Abstract
Neisseria gonorrhoeae is an obligate human pathogen that causes the common sexually transmitted infection gonorrhea. Gonococcal infections cause significant morbidity, particularly among women, as the organism ascends to the upper reproductive tract, resulting in pelvic inflammatory disease, ectopic pregnancy, and infertility. In the last few years, antibiotic resistance rates have risen dramatically, leading to severe restriction of treatment options for gonococcal disease. Gonococcal infections do not elicit protective immunity, nor is there an effective vaccine to prevent the disease. Thus, further understanding of the expression, function, and regulation of surface antigens could lead to better treatment and prevention modalities in the future. In the current study, we determined that an iron-repressed regulator, MpeR, interacted specifically with the DNA sequence upstream of fetA and activated FetA expression. Interestingly, MpeR was previously shown to regulate the expression of gonococcal antimicrobial efflux systems. We confirmed that the outer membrane transporter FetA allows gonococcal strain FA1090 to utilize the xenosiderophore ferric enterobactin as an iron source. However, we further demonstrated that FetA has an extended range of substrates that encompasses other catecholate xenosiderophores, including ferric salmochelin and the dimers and trimers of dihydroxybenzoylserine. We demonstrated that fetA is part of an iron-repressed, MpeR-activated operon which putatively encodes other iron transport proteins. This is the first study to describe a regulatory linkage between antimicrobial efflux and iron transport in N. gonorrhoeae. The regulatory nidus that links these systems, MpeR, is expressed exclusively by pathogenic neisseriae and is therefore expected to be an important virulence factor.
INTRODUCTION
Neisseria gonorrhoeae is an obligate human pathogen that infects primarily the urogenital or anorectal mucosa following intimate contact. N. gonorrhoeae is the etiological agent of gonorrhea, which is the second most commonly reported notifiable infectious disease in the United States. In 2009, the Centers for Disease Control and Prevention reported a total of 301,174 cases of gonorrhea in the United States (14); however, this is thought to be a conservative estimate due to underreporting. In men, a gonococcal infection is characterized by acute urethritis with symptoms that include purulent discharge and dysuria. It is estimated that up to 80% of women infected with N. gonorrhoeae are asymptomatic or present with very minor symptoms (45). Women with symptomatic disease experience cervicitis and vaginal discharge. When left untreated, due to the asymptomatic nature of the infection in women, the bacteria can ascend to the upper female genital tract. This ascending infection can result in pelvic inflammatory disease, which may lead to ectopic pregnancy or infertility (47, 61). The Centers for Disease Control currently recommends only extended-spectrum cephalosporins for treatment due to increased antimicrobial resistance to all previously recommended therapies (15, 33, 39). Unfortunately, resistance to this class of antimicrobial agent has already emerged (14). N. gonorrhoeae infections do not elicit protective immunity, and there is evidence that gonococcal infections increase the spread of HIV (18, 44). Since gonococcal disease poses a significant public health challenge, it is important to understand the pathogenesis of N. gonorrhoeae in order to identify new therapies.
Iron is an essential nutrient for most microorganisms, including the neisseriae (10). Many microorganisms acquire iron from the human host by synthesizing and secreting siderophores. Siderophores are low-molecular-weight iron-chelating molecules that scavenge iron from the environment or host iron binding proteins. N. gonorrhoeae does not synthesize siderophores but instead obtains iron directly from human iron binding proteins, including transferrin, lactoferrin, and hemoglobin, in a receptor-mediated mechanism (7, 17, 19, 37). Expression of either the transferrin or lactoferrin receptor by N. gonorrhoeae is necessary to establish infection in human male volunteers (2, 20). Gonococci can also hijack siderophores produced by other bacteria, which is known as xenosiderophore utilization. It has been previously demonstrated that strains of gonococci can obtain iron from the xenosiderophores enterobactin, aerobactin, and salmochelin, which are all synthesized by enteric bacteria (12, 62, 65).
Iron acquisition is tightly regulated, since excess iron can promote Haber-Weiss-Fenton chemistry, creating highly reactive, toxic hydroxyl radicals within the cell (30). In many bacteria, including N. gonorrhoeae, the ferric uptake regulator (Fur) acts as a transcriptional regulator for iron acquisition genes. Under iron-replete conditions, a dimer of Fur binds to its corepressor, ferrous iron, and assumes a DNA binding conformation. The Fur-Fe2+ complex binds to a specific DNA sequence, called the Fur box, found in the promoter regions of iron-regulated genes (4). Once Fur is bound to the Fur box, it blocks gene transcription. As intracellular iron stores become depleted, apo-Fur dissociates from the Fur box, allowing RNA polymerase to bind to the promoter and transcribe the Fur-regulated gene (22). Gonococcal Fur not only regulates iron acquisition genes but also affects the expression of a broad range of genes, including those that encode Opa proteins, NADH dehydrogenase, sodium pumps, and other transcriptional regulators (34). The regulation of genes involved in iron acquisition, adhesion, and metabolism establishes Fur as a global regulator (34, 58).
Fur may not be the sole transcriptional regulator of iron acquisition systems in N. gonorrhoeae. AraC-like regulators operate as both positive and negative regulators of iron acquisition systems in other microorganisms, including Pseudomonas aeruginosa, Yersinia pestis, and Bordetella pertussis (5, 23, 32, 53). In these microorganisms, the AraC-like regulator functions as a transcriptional regulator of siderophore biosynthesis and acquisition genes. The mechanism of AraC-like regulation of siderophore genes involves the cognate siderophore functioning as a coinducer. The AraC-like regulator is under the transcriptional control of Fur, and therefore AraC-like regulation occurs under iron-depleted conditions. The gonococcal genome encodes multiple AraC-like regulators, and it has been recently demonstrated that one of these regulators, MpeR (25), is Fur regulated (34).
In this study, we demonstrated that MpeR, an iron-regulated AraC-like regulator, is required for upregulated expression of the outer membrane transporter FetA in gonococcal strain FA1090. We also establish that fetA is part of an iron-regulated operon that encodes a periplasmic binding protein and components of a putative ABC transport system; however, only fetA and not the downstream genes require MpeR for detectable expression. Furthermore, we determined that gonococcal strain FA1090 acquires iron from enterobactin, enterobactin derivatives, and salmochelin S2 in a FetA- and TonB-dependent manner. Expression of MpeR was necessary to achieve maximal growth on these siderophores, but none of the utilizable iron sources appear to act as a coinducer for MpeR-dependent activation of fetA.
MATERIALS AND METHODS
Bacterial growth conditions.
Gonococcal strains were maintained on GC medium base (GCB) (Difco) containing Kellogg's supplement 1 (35) and 12 μM ferric nitrate. For mutant selection, strains were grown on medium supplemented with 50 μg/ml kanamycin or 1 μg/ml erythromycin. For iron-depleted growth conditions, gonococci were grown in a defined medium that was treated with Chelex 100 (Bio-Rad) (CDM) (66). For iron-replete conditions, gonococcal strains were grown in CDM for one mass doubling before the addition of 12 μM ferric nitrate. For growth in the presence of siderophores, gonococci were grown in liquid CDM for one mass doubling, and then ferric siderophores were added at a concentration of 10 μM. All liquid cultures were grown at 37°C with 5% CO2. For large-scale preparation of gonococcal membrane proteins, GCB broth containing Kellogg's supplement 1 was inoculated with plate-grown, nonpiliated gonococci as described previously (19). For iron-depleted conditions, the iron chelator Desferal (deferroxamine mesylate; Sigma-Aldrich) was added to a final concentration of 50 μM; for iron-replete conditions, 12 μM ferric nitrate was added.
Construction of gonococcal mutants.
All strains and plasmids used in this study are listed in Table 1. Gonococcal strain FA1090 has been described previously (9). To create gonococcal strain MCV304, the mpeR gene was inactivated by the insertion of a kanamycin resistance cassette (aphA-3) as previously described (25). All oligonucleotide primers used in this study are described in Table 2. Primers 5′mpeR and 3′mpeR were used to PCR amplify chromosomal DNA from mpeR::aphA-3 strain FA19 (25). The amplicon was then used to transform gonococcal strain FA1090. To select for allelic exchange, FA1090 transformants were plated on GCB agar containing kanamycin. Strain FA6959 was previously constructed (12) by insertional inactivation of the FA1090 fetA gene with a polar Ω cassette (54). For the construction of the mpeRC complemented strain (MCV305), the mpeR coding sequence from gonococcal strain FA19 was amplified using primers 5′pMpeR pac and 3′GC4 MpeR. The resulting amplicon contains the mpeR gene and 250 bp upstream of the mpeR start codon. The amplicon was inserted between the PacI and PmeI sites of pGCC3 (59). The resulting plasmid, pGCC3-mpeR, was then digested with ClaI, and the fragment containing mpeR, lctP, aspC, and the erythromycin resistance cassette was purified and used to transform MCV304. Transformants were selected on GCB agar supplemented with erythromycin. The resulting complemented strain, MCV305, contains the original mpeR mutation and an ectopically inserted copy of the wild-type mpeR gene, preceded by 250 bp of upstream regulatory signals. The fetA mpeR double mutant strain, MCV306, was constructed by transforming FA6959 with the mpeR::aphA-3 amplicon as described for the construction of MCV304. GCB agar supplemented with kanamycin was used for selection.
