Abstract
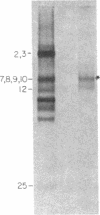
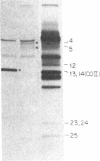
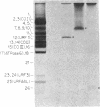
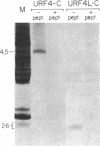
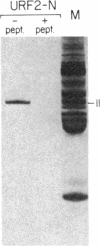
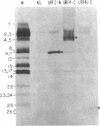
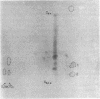
In previous work, antibodies prepared against chemically synthesized peptides predicted from the DNA sequence were used to identify the polypeptides encoded in three of the eight unassigned reading frames (URFs) of human mitochondrial DNA (mtDNA). In the present study, this approach has been extended to other human mtDNA URFs. In particular, antibodies directed against the NH2-terminal octapeptide of the putative URF2 product specifically precipitated component 11 of the HeLa cell mitochondrial translation products, the reaction being inhibited by the specific peptide. Similarly, antibodies directed against the COOH-terminal nonapeptide of the putative URF4 product reacted specifically with components 4 and 5, and antibodies against a COOH-terminal heptapeptide of the presumptive URF4L product reacted specifically with component 26. Antibodies against the NH2-terminal heptapeptide of the putative product of URF5 reacted with component 1, but only to a marginal extent; however, the results of a trypsin fingerprinting analysis of component 1 point strongly to this component as being the authentic product of URF5. The polypeptide assignments to the mtDNA URFs analyzed here are supported by the relative electrophoretic mobilities of proteins 11, 4-5, 26, and 1, which are those expected for the molecular weights predicted from the DNA sequence for the products of URF2, URF4, URF4L, and URF5, respectively. With the present assignment, seven of the eight human mtDNA URFs have been shown to be expressed in HeLa cells.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anderson S., Bankier A. T., Barrell B. G., de Bruijn M. H., Coulson A. R., Drouin J., Eperon I. C., Nierlich D. P., Roe B. A., Sanger F. Sequence and organization of the human mitochondrial genome. Nature. 1981 Apr 9;290(5806):457–465. doi: 10.1038/290457a0. [DOI] [PubMed] [Google Scholar]
- Anderson S., de Bruijn M. H., Coulson A. R., Eperon I. C., Sanger F., Young I. G. Complete sequence of bovine mitochondrial DNA. Conserved features of the mammalian mitochondrial genome. J Mol Biol. 1982 Apr 25;156(4):683–717. doi: 10.1016/0022-2836(82)90137-1. [DOI] [PubMed] [Google Scholar]
- Bibb M. J., Van Etten R. A., Wright C. T., Walberg M. W., Clayton D. A. Sequence and gene organization of mouse mitochondrial DNA. Cell. 1981 Oct;26(2 Pt 2):167–180. doi: 10.1016/0092-8674(81)90300-7. [DOI] [PubMed] [Google Scholar]
- Bonitz S. G., Coruzzi G., Thalenfeld B. E., Tzagoloff A., Macino G. Assembly of the mitochondrial membrane system. Structure and nucleotide sequence of the gene coding for subunit 1 of yeast cytochrme oxidase. J Biol Chem. 1980 Dec 25;255(24):11927–11941. [PubMed] [Google Scholar]
- Burger G., Werner S. The mitochondrial URF1 gene in Neurospora crassa has an intron that contains a novel type of URF. J Mol Biol. 1985 Nov 20;186(2):231–242. doi: 10.1016/0022-2836(85)90100-7. [DOI] [PubMed] [Google Scholar]
- Ching E., Attardi G. High-resolution electrophoretic fractionation and partial characterization of the mitochondrial translation products from HeLa cells. Biochemistry. 1982 Jun 22;21(13):3188–3195. doi: 10.1021/bi00256a024. [DOI] [PubMed] [Google Scholar]
- Chomyn A., Mariottini P., Cleeter M. W., Ragan C. I., Matsuno-Yagi A., Hatefi Y., Doolittle R. F., Attardi G. Six unidentified reading frames of human mitochondrial DNA encode components of the respiratory-chain NADH dehydrogenase. Nature. 1985 Apr 18;314(6012):592–597. doi: 10.1038/314592a0. [DOI] [PubMed] [Google Scholar]
- Chomyn A., Mariottini P., Gonzalez-Cadavid N., Attardi G., Strong D. D., Trovato D., Riley M., Doolittle R. F. Identification of the polypeptides encoded in the ATPase 6 gene and in the unassigned reading frames 1 and 3 of human mtDNA. Proc Natl Acad Sci U S A. 1983 Sep;80(18):5535–5539. doi: 10.1073/pnas.80.18.5535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clary D. O., Wahleithner J. A., Wolstenholme D. R. Sequence and arrangement of the genes for cytochrome b, URF1, URF4L, URF4, URF5, URF6 and five tRNAs in Drosophila mitochondrial DNA. Nucleic Acids Res. 1984 May 11;12(9):3747–3762. doi: 10.1093/nar/12.9.3747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clary D. O., Wolstenholme D. R. Genes for cytochrome c oxidase subunit I, URF2, and three tRNAs in Drosophila mitochondrial DNA. Nucleic Acids Res. 1983 Oct 11;11(19):6859–6872. doi: 10.1093/nar/11.19.6859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hensgens L. A., Brakenhoff J., De Vries B. F., Sloof P., Tromp M. C., Van Boom J. H., Benne R. The sequence of the gene for cytochrome c oxidase subunit I, a frameshift containing gene for cytochrome c oxidase subunit II and seven unassigned reading frames in Trypanosoma brucei mitochondrial maxi-circle DNA. Nucleic Acids Res. 1984 Oct 11;12(19):7327–7344. doi: 10.1093/nar/12.19.7327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kyte J., Doolittle R. F. A simple method for displaying the hydropathic character of a protein. J Mol Biol. 1982 May 5;157(1):105–132. doi: 10.1016/0022-2836(82)90515-0. [DOI] [PubMed] [Google Scholar]
