Abstract
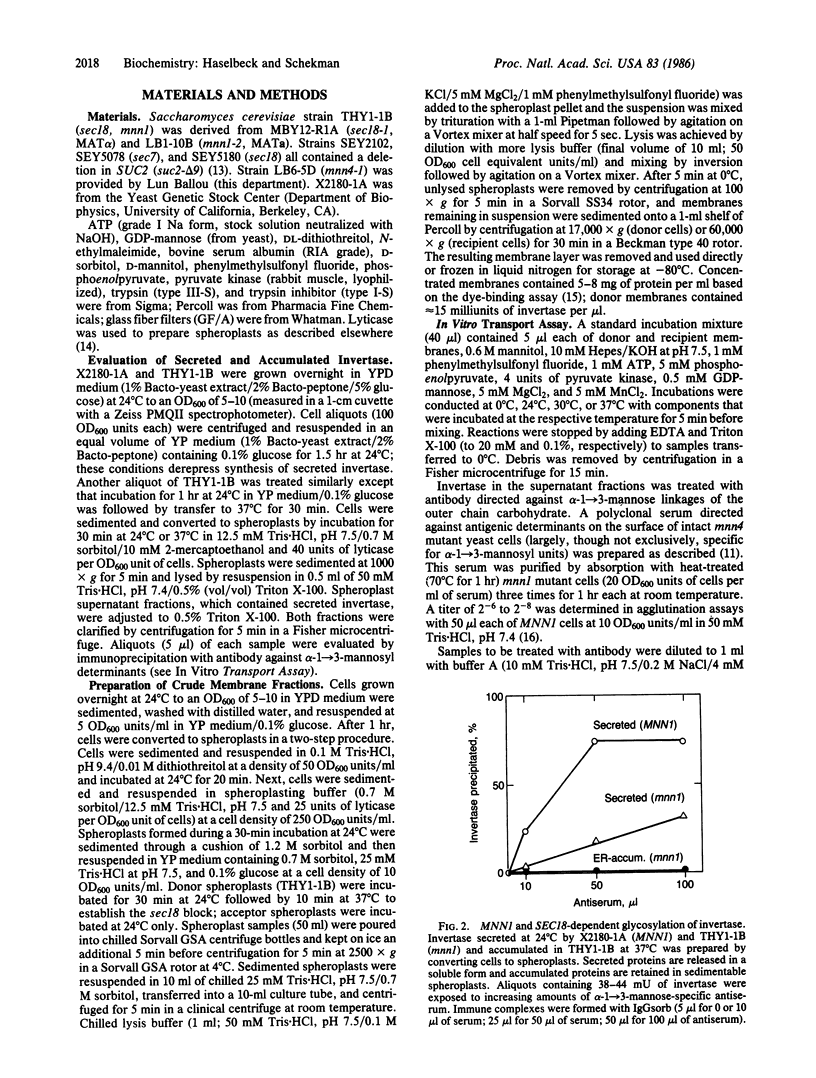
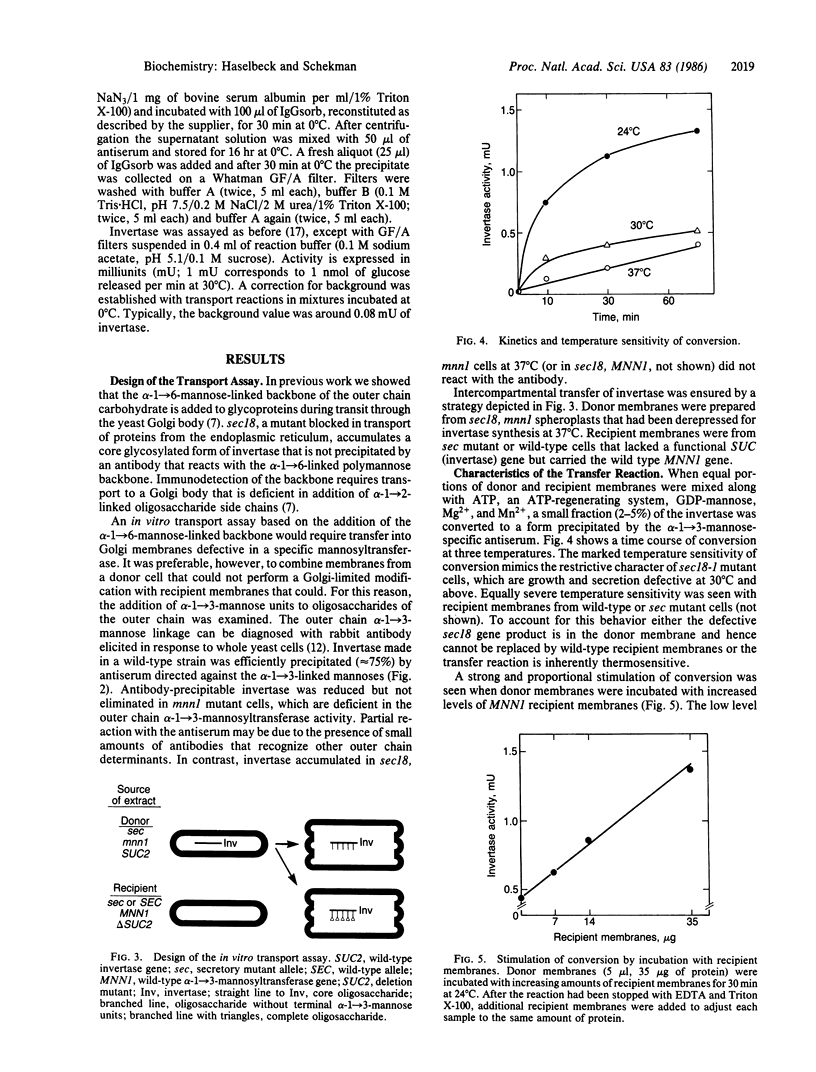
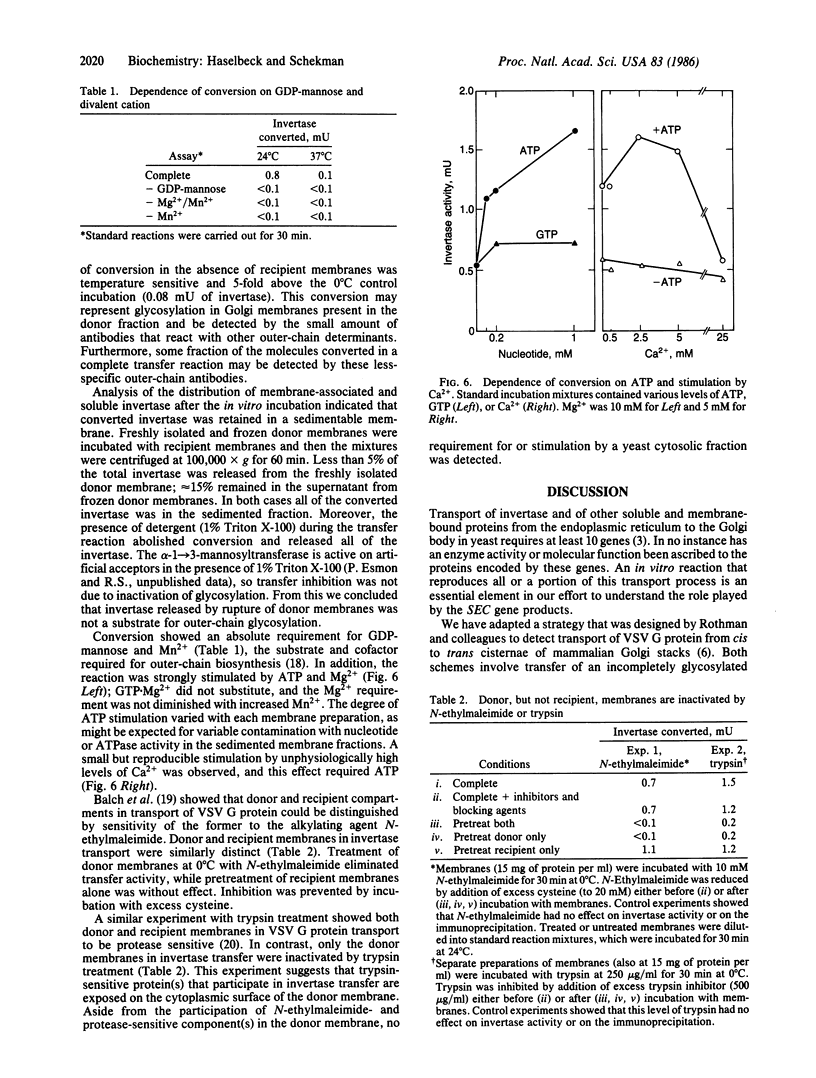
Core glycosylated proteins formed in the yeast endoplasmic reticulum (ER) are transported to the Golgi body, where oligosaccharides are elongated by addition of outer-chain carbohydrate. The transport process is blocked in a temperature-sensitive secretion mutant (sec18) of Saccharomyces cerevisiae, which accumulates core glycosylated invertase (product of SUC2; EC 3.2.1.26) in the ER. To approach the molecular mechanism of this transport process, we have devised a reaction in which core glycosylated invertase, accumulated in sec18 cells, is transferred to the Golgi body in vitro. For this purpose, membranes from sec18, SUC2 cells that are also defective in an outer chain alpha-1----3-mannosyltransferase (mnnl) are mixed with membranes from a strain that contains the transferase but is deficient in invertase (MNNl, delta SUC2). Transfer is detected by the acquisition of outer-chain alpha-1----3-linked mannose residues dependent on both donor and recipient membranes. The reaction is temperature and detergent sensitive and requires ATP, GDP-mannose, Mg2+, and Mn2+, and the product invertase remains associated with sedimentable membranes. Treatment of donor, but not acceptor, membranes with N-ethylmaleimide or trypsin inactivates transfer competence. These characteristics suggest that the ER, or a vesicle derived from the ER, contributes invertase to a chemically distinct compartment where mannosyl modification is executed.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Antalis C., Fogel S., Ballou C. E. Genetic control of yeast mannan structure. Mapping the first gene concerned with mannan biosynthesis. J Biol Chem. 1973 Jul 10;248(13):4655–4659. [PubMed] [Google Scholar]