Table 1.
Strains and plasmids used in this study
| Strain or plasmid | Genotype and/or relevant characteristicsa | Reference |
|---|---|---|
| Strains | ||
| FA1090 | Wild type (ΔlbpA, HpuAB off) | 9 |
| FA6959 | FA1090 fetA::Ω (Strr Spcr) | 12 |
| MCV656 | FA1090 tonB::Ω (Strr Spcr) | 29 |
| MCV304 | FA1090 mpeR::aphA-3 (Strr Spcr Kmr) | This study |
| MCV305 | mpeRC (Strr Spcr Kmr Ermr) | This study |
| MCV306 | fetA::Ω, mpeR::aphA-3 (Strr Spcr Kmr) | This study |
| Plasmids | ||
| pGCC3-mpeR | pGCC3 containing the full-length mpeR gene and 250 bp of upstream sequence | This study |
| pVCU366 | pCR 2.1 containing 800 bp of fetA through fetB amplified by oVCU498 and oVCU499 | This study |
Strr, streptomycin resistance; Spcr, spectinomycin resistance; Kanr, kanamycin resistance; Ermr, erythromycin resistance.
Table 2.
Oligonucleotides used in this study
| Oligonucleotide | Amplicon | Sequence (5′ to 3′) |
|---|---|---|
| 5′mpeR | mpeR::aphA-3 | ATGAACACCGCCGCCATCT |
| 3′mpeR | mpeR::aphA-3 | GCACTTTTTCACATCCGAAGG |
| 5′pMpeR pac | mpeRC construct | GGTTAATTAACGAAACAACCTGCAGAAACC |
| 3′GC4 MpeR | mpeRC construct | GGTTTAAACTCAGCACTTTTTCACATCCGA |
| malEmpeRF | mpeR coding region | CACTGGGGATCCATGAATACCGCCGCCATCT |
| malEmpeRR | mpeR coding region | CACTGGCTGCAGTCAGCACTTTTTCACATCCGA |
| 5′fetAup | 500-bp upstream fetA probe | GCCCGAACGGTTCGGACAAATT |
| 3′fetAup | 500-bp upstream fetA probe | TTGTTCGTCCTTTTGAGTGT |
| 3′fetAupint | fetA1 upstream probe | TCACTTGGTGCTTCAGCACC |
| 5′fetAupint | fetA2 upstream probe | GGTGCTGAAGCACCAAGTGA |
| rnpB1F | rnpB | CGGGACGGGCAGACAGTCGC |
| rnpB1R | rnpB | GGACAGGCGGTAAGCCGGGTTC |
| oVCU484 | mpeR RT-PCR, forward | GCGTTTCCCACCGAAATCCACAAT |
| oVCU485 | mpeR RT-PCR, reverse | AGCGTAATAATCGGGCGGAGAGTT |
| oVCU486 | fetA RT-PCR, forward | AAAGATTACGAAGCCGGCAAAGGC |
| oVCU487 | fetA RT-PCR, reverse | TTCAACAGGGTTTGTTCGGCAAGG |
| oVCU498 | fetA-fetB, forward | CCAACGCTGCACCAATACCCTGC |
| oVCU499 | fetA-fetB, reverse | CCTTCAGCTTGTCGGCTTCCGCCTG |
| oVCU500 | fetB-ng2091, forward | CAGGCGGAAGCCGACAAGCTGAACG |
| oVCU501 | fetB-ng2091, reverse | GCACAGGCTGACGGCAAACAATACC |
| oVCU513 | ng2091-ng2090, forward | GGTATTGTTTGCCGTCAGCCTGTCG |
| oVCU516 | ng2091-ng2090, reverse | GCTCGCTCGGGACGGTATTGAATC |
| oVCU515 | ng2090-ng2089, forward | GACTTTGTCTTGCACCTGCGCCTG |
| oVCU518 | ng2090-ng2089, reverse | CAGTTCGCGGACGATGACGAACTG |
| oVCU517 | ng2089-ng2088, forward | CTGTGCGGACATTCCTGCCCGAC |
| oVCU553 | ng2089-ng2088, reverse | TGCTTTGGCAGAACACCATCGCAA |
| oVCU110 | 16S rRNA gene, forward | TATCGGAACGTACCGGGTAGC |
| oVCU111 | 16S rRNA gene, reverse | GTATTACCGCGGCTGCTGGCA |
| oVCU529 | fetA quantitative RT-PCR, forward | ATCCAACACCAATTTGGCGTA |
| oVCU530 | fetA quantitative RT-PCR, reverse | CATCGGCGGAATAGCGTTT |
| oVCU461 | mpeR quantitative RT-PCR, forward | TCTACCGCCAGTACCAAACC |
| oVCU462 | mpeR quantitative RT-PCR, reverse | GGCTGAAATTGTGGATTTCG |
| oVCU457 | rmpM quantitative RT-PCR, forward | GGAGCAGGCTCCTCAATATG |
| oVCU458 | rmpM quantitative RT-PCR, reverse | TAAAGTCGGTATGGCCTTCG |
| oVCU576 | ng2091 quantitative RT-PCR, forward | GTGAGCGTGAATTTGGGTTT |
| oVCU577 | ng2091 quantitative RT-PCR, reverse | TATTGCCGACCGTTACAATG |
| oVCU676 | fetB primer extension | CAGGGTCGGAATTTTGCGGCGAACAC |
Isolation of cell fractions containing membrane proteins.
As described previously (19), gonococci were pelleted after large-scale growth under iron-replete or iron-depleted conditions. Gonococcal cells were then resuspended in 10 mM HEPES and passed through a French pressure cell once at 20,000 lb/in2. Intact gonococcal cells were removed by centrifugation at 8,500 × g, and membrane proteins were pelleted by centrifugation at 140,000 × g for 1 h. Total membrane protein fractions were resuspended in 10 mM HEPES, and protein concentrations were determined by bicinchoninic acid assay (Pierce).
Separation of total membrane protein fractions and MS analysis.
Membrane protein fractions, isolated as described above, were solubilized, and proteins were separated on a 7.5% polyacrylamide gel. Proteins were visualized by Coomassie blue staining. One band of interest was extracted from the stained gel and submitted to the VCU Mass Spectrometry Resource Center for identification. The sample was digested overnight with trypsin, and the resulting peptides were extracted. The peptides were analyzed on a liquid chromatography-mass spectrometry (LC-MS) system that consisted of a Thermo Electron Deca XP Plus mass spectrometer with a nanospray ion source interfaced with a reversed-phase capillary column.
Immunodetection of FetA and TbpA.
For detection of FetA in total membrane protein preparations, aliquots containing 20 μg of protein were resolved using SDS-PAGE. For detection of FetA in whole-cell lysates, gonococcal strains were grown in the presence of iron-containing catechols for 6 h; every 2 h, aliquots were removed and standardized to culture density. Cells were pelleted, lysed with Laemmli solubilizing buffer (36), and stored at −20°C. Before use, 5% β-mercaptoethanol was added to all preparations prior to heating at 95°C for 3 min. After SDS-PAGE (19), proteins were electroblotted to nitrocellulose membranes in 20 mM Tris base, 150 mM glycine, and 20% methanol (64) within a submerged transfer apparatus (Bio-Rad). For detection of FetA, membranes were blocked with 5% skim milk in low-salt Tris-buffered saline (LS-TBS). FetA blots were then probed with a FetA-specific monoclonal antibody (13) and washed with LS-TBS, followed by a secondary goat anti-mouse antibody conjugated to alkaline phosphatase (Bio-Rad). Blots were developed using nitroblue tetrazolium and 5-bromo-4-chloro-3-indolylphosphate (Sigma).
For detection of TbpA in whole-cell lysates, proteins were separated by SDS-PAGE as described above, followed by electroblotting to nitrocellulose. Membranes were blocked with 5% bovine serum albumin (Roche) in high-salt TBS plus 0.05% Tween 20 (Sigma). TbpA blots were then probed with a polyclonal TbpA-specific antiserum (19) and washed with high-salt TBS plus 0.05% Tween, followed by a secondary goat anti-rabbit antibody conjugated to alkaline phosphatase (Bio-Rad). Blots were developed using nitroblue tetrazolium and 5-bromo-4-chloro-3-indolylphosphate (Sigma).
RNA isolation.
Gonococcal strains were grown in CDM under iron-depleted and iron-replete conditions as described above. After one mass doubling, 12 μM FeNO3 (iron-replete conditions) or no additional iron (iron-depleted conditions) was added, and the cultures were grown for an additional 2 h. Total RNA was isolated from cultures using the RNeasy minikit as directed by the manufacturer (Qiagen). Purified RNA was treated twice with RNase-free DNase as directed by the manufacturer (Qiagen). SUPERase-In (Ambion) was added before storage at −80°C.
Qualitative RT-PCR.
Portions of the 16S rRNA gene, fetA, mpeR, and intergenic regions between the fet genes were amplified using the Thermoscript reverse transcriptase PCR (RT-PCR) system (Invitrogen) as described previously (29). A portion of this reaction product was used as the template for PCR amplification with Platinum Taq polymerase (Invitrogen) according to the manufacturer's protocol. Oligonucleotide sequences of the primers used in this analysis are listed in Table 2. After an initial denaturation step at 94°C for 3 min, DNA was amplified for 30 cycles. Each cycle consisted of 1 min at 94°C, 30 s at 60°C, and 1.5 min at 72°C, followed by a final extension step for 10 min at 72°C. To detect any DNA contamination of RNA preparations, parallel RT-PCRs were conducted in the absence of reverse transcriptase. Amplicons resulting from the RT-PCRs were detected by ethidium bromide staining of agarose gels.
Real-time RT-PCR.
cDNA was generated by reverse transcription of 100 ng of total RNA using the Accuscript high-fidelity first-strand cDNA synthesis kit (Stratagene) according to the manufacturer's protocol. Synthesized cDNA was used as the PCR template. The SensiMix SYBR No-ROX kit (Bioline) and CFX96 real-time system (Bio-Rad) were employed for the real-time RT-PCRs. Oligonucleotide sequences of the primers used in this analysis are listed in Table 2. The polar Ω insertion in fetA was located between the binding sites for the primers used to detect fetA expression; the aphA-3 cassette was located downstream of the primer binding sites for mpeR expression analysis. The cDNA-SensiMix mixture was initially heated to 94°C for 10 min and subjected to 40 cycles of 95°C for 15 s, 60°C for 30 s, and 72°C for 30 s. Transcription of rmpM was employed as an internal control. For each experimental condition, fetA, mpeR, and ng2091 transcripts were normalized to rmpM levels. The relative threshold cycle (CT) method (41) was employed to compare normalized expression levels under different conditions. Three biological replicates were analyzed, each conducted in triplicate.
Identification of the fetB transcriptional start site by primer extension analysis.
In order to identify the fetB transcriptional start site, RNA was harvested from FA1090 grown under iron-replete or iron-depleted conditions as described above. The reverse primer, oVCU676 (Table 2), was radiolabeled with γ-32P using T4 polynucleotide kinase; 5 μg of RNA was incubated with the radiolabeled primer and reverse transcriptase to generate the primer extension product. In order to generate reference sequence products, plasmid pVCU366 (Table 1) was sequenced using the reverse primer, oVCU676, and the SequiTherm EXCEL II DNA-sequencing kit (Epicenter) as described previously (57). The sequencing template plasmid, pVCU366, was generated by amplification of wild-type FA1090 genomic DNA with the primers oVCU498 and oVCU499 (Table 2). The resulting amplicon contained the fetB promoter region and was cloned into pCR 2.1 (Invitrogen). Both the primer extension product and the reference sequence were subjected to electrophoresis on a 6% acrylamide sequencing gel, which was dried and subjected to autoradiography for visualization.
Electrophoretic mobility shift assay (EMSA).
MpeR was fused in frame at its amino terminus to the maltose binding protein (MBP) using the pMal-c2x fusion vector (New England Biolabs). For this purpose, the mpeR coding region was PCR amplified from FA19 chromosomal DNA, which was isolated as described previously (43) using primers 5′malEmpeRF and 3′malEmpeRR (Table 2). The resulting amplicon was purified using the QIAquick PCR purification kit (Qiagen). Both the vector and PCR product were digested with PstI and BamHI (New England BioLabs) and ligated using T4 DNA ligase (New England Biolabs). Escherichia coli transformants harboring the construct were selected on LB agar (Difco) plates containing 100 μg/ml of ampicillin. Both strands of the cloned insert were sequenced to ensure fidelity of the PCR amplification reaction and in-frame fusion with malE. Growth of the E. coli transformant bearing the plasmid construct, induction of expression, and purification of MBP-MpeR were performed as described previously for an MBP-MtrR fusion (38).
For the EMSA studies, the 500-bp intergenic region immediately upstream of the FetA start codon was PCR amplified from FA1090 chromosomal DNA using primers 5′fetAup and 3′fetAup (Table 2). This upstream region was further divided into two smaller products by PCR amplification. The 5′fetAup and 3′fetAupint primers (Table 2) resulted in fetA1, and the 5′fetAupint and 3′fetAup primers (Table 2) were used to amplify the fetA2 region. These three PCR products were purified using the QIAquick PCR purification kit (Qiagen) and end labeled with γ-32P (Perkin-Elmer) and T4 polynucleotide kinase (New England Biolabs). The radiolabeled PCR products were purified by excising the DNA from nondenaturing polyacrylamide gels and recovered by crush-soak elution overnight at 37°C into 750 μl of PB buffer from the QIAquick PCR purification kit (Qiagen). The radiolabeled DNA-buffer mixture was centrifuged at 15,800 × g for 10 min, and the resulting supernatant was removed and added to a QIAquick PCR purification kit (Qiagen) column, which was then washed with 750 μl PE buffer. The DNA was eluted in 100 μl of nuclease-free water (Ambion). Five nanograms of each radiolabeled probe was incubated with 10 μg of MBP-MpeR for 30 min at room temperature.
The specificity of the MpeR-fetA promoter interaction was evaluated by adding either specific or nonspecific unlabeled competitor DNA to the binding reaction mixtures. The specific unlabeled competitor DNA was generated by PCR amplification of the fetA1 promoter region as described above. The fetA2 sequence was also amplified as described above and used as an unlabeled competitor. The nonspecific competitor DNA was generated by PCR amplification of a portion of the rnpB gene using the primers rnpB1F and rnpB1R (Table 2). All binding reaction mixtures were incubated in DNA binding buffer [20 mM Tris-HCl, 200 mM NaCl, 1 mM dithiothreitol (DTT), 1 μg/ml poly(dI-dC)] for 30 min at room temperature. All samples were subjected to electrophoresis on a 5% (wt/vol) polyacrylamide gel at 4°C. After electrophoresis, the gel was dried on Whatman filter paper and exposed to X-ray film for autoradiography.
Preparation of ferric siderophores.
The siderophores used in this study were purchased from EMC Microcollections (Tübingen, Germany). Siderophores were resuspended to a final concentration of 1 mg/ml in sterile deionized water and ferrated to 80% saturation using FeCl3 (62). In some experiments, the siderophores were purchased preferrated and the lyophilized ferric siderophores were dissolved in methanol (enterobactin) or water prior to final dilution in water. The results of the siderophore utilization assays were the same regardless of whether the siderophores were purchased in the ferrated state or ferrated immediately before use. The following siderophores were tested for growth support of gonococcal strain FA1090: ornibactin, aerobactin, ferrichrysin, ferrirubin, coprogen, neocoprogen, enterobactin, dihydroxybenzoylserine (DHBS) monomer, DHBS dimer, DHBS trimer, salmochelin S4, and salmochelin S2.
Xenosiderophore utilization assays.
Plate bioassays to evaluate xenosiderophore utilization were performed using CDM plates supplemented with 2.5 μM apo-bovine transferrin to chelate excess iron. As described previously (62), strains were inoculated onto plates using a sterile Dacron swab (Puritan). A sterile Pasteur pipette was used to bore a well into the agar. Subsequently, 10 μl of the diluted ferric siderophore solution (100 μg/ml) was added to each well. Ten microliters of apo-bovine transferrin at a concentration of 10 mg/ml was used as a negative control, and 10 μl of ferric citrate (10 μM) was used as a positive control. Plates were incubated at 37°C in a 5% CO2 atmosphere for 24 h and then evaluated for ferric siderophore-dependent growth.
Statistical analysis.
Statistical significance of xenosiderophore-dependent growth data was determined by using a two-tailed, unpaired Student t test. P values for specific comparisons are reported in the figure legends. For real-time RT-PCR data (see Table 3), the relative CT method (41) was utilized to calculate the fold change for each comparison. The average fold change values were calculated from three independently conducted RT-PCRs and are representative of results generated from three independent RNA preparations (biological replicates).
Table 3.
Effects on gene expression measured by real-time RT-PCR
| Comparisona | Normalized target gene expression ratiob |
||
|---|---|---|---|
| fetA | mpeR | ng2091 | |
| WT−Fe/WT+Fe | 97 (103-83) | 198 (301-106) | 16.3 (17-16) |
| WT−Fe/FA6959−Fe | 194 (209-167) | 2.2 (3.1-.6) | 23 (28-16) |
| WT−Fe/MCV304−Fe | 117 (185-33) | 62 (123-46) | 22.3 (24.22) |
| WT−Fe/MCV305−Fe | 9.6 (15-5) | 1.9 (2.4-1.3) | 7.1 (9.7-5) |
| WT−Fe/MCV306−Fe | 108 (154-70) | 37 (45-26) | 11.5 (16-9) |
−Fe, iron-depleted conditions; +Fe, iron-replete conditions.
The average fold change for each comparison is shown, with the range detected among three independently conducted real time RT-PCR assays shown in parentheses.
RESULTS
The mpeR mutant displays differential protein expression under iron-depleted conditions.
The mpeR gene was originally identified by Folster and Shafer as encoding an AraC-like regulator that plays a role in the coordinate expression of hydrophobic agent efflux pumps (25). We began investigating whether MpeR plays other regulatory roles in gonococci due to its proximity to a gene that encodes an uncharacterized TonB-dependent transporter, TdfF (29). We initially determined that mpeR was iron regulated, which was recently confirmed by Jackson et al. via microarray analysis, Fur titration assay (FURTA), and the presence of a Fur box in its promoter (34). We hypothesized that since MpeR is iron regulated, it could also regulate iron acquisition systems in gonococci, particularly the one putatively encoded by the tdfF locus. To test this hypothesis, the wild type (FA1090) and mpeR mutant (MCV304) were grown under iron-depleted and iron-replete conditions, and total membrane protein fractions were isolated. Using SDS-PAGE analysis, we determined that an 80-kDa protein was expressed by the wild-type strain only under iron-depleted conditions but this protein was not detectable in the mpeR mutant strain (Fig. 1A). The 80-kDa band was excised from the gel and analyzed by mass spectrometry, which unambiguously identified the protein as FetA. Western blot analysis of total membrane proteins isolated from the wild type (FA1090), the mpeR mutant (MCV304), the fetA mutant (FA6959), and the mpeRC strain (MCV305) grown under iron-depleted and iron-replete conditions was used to confirm the mass spectrometry results. The wild-type and complemented strains grown under iron-depleted conditions expressed detectable levels of FetA, whereas neither the fetA mutant nor the mpeR mutant expressed this protein (Fig. 1B). From this analysis, we concluded that the iron-regulated AraC-like regulator MpeR controlled the expression of FetA in gonococcal strain FA1090.
Fig. 1.
FetA expression is regulated by MpeR. (A) SDS-PAGE analysis of protein expression. Total membrane proteins were isolated from wild-type (WT) (FA1090) and mpeR mutant (MCV304) strains grown under iron-depleted (−) and iron-replete (+) conditions for 4 h. Proteins were separated on a 7.5% acrylamide gel. The arrow on the left indicates the band that was excised and identified as FetA by mass spectrometry analysis. The positions of molecular mass markers are indicated on the right. (B) Western blot analysis of FetA expression. The WT (FA1090), mpeR mutant (MCV304), fetA mutant (FA6959), and complemented mpeRC (MCV305) strains were grown under iron-depleted (−) and iron-replete (+) conditions. Total membrane proteins from each strain were isolated and standardized before being separated by SDS-PAGE and then transferred to nitrocellulose. Blots were probed with an anti-FetA monoclonal antibody.
fetA transcription requires MpeR expression and iron-depleted growth conditions.
fetA, previously called frpB, encodes a TonB-dependent outer membrane transporter (FetA) that is immunogenic and subject to Fur regulation (6, 12, 51, 63). FetA was renamed by Carson et al. when they discovered that this transporter was necessary for efficient ferric enterobactin transport (12). To determine whether MpeR transcriptionally regulates fetA, endpoint relative RT-PCR was utilized to detect fetA and mpeR transcripts. RNA was isolated from gonococcal strains grown under iron-depleted and iron-replete conditions. Transcripts from fetA and mpeR were detected in the wild-type strain preferentially under iron-depleted conditions (Fig. 2), consistent with previous published studies (13, 34). The fetA transcript was not detected in the mpeR mutant (MCV304). In the mpeRC strain (MCV305), both mpeR and fetA transcripts were detected (Fig. 2), indicating that the MpeR effect on fetA expression was restored by complementation. Expression of mpeR in the mpeRC strain (MCV305) was iron regulated (Fig. 2), consistent with the presence of iron-sensitive regulatory signals in the sequence upstream of the MpeR start codon, which were included in the complementation construct. Overall, these data allow us to conclude that MpeR is necessary for fetA expression under iron-depleted conditions.
Fig. 2.
MpeR activates fetA transcription under iron-depleted conditions. The WT (FA1090), fetA mutant (FA6959), mpeR mutant (MCV304), and complemented mpeRC (MCV305) strains were grown under iron-depleted (−) and iron-replete (+) conditions. RNA samples isolated from each gonococcal strain were analyzed for expression of fetA and mpeR by RT-PCR. The 16S rRNA gene (16s)was used as a positive control because it was constitutively expressed under all growth conditions. Expression of the 16S rRNA gene in the absence of reverse transcriptase [16s(−)] was used as a negative control.
Real-time RT-PCR was employed to confirm the endpoint RT-PCR results and to quantify the level of gene expression. All CT values were normalized to rmpM gene expression, and then the fold change was determined using the relative CT method (41). The rmpM gene encodes an outer membrane protein that is constitutively expressed under all growth conditions tested in this study. In Table 3, the average fold change is shown, representing the comparison between expression under iron-depleted and iron-replete conditions or between expression by the wild type and the mutant under iron-depleted conditions. As expected, the fold change in expression under iron-depleted versus iron-replete conditions was large for both fetA and mpeR (97-fold for fetA and 198-fold for mpeR) (Table 3). When the wild type and its isogenic fetA mutant were compared, there was 194-fold more fetA expression in the wild type. mpeR expression was unaffected in the fetA mutant. When the wild type and the mpeR mutant were compared, there was a 117-fold change in fetA expression (Table 3), which is consistent with the inability to detect fetA transcripts in the mpeR mutant as observed by qualitative RT-PCR (Fig. 2). As shown in Table 3, the restoration of the mpeR gene in the mpeRC strain resulted in mpeR expression levels that approached those of the wild type (1.9-fold difference between the wild type and the mpeRC strain). Commensurate with the return of mpeR expression, we detected an increase in FetA expression in the mpeRC strain relative to the mpeR mutant (Table 3), again supporting the relative RT-PCR data (Fig. 2). Together, the expression data support the previous observations that both fetA and mpeR are iron regulated (6, 34) and furthermore demonstrate that mpeR expression was not affected by the fetA mutation. The results from the relative and real-time expression studies establish that when mpeR was absent, fetA expression decreased, and when mpeR expression was restored by complementation with a wild-type copy, fetA levels returned to near-wild-type levels under iron-depleted conditions. Cumulatively, these data indicate that MpeR is necessary for fetA transcription under iron-depleted conditions.
Specific binding of MpeR to DNA upstream of fetA.
In order to determine whether MpeR-dependent fetA regulation occurred as a result of direct interaction, we used EMSA to test whether MpeR bound to target DNA sequences upstream of the FetA start codon. Results from preliminary EMSA experiments using increasing amounts of MBP-MpeR (1 to 10 μg) incubated with the radiolabeled fetA probe showed shifting of a DNA fragment of 500 bp in length, whereas MBP alone showed no shift (data not shown). This upstream region was further divided into two smaller DNA fragments of 250 bp in length (Fig. 3A), both of which were tested for MpeR interaction by EMSA. The probe fragment corresponding to the most upstream portion of the intergenic region (fetA1) (Fig. 3A) was shifted by addition of MpeR protein (Fig. 3B), while MBP alone did not shift the fetA1 fragment (data not shown). The probe fragment corresponding to the sequence immediately preceding the fetA start codon (fetA2) (Fig. 3B) was not shifted in the presence of MpeR (data not shown). In order to determine whether MpeR binding upstream of fetA was specific, a competitive EMSA experiment was performed in which increasing amounts (2× to 20×) of excess unlabeled specific (fetA) or nonspecific (rnpB) DNA fragments were added to the binding reaction. The results demonstrate that the specific competitor, but not the nonspecific competitor, reduced binding of MpeR to the labeled fetA probe (Fig. 3B). The unlabeled fetA2 sequence modestly competed with the binding of MpeR to the fetA1 probe, suggesting that MpeR may bind preferentially to the fetA1 sequence and with a lower affinity to the fetA2 sequence. Cumulatively, these results allow us to conclude that binding of MpeR to the DNA sequence upstream of fetA is specific and that its capacity to activate fetA expression is likely by a direct mechanism.
Fig. 3.
MpeR binds upstream of fetA in a specific manner. (A) Sequence of the 500-bp intergenic region immediately upstream of fetA. The sequence highlighted in teal is contained within the fetA1 probe employed for the EMSA shown in panel B. The sequence highlighted in yellow is contained within the fetA2 competitor DNA. The sequence highlighted in blue is contained in both fetA1 and fetA2 amplicons. The Fur binding site (34) is highlighted in gray. The previously mapped (14) promoter elements (underlined) and transcriptional start site (asterisk) are identified. The start codon for FetA is shown in red. (B) Five nanograms of the 250-bp labeled fetA1 probe (lane 1) was incubated with 10 μg of MBP-MpeR in the absence of unlabeled competitor (lane 2) or in the presence of 2×, 10×, or 20× excess unlabeled competitor DNA. Lanes 3 to 5 contain reaction mixtures including increasing concentrations of the specific competitor (fetA1), lanes 6 to 8 contain mixtures including increasing concentrations of fetA2, and lanes 9 to 11 contain mixtures including increasing concentrations of the nonspecific competitor rnpB.
fetA is part of an iron-responsive operon that is differentially affected by MpeR expression.
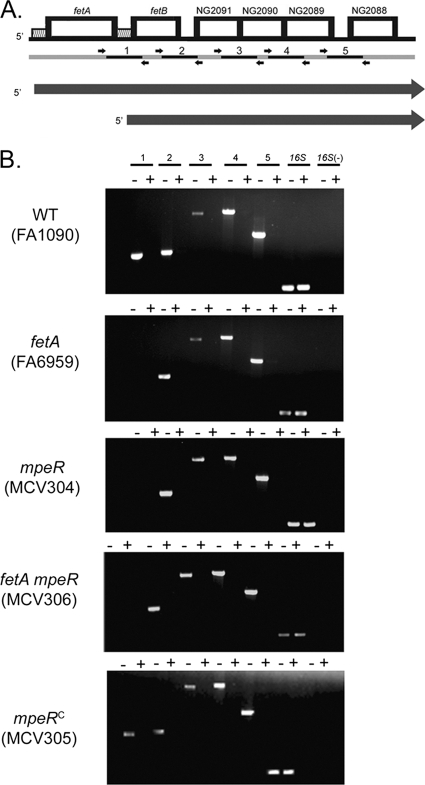
The DNA region downstream of fetA encodes putative xenosiderophore acquisition genes (Fig. 4A). The genes downstream of fetA include the following: fetB, which encodes a putative periplasmic binding protein; ng2091 and ng2090, which encode two predicted permease proteins; and ng2088, encoding an ATP binding protein. The last three genes are therefore predicted to form an ABC transport complex. ng2089 encodes an uncharacterized protein. Most siderophore acquisition systems are cotranscribed, and all genes involved in the system are coordinately regulated. Therefore, endpoint RT-PCR was utilized to determine if fetA and the downstream genes were cotranscribed and also activated by MpeR. Primers were designed to amplify the intergenic regions between each gene in the hypothetical operon (Fig. 4A). RNA was isolated from gonococcal strains grown under iron-depleted and iron-replete conditions. In the wild-type strain (FA1090), all amplicons were detected under iron-depleted conditions (Fig. 4B), indicating that fetA and downstream genes are part of an iron regulated operon. Interestingly, the fetA mutant, which contains a polar Ω (54) insertion in the fetA gene, maintained the ability to express all of the downstream putative genes (Fig. 4B). Similar results were observed for the mpeR mutant, in which genes fetB to ng2088 were cotranscribed under iron-depleted conditions (Fig. 4B). A fetA mpeR double mutant strain was also employed to investigate cotranscription of the ABC transport genes. Again, the downstream putative transport genes, in the absence of both fetA and mpeR, were all transcribed under iron-depleted conditions (Fig. 4B). Finally, when the mpeR mutant was complemented with the wild-type mpeR gene, fetA and the downstream cotranscripts were detected under iron-depleted conditions, similar to the case for the wild-type strain. Based on these RT-PCR results, we propose that the wild-type strain produces two different transcripts under iron-depleted conditions (Fig. 4A). One transcript encodes fetA to ng2088, whereas the other transcript encodes fetB to ng2088, expression of which is iron regulated but not qualitatively affected by MpeR (Fig. 4A). The fetA transcriptional start site was previously mapped (13) and is identified in Fig. 3. To confirm the presence of a second transcriptional start site upstream of fetB, we employed primer extension analysis. As shown in Fig. 5, we identified a transcriptional start site downstream of several possible −10 sequences, within the previously identified Fur binding site (34). The transcript starting with fetB was modestly iron regulated, and the initiation site was located 72 nucleotides upstream of the FetB start codon.
Fig. 4.
FetA is encoded as part of a multigene operon. (A) Genetic locus, including fetA and downstream genes. Genes are depicted by boxes. Hatched regions 5′ of fetA and fetB indicate approximate locations of Fur boxes (34). Below the chromosomal locus are small dark arrows indicating primer locations. Numbered black bars denote the amplicons generated from each primer set. Long, dark gray arrows indicate the lengths and start positions of two proposed transcripts. (B) RT-PCR analysis of the fet operon. RNA was isolated from the indicated gonococcal strains which were grown under iron-depleted (−) and iron-replete (+) conditions. Amplicon numbers correspond to the diagram in panel A. The 16S rRNA gene (16S) was used as a positive control because it is constitutively expressed under all conditions tested. Expression of the 16S rRNA gene in the absence of reverse transcriptase [16S(−)] was used as a negative control.
Fig. 5.
Identification of the fetB transcriptional start site. (A) Sequence of the intergenic region immediately upstream of fetB. The ATG at the end of the sequence represents the FetB start codon. Several overlapping potential −10 promoter elements are underlined. The Fur binding site (34) is highlighted in gray. The transcriptional start site identified in this analysis is identified by the asterisk. (B) Primer extension products generated from RNA samples harvested from wild-type gonococcal strain FA1090 grown under iron-replete (+Fe) and iron-depleted (−Fe) conditions. Equivalency of the amount of RNA template in each sample was confirmed by ethidium bromide staining of RNA separated on an agarose gel. For comparison, the sequencing reaction using the same primer as was used for the primer extension reaction is shown on the left. The T residue highlighted by the asterisk marks the point of transcript initiation on the noncoding strand.
To establish whether mpeR or fetA interruption quantitatively affected expression of the downstream ABC transport genes, real-time RT-PCR was employed. ng2091 is the first gene transcribed downstream of the gene encoding the putative periplasmic binding protein (fetB) (Fig. 4A). The same RNA samples isolated for qualitative RT-PCR (Fig. 4B) were used for real-time RT-PCR analysis. All CT values were normalized to rmpM gene expression. In Table 3, the average fold change is shown, representing the comparison between expression under iron-depleted and iron-replete conditions or expression by the wild type and the mutant under iron-depleted conditions. As shown in Table 3, ng2091 gene expression was iron repressed; 16.3-fold more transcript was detected under iron-depleted conditions. When the wild-type and mutant strains were compared, we observed that ng2091 gene expression levels were 23-fold higher in the wild type than in the fetA mutant. Similarly, ng2091 expression was 22.3-fold higher in the wild type than in the mpeR mutant. The wild-type strain expressed 11.5-fold more ng2091 transcript than the fetA mpeR double mutant (Table 3). The mpeRC strain expressed more ng2091 transcript than the mpeR mutant, as expected. Cumulatively, these results indicate that fetA is part of an iron-regulated operon encoding a putative periplasmic binding protein and ABC transport system. The quantitative expression studies suggest that the ABC transport system is not as tightly iron regulated as is either fetA or mpeR, which is consistent with the transcriptional start site mapping data presented in Fig. 5. Furthermore, the real-time RT-PCR data indicate that expression of ng2091 is activated by MpeR but that the extent of this activation is not as great as was detected for fetA.
Xenosiderophore iron acquisition by gonococcal strain FA1090.
N. gonorrhoeae does not produce siderophores; however, the pathogen is capable of utilizing siderophores synthesized by other microorganisms (12, 62, 65). In gonococcal strain FA1090, Carson et al. demonstrated that FetA functions as a receptor for the xenosiderophore enterobactin (12). Enterobactin is a cyclic catecholate siderophore composed of three 2,3-dihydroxybenzoylserine (DHBS) subunits and was first characterized in E. coli and Salmonella (49, 52). Enterobactin derivatives, including the DHBS monomer (D1), the DHBS dimer (D2), and the DHBS trimer (D3), have also been identified as siderophores secreted by E. coli (31, 48). Carson et al. determined that enterobactin is utilized by gonococcal strain FA1090 in a FetA- and TonB-dependent manner (12). We tested whether strain FA1090 could also utilize other xenosiderophores in a similar manner. Since MpeR activates fetA expression, MpeR was predicted to affect catecholate utilization in gonococcal strain FA1090 as well.
Plate bioassays were employed to measure siderophore-dependent growth of gonococcal strain FA1090. We tested a variety of siderophores, including ornibactin, aerobactin, ferrichrysin, ferrirubin, coprogen, neocoprogen, enterobactin, DHBS, and salmochelin, for growth support. We found that ferrichrysin, ferrirubin, coprogen, and neocoprogen did not support growth at all. Ornibactin and aerobactin supported growth but only in a TonB-independent fashion (data not shown). Thus, we focused on the catecholate-type siderophores, including enterobactin, DHBS monomer (D1), DHBS dimer (D2), DHBS trimer (D3), salmochelin S4 (a cyclic, diglucosylated form of enterobactin), and the linear derivative of salmochelin (S2) (46). We evaluated xenosiderophore utilization in the wild type, the tonB mutant, the fetA mutant, the mpeR mutant, the mpeRC strain, and the fetA mpeR double mutant. Ferric citrate was used as a positive control, and bovine transferrin was used as a negative control since gonococci cannot utilize iron bound to non-human transferrin (3, 37). Figure 6 shows the average growth zone (in millimeters) detected around each iron source, with the standard deviation reflecting the variability within four or five independently conducted experiments, each of which was performed in triplicate. As previously shown by Carson et al. (12), gonococcal strain FA1090 grew in the presence of enterobactin (Fig. 6). Enterobactin-dependent growth of the fetA and tonB mutants was reduced to just above background levels (Fig. 6, dashed line). Enterobactin-dependent growth of the mpeR mutant was also decreased relative to that of the wild-type strain, although growth inhibition was not as great as was seen with the fetA and tonB mutant strains. Enterobactin-dependent growth was restored in the mpeRC strain to near-wild-type levels, consistent with the recovery of MpeR regulatory function in this strain. The fetA mpeR double mutant was severely restricted in enterobactin-dependent growth. These findings are consistent with those of Carson et al. (12) but further extend our observation that MpeR activates FetA expression and leads to an enhanced ability to utilize the xenosiderophore enterobactin.
Fig. 6.
Xenosiderophore utilization by gonococcal strain FA1090. CDM plates were supplemented with apo-bovine transferrin, and wells within the plates were inoculated with the following siderophores: ENT, enterobactin; D1, dihydroxybenzoylserine (DHBS); D2, the dimer form of DHBS; D3, the trimer form of DHBS; S2, the linear derivative of salmochelin; and S4, the cyclized form of salmochelin. Ferric citrate (+) was used as the positive control, and apo-bovine transferrin (−) was used as the negative control. Each bar indicates the average growth in millimeters around each siderophore source; the averages and standard deviations were determined from seven independent experiments, each conducted in triplicate. Bars represent average growth zones for the wild-type strain FA1090, fetA mutant strain FA6959, tonB mutant strain MCV656, the mpeR mutant strain, the mpeRC complemented strain, and the fetA mpeR double mutant strain. The horizontal dashed line indicates the diameter of the well containing each iron source. Pairwise comparisons between the wild-type and mutant strains resulted in the following P values: *, P < 0.001; #, P = 0.0124; ^, P = 0.0197.
As shown in Fig. 6, our analysis additionally demonstrates that gonococcal strain FA1090 can employ the enterobactin derivatives D2 and D3 in a FetA- and TonB-dependent manner. As was detected with enterobactin, use of these enterobactin derivatives was also maximized when MpeR was expressed. The mpeR mutant was capable of less growth around D2 and D3 (Fig. 6) than the wild-type strain, and complementation of the mpeR mutation led to an increase in D2- and D3-dependent growth. Interestingly, there was also a significant decrease in salmochelin S2-dependent growth of both the fetA mutant and the tonB mutant, indicating that FA1090 utilized salmochelin S2 in a FetA-and TonB-dependent manner as well. These results demonstrate that gonococcal strain FA1090 is capable of employing enterobactin, the DHBS dimer (D2) and trimer (D3) derivatives, and salmochelin S2 as iron sources. FetA, TonB, and MpeR are critical for this process, consistent with the ability of MpeR to induce expression of FetA. This is the first demonstration that gonococcal strain FA1090 utilizes DHBS dimers and trimers and salmochelin S2 as iron sources and that this uptake pathway depends upon expression of FetA, TonB, and MpeR activation. While salmochelin S4 was not utilized by any FA1090 variants tested in this analysis, the DHBS monomer was employed by all strains, both mutant and wild type. These results imply that FA1090 does not have the capacity to internalize salmochelin S4 but can import DHBS in a TonB- and FetA-independent pathway.
Xenosiderophores that support growth do not serve as coinducers for MpeR-dependent fetA activation.
AraC-like transcriptional regulators are distinguished by a C-terminal helix-turn-helix motif, which is responsible for DNA binding. These regulators also contain an N-terminal binding site to which activator molecules bind, conferring specificity on regulation (26). MpeR, an AraC-like regulator, enhances fetA transcription (Fig. 2 and 4), and MpeR binds to the region upstream of the fetA gene (Fig. 3). Gonococcal strain FA1090 can utilize enterobactin, DHBS, and salmochelin S2 as iron sources (Fig. 6). Given these findings, we tested whether these xenosiderophores could serve as coinducers for MpeR-dependent FetA activation. Western blot analysis was utilized to investigate FetA expression when gonococci were grown in the presence of ferrated catecholates. The wild-type strain was grown in the presence of the ferrated forms of enterobactin, DHBS monomer (D1), DHBS dimer (D2), DHBS trimer (D3), salmochelin S4, and salmochelin (S2). FA1090 grown in the presence of all tested siderophores except salmochelin S4 exhibited FetA expression levels similar to those detected under iron-replete conditions (Fig. 7A). The wild-type strain grown in the presence of S4 exhibited FetA expression levels similar to those expressed when grown under iron-depleted conditions (Fig. 7A). To determine whether the decrease in FetA expression was siderophore specific or due to iron status, we analyzed TbpA expression from the same cultures. TbpA is a Fur-regulated outer membrane transporter of iron from human transferrin (19). TbpA expression mirrored that of FetA as a function of growth on xenosiderophores (Fig. 7A). As shown in Fig. 6, salmochelin S4 did not support the growth of FA1090. Thus, the increase in FetA expression in the presence of salmochelin S4 is due to iron stress rather than to S4 serving as a coinducer for MpeR in the activation of FetA. Furthermore, the other siderophores and derivatives that supported growth resulted in high internal iron pools and consequently resulted in repression of FetA. We repeated this experiment, replacing the ferrated siderophores with the iron-free forms during gonococcal growth (Fig. 7B). As seen with the ferrated xenosiderophores, there was no evidence of siderophore-dependent induction, as all strains expressed FetA at levels similar to those detected under iron-depleted growth conditions. Cumulatively, these results suggest that FetA expression is sensitive to the iron status of the cell and that the presence of the xenosiderophores, in either ferrated or iron-free form, did not further influence FetA expression.
Fig. 7.
FetA expression is not induced by the presence of xenosiderophores. (A) WT strain FA1090 was grown in CDM with the indicated ferrated xenosiderophores (final concentration of 10 μM) as the sole iron source: ENT, enterobactin; D1, dihydroxybenzoylserine (DHBS); D2, the dimer form of DHBS; D3, the trimer form of DHBS; S2, the linear derivative of salmochelin; and S4, the cyclized form of salmochelin. As controls, the WT strain was grown in the absence of iron (−) or with ferric nitrate (+) but without the addition of siderophores. Aliquots collected at 2, 4, and 6 h (indicated above the blots) were lysed and subjected to SDS-PAGE. After separation, proteins were transferred to nitrocellulose. Blots were probed with anti-FetA (top) or anti-TbpA (bottom) antibodies. (B) As in panel A, except the WT strain FA1090 was grown in CDM with the indicated xenosiderophores in the iron-free or apo form.
DISCUSSION
MpeR was first identified by Folster and Shafer as a homolog of other AraC-like regulators (25). MpeR was originally described as a transcriptional regulator of the mtrF gene, which encodes a protein that modulates antimicrobial efflux pump activity in gonococcal strain FA19. In the present study, we determined that in contrast to its repressive action on mtrF, MpeR activates fetA transcription under iron-depleted conditions. While MpeR clearly plays a role, other regulators or cofactors may also be involved in controlling fetA expression. Importantly, this is the first example of an AraC-like regulator that is involved in the regulation of an outer membrane xenosiderophore transporter in N. gonorrhoeae. Thus, the transcriptional regulatory activities of MpeR affect at least two important properties needed for survival of gonococci during infection: efflux of host-derived antimicrobials by the Mtr system (25) and xenosiderophore iron acquisition via FetA. In the context of the present work, we suggest that MpeR regulation of fetA may aid in gonococcal immune evasion. Anti-FetA antibodies are present in sera from patients convalescing from meningococcal disease, and these antibodies are cross-reactive against gonococcal strains (1, 8). In addition, monoclonal antibodies against FetA are bactericidal in the presence of human complement (51). Therefore, continuous, unregulated FetA expression during the entirety of an infection is expected to elicit a host response that would inhibit the bacteria from thriving in vivo. Thus, fetA expression is expected to be tightly controlled.
In the current study, we determined that enterobactin and DHBS dimers (D2) and trimers (D3) were utilized by strain FA1090 in a FetA- and TonB-dependent manner. MpeR-dependent activation of FetA enhanced the ability of FA1090 to employ these xenosiderophores as sole iron sources. While the ability of N. gonorrhoeae to utilize enterobactin had been previously recognized (12), this is the first demonstration of gonococcal use of DHBS dimers (D2) and trimers (D3) as iron sources. Like enterobactin, these enterobactin derivatives were employed by wild-type FA1090 in a FetA-, TonB-, and MpeR-dependent mechanism. This is somewhat surprising, since these siderophores are internalized via distinct TonB-dependent transporters in other Gram-negative bacteria (27, 67) Moreover, strain FA1090 can also employ the xenosiderophore salmochelin S2 in the same FetA-dependent pathway. The broad specificity of FetA for all four xenosiderophores is remarkable, as their import into E. coli is facilitated by four distinct transporters, i.e., FepA, Cir, FiuA, and IroN (67). Carson et al. (12) noted that the sequence of FetA from gonococcal strain FA1090 retained those residues known to be important for enterobactin binding to FepA (16); however, the spacing between the conserved residues was distinct. In addition, the binding affinity of FetA for enterobactin was found to be much weaker (12) than that described for E. coli FepA (11). Thus, we propose that FetA has evolved as a gonococcal transporter capable of importing a broad spectrum of catecholate-type xenosiderophores, perhaps at the expense of high-affinity interactions with any single siderophore.
Salmochelin is known as a “stealth siderophore” and is derived by glucosylation of enterobactin via the products of the iroA locus (46). Virulent pathogens, including uropathogenic E. coli, Salmonella enterica, and Shigella dysenteriae, harbor the iroA locus, sometimes on pathogenicity islands (50). Modification of the enterobactin molecule by addition of two glucose molecules leads to increased hydrophilicity and to the ability of salmochelin to evade the host's innate immune response. In the human host, enterobactin is sequestered and made ineffectual by the innate immunity protein siderocalin (also known as lipocalin 2), whereas salmochelin is not (24, 28). Thus, salmochelin production by pathogens allows for efficient iron acquisition in the presence of siderocalin (56), which is found in lymphocytes. Salmochelin S4 is a cyclic form of the siderophore, and salmochelin S2 is the linear derivative of S4; both forms coordinate iron and can be employed as “stealth siderophores.” Gonococcal strain FA1090 did not utilize iron from salmochelin S4 but did obtain iron from S2. As microbial producers of enterobactin, DHBS and salmochelin inhabit the same niche as the gonococcus (21, 55), it seems likely that the ability to hijack these siderophores, in the presence of neutrophil-derived siderocalin, enhances the survival of N. gonorrhoeae in vivo.
Interestingly, our laboratory has observed differences in xenosiderophore utilization among gonococcal strains. Strain FA19 utilizes enterobactin, D1, and S2 in a TonB- and FetA-independent mechanism that requires expression of the FbpABC system (62). The mechanism by which these siderophores cross the outer membrane was not precisely defined, but we proposed that entry may be via nonspecific diffusion through abundant porin proteins (62). In the current study, we demonstrated that FA1090 utilizes D1 in a TonB- and FetA-independent mechanism. However, FA1090 utilizes enterobactin, D2, D3, and salmochelin S2 in a FetA- and TonB-dependent manner. Our hypothesis for the differences in xenosiderophore utilization phenotypes between strains is that there are two different pathways by which iron from xenosiderophores can be transported into the gonococcus. One pathway is TonB and FetA dependent, while the other pathway is TonB independent and requires the FbpABC system. In gonococcal strain FA1090, the fetA and tonB mutants were capable of significantly less growth with enterobactin, D2 D3, and S2 than the wild-type strain; however, both mutants exhibit xenosiderophore-dependent growth above background (dashed line in Fig. 6). Growth was diminished but not abolished in the tonB and fetA mutant strains; therefore, we hypothesize that the FbpABC-dependent, Ton-independent pathway is also employed by these mutants in the FA1090 background. In further support of this hypothesis, Carson et al. demonstrated that both fetA and fetB mutants of FA1090 exhibited a decrease in growth in the presence of enterobactin but that growth was not abolished (12). Similar results were also observed with the tonB mutant in the previous study (12). Thus, some iron was transported into the fet and tonB mutants in an energy- and fet operon-independent pathway. The difference in xenosiderophore-dependent growth between FA19 and FA1090 is likely due to differential use of these two distinct pathways. Gonococcal strain FA19 seems to be limited to use of the TonB-independent pathway that employs the FbpABC system for iron transit through the periplasm and cytoplasmic membrane.
In N. gonorrhoeae, the fetA gene is the only TonB-dependent transporter gene that is also carried with a periplasmic binding protein gene and a complete set of ABC transport genes (fetB to ng2088). The data presented in the current study suggest that the putative periplasmic binding protein and ABC transport system genes are cotranscribed with fetA but also independent of FetA and MpeR. Expression of the shorter transcript carrying fetB to ng2088 was detected only in gonococci grown under iron-depleted conditions. A Fur box upstream of fetB was identified by FURTA and EMSA (34), consistent with our finding of a separate iron-regulated transcript including the fetB to ng2088 genes. The FetA-independent but iron-regulated transcription of the ABC transport genes suggests that the ABC transport system could be utilized by other TonB-dependent transporters, in addition to FetA.
Enterobactin, D2, D3, and salmochelin S2 were acquired by gonococcal strain FA1090, which led us to consider the possibility that one or all of these xenosiderophores could serve as a coinducer of MpeR for the activation of fetA expression. However, contrary to our hypothesis, FetA expression was not altered by the presence of any of the siderophores tested, regardless of their iron status. We are currently entertaining three possible mechanisms to explain these results. First, MpeR may not require a coinducing molecule in order to activate FetA expression. Second, because MpeR is involved in modulating the expression of proteins involved in antimicrobial efflux, the coinducing agent for MpeR-dependent regulation could be related to efflux. In this context, since the Mtr system is necessary for efflux of host-derived antimicrobials, MpeR might sense an efflux substrate as a signal of location within the host. Third, other catechols might serve as coinducers with MpeR. Given the broad specificity of FetA, it is possible that an as-yet-unrecognized catecholate molecule could serve as an inducer and also provide iron in a FetA-dependent manner.
The mpeR gene has been identified only in the genomes of the pathogenic neisseriae and is absent from the genomes of commensal neisseriae (42, 60). The MpeR proteins are highly conserved, sharing 97 to 100% sequence identity among the pathogenic Neisseria species (data not shown). This conserved, pathogen-specific regulator controls the expression of FetA, which is a pan-Neisseria transporter (42). In Staphylococcus aureus, different community-acquired strains vary in their virulence. The difference among strains in pathogenic potential is linked to increased expression of core genome-carried virulence genes (40). It was hypothesized by Li et al. (40) that global regulators of virulence genes are responsible for variable virulence among strains. Similarly, commensal neisseriae have an extensive repertoire of virulence alleles that are also expressed by pathogenic neisseriae and other bacterial genera (42). Thus, differential regulation of these alleles, rather than their presence or absence, may contribute to increased virulence in the pathogenic neisseriae. Perhaps MpeR-dependent activation of fetA and other potential transporters enhances virulence in pathogenic neisseriae relative to the commensals.
In conclusion, in this study we demonstrated that MpeR, a pathogen-specific regulator in N. gonorrhoeae, enhances expression of the siderophore receptor FetA under iron-depleted conditions. A second, internal transcriptional start site was identified upstream of fetB, positioned near several potential −10 promoter elements and overlapped by the Fur binding site. Additional studies will be required, however, to fully define the mechanisms that control fetB transcription and determine whether other regulatory proteins are involved. We also determined that enterobactin, D2, D3, and salmochelin S2 were utilized in a FetA- and TonB-dependent mechanism in strain FA1090. Expression of MpeR enhanced the ability of strain FA1090 to utilize enterobactin and salmochelin, consistent with MpeR-dependent activation of FetA. None of the catecholates that supported growth in this study appeared to act as a coinducer for MpeR-dependent activation of fetA. To our knowledge, this study represents the first description of a pathogen-specific regulator in N. gonorrhoeae that activates expression of a pan-Neisseria TonB-dependent transporter. In addition, the current study is the first to link regulation of iron transport and antimicrobial efflux systems through the action of a pathogenic Neisseria-specific regulator.
ACKNOWLEDGMENTS
Funding for this work was provided by U.S. Public Health Service grants AI065555 and AI084400 to C.N.C. and R37 AI021150 to W.M.S. from the National Institute of Allergy and Infectious Diseases at the National Institutes of Health. W.M.S. was supported in part by a Senior Research Career Scientist Award from the Department of Veterans Affairs. Both C.N.C. and W.M.S. are supported by SE STI Center grant U19 AI31496 from the National Institute of Allergy and Infectious Diseases. Mass spectrometry was performed by Kristina Nelson at the VCU Mass Spectrometry Resource Center with funding from the VCU College of Humanities and Science.
We gratefully acknowledge Fred Sparling for providing the FetA-specific antibody and the fetA mutants. We also thank H. Strange for conducting some of the initial plate bioassays, V. Stringer for excellent technical assistance with the EMSA experiments, and V. Dhulipala for critical technical contributions to the primer extension experiments.
Footnotes
Published ahead of print on 26 September 2011.
REFERENCES
- 1. Ala'Aldeen D. A., Wall R. A., Borriello S. P. 1990. Immunogenicity and cross-reactivity of the 70-Kda iron-regulated protein of Neisseria meningitidis in man and animals. J. Med. Microbiol. 32: 275–281 [DOI] [PubMed] [Google Scholar]
- 2. Anderson J. E., Hobbs M. M., Biswas G. D., Sparling P. F. 2003. Opposing selective forces for expression of the gonococcal lactoferrin receptor. Mol. Microbiol. 48: 1325–1337 [DOI] [PubMed] [Google Scholar]
- 3. Archibald F. S., Simonson C., DeVoe I. W. 1981. Comparison of iron binding and uptake from FeCl3 and Fe-citrated by Neisseria meningitidis. Can. J. Microbiol. 27: 1066–1070 [DOI] [PubMed] [Google Scholar]
- 4. Bagg A., Neilands J. B. 1987. Ferric uptake regulation protein acts as a repressor, employing iron (II) as a cofactor to bind the operator of an iron transport operon in Escherichia coli. Biochemistry 26: 5471–5477 [DOI] [PubMed] [Google Scholar]
- 5. Beaumont F. C., Kang H. Y., Brickman T. J., Armstrong S. K. 1998. Identification and characterization of alcR, a gene encoding an AraC-like regulator of alcaligin siderophore biosynthesis and transport in Bordetella pertussis and Bordetella bronchiseptica. J. Bacteriol. 180: 862–870 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Beucher M., Sparling P. F. 1995. Cloning, sequencing, and characterization of the gene encoding FrpB, a major iron-regulated, outer membrane protein of Neisseria gonorrhoeae. J. Bacteriol. 177: 2041–2049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Biswas G. D., Sparling P. F. 1995. Characterization of lbpA, the structural gene for a lactoferrin receptor in Neisseria gonorrhoeae. Infect. Immun. 63: 2958–2967 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Black J. R., Dyer D. W., Thompson M. K., Sparling P. F. 1986. Human immune response to iron-repressible outer membrane proteins of Neisseria meningitidis. Infect. Immun. 54: 710–713 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Black W. J., Schwalbe R. S., Nachamkin I., Cannon J. G. 1984. Characterization of Neisseria gonorrhoeae protein II phase variation by use of monoclonal antibodies. Infect. Immun. 45: 453–457 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Briat J. F. 1992. Iron assimilation and storage in prokaryotes. J. Gen. Microbiol. 138: 2475–2483 [DOI] [PubMed] [Google Scholar]
- 11. Cao Z., Qi Z., Sprencel C., Newton S. M., Klebba P. E. 2000. Aromatic components of two ferric enterobactin binding sites in Escherichia coli FepA. Mol. Microbiol. 37: 1306–1317 [DOI] [PubMed] [Google Scholar]
- 12. Carson S. D., Klebba P. E., Newton S. M., Sparling P. F. 1999. Ferric enterobactin binding and utilization by Neisseria gonorrhoeae. J. Bacteriol. 181: 2895–2901 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Carson S. D., Stone B., Beucher M., Fu J., Sparling P. F. 2000. Phase variation of the gonococcal siderophore receptor FetA. Mol. Microbiol. 36: 585–593 [DOI] [PubMed] [Google Scholar]
- 14. CDC 2010. Sexually transmitted disease surveillance 2009 supplement, gonococcal isolate surveillance project (GISP) annual report 2009. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, Atlanta, GA: [Google Scholar]
- 15. CDC 2007. Update to CDC's sexually transmitted diseases treatment guidelines, 2006: fluoroquinolones no longer recommended for treatment of gonococcal infections. MMWR Morb. Mortal. Wkly. Rep. 56: 625–63617597693 [Google Scholar]
- 16. Chakraborty R., Lemke E. A., Cao Z., Klebba P. E., van der Helm D. 2003. Identification and mutational studies of conserved amino acids in the outer membrane receptor protein, FepA, which affect transport but not binding of ferric-enterobactin in Escherichia coli. Biometals 16: 507–518 [DOI] [PubMed] [Google Scholar]
- 17. Chen C. J., Sparling P. F., Lewis L. A., Dyer D. W., Elkins C. 1996. Identification and purification of a hemoglobin-binding outer membrane protein from Neisseria gonorrhoeae. Infect. Immun. 64: 5008–5014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Cohen M. S., et al. 1997. Reduction of concentration of HIV-1 in semen after treatment of urethritis: implications for prevention of sexual transmission of HIV-1. AIDSCAP Malawi Research Group. Lancet 349: 1868–1873 [DOI] [PubMed] [Google Scholar]
- 19. Cornelissen C. N., et al. 1992. Gonococcal transferrin-binding protein 1 is required for transferrin utilization and is homologous to TonB-dependent outer membrane receptors. J. Bacteriol. 174: 5788–5797 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Cornelissen C. N., et al. 1998. The transferrin receptor expressed by gonococcal strain FA1090 is required for the experimental infection of human male volunteers. Mol. Microbiol. 27: 611–616 [DOI] [PubMed] [Google Scholar]
- 21. El Aila N. A., et al. 2009. Identification and genotyping of bacteria from paired vaginal and rectal samples from pregnant women indicates similarity between vaginal and rectal microflora. BMC Infect. Dis. 9: 167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Escolar L., Perez-Martin J., de Lorenzo V. 1999. Opening the iron box: transcriptional metalloregulation by the Fur protein. J. Bacteriol. 181: 6223–6229 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Fetherston J. D., Bearden S. W., Perry R. D. 1996. YbtA, an AraC-type regulator of the Yersinia pestis pesticin/yersiniabactin receptor. Mol. Microbiol. 22: 315–325 [DOI] [PubMed] [Google Scholar]
- 24. Fischbach M. A., et al. 2006. The pathogen-associated iroA gene cluster mediates bacterial evasion of lipocalin 2. Proc. Natl. Acad. Sci. U. S. A. 103: 16502–16507 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Folster J. P., Shafer W. M. 2005. Regulation of mtrF expression in Neisseria gonorrhoeae and its role in high-level antimicrobial resistance. J. Bacteriol. 187: 3713–3720 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Gallegos M. T., Schleif R., Bairoch A., Hofmann K., Ramos J. L. 1997. Arac/XylS family of transcriptional regulators. Microbiol. Mol. Biol. Rev. 61: 393–410 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Garcia E. C., Brumbaugh A. R., Mobley H. L. 2011. Redundancy and specificity of Escherichia coli iron acquisition systems during urinary tract infection. Infect. Immun. 79: 1225–1235 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Goetz D. H., et al. 2002. The neutrophil lipocalin NGAL is a bacteriostatic agent that interferes with siderophