have been emphasized on the engineering of surface property (physical and chemical characteristics) to reduce protein adsorption and cell interactions and subsequently improve implant biocompatibility. This brief review is aimed to summarize the past efforts and our recent knowledge about the influence of surface functionality on protein:cell:biomaterial interactions. It is our belief that detailed understandings of bioactivity of surface functionality provide an easy, economic, and specific approach for the future rational design of implantable medical devices with desired tissue reactivity and, hopefully, wound healing capability.
Keywords: functional groups, biomaterials, biocompatibility, proteins, cells
Introduction
An aging population, coupled with continued medical advances, has driven the increased utilization of medical implants and demand for new medical devices in the years ahead. Unfortunately, blood-contact medical devices may trigger a variety of iatrogenic reactions, including surface mediated thrombosis, complement activation, and device-centered infection [1–2]. There are also significant adverse reactions involved with tissue-contact medical implants. These include (1) local implant-associated inflammation, including temporomandibular [3–4] and other joint related implants [4–8], (2) phagocyte-mediated oxidation and ”stress cracking” of the implanted materials [9–10], (3) implant degradation and tissue fibrosis surrounding mammary prosthesis and many other types of implants [11–13], (4) poor integration of fibrotic tissues artificial ligaments using artificial (Woven Dacron®) or biological (such as collagen fiber) materials [14–16], (5) the formation of thick fibrotic capsules surrounding implants contribute to the failure of many types of devices, such as biosensors [17–18], joint implants [19–20], breast implants [21–22], encapsulated tissues/cells [23–25], drug delivery systems [26], and eye implants [27–29]. Unfortunately, the mechanisms governing biomaterial-mediated tissue responses and the possible role of surface properties affecting such responses are mostly unclear.
At first glance, these inflammatory responses to inert, non-immunogenic, and non-toxic materials are difficult to understand. We and many others have found that, shortly after implantation (minutes to hours), biomaterial implants are covered with a layer of host proteins prior to the accumulation of inflammatory cells [13,30–32]. Thus, it is generally believed that the composition and state of these adsorbed proteins play a pivotal role in subsequent host coagulation and immune reactions [13,30,32–33]. Therefore, the ability to control both (1) the amount and composition of host protein adhering to a surface, as well as (2) the degree of adsorbed proteins’ conformational changes (exposing inflammatory epitopes), is the primary goal of modified biomaterial surfaces.
Many surface modification techniques have been developed during the past twenty years. The modifications of surface wettablility, hydrophobicity and surface charges have been shown to alter the extent of protein adsorption. Overall, increasing surface hyrdophilicity has been shown to improve the biocompatibility of the medical devices at least in vitro. Recently more effort has been placed on creating surfaces uniformly coated with different chemical functionalities in an attempt to determine more specifically what surface chemical entities, beyond hydrophobic or hydrophilic characteristics, can improve biomaterial compatibility. Though promising in vitro results have been widely documented in literature, surface functionalities have shown little success in exerting a significant influence on tissue responses in vivo. The purpose of this article is to review past and recent work in the area of surface group functional modification of biomaterials, and what effect these groups have on protein and cellular interactions with surface functionalities.
How does biomaterial trigger foreign body reactions?
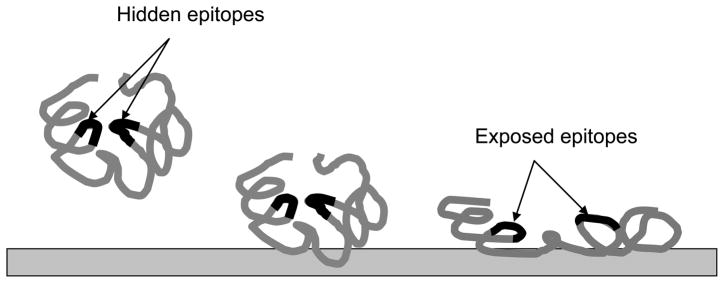
Commonly used biomaterials are mostly hydrophobic and have high affinity to a wide variety of proteins. Shortly after implantation, biomaterials are covered with a layer of plasma proteins, predominately albumin, fibrinogen, IgG, fibronectin, and von Willebrand factor [30,32–35]. These adsorbed proteins, possibly via hydrophobic interactions, tend to assume an altered conformation and to expose hydrophobic domains which become tightly adherent to hydrophobic biomaterial surfaces [36–41]. Such surface-mediated conformational changes of adsorbed proteins have been demonstrated by several different techniques including resistance to sodium dodecyl sulfate (SDS) elution [42–43], scanning angle reflectometry [12,44–45], ‘scanning force’ microscopy [46–48] and attenuated total reflectance Fourier transform infrared spectroscopy [37,49]. The conformational changes of adsorbed protein are thought to be responsible for initiating adverse reactions such as inflammation, coagulation, and foreign body response [39,50–52]. It is believed that these protein:biomaterial interactions prompt the exposure of hidden protein structures and sequences that serve as receptor sites for inflammatory cells, which then initiate the foreign body reactions (Figure 1).
Figure 1.
Schematic drawing to depict the protein:biomaterial interactions which often lead to conformational changes of adsorbed proteins and the subsequent exposure of hidden proinflammatory epitopes.
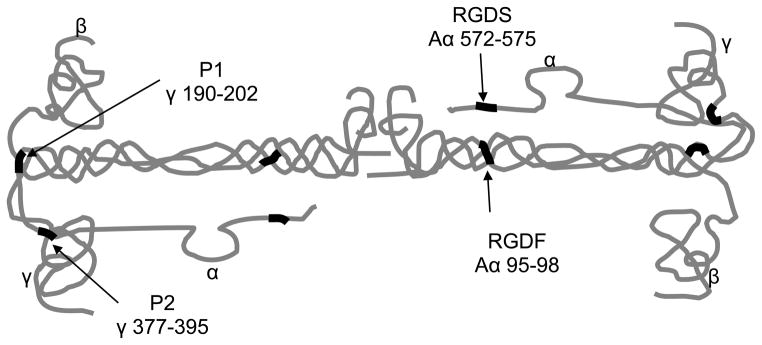
Although the influence of material surface properties on subsequent tissue responses is not totally understood, it is widely documented that polymer surface properties can affect the amounts and types of bound proteins, as well as the conformation, orientation or binding strength of the adsorbed protein [12,31,38,53–54]. Adsorbed proteins can either retain a structure close to that in solution or may conformationally adjust in response to local environments [55]. This time-dependent conformational readjustment is generally believed to affect the cellular responses and subsequent tissue reactions [12,31,38,53]. In the case of fibrinogen, the resistance to SDS elution has been correlated with slow, progressive changes in state of the surface protein on biomedical polymers such as polyetherurethanes [36]. After polyethylene terephthalate (PET) film has been incubated with fibrinogen for 4 hours, more than 60% of the protein is resistant to SDS elution [56]. Albumin and IgG behave similarly [57]. A recent study employing grazing angle infrared analysis was conducted to determine the interactions of fibrinogen and albumin adsorption onto hydrophobic and hydrophilic surfaces. Results showed within 1 hour of fibrinogen adsorption, changes in secondary structure (amide I band) could be seen suggesting that changes in fibrinogen structure were due to protein-surface interactions [58–59]. The degree of secondary structure alteration was shown to be dependent on the properties of the surface (hydrophilic versus hydrophobic) [60]. It has been shown that, upon adsorption to plastic tissue culture surfaces (which are not precisely analogous to most hydrophobic implant surfaces), fibrinogen changes conformation and exposes multiple receptor-induced binding sites (RIBS). Two of these RIBS epitopes have been localized to residues gamma112–119 (recognized by mAb 9F9) and Aα 95–98 (RGDF; recognized by mAb 155B16). Please see Figure 2 for the locations of some of these epitopes which are known to interact with immune cells. A similar exposure of these RIBS epitopes and fibrin-specific epitopes may also occur upon adsorption and conformational change of fibrinogen on implantable biomaterials [61–63].
Figure 2.
Schematic drawing of fibrin(ogen) showing the approximate locations of some of the epitopes which have been implicated to be responsible for triggering foreign body reactions. The potential inflammatory epitopes include P1 (γ190–202), P2 (γ377–395), RGDS (Aα 572–575), and RGDF (Aα 95–98).
Taken together, these findings indicate that upon interaction with implant surfaces, fibrinogen (i) becomes progressively more adherent, (ii) shows signs of changes in conformation (?denaturation?), particularly involving the D domain, and (iii) exposes previously occult sequences (e.g., RIBS, P1 and P2). Based on these earlier observations, it is likely that material properties can be modified to reduce the extent of neo-epitopes’ exposure and subsequent cell and tissue responses.
Influence of surface chemistry on biocompatibility
Surface modification techniques
In the search for “perfect” surfaces, material scientists have been modifying surface characteristics by a plethora of techniques including physical modifications, chemical modifications, and radiation [64–65]. In general, the modification of material surface properties (including chemistry, wettability, domain composition and morphology) has been shown to influence protein adsorption and subsequent cellular responses to biomaterials in vitro. Unfortunately, due to the lack of well-defined surfaces (that differ only in one or two properties) and well-characterized animal implantation models, many early studies have produced little insight into the influence of surface properties on the pathogenesis of foreign body reaction. For creating homogenous and well-defined surfaces, self-assembled monolayer techniques, chemical graft modification, and plasma medication techniques have been developed and used in many recent studies. Each method has its unique benefits and drawbacks.
Chemical graft modification has been employed to chemically immobilize compounds onto the surface of a biomaterial. This method usually involves covalent conjugation of either a protein or monomer to the surface to alter surface chemistry, avoiding drawbacks of protein/monomer detachment by providing long-term stability. The process involves activating the surface with reactive groups followed by grafting the desired functionality to the surface. Many different methods fall into this category and differ only by the way in which the surface is activated, including chemical reactions, UV, radiation exposure, plasma, and ozone exposure [66]. Chemical grafting has been used to mount heparin and PEO to surfaces to increase blood compatibility. Drawbacks, especially when grafting proteins, include the loss of protein mobility on the surface, and possibly, presentation of the protein in an unfamiliar conformation on the surface. Toxic monomer residues may also be left in the grafted surface. Some issues can be addressed by physical adsorption techniques, which usually involve dip coating a material to form a film with desired properties on the surface. While physical adsorption may help reduce toxic monomer residues, issues with binding between the materials arise as well as instability of immobilization when using proteins.
Addressing the drawbacks in chemical and physical modification, self-assembled monolayers (SAMs) were developed as a method to more precisely control the density and conformation of a single or multiple specific functional groups on a surface. The general process of producing SAMs is to first activate the bulk material surface, then graft polymerize onto the activated surface. SAMs can provide flat and chemically well defined surfaces [67], with additional benefits of a surface near thermodynamic equilibrium with closely packed, well-ordered functionalities on the surface [66]. SAMs tend to provide functionalities with control over pattern and densities. Selection of terminal group can also provide a site for further functionalization of the coated surface. Surface functionality via SAMs have been used to investigate in vivo inflammatory and foreign body responses of implanted biomaterials [68–69] as well as many other processes, giving insight into how a particular functional group effects a particular process. However, SAMs are limited to gold-coated or silver coated surfaces.
An increasing popular method of altering surface functionality of biomaterials is plasma modification. Plasma, which can be regarded as the fourth state of matter, is composed of highly excited atomic, molecular, ionic, and radical species. It is typically obtained when gases are excited into energetic states by radio frequency, microwave, or electrons from a hot filament discharge [70]. The process provides an economical and effective way to infer functionality to a surface, and is compatible with most materials currently being investigated in medicine including metals [71], polymers [72], and ceramics [73]. One of the great benefits of plasma modification is the ability to uniformly modify surfaces regardless of geometry, allowing for modification of micro and nanoparticles [74], films [72], or 3D components required in tissue engineering and artificial organs [75]. In addition, the use of pulsed plasmas, in lieu of the conventional continuous-wave operational mode, provides an exceptionally high level of film chemistry controllability during the coating process [76–80].
An increasing number of chemical functionalities have been placed on implant surfaces. The influence of these functionalities on protein adsorption, cellular and tissue reactions has also been investigated. Highlighted in the next section, is a summation of the recent understanding of surface chemistry as related to protein adsorption, cellular and tissue reactions.
Effects of Surface Chemistry on Protein:Surface Interactions
Surface properties affect the extent of protein adsorption, denaturation, and epitope exposure (specifically γ190–202 [P1] and γ377–395 [P2]) [38,81]. To reduce unwanted protein:surface interactions, intensive research efforts have been placed on engineering surfaces with uniform functional groups to “repel” protein. The typical characteristics of protein-resistant functional groups include: hydrophilic nature, presence of hydrogen bond acceptors, no hydrogen bond donors, and neutral charge [82], though there appear to be exceptions. It is generally believed that the identification of protein resistant functional coating can help to tailor material surfaces to control protein adhesion and their interactions following adsorption, and can thus enhance biocompatibility.
The effect of surface functionality on fibrinogen adsorption has been investigated vigorously in recent years [83–86]. Computer simulations have show that water is essentially able to successfully compete with fibrinogen over adsorption to hydroxyl coated surfaces, reaffirming results indicating that fibrinogen binds tightly to hydroxyl (-OH) surfaces [84]. It has also been suggested that an optimum concentration of -OH groups can enhance the affinity for albumin binding over fibrinogen [86]. Fibrinogen was however able to hydrophobically interact with methyl functionalized surfaces, in agreement with previous studies that suggest abundant adsorption of fibrinogen to methyl (-CH3) functionalized surfaces [85]. Amine (-NH2) surfaces were able to form hydrogen bonds with fibrinogen, tethering it to the amine functionalized surface. In the case of carboxyl (-COOH) groups, fibrinogen was not able to out-compete water to interact with the carboxyl surface [81]. A similar result was also seen in another investigation in which positive, negative, hydrophobic, and hydrophilic surfaces were adsorbed with equivalent amounts of fibrinogen. Negatively charged surfaces, such as -COOH, formed little to no fibrin compared to other surfaces [83]. Further research involving mixed functionalities as well as increased in vivo investigations are needed to develop a more comprehensive understanding of surface functionality and protein interactions.
To determine the possible role(s) of functional group density in modulating protein:biomaterial interactions, we have generated surfaces bearing different densities of –OH and –NH2 functional groups using Radio Frequency Glow Discharge plasma polymerization technique. As expected, increasing the density of surface -OH and -NH2 improves surface hydrophilicity. In the case of surface:fibrinogen interactions, -OH bearing surfaces retained progressively less protein, whereas the -NH2 surfaces adsorbed progressively more protein as the surface density of -OH and -NH2 groups increased, respectively. This data confirms that the amount of spontaneously adsorbed protein is a function of surface chemistry. Subsequent studies have been done to determine whether the species and density of surface functional groups affect protein conformational changes (also called as denaturation). Our results confirm that even these modest changes in surface amine density strongly influence fibrinogen adsorption and P1/P2 epitope exposure.
Surface chemistry of biomaterials has also been shown to participate in complement cascades through both the classical and alternative pathway, leading to the recruitment and activation of phagocytes and the adherence and activation of leukocytes [12]. This was first identified in hemodialysis membranes [87] and activation was shown to occur on surface with many different functionalities. Results have suggested that IgG undergoes a more extensive denaturation on hydrophobic as opposed to hydrophilic surfaces, resulting in exposure of active sites which bind complement proteins [88]. It has also been suggested that the chemical functionalities themselves can covalently bind complement proteins, harboring them from regulatory proteins [89]. Hydroxyl (-OH) functionalized surfaces have been shown to selectively adsorb IgG from serum, ultimately leading to C3 deposition on the surface [90]. In contrast, surfaces functionalized with -NH2 and -COOH showed only slight activation in comparison to -OH functionalized surfaces [69]. In vivo studies using SAMs with -OH functionality showed increased leukocyte response [91]. This process is composed of at least three continuous steps- the surface –OH-mediated complement activation, complement fragment C5a-triggered leukocyte activation, and leukocyte adhesion via binding to surface-bound complement fragment C3b [92].
Influence of surface functional groups on cellular responses
Substantial research efforts have been placed on studying the influence of surface functionality in the cellular response to biomaterials. Surface functional groups can influence cell growth [93–94], likely due to the fact that surface chemical functionality affects adsorbed protein and subsequent protein:cell interactions. In general, hydrophilic functionality provides low interfacial free energy resulting in reduced protein adsorption, cell adhesion, and blood compatibility [54,95]. It is well established that proteins tend to bind to hydrophilic surfaces in a lower amount and less tightly than to hydrophobic surfaces [96–98]. Such reduction of protein adsorption affects subsequent cellular responses. For example, in an interpenetrating network study, it was seen that increasing hydrophilic monomers lead to a decrease in fibrinogen adsorption and an increase in albumin adsorption [99], as well as a gradual decrease in platelet adhesion [100]. Decreasing platelet adhesion has also been attributed to coatings that are non-ionic and neutral [101–102]. It has also been suggested that hydrophilic surfaces provide significant inhibition of leukocyte adhesion and macrophage fusion [103], and may result in decreased cytokine secretion and attenuated inflammatory reactions [104]. In contrast, the addition of –CH3 groups, imparting hydrophobic functionality, has been shown to increase leukocyte adhesion [105]. The adhesion of leukocytes to solid surfaces depends on many different factors such as surface chemistry, charge or hydrophilicity, and protein adsorption [105].
Though the influence of surface hydrophobicity on protein adsorption and cellular responses is well documented, recent studies have revealed that surface functional groups also affect protein and cell behavior [106–109]. The most common functionalities investigated with relation to biomaterial interactions are the carboxyl (-COOH), hydroxyl (-OH), amino (-NH2), and methyl (-CH3) groups. Many recent reports have investigated the effect of these groups on the binding of adhesive proteins and subsequent cellular interactions. The results of their findings are summarized below.
Carboxyl (-COOH) functional group-bearing surface
Biomaterial-bearing -COOH displays a negative charged functionality on material surfaces [109]. Studies on protein adsorption have shown that fibronectin and albumin are more easily eluted from surfaces coated with –COOH [93]. This was tied into the fact that this functionality, compared to other common surface functional groups, adsorbed FN and albumin at ratio significantly different to other coatings. Surfaces with –COOH also have shown an increase in cell growth. A more recent study showed that this phenomenon is dependent upon the concentration of –COOH on the surface, as an increase in functional group density results in a higher negative charge on the surface, which was shown to inhibit cell growth [94]. Carboxyl functional surfaces, pre-treated with FN, showed high levels of two FN domains (α5β1 and αvβ3) associated with structural and signaling components related to focal adhesions [109]. On the other hand, -COOH-mediated αvβ3 exposure has been shown to inhibit osteoblast differentiation and mineralization. Similarly, increase in αvβ3 integrin expression on –COOH surfaces was correlated with a low level of myoblast differentiation though cell proliferation levels were high [110].
Hydroxyl (-OH) functional group-coated surfaces
The hydroxyl group functionality (-OH) represents a neutral, hydrophilic surface. Early research into surface functionality suggested that an increase in oxygen containing functionalities was proportional to cell growth [111], sparking further research in oxygen containing functional groups such as –OH. FN adsorbed onto -OH functional groups (in comparison with –CH3 functional group) show high levels of α5β1 levels leading to increased cell adhesion strength as well as increased levels of structural signaling components related to focal adhesions [109]. Further investigation with osteoblasts showed high levels of differentiation and mineralization with –OH functionality as opposed to other functional groups. Rather contradictory, -OH functionality has been shown to have reduced plasma protein adsorption and thus higher platelet compatibility [112]. It has been suggested that the charge neutrality and hydrophilic nature of the -OH functionality has low protein affinity, and thus protein repelling properties [113].
Amine (-NH2) functional group-rich surfaces
Amine group (-NH2) functionality displays a positive charge to the biomaterial surface. Studies using FN and osteopontin (OPN) show favorable protein conformations after adsorption to the positively charge -NH2 surface [109,114]. Particularly, -NH2 surfaces promote the exposure of high density bound receptors as well as focal adhesion components by adsorbed fibronectin [109]. These favorable protein adsorption profiles often lead to increased endothelial cell growth [114] and enhanced differentiation and mineralization of osteoblast cells [109,115]. Preferable adhesion, growth, and matrix formation was also shown with fibroblasts on –NH2 surfaces compared with other coatings [116–117]. Further analysis showed than within 45 minutes, cells cultured on -NH2 had already begun the formation of focal adhesion plaques, linked to increased cell spreading on the surfaces [116]. However, in a study using myoblasts, -NH2 functionality was shown to promote high levels of myoblast proliferation with low levels of differentiation [110].
Methyl (-CH3) functional group-bearing surfaces
The -CH3 group is the major component of commonly used polymeric materials and provides a hydrophobic surface on biomaterials. It is generally accepted that the hydrophobic –CH3 functionality promotes protein adsorption, usually in conformations unfavorable for desired cellular interactions [109]. As a result, -CH3 functionality has also shown increased fibrinogen binding, platelet accumulation and thus poor blood compatibility [112]. A study measuring the adhesion strength of fibrinogen, albumin, and IgG showed that all three proteins adhered with the highest strength to -CH3 bearing surfaces [118]. Overall, these results suggest that -CH3 surfaces will likely have unfavorable surface reactions with cells due to the magnitude of tightly bound proteins, and the likelihood that the bound proteins will expose sites responsive to inflammatory cells.
Surfaces with Mixed Functionality
In the recent years, research has progressed to the evaluation of mixed functional group chemistries on surfaces. The goal of these studies is to attempt to combine favorable properties of both functionalities onto one surface to enhance biocompatibility. Interestingly, the properties of each functional group seem to almost equally contribute to the overall properties of the surface. Using a SAM combining -NH2 (positive charge) and -COOH (negative charge) functionality in different proportions, it was seen that at equal molar fractions the lowest platelet adsorption was seen [119]. This equal proportion of the functionalities left the surface with a near neutral charge. This finding highlights the possible importance of surface neutrality related to blood compatibility of biomaterials.
Another study, investigating -OH and -CH3 mixed SAMs, found a decrease in platelet adhesion and activation as well as decreased fibrinogen adsorption with increasing hydrophilicity, attributed to the -OH functionality [120]. This study further highlights the importance of a wettable surface in decreasing blood platelet interactions. With regards to surface adhesion, it was seen that hydrophobic SAMs combining -COOH and -CH3 groups had a higher adhesion than hydrophilic -COOH and -OH SAMs [121]. A study using -PO3H2 functionality with -CH3 showed that though total number of platelets increased with increasing -PO3H2 content, the number of activated platelets decreased [122]. However, when considering -PO3H2 and -OH mixed SAMs, the number of activated platelets increased with increasing -PO3H2 with the lowest adhesion seen at almost equivalent proportion of the functionalities. Highlighting the importance of the fact that OH functionality has been shown to have higher platelet compatibility than -CH3 due to its hydrophilic nature and neutral charge. This result also suggests that the platelet compatibility of -PO3H2 (low hydrophilic and anionic) is somewhere between -OH and -CH3 [122]. At this time, there has been no work published with mixed SAMs in vivo. The in vivo interactions of mixed surface functionality would likely provide valuable information and fill a large information void on the effects of surface functionality in vivo.
Effect of surface functionality on tissue responses
Despite a significant body of in vitro results, there are very few works which had focused on the effects of surface functional groups in vivo. Almost all in vivo studies were carried out in an acute implantation model. The results from those works have supported that surface functionality may alter biomaterial-mediated acute inflammatory responses in vivo [108,123–124]. Using an intraperitoneal implantation model with RFGD functionalized disks, our early results have shown that surface functionality prompts different extents of acute inflammatory responses in the order: -NH2 > CH3 -CFx > -OH > siloxyl group [108]. A recent study was carried out using using SAMs to produce hydrophobic –CH3 and hydrophilic -OH and -COOH surfaces. The results have shown that - CH3 surface functionality slightly increases the thickness of the fibrous capsule formed around implants as well as higher recruitment of inflammatory cells in comparison to -OH and -COOH functionalities [68]. In a separate study it was seen that –CH3 terminated SAMs lead to an increase in leukocyte and phagocyte migration to the implant site compared to the gold surface alone [124].
Surprisingly, almost all previous studies failed to show the effects of surface chemistry on chronic fibrotic responses in vivo [68,108,125]. We speculated that the relatively ineffectiveness of surface functionality on long-term in vivo cellular responses may simply reflect insufficient cell:surface interactions. Specifically, in an in vitro setting, cells are seeded on top of functional group and surface functionality, thus, exerting maximal effect on cellular responses. On the other hand, in an in vivo environment, the functional groups on non-porous implants can only interact with the first layer of the cells which represent only a small portion of the surrounding cells/tissue. These relatively poor cell-functional group interactions thus minimize the effect of surface functionality on cellular responses. Thus we hypothesized that possibly the influence of implant surface chemistry may be substantially enhanced by increasing the interaction between functional groups and cells in applications such as tissue scaffolds and drug releasing microspheres. To test this hypothesis, we have examined, in vivo, responses to microspheres bearing different, molecularly tailored, surface functionalities. Spherical particles were chosen for this purpose to maximize the surface to volume ratio of the implants and also to mimic microspheres used for controlled drug release. To test this hypothesis, we have recently reported a study using functionalized micron-sized particles, created and tested for their ability in modulating tissue responses to biomaterial implants. In this work, the surfaces of polypropylene particles were controllably coated with four different functional groups, specifically –OH, -NH2, -CFx and –COOH, using a RFGD plasma polymerization technique [74]. The effect of these surface functionalities on host tissue responses were then evaluated in vivo. Surfaces with hydrophilic –OH and –NH2 surface groups induced the thickest fibrous capsule accompanied with the greatest cellular infiltration into the implants. As previously mentioned, hydrophilic groups have been shown to reduce protein adsorption, though these studies used thin films consisting of functionality derived from SAMs. In contrast, surfaces with –CFx and –COOH exhibited the least inflammatory/fibrotic responses and cellular infiltrations. The results suggest the surface/volume ratio of the implant, as well as surface functionality, may play an important role in influencing tissue responses and biocompatibility [74].
Role of Surface Chemistry in Tissue Engineering
Investigations into the effects of surface functionality in tissue engineering are still in its infancy. Recently groups have focused on the incorporation or modification of scaffold surfaces to enhance hydrophilicty. As previously mentioned, knowledge of how functional groups effect cell adhesion properties can be used to modify scaffold surfaces to enhance cell adhesion while also reducing inflammatory response to the scaffold material. Currently most research involving tissue engineering scaffolds is focused on the addition of ECM derived materials to enhance cell growth and differentiation (126). Surface functional groups are being used primary to facilitate the chemical addition of specific membrane ligands to promote cell adhesion. As knowledge of the in vivo effects of surface functional group coating of biomaterial surfaces expands, we will likely see increased research and application of surface group functionalized scaffolds.
Interestingly, the results from recent studies suggest that surface functionality can induce cell differentiation. A study using Caco-2 cells showed that there was a relationship between perfluoronated functional coating and degree of cell differentiation [127]. In a separate study, –COOH-mediated αvβ3 integrin exposure has been shown to inhibit osteoblast differentiation and mineralization [112]. Investigation of -OH functionality with adhered osteoblasts showed high levels of differentiation and mineralization with –OH, and -NH2 functionality as opposed to other functional groups including -COOH, -SO4, and –CH3 terminal groups [109,112,115]. There remain many possibilities as to the characterization of protein adherence to functional coatings and the subsequent effects of these proteins and their configuration on cell differentiation.