- Balch W. E., Dunphy W. G., Braell W. A., Rothman J. E. Reconstitution of the transport of protein between successive compartments of the Golgi measured by the coupled incorporation of N-acetylglucosamine. Cell. 1984 Dec;39(2 Pt 1):405–416. doi: 10.1016/0092-8674(84)90019-9. [DOI] [PubMed] [Google Scholar]
- Balch W. E., Glick B. S., Rothman J. E. Sequential intermediates in the pathway of intercompartmental transport in a cell-free system. Cell. 1984 Dec;39(3 Pt 2):525–536. doi: 10.1016/0092-8674(84)90459-8. [DOI] [PubMed] [Google Scholar]
- Balch W. E., Rothman J. E. Characterization of protein transport between successive compartments of the Golgi apparatus: asymmetric properties of donor and acceptor activities in a cell-free system. Arch Biochem Biophys. 1985 Jul;240(1):413–425. doi: 10.1016/0003-9861(85)90046-3. [DOI] [PubMed] [Google Scholar]
- Byrd J. C., Tarentino A. L., Maley F., Atkinson P. H., Trimble R. B. Glycoprotein synthesis in yeast. Identification of Man8GlcNAc2 as an essential intermediate in oligosaccharide processing. J Biol Chem. 1982 Dec 25;257(24):14657–14666. [PubMed] [Google Scholar]
- Emr S. D., Schekman R., Flessel M. C., Thorner J. An MF alpha 1-SUC2 (alpha-factor-invertase) gene fusion for study of protein localization and gene expression in yeast. Proc Natl Acad Sci U S A. 1983 Dec;80(23):7080–7084. doi: 10.1073/pnas.80.23.7080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Esmon B., Esmon P. C., Schekman R. Early steps in processing of yeast glycoproteins. J Biol Chem. 1984 Aug 25;259(16):10322–10327. [PubMed] [Google Scholar]
- Esmon B., Novick P., Schekman R. Compartmentalized assembly of oligosaccharides on exported glycoproteins in yeast. Cell. 1981 Aug;25(2):451–460. doi: 10.1016/0092-8674(81)90063-5. [DOI] [PubMed] [Google Scholar]
- Fries E., Rothman J. E. Transport of vesicular stomatitis virus glycoprotein in a cell-free extract. Proc Natl Acad Sci U S A. 1980 Jul;77(7):3870–3874. doi: 10.1073/pnas.77.7.3870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldstein A., Lampen J. O. Beta-D-fructofuranoside fructohydrolase from yeast. Methods Enzymol. 1975;42:504–511. doi: 10.1016/0076-6879(75)42159-0. [DOI] [PubMed] [Google Scholar]
- Nakajima T., Ballou C. E. Yeast manno-protein biosynthesis: solubilization and selective assay of four mannosyltransferases. Proc Natl Acad Sci U S A. 1975 Oct;72(10):3912–3916. doi: 10.1073/pnas.72.10.3912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Novick P., Ferro S., Schekman R. Order of events in the yeast secretory pathway. Cell. 1981 Aug;25(2):461–469. doi: 10.1016/0092-8674(81)90064-7. [DOI] [PubMed] [Google Scholar]
- Novick P., Field C., Schekman R. Identification of 23 complementation groups required for post-translational events in the yeast secretory pathway. Cell. 1980 Aug;21(1):205–215. doi: 10.1016/0092-8674(80)90128-2. [DOI] [PubMed] [Google Scholar]
- Palade G. Intracellular aspects of the process of protein synthesis. Science. 1975 Aug 1;189(4200):347–358. doi: 10.1126/science.1096303. [DOI] [PubMed] [Google Scholar]
- Parodi A. J. Biosynthesis of yeast mannoproteins. Synthesis of mannan outer chain and of dolichol derivatives. J Biol Chem. 1979 Sep 10;254(17):8343–8352. [PubMed] [Google Scholar]
- Schauer I., Emr S., Gross C., Schekman R. Invertase signal and mature sequence substitutions that delay intercompartmental transport of active enzyme. J Cell Biol. 1985 May;100(5):1664–1675. doi: 10.1083/jcb.100.5.1664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scott J. H., Schekman R. Lyticase: endoglucanase and protease activities that act together in yeast cell lysis. J Bacteriol. 1980 May;142(2):414–423. doi: 10.1128/jb.142.2.414-423.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vincent R., Nadeau D. A micromethod for the quantitation of cellular proteins in Percoll with the Coomassie brilliant blue dye-binding assay. Anal Biochem. 1983 Dec;135(2):355–362. doi: 10.1016/0003-2697(83)90696-6. [DOI] [PubMed] [Google Scholar]