- Laudano A. P., Doolittle R. F. Studies on synthetic peptides that bind to fibrinogen and prevent fibrin polymerization. Structural requirements, number of binding sites, and species differences. Biochemistry. 1980 Mar 4;19(5):1013–1019. doi: 10.1021/bi00546a028. [DOI] [PubMed] [Google Scholar]
- Macreadie I. G., Novitski C. E., Maxwell R. J., John U., Ooi B. G., McMullen G. L., Lukins H. B., Linnane A. W., Nagley P. Biogenesis of mitochondria: the mitochondrial gene (aap1) coding for mitochondrial ATPase subunit 8 in Saccharomyces cerevisiae. Nucleic Acids Res. 1983 Jul 11;11(13):4435–4451. doi: 10.1093/nar/11.13.4435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mariottini P., Chomyn A., Attardi G., Trovato D., Strong D. D., Doolittle R. F. Antibodies against synthetic peptides reveal that the unidentified reading frame A6L, overlapping the ATPase 6 gene, is expressed in human mitochondria. Cell. 1983 Apr;32(4):1269–1277. doi: 10.1016/0092-8674(83)90308-2. [DOI] [PubMed] [Google Scholar]
- Matheka H. D., Enzmann P. J., Bachrach H. L., Migl B. The influence of sodium dodecyl sulfate from different sources on the separation of virus proteins in polyacrylamide gel electrophoresis. Anal Biochem. 1977 Jul;81(1):9–17. doi: 10.1016/0003-2697(77)90592-9. [DOI] [PubMed] [Google Scholar]
- Nobrega F. G., Tzagoloff A. Assembly of the mitochondrial membrane system. DNA sequence and organization of the cytochrome b gene in Saccharomyces cerevisiae D273-10B. J Biol Chem. 1980 Oct 25;255(20):9828–9837. [PubMed] [Google Scholar]
- Ojala D., Merkel C., Gelfand R., Attardi G. The tRNA genes punctuate the reading of genetic information in human mitochondrial DNA. Cell. 1980 Nov;22(2 Pt 2):393–403. doi: 10.1016/0092-8674(80)90350-5. [DOI] [PubMed] [Google Scholar]
- Oliver N. A., Greenberg B. D., Wallace D. C. Assignment of a polymorphic polypeptide to the human mitochondrial DNA unidentified reading frame 3 gene by a new peptide mapping strategy. J Biol Chem. 1983 May 10;258(9):5834–5839. [PubMed] [Google Scholar]
- Pepe G., Holtrop M., Gadaleta G., Kroon A. M., Cantatore P., Gallerani R., De Benedetto C., Quagliariello C., Sbisà E., Saccone C. Non-random patterns of nucleotide substitutions and codon strategy in the mammalian mitochondrial genes coding for identified and unidentified reading frames. Biochem Int. 1983 Apr;6(4):553–563. [PubMed] [Google Scholar]
- Roe B. A., Ma D. P., Wilson R. K., Wong J. F. The complete nucleotide sequence of the Xenopus laevis mitochondrial genome. J Biol Chem. 1985 Aug 15;260(17):9759–9774. [PubMed] [Google Scholar]
- Sebald W., Machleidt W., Otto J. Products of mitochondrial protein synthesis in Neurospora crassa. Determination of equimolar amounts of three products in cytochrome oxidase on the basis of amino-acid analysis. Eur J Biochem. 1973 Oct 5;38(2):311–324. doi: 10.1111/j.1432-1033.1973.tb03064.x. [DOI] [PubMed] [Google Scholar]
- Storrie B., Attardi G. Expression of the mitochondrial genome in HeLa cells. 13. Effect of selective inhibition of cytoplasmic or mitochondrial protein synthesis on mitochondrial nucleic acid synthesis. J Mol Biol. 1972 Nov 14;71(2):177–199. doi: 10.1016/0022-2836(72)90345-2. [DOI] [PubMed] [Google Scholar]
- Swaney J. B., Vande Woude G. F., Bachrach H. L. Sodium dodecylsulfate-dependent anomalies in gel electrophoresis: alterations in the banding patterns of foot-and-mouth disease virus polypeptides. Anal Biochem. 1974 Apr;58(2):337–346. doi: 10.1016/0003-2697(74)90201-2. [DOI] [PubMed] [Google Scholar]
- Tzagoloff A. Assembly of the mitochondrial membrane system. IV. Role of mitochondrial and cytoplasmic protein synthesis in the biosynthesis of the rutamycin-sensitive adenosine triphosphatase. J Biol Chem. 1971 May 10;246(9):3050–3056. [PubMed] [Google Scholar]
- de Bruijn M. H. Drosophila melanogaster mitochondrial DNA, a novel organization and genetic code. Nature. 1983 Jul 21;304(5923):234–241. doi: 10.1038/304234a0. [DOI] [PubMed] [Google Scholar]
- de la Cruz V. F., Neckelmann N., Simpson L. Sequences of six genes and several open reading frames in the kinetoplast maxicircle DNA of Leishmania tarentolae. J Biol Chem. 1984 Dec 25;259(24):15136–15147. [PubMed] [Google Scholar]