Functionalized surfaces to improve drug delivery
Surface chemical effect on reducing diffusion barrier and improving drug release
Like many other implantable medical devices, drug release implants are often surrounded by thick layer of collagenous fibrotic tissue a few days (weeks) after implantation. The fibrotic tissue may serve as diffusion barrier and, thus, reduce the drug transport from implanted drug release devices to the circulation (Figure 3). Therefore, it is generally believed that the reduction of biomaterial-mediated fibrotic reactions may improve the drug release properties [128–130]. Results using SAMs on gold have shown that implants coated with -COOH and -OH functionality produce thinner fibrous capsules than -CH3 functionality [68]. Results from our laboratory using polypropylene functionalized microsphere implants also showed thin capsules for -COOH functionality. However, significantly increased fibrous capsule formation was found surrounding implants with -OH functionality [74]. More research is needed to determine the relationship between surface functional groups and resulting drug release properties.
Figure 3.
Prominent fibrotic tissue formation was found surrounding poly-L-lactic acid particles which were subcutaneously implanted in Balb/C mice for 2 weeks. The presence of collagenous fibrotic tissue create diffusion barrier for drug transport from implanted particles to blood stream.
Surface functionality on improving drug cellular uptake
A recent study has also suggested that surface functionality of drug delivery vehicles affects both the rate and mechanism of cell uptake. Using dendrimers with terminal groups of -NH2, -OH, and PEG, results have shown that -NH2 surfaces were able to enter cancer cells at a higher rate than -OH and PEG functionalized surfaces. It is suggested that the favorable interactions of the cationic -NH2 surface with the cell membrane allow quicker passage of drug delivery vehicles into the cell. It is likely that the -OH and PEG functionalities, due to their anionic nature, increase affinity to the cell membrane, and then taken in by an endocytotic route [131]. Another study investigating -OH, -NH2, and -COOH terminal groups on dendrimers showed that -COOH and -OH functional surfaces tend to have increased residence times in vivo, which may be attributed to their ability to resist recognition by the body through protein adsorption, as well as cell uptake properties due to surface functionality [132].
Surface coating has been shown to effect cellular uptake of nanoparticles [133–134]. In general, micro and nanoparticles with a hydrophilic shell show decreased protein adsorption and increased circulation time [135]. Particles modified with a PEG shell, with a hydrophilic –OH surface functionality, can reduce particle uptake by mononuclear phagocytes [136]. A recent study, examining the effects of altering surface charge on particle uptake, showed that increasing –NH2 group functionality on the particle surface has a correlation to cell uptake [137]. This phenomenon was also shown to be cell dependant, with cancerous cell lines taking up more particles with an increase in –NH2 group density on the surface. An interesting trend was seen with hydrophilic PVA surface modification with –OH functionality. In a study comparing uncoated PLGA versus -OH coated PLGA, it was seen that the addition of the hydrophilic coating led to a 2 fold increase in cellular uptake [138]. A recent study has shown that -COOH functional surface enhances cell uptake and the amount of nanoparticle uptake could be correlated to the amount of -COOH functionality on the nanoparticle surface. Such interesting phenomenon might be due to favorable interactions of the cell with the negatively charged coating [139].
Conclusion
A significant body of research clearly demonstrates that in vivo tissue compatibility may be modified by varying surface chemistry. Many recent works have uncovered that the species, density and composition of surface functional groups play an important role of controlling protein, cell and tissue reactions to material implants (Table 1). More in vivo studies are needed to define the interactions between surface functionality and host responses. It is our belief that such knowledge may help the development of molecularly tailored surface to improve the function and safety of many medical devices, including drug release devices, encapsulated cells and implantable sensors.
Table 1.
Effect of surface functionality on protein adsorption, cell behavior and tissue responses.
| Surface Functional Group | Protein, Cell, and Tissue Properties |
|---|---|
| Carboxyl (-COOH) hydrophilic, negatively charged |
|
| Hydroxyl (-OH) hydrophilic, neutral charge | |
| Amine (-NH2) hydrophilic, positive charge |
|
| Methyl (-CH3) hydrophobic, neutral charge | |
| Mixed -NH2 & -COOH positive & negative |
|
| Mixed -OH & -CH3 hydrophobic & hydrophilic |
|
| Mixed -PO3H2 & -OH negative & neutral |
|
Acknowledgments
This work was supported by a NIH grant RO1 GM074021, a Texas Higher Education Coordinating Board’s Advanced Technology Program (003594-0003-2006), and an AHA Established Investigator Award.
Abbreviations
- SDS
Sodium dodecyl sulfate
- PET
polyethylene terephthalate
- RIBS
receptor-induced binding sites
- (SAMs)
Self-assembled monolayers
- -OH
Hydroxyl
- -CH3
methyl
- -COOH
carboxyl
- -NH2
amine
References
- 1.Schmidt DR, Kao WJ. The interrelated role of fibronectin and interleukin-1 in biomaterial-modulated macrophage function. Biomaterials. 2007;28:371–382. doi: 10.1016/j.biomaterials.2006.08.041. [DOI] [PubMed] [Google Scholar]
- 2.Nilsson B, Ekdahl FN, Mollnes TE, Lambris JD. The role of complement in biomaterial-induced inflammation. Mol Immun. 2007;44:82–94. doi: 10.1016/j.molimm.2006.06.020. [DOI] [PubMed] [Google Scholar]
- 3.Trumpy I, Lyberg T. In vivo deterioration of proplast-teflon temporomandibular joint interpositional implants: A scanning electron microscopic and energy-dispersive X-ray analysis. J Oral Maxillofac Surg. 1993;51:624–629. doi: 10.1016/s0278-2391(10)80259-9. [DOI] [PubMed] [Google Scholar]
- 4.Henry CH, Wolford LM. Treatment outcomes for temporomandibular joint reconstruction after Proplast-Teflon implant failure. J Oral Maxillofac Surg. 1993;51:352–358. doi: 10.1016/s0278-2391(10)80343-x. [DOI] [PubMed] [Google Scholar]
- 5.Thornhill TS, Ozuna RM, Shortkroff S, Keller K, Sledge CB, Spector M. Biochemical and histological evaluation of the synovial-like tissue around failed (loose) total joint replacement prostheses in human subjects and a canine model. Biomaterials. 1990;11:69–72. [PubMed] [Google Scholar]
- 6.Jasty M. Clinical reviews: Particulate debris and failure of total hip replacements. J Applied Biomaterials. 1993;4:273–276. doi: 10.1002/jab.770040310. [DOI] [PubMed] [Google Scholar]
- 7.Murray DW, Ruchton N. Macrophages stimulate bone resorption when they phagocytose particles. J Bone Joint Surg. 1990;72B:988–992. doi: 10.1302/0301-620X.72B6.2246303. [DOI] [PubMed] [Google Scholar]
- 8.Athanasou NA, Quinn J, Bulstrode CJK. Resorption of bone by inflammatory cells derived from the joint capsule of hip arthroplasties. J Bone Joint Surg (Am) 1992;74B:57–62. doi: 10.1302/0301-620X.74B1.1732267. [DOI] [PubMed] [Google Scholar]
- 9.Sutherland K, Mahoney JR, II, Coury A, Eaton JW. Degradation of biomaterials by phagocyte-derived oxidants. J Clin Invest. 1993;92:2360–2367. doi: 10.1172/JCI116841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhao Q, Topham NS, Anderson JM, Hiltner A, Lodoen G, Payet CR. Foreign-body giant Cells and polyurethane biostability: In vivo correlation of cell adhesion and surface cracking. J. Biomed. Mater. Res. 1991;25:177–183. doi: 10.1002/jbm.820250205. [DOI] [PubMed] [Google Scholar]
- 11.Poeppl N, Schreml S, Lichtenegger F, Lenich A, Eisenmann-Klein M, Prantl L. Does the surface structure of implants have an impact on the formation of a capsular contracture? Aesthetic Plastic Surg. 2007;31:133–139. doi: 10.1007/s00266-006-0091-y. [DOI] [PubMed] [Google Scholar]
- 12.Tang L, Hu W. Molecular Determinants of Biocompatibility. Expert Rev Med Devices. 2005;2:493–500. doi: 10.1586/17434440.2.4.493. [DOI] [PubMed] [Google Scholar]
- 13.Tang L, Eaton JW. Molecular determinants of acute inflammatory responses to biomaterials. Chapter 20. In: Zilla PP, Greisler HP, editors. Tissue engineering of prosthetic vascular grafts. RG Landes Company; Austin, Texas: 1999. pp. 207–218. [Google Scholar]
- 14.Schroven IT, Geens S, Becker L, Lagrange W, Fabry G. Experience with the Leeds-Keio ligament for anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 1994;2:214–218. doi: 10.1007/BF01845590. [DOI] [PubMed] [Google Scholar]
- 15.Schiavone PA, Denti M, Franzese S, Monteleone M. The bone-ligament junction: a Comparison between biological and artificial ACL reconstruction. Knee Surg Sports Traumatol Arthrosc. 1993;1:9–12. doi: 10.1007/BF01552151. [DOI] [PubMed] [Google Scholar]
- 16.Prescott RJ, Ryan WG, Bisset DL. Histopathological features of failure prosthetic Leed-Keio anterior cruciate ligaments. J Clin Pathol. 1994;47:375–376. doi: 10.1136/jcp.47.4.375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Turner RF, Harrison DJ, Rajotte RV. Preliminary in vivo biocompatibility studies on prefluorosulphonic acid polymer membranes for biosensor applications. Biomaterials. 1991;12:361–368. doi: 10.1016/0142-9612(91)90003-s. [DOI] [PubMed] [Google Scholar]
- 18.Clark HR, Barbari TA, Rao G. Modeling the response time of an in vivo glucose affinity sensor. Biotechnol, Prog. 1999;15:259–266. doi: 10.1021/bp990003j. [DOI] [PubMed] [Google Scholar]
- 19.Carro LP, Suarez GG. Intracondylar notch fibrous nodule after total knee replacement. Arthroscopy. 1999;15:103–105. doi: 10.1053/ar.1999.v15.015010. [DOI] [PubMed] [Google Scholar]
- 20.Kryshtalsky B, Weinberg S. Surgical arthroscopy of the temporomandibular joint. Ont Dent. 1996;73:40–42. [PubMed] [Google Scholar]
- 21.Domanskis EJ, Owsley JQ. Histological of the etiology of capsule contracture following augmentation mammaplasty. Plastic Reconstr Surg. 1976;58:689–693. doi: 10.1097/00006534-197612000-00006. [DOI] [PubMed] [Google Scholar]
- 22.Van Diest PJ, Beekman WH, Hage JJ. Pathology of silicone leakage from breast implants. J Clin Pathol. 1998;51:493–497. doi: 10.1136/jcp.51.7.493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Altman JJ, Houlbert D, Callard P, McMillan P, Solomon BA, Rosen J, Galletti PM. Long term plasma glucose normalization in experimental diabetic rats with macroencapsulated implants of benign human insulinomas. Diabetes. 1986;35:625–633. doi: 10.2337/diab.35.6.625. [DOI] [PubMed] [Google Scholar]
- 24.Archer J, Kaye R, Mutter G. Control of streptozotocin diabetes in Chinese Hamsters by cultured mouse islet cells without immunosuppression: a preliminary report. J Surg Res. 1980;28:77–85. doi: 10.1016/0022-4804(80)90086-4. [DOI] [PubMed] [Google Scholar]
- 25.Clayton HA, James RF, London NJ. Islet microencapsulation: a review. Acta Diabetol. 1993;30:181–189. doi: 10.1007/BF00569928. [DOI] [PubMed] [Google Scholar]
- 26.Butler K, Benghuzzi H, Tucci M, Cason Z. A comparison of fibrous tissue formation surrounding intra-peritoneal and subcutaneous implantation of ALCAP, HA, and TCP ceramic devices. Biomed Sci Instrum. 1997;34:18–23. [PubMed] [Google Scholar]
- 27.Amon M, Menapace R. Long-term results and biocompatibility of heparin-surface-modified intraocular lenses. J Cataract Refract Surg. 1993;19:258–262. doi: 10.1016/s0886-3350(13)80953-4. [DOI] [PubMed] [Google Scholar]
- 28.Amon M, Menapace R, Radax U, Papapanos P. Clinical results with three different kinds of small optic PMMA-IOLs. Int Ophthalmol. 1994;18:9–13. doi: 10.1007/BF00919407. [DOI] [PubMed] [Google Scholar]
- 29.Nishi O, Nishi K, Sakka Y, Sakuraba T, Maeda S. Intercapsular cataract surgery with lens epithelial cell removal. Part IV: Capsular fibrosis induced by poly(methylmethacrylate) J Cataract Refract Surg. 1991;17:471–477. doi: 10.1016/s0886-3350(13)80854-1. [DOI] [PubMed] [Google Scholar]
- 30.Anderson JM, Bonfield TL, Ziats NP. Protein adsorption and cellular adhesion and activation on biomedical polymers. Int J Artif Org. 1990;13:375–382. [PubMed] [Google Scholar]
- 31.Wilson CJ, Clegg RE, Leavesley DI, Pearcy MJ. Mediation of biomaterial-cell interactions by adsorbed protein: a review. Tissue Engineering. 2005;11:1–18. doi: 10.1089/ten.2005.11.1. [DOI] [PubMed] [Google Scholar]
- 32.Shen M, Garcia I, Maier RV, Horbett TA. Effects of adsorbed protein and surface chemistry on foreign body giant cell formation, tumor necrosis factor alpha release, and pro-coagulant activity of monocytes. J Biomed Mater Res. 2004;70A:533–541. doi: 10.1002/jbm.a.30069. [DOI] [PubMed] [Google Scholar]
- 33.Nath N, Hyun J, Ma H, Chilkoti A. Surface engineering strategies for control of protein and cell interactions. Surf Sci. 2004;570:98–110. [Google Scholar]
- 34.Pankowsky DA, Ziats NP, Topham NS, Ratnoff OD, Anderson JM. Morphologic characteristics of adsorbed human plasma proteins on vascular grafts and biomaterials. J Vasc Surg. 1990;11:599–606. [PubMed] [Google Scholar]
- 35.Andrade JD, Hlady VL. Plasma protein adsorption: The big twelve. Ann NY Acad Sci. 1987;516:158–172. doi: 10.1111/j.1749-6632.1987.tb33038.x. [DOI] [PubMed] [Google Scholar]
- 36.Lenk TJ, Horbett TA, Ratner BD, Chittur KK. Infrared spectroscopic studies of time-dependent changes in fibrinogen adsorbed to polyurethanes. Langmuir. 1991;7:1755–1764. [Google Scholar]
- 37.Lu DR, Park K. Effect of surface hydrophobicity on the conformational changes of adsorbed fibrinogen. J Colloid Interfacial Sci. 1991;144:271–281. [Google Scholar]
- 38.Hu WJ, Eaton JW, Tang L. Molecular basis of biomaterial-mediated foreign body reactions. Blood. 2001;98:1231–1238. doi: 10.1182/blood.v98.4.1231. [DOI] [PubMed] [Google Scholar]
- 39.Collier TO, Anderson JM. Protein and surface effects on monocyte and macrophage adhesion, maturation, and survival. J Biomed Mater Res. 102(60):487–496. doi: 10.1002/jbm.10043. [DOI] [PubMed] [Google Scholar]
- 40.Heuberger M, Drobek T, Spencer ND. Interaction forces and morphology of a protein -resistant poly(ethylene glycol) layer. Biophy J. 2005;88:495–504. doi: 10.1529/biophysj.104.045443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Evans-Nguyen KM, Fuierer RR, Fitchett BD, Tolles LR, Conboy JC, Schoenfisch MH. Changes in adsorbed fibrinogen upon conversion to fibrin. Langmuir. 2006;22:5115–5121. doi: 10.1021/la053070y. [DOI] [PubMed] [Google Scholar]
- 42.Chinn JA, Posso SE, Horbett TA, Ratner BD. Postadsorption transitions in fibrinogen adsorbed to polyurethanes: Changes in antibody binding and sodium dodecyl sulfate elutibility. J Biomed Mater Res. 1992;26:757–778. doi: 10.1002/jbm.820260606. [DOI] [PubMed] [Google Scholar]
- 43.Rapoza RJ, Horbett TA. Changes in the SDS elutability of fibrinogen adsorbed from plasma to polymers. J Biomat Sci Polym Ed. 1989;1:99–110. doi: 10.1163/156856289x00091. [DOI] [PubMed] [Google Scholar]
- 44.Schaaf P, Dejardin P. Structural changes within an adsorbed fibrinogen layer during the adsorption process: A study by scanning angle relectometry. Colloids Surf. 1988;31:89–100. [Google Scholar]
- 45.Ladam G, Schaaf P, Decher G, Voegel JC, Cuisinier JG. Protein Adsorption on auto-assembled polyelectrolyte films. Biomolecular Eng. 2002;19:273–280. doi: 10.1016/s1389-0344(02)00031-x. [DOI] [PubMed] [Google Scholar]
- 46.Wigren R, Elwing H, Erlandsson R, Welin S, Lundstrom I. Structure of adsorbed fibrinogen obtained by scanning force microscopy. FEBS Letts. 1991;280:225–258. doi: 10.1016/0014-5793(91)80298-h. [DOI] [PubMed] [Google Scholar]
- 47.Gettens RTT, Bai Z, Gilbert JL. Quantification of the kinetics and thermodynamics of protein adsorption using atomic force microscopy. J Biomed Mater Res. 2005;72A:246–257. doi: 10.1002/jbm.a.30218. [DOI] [PubMed] [Google Scholar]
- 48.Takahara A, Hara Y, Kojio K, Kajiyama T. Plasma protein adsorption behavior onto the surface of phase-separated organosilane monolayers on the basis of scanning force microscopy. Colloids Surf. 2002;23:141–152. [Google Scholar]
- 49.Tantipolphan R, Rades T, McQuillan AJ, Medlicott NJ. Adsorption of bovine serum albumin (BSA) onto lecithin studied by attenuated total reflectance Fourier transform infrared (ATR-FTIR) spectroscopy. Intern J Pharmaceutics. 2006;337:40–47. doi: 10.1016/j.ijpharm.2006.12.021. [DOI] [PubMed] [Google Scholar]
- 50.Andrade JD, Hlady VL, Van Wagenen RA. Effects of plasma protein adsorption on protein conformation and activity. Pure Appl Chem. 1984;56:1345–1350. [Google Scholar]
- 51.Absolom DR, Zingg W, Policova Z, Neumann AW. Determination of the surface tension of protein coated mate-rials by means of the advancing solidification front technique. Trans Am Soc Intern Organs. 1983;29:146–151. [PubMed] [Google Scholar]
- 52.Dadsetan M, Jones JA, Hiltner A, Anderson JM. Surface chemistry mediates adhesive structure, cytoskeletal organization, and fusion of macrophages. J Biomed Mater Res. 2004;71A:439–448. doi: 10.1002/jbm.a.30165. [DOI] [PubMed] [Google Scholar]
- 53.Horbettl TA, Lew KR. Residence time effects on monoclonal antibody binding to adsorbed fibrinogen. J Biomater Sci Polym Ed. 1994;6:15–33. doi: 10.1163/156856295x00724. [DOI] [PubMed] [Google Scholar]
- 54.Wang YX, Robertson JL, Spillman WB, Claus RO. Effects of the chemical structure and the surface properties of polymeric biomaterials and their biocompatibility. Pharmaceutical Res. 2004;21:1362–1373. doi: 10.1023/b:pham.0000036909.41843.18. [DOI] [PubMed] [Google Scholar]
- 55.Lhoest JB, Wagner MS, Tidwell CD, Castner DG. Characterization of adsorbed protein films by time of flight secondary ion mass spectrometry. J Biomed Mater Res. 2001;57:432–440. doi: 10.1002/1097-4636(20011205)57:3<432::aid-jbm1186>3.0.co;2-g. [DOI] [PubMed] [Google Scholar]
- 56.Tang L, Eaton JW. Fibrin(ogen) mediates acute inflammatory responses to biomaterials. J Exp Med. 1993;178:2147–2156. doi: 10.1084/jem.178.6.2147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Safranj A, Kiaei D, Hoffman AS. Antibody immobilization onto glow discharge treated polymers. Biotech Prog. 1991;7:173–177. doi: 10.1021/bp00008a012. [DOI] [PubMed] [Google Scholar]
- 58.Roach P, Farrar D, Perry CC. Interpretation of protein adsorption: surface-induced conformational changes. J Am Chem Soc. 2005;127:8168–8173. doi: 10.1021/ja042898o. [DOI] [PubMed] [Google Scholar]
- 59.Gallagher WM, Lynch I, Allen LT, Miller I, Penney SC, O’Connor DP, Pennington S, Keenan AK, Dawson KA. Molecular basis of cell-biomaterial interaction: Insights gained from transcriptomic and proteomic studies. Biomaterials. 2006;27:5871–5882. doi: 10.1016/j.biomaterials.2006.07.040. [DOI] [PubMed] [Google Scholar]
- 60.Clarke ML, Wang J, Chen Z. Conformational changes of fibrinogen after adsorption. J Phys Chem B. 2005;109:22027–22035. doi: 10.1021/jp054456k. [DOI] [PubMed] [Google Scholar]
- 61.Shiba E, Lindon JN, Kushner L, Matsueda GR, Hawiger J, Kloczewiak M, Kudryk B, Salzman EW. Antibody-detectable changes in fibrinogen adsorption affecting platelet activation on polymer surfaces. Am J Physiol. 1991;260:C965–C974. doi: 10.1152/ajpcell.1991.260.5.C965. [DOI] [PubMed] [Google Scholar]
- 62.Smolarczyk K, Boncela K, Szymanski J, Gils A, Cierniewski CS. Fibrinogen contains cryptic PAI-1 binding sites that are exposed on binding to solid surfaces or limited proteolysis. Arteriosclerosis Thromb Vasc Biol. 2005;25:2679–2684. doi: 10.1161/01.ATV.0000189305.84297.8b. [DOI] [PubMed] [Google Scholar]
- 63.Ugarova TP, Budzynski AZ, Shattil SJ, Ruggeri ZM, Ginsberg MH, Plow EF. Conformational changes in fibrinogen elicited by its interactions with platelet membrane glycoprotein GPIIb-IIIa. J Biol Chem. 1993;268:21080–21087. [PubMed] [Google Scholar]
- 64.Sheu MS, Hudson DM, Loh IH. Biomaterial surface modification using plasma gas discharge processes. In: Wise DL, Trantolo DJ, Altobelli DE, Yaszemski MJ, Gresser JD, Schwartz ER, editors. Encyclopedic handbook of biomaterials and bioengineering, Part A: Materials. Marcel Dekker, Inc; New York: 1995. pp. 865–894. [Google Scholar]
- 65.Mao C, Qiu Y, Sang H, Mei H, Zhu A, Shen J, Lin S. Various approaches to modify biomaterial surfaces for improving hemocompatibility. Advance Colloid Interface Sci. 2004;110:5–17. doi: 10.1016/j.cis.2004.02.001. [DOI] [PubMed] [Google Scholar]
- 66.Ruckenstein E, Li ZF. Surface modification and functionalization through the self-assembled monolayer and graft polymerization. Advances Colloid Interface Sci. 2005;113:43–63. doi: 10.1016/j.cis.2004.07.009. [DOI] [PubMed] [Google Scholar]
- 67.Arima Y, Iwata H. Effect of wettability and surface functional groups on protein adsorption and cell adhesion using well-defined mixed self-assembled monolayers. Biomaterials. 2007;28:3074–3082. doi: 10.1016/j.biomaterials.2007.03.013. [DOI] [PubMed] [Google Scholar]
- 68.Barbosa JN, Madureira P, Barbosa MA, Aguas AP. The influence of functional groups of self-assembled monolayers on fibrous capsule formation and cell recruitment. J Biomed Mater Res. 2006;76A:737–743. doi: 10.1002/jbm.a.30602. [DOI] [PubMed] [Google Scholar]
- 69.Tang L, Lui L, Elwing H. Complement activation and inflammation triggered by model biomaterial surfaces. J Biomed Mater Res. 1998;41:330–340. doi: 10.1002/(sici)1097-4636(199808)41:2<333::aid-jbm19>3.0.co;2-l. [DOI] [PubMed] [Google Scholar]
- 70.Chu PK, Chen JY, Wang LP, Huang N. Plasma-surface modification of biomaterials. Mater Sci Eng Reports. 2002;36(5):143–206. [Google Scholar]
- 71.Yamamoto H, Shibata Y, Miyazaki T. Anode glow discharge plasma treatment of titanium plates facilitates adsorption of extracellular matrix proteins to the plates. J Dental Res. 2005;84(7):668–671. doi: 10.1177/154405910508400717. [DOI] [PubMed] [Google Scholar]
- 72.Ryu GH, Yang WS, Roh HW, Lee IS, Kim JK, Lee GH, Lee DH, Park BJ, Lee MS, Park JC. Plasma surface modification of poly (D,L-lactic-co-glycolic acid) (65/35) film for tissue engineering. Surf Coatings Techn. 2005;193:60–64. [Google Scholar]
- 73.Dyshlovenko S, Pawlowski L, Smurov I, Veiko V. Pulsed laser modification of plasma-sprayed coatings: Experimental processing of hydroxyapatite and numerical simulation. Surf Coatings Techn. 2006;201:2248–2255. [Google Scholar]
- 74.Kamath S, Bhattacharyya D, Padukudru C, Timmons RB, Tang L. Surface chemistry influence implant mediated host tissue responses. J Biomed Mater Res. 2007 doi: 10.1002/jbm.a.31649. (in press) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Kaar JL, Oh HI, Russell AJ, Federspiel WJ. Towards improved artificial lungs through biocatalysis. Biomaterials. 2007;28(20):3131–3139. doi: 10.1016/j.biomaterials.2007.03.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Savage CR, Lin JW, Timmons RB. Molecular control of surface film compositions via pulsed RF plasma polymerization of perfluoropropylene oxide. Chem Mater. 1991;3:575. [Google Scholar]
- 77.Savage CR, Lin JW, Timmons RB. Spectroscopic characterization of films obtained in pulsed radio-frequency plasma discharges of fluorocarbon monomers. In: Urban ME, Craver CD, editors. Structure-property relations in polymers. Vol. 236. 1993. pp. 745–768. Advances in Chem Series. [Google Scholar]
- 78.Rinsch CL, Eberhart RC, Savage CR, Timmons RB. Film chemistry control during the plasma polymerization of allylamine. Polym Preprints (ACS) 1995;36:95–97. [Google Scholar]
- 79.Rinsch CL, Chen X, Panchalingam V, Eberhart RC, Wang JH, Timmons RB. Pulsed radio frequency plasma polymerization of allyl alcohol: controlled deposition of surface hydroxyl groups. Langmuir. 1996;12:2995–3002. [Google Scholar]
- 80.Timmons RB, Wang JH. Molecular tailoring of surfaces. 5,876,753. US Patent. 1999
- 81.Agashe M, Raut V, Stuart SJ, Latour RA. Molecular simulation to characterize the adsorption behavior of a fibrinogen gamma-chain fragment. Langmuir. 2005;21(3):1103–1117. doi: 10.1021/la0478346. [DOI] [PubMed] [Google Scholar]
- 82.Ostuni E, Chapman RG, Holmlin RE, Takayama S, Whitesides GM. A survey of structure-property relationships of surfaces that resist adsorption of protein. Langmuir. 2001;17(18):5605–5620. doi: 10.1021/la0015258. [DOI] [PubMed] [Google Scholar]
- 83.Evans-Nguyen KM, Tolles LR, Gorkun OV, Lord ST, Schoenfisch MH. Interactions of thrombin with fibrinogen adsorbed on methyl-, hydroxyl-, amine-, and carboxyl-terminated self-assembled monolayers. Biochemistry. 2005;44(47):15561–15568. doi: 10.1021/bi0514358. [DOI] [PubMed] [Google Scholar]
- 84.Benesch J, Svedhem S, Svensson SC, Valiokas R, Liedberg B, Tengvall P. Protein adsorption to oligo(ethylene glycol) self-assembled monolayers: experiments with fibrinogen, heparinized plasma, and serum. J Biomater Sci Polym Ed. 2001;12(6):581–597. doi: 10.1163/156856201316883421. [DOI] [PubMed] [Google Scholar]
- 85.Tegoulia VA, Rao W, Kalambur AT, Rabolt JF, Cooper SL. Surface properties, fibrinogen adsorption, and cellular interactions of a novel phosphorylcholine-containing self-assembled monolayer on gold. Langmuir. 2001;17(14):4396–4404. [Google Scholar]
- 86.Martins MC, Ratner BD, Barbosa MA. Protein adsorption on mixtures of hydroxyl- and methyl-terminated alkanethiols self-assembled monolayers. J Biomed Mater Res. 2003;67A(1):158–171. doi: 10.1002/jbm.a.10096. [DOI] [PubMed] [Google Scholar]
- 87.Craddock PR, Fehr J, Dalmasso AP, Brighan KL, Jacob HS. Hemodialysis leucopenia. Pulmonary vascular leukostasis resulting from complement activation by dialyzer cellophane membranes. J Clin Invest. 1977;59(5):879–888. doi: 10.1172/JCI108710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Lui L, Elwing H. Complement activation on solid surfaces as determined by C3 deposition and hemolytic consumption. J Biomed Mater Res. 1994;28(7):767–773. doi: 10.1002/jbm.820280703. [DOI] [PubMed] [Google Scholar]
- 89.Haeffner-Cavaillon N, Fischer E, Bacle F, Carreno MP, Maillet R, Cavaillon JM, Kazatchkine MD. Complement activation and induction of interleukin-1 production during hemodialysis. Contri Nephrol. 1988;62:86–98. doi: 10.1159/000415479. [DOI] [PubMed] [Google Scholar]
- 90.Lui L, Elwing H. Complement activation on thiol-modified gold surfaces. J Biomed Mater Res. 1996;30(4):535–541. doi: 10.1002/(SICI)1097-4636(199604)30:4<535::AID-JBM12>3.0.CO;2-G. [DOI] [PubMed] [Google Scholar]
- 91.Kalltorp M, Oblogina S, Jacobsson S, Karlsson A, Tengvall P, Thomsen P. In vivo cell recruitment, cytokine release, and chemiluminescence response at gold, and thiol functionalized surfaces. Biomaterials. 1999;20(22):2123–2137. doi: 10.1016/s0142-9612(99)00115-5. [DOI] [PubMed] [Google Scholar]
- 92.Sperling C, Maitz MF, Talkenberger S, Gouzy MF, Groth T, Werner C. In vitro blood reactivity to hydroxylated and non-hydroxylated polymer surfaces. Biomaterials. 2007 doi: 10.1016/j.biomaterials.2007.04.041. (in press) [DOI] [PubMed] [Google Scholar]
- 93.Tidwell CD, Ertel SI, Ratner BD, Tarasevich BJ, Atre S, Allara DL. Endothelial cell growth and protein adsorption on terminally functionalized, self-assembled monolayers of alkanethiolates on gold. Langmuir. 1997;13:3404–3413. [Google Scholar]
- 94.Ohya Y, Matsunami H, Ouchi T. Cell growth on porous sponges prepared from poly(depsipeptide-co-lactide) having various functional groups. J Biomater Sci Polym Ed. 2004;15(1):111–123. doi: 10.1163/156856204322752264. [DOI] [PubMed] [Google Scholar]
- 95.Wang J, Pan CJ, Huang N, Sun H, Yang P, Leng XY, Chen JY, Wan GJ, Chu PK. Surface characterization and blood compatibility of poly(ethylene terephthalate) modified by plasma surface grafting. Surf Coatings Tech. 2005;196(1–3):307–311. [Google Scholar]
- 96.Higuchi A, Shirano K, Harashima M, Yoon BO, Hara M, Hattori M, Imamura K. Chemically modified polysulfone hollow fibers with vinylpyrrolidone having improved blood compatibility. Biomaterials. 2002;23(13):2659–2666. doi: 10.1016/s0142-9612(01)00406-9. [DOI] [PubMed] [Google Scholar]
- 97.MacDonald DE, Deo N, Markovic B, Stranick M, Somasundaran P. Adsorption and dissolution behavior of human plasma fibronectin on thermally and chemically modified titanium dioxide particles. Biomaterials. 2002;23(4):1269–1279. doi: 10.1016/s0142-9612(01)00317-9. [DOI] [PubMed] [Google Scholar]
- 98.Hess H, Vogel V. Molecular shuttles based on motor proteins: active transport in synthetic environments. J Biotech. 2001;82(1):67–85. doi: 10.1016/s1389-0352(01)00029-0. [DOI] [PubMed] [Google Scholar]
- 99.Abraham GA, de Quieroz AA, Roman JS. Hydrophilic hybrid IPNs of segmented polyurethanes and copolymers of vinylpyrrolidone for applications in medicine. Biomaterials. 2001;22(14):1971–1985. doi: 10.1016/s0142-9612(00)00381-1. [DOI] [PubMed] [Google Scholar]
- 100.Lee JH, Khang G, Lee JW, Lee HB. Platelet adhesion onto chargeable functional group gradient surfaces. J Biomed Mater Res. 1998;40:180–186. doi: 10.1002/(sici)1097-4636(199805)40:2<180::aid-jbm2>3.0.co;2-h. [DOI] [PubMed] [Google Scholar]
- 101.Lin JC, Chuang WH. Synthesis, surface characterization, and platelet reactivity evaluation for the self-assembled monolayer of alkanethiol with sulfonic acid functionality. J Biomed Mater Res. 2000;51(3):413–423. doi: 10.1002/1097-4636(20000905)51:3<413::aid-jbm16>3.0.co;2-l. [DOI] [PubMed] [Google Scholar]
- 102.Tsai MY, Lin JC. Surface characterization and platelet adhesion studies of self-assembled monolayer with phosphate ester and phosphonic acid functionalities. J Biomed Mater Res. 2001;55(4):554–565. doi: 10.1002/1097-4636(20010615)55:4<554::aid-jbm1049>3.0.co;2-v. [DOI] [PubMed] [Google Scholar]
- 103.Jenney CR, Anderson JM. Adsorbed serum proteins responsible for surface dependent human macrophage behavior. J Biomed Mater Res. 2000;49(4):435–447. doi: 10.1002/(sici)1097-4636(20000315)49:4<435::aid-jbm2>3.0.co;2-y. [DOI] [PubMed] [Google Scholar]
- 104.Brodbeck WG, Voskerician G, Ziats NP, Nakayama Y, Matsuda T, Anderson JM. In vivo leukocyte cytokine mRNA responses to biomaterials are dependent on surface chemistry. J Biomed Mater Res. 2003;64A:320–329. doi: 10.1002/jbm.a.10425. [DOI] [PubMed] [Google Scholar]
- 105.Barbosa JN, Barbosa MA, Aguas AP. Adhesion of human leukocytes to biomaterials: An in vitro study using alkanethiolate monolayers with different chemically functionalized surfaces. J Biomed Mater Res. 2003;65A:429–434. doi: 10.1002/jbm.a.10488. [DOI] [PubMed] [Google Scholar]
- 106.Beyer D, Knoll W, Ringsdorf H, Wang JH, Timmons RB, Sluka P. Reduced protein adsorption on plastics via direct plasma deposition of triethylene glycol monoallyl ether. J Biomed Mater Res. 1997;36:181–189. doi: 10.1002/(sici)1097-4636(199708)36:2<181::aid-jbm6>3.0.co;2-g. [DOI] [PubMed] [Google Scholar]
- 107.Scotchford CA, Gilmore CP, Cooper E, Leggett GJ, Downes S. Protein adsorption and human osteoblast-like cell attachment and growth on alkylthiol on gold self-assembled monolayers. J Biomed Mater Res. 2002;59:84–99. doi: 10.1002/jbm.1220. [DOI] [PubMed] [Google Scholar]
- 108.Tang L, Wu Y, Timmons RB. Fibrinogen adsorption and host tissue responses to plasma functionalized surfaces. J Biomed Mater Res. 1998;42(1):156–163. doi: 10.1002/(sici)1097-4636(199810)42:1<156::aid-jbm19>3.0.co;2-j. [DOI] [PubMed] [Google Scholar]
- 109.Keselowsky BG, Collard DM, Garcia AJ. Surface chemistry modulates focal adhesion composition and signals through changes in integrin binding. Biomaterials. 2004;25:5947–5954. doi: 10.1016/j.biomaterials.2004.01.062. [DOI] [PubMed] [Google Scholar]
- 110.Lan MA, Gersbach CA, Michael KE, Keselowsky BG, Garcia AJ. Myoblast proliferation and differentiation on fibronectin-coated self assembled monolayers presenting different surface chemistries. Biomaterials. 2004;26:4523–4531. doi: 10.1016/j.biomaterials.2004.11.028. [DOI] [PubMed] [Google Scholar]
- 111.Ertel SI, Ratner BD, Horbett TA. Radiofrequency plasma deposition of oxygen-containing films on polystyrene and poly(ethylene terephthalate) substrates improves endothelial cell growth. J Biomed Mater Res. 1990;24(12):1637–1659. doi: 10.1002/jbm.820241207. [DOI] [PubMed] [Google Scholar]
- 112.Lestelius M, Liedberg B, Tengvall P. In vitro plasma protein adsorption on ω-functionalized alkanethiolate self-assembled monolayers. Langmuir. 1997;13(22):5900–5908. [Google Scholar]
- 113.Lindblad M, Lestelius M, Johansson A, Tengvall P, Tomsen P. Cell and soft tissue interactions with methyl- and hysroxyl-terminated alkane thiols on gold surfaces. Biomaterials. 1997;18(15):1059–1068. doi: 10.1016/s0142-9612(97)00029-x. [DOI] [PubMed] [Google Scholar]
- 114.Liu L, Chen S, Giachelli CM, Ratner BD, Jiang S. Controlling osteopontin orientation on the surfaces to modulate endothelial cell adhesion. J Biomed Mater Res. 2005;74A:23–31. doi: 10.1002/jbm.a.30221. [DOI] [PubMed] [Google Scholar]
- 115.Keselowsky BG, Collard DM, Garcia AJ. Integrin binding specificity regulates biomaterial surface chemistry effects on cell differentiation. PNAS. 2005;102(17):5953–5957. doi: 10.1073/pnas.0407356102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Faucheux N, Schweiss R, Lutxzow K, Werner C, Groth T. Self-assembled monolayers with different terminating groups as model substrates for cell adhesion studies. Biomaterials. 2004;25:2721–2730. doi: 10.1016/j.biomaterials.2003.09.069. [DOI] [PubMed] [Google Scholar]
- 117.Altankov G, Richau K, Groth TH. The role of surface zeta potential and substratum chemistry for regulation of dermal fibroblasts interaction. Materialwissenschaft und Werkstofftechnik. 2003;34(12):1120–1128. [Google Scholar]
- 118.Kidoaki S, Matsuda T. Adhesion forces of the blood plasma proteins on self-assembled monolayer surfaces of alkanethiolates with different functional groups measured by an atomic force microscope. Langmuir. 1999;15:7639–7646. [Google Scholar]
- 119.Chuang WH, Lin JC. Surface characterization and platelet adhesion studies for the mixed self-assembled monolayers with amine and carboxylic acid terminated functionalities. J Biomed Mater Res. 2007 doi: 10.1002/jbm.a.31193. (in press) [DOI] [PubMed] [Google Scholar]
- 120.Rodrigues SN, Goncalves IC, Martins MCL, Barbosa MA, Ratner BD. Fibrinogen adsorption, platelet adhesion, and activation on mixed hydroxyl-/methyl-terminated self-assembled monolayers. Biomaterials. 2006;27:5357–5367. doi: 10.1016/j.biomaterials.2006.06.010. [DOI] [PubMed] [Google Scholar]
- 121.Sperling C, Schweiss RB, Streller U, Werner C. In vitro hemocombatibility of self-assembled monolayers displaying various functional groups. Biomaterials. 2005;26:6547–6557. doi: 10.1016/j.biomaterials.2005.04.042. [DOI] [PubMed] [Google Scholar]
- 122.Tsai MY, Sun YT, Lin JC. Surface characterization and platelet compatibility evaluation of the binary mixed self-assembled monolayers. J Colloid Interface Sci. 2007;308:474–484. doi: 10.1016/j.jcis.2007.01.015. [DOI] [PubMed] [Google Scholar]
- 123.Barbosa JN, Barbosa MA, Aguas AP. Inflammatory responses and cell adhesion to self-assembled monolayers of alkanethiolates on gold. Biomaterials. 2004;25:2557–2563. doi: 10.1016/j.biomaterials.2003.09.047. [DOI] [PubMed] [Google Scholar]
- 124.Barbosa JN, Madureira P, Barbosa MA, Aguas AP. The attraction of Mac-1+ phagocytes during acute inflammation by methyl-coated self-assembled monolayers. Biomaterials. 2005;26:3021–3027. doi: 10.1016/j.biomaterials.2004.09.002. [DOI] [PubMed] [Google Scholar]
- 125.Ratner BD, Bryant SJ. Biomaterials: where we have been and where we are going. Ann Rev Biomed Eng. 2004;6:41–75. doi: 10.1146/annurev.bioeng.6.040803.140027. [DOI] [PubMed] [Google Scholar]
- 126.Duan X, McLaughlin C, Griffith M, Sheardown H. Biofunctionalization of collagen for improved biological response: scaffolds for corneal tissue engineering. Biomaterials. 2007;28(1):78–88. doi: 10.1016/j.biomaterials.2006.08.034. [DOI] [PubMed] [Google Scholar]
- 127.Piana C, Gull I, Gerbes S, Gerdes R, Mills C, Samitier J, Wirth M, Gabor F. Influence of surface modification on vitality and differentiation of Caco-2 cells. Differentiation. 2007;75(4):308–317. doi: 10.1111/j.1432-0436.2006.00141.x. [DOI] [PubMed] [Google Scholar]
- 128.Ratner BD. Reducing capsular thickness and enhancing angiogenesis around implant drug release systems. J Controlled Release. 2002;78(1–3):211–218. doi: 10.1016/s0168-3659(01)00502-8. [DOI] [PubMed] [Google Scholar]
- 129.Fournier E, Passirani C, Montero-Menei CN, Benoit JP. Biocompatibility of implantable synthetic polymeric drug carriers: Focus on brain biocompatibility. Biomaterials. 2003;24(19):3311–3331. doi: 10.1016/s0142-9612(03)00161-3. [DOI] [PubMed] [Google Scholar]
- 130.Rihova B. Immunocompatibility and biocompatibility of cell delivery systems. Advan Drug Delivery Rev. 2000;42(1–2):65–80. doi: 10.1016/s0169-409x(00)00054-5. [DOI] [PubMed]
- 131.Kannan S, Kohle P, Raykova V, Glibatec M, Kannan RM, Lieh-Lai M, Bassett D. Dynamics of cell entry and drug delivery by dendritic polymers into human lung epithelial carcinoma cells. J Biomater Sci Polym Ed. 2004;15(3):311–330. doi: 10.1163/156856204322977201. [DOI] [PubMed] [Google Scholar]
- 132.Vandamme TF, Brodbeck L. Poly(amidoamine) dendrimers as ophthalmic vehicles for ocular delivery of pilocarpine nitrate and tropicamide. J Controlled Release. 2005;102(1):23–38. doi: 10.1016/j.jconrel.2004.09.015. [DOI] [PubMed] [Google Scholar]
- 133.Wilhelm C, Billotey C, Roger J, Pons JN, Bacri JC, Gazeau F. Intracellular uptake of anionic superparamagnetic nanoparticles as a function of their surface coating. Biomaterials. 2003;24(6):1001–1011. doi: 10.1016/s0142-9612(02)00440-4. [DOI] [PubMed] [Google Scholar]
- 134.Shikata F, Tokumitsu H, Ichikawa H, Fukumori Y. In vitro cellular accumulation of gadolinium incorporated in chitosan nanoparticles designed for neutron-capture therapy of cancer. Eur J Pharmac Biopharmaceutics. 2002;53(1):57–63. doi: 10.1016/s0939-6411(01)00198-9. [DOI] [PubMed] [Google Scholar]
- 135.Redhead HM, Davis SS, Illum L. Drug delivery in poly(lactide-co-glycolide) nanoparticles surface modified with poloxamer 407 and poloxamer 908: In vitro characterization and in vivo evaluation. J Controlled Release. 2001;70(3):353–363. doi: 10.1016/s0168-3659(00)00367-9. [DOI] [PubMed] [Google Scholar]
- 136.Choi SW, Kim WS, Kim JH. Surface modification of functional nanoparticles for controlled drug delivery. J Dispersion Sci Techn. 2003;24(3–4):475–487. [Google Scholar]
- 137.Lorenz MR, Holzapfel V, Musyanovych A, Nothelfer K, Walther P, Frank H, Landfester K, Schrezenmeier H, Mailander V. Uptake of functionalized, fluorescently-labeled polymeric particles in different cell lines and stem cells. Biomaterials. 2006;27(14):2820–2828. doi: 10.1016/j.biomaterials.2005.12.022. [DOI] [PubMed] [Google Scholar]
- 138.Win KY, Feng SS. Effects of particle size and surface coating on cellular uptake of polymeric nanoparticles for oral delivery of anticancer drugs. Biomaterials. 2005;26(15):2713–2722. doi: 10.1016/j.biomaterials.2004.07.050. [DOI] [PubMed] [Google Scholar]
- 139.Holzapfel V, Lorenz M, Weiss CK, Schrezenmeier H, Landfester K, Mailander V. Synthesis and biomedical application of functionalized fluorescent and magnetic dual reporter nanoparticles as obtained in a miniemulsion process. J Physics Condensed Matter. 2006;18:S2581–S2594. [Google Scholar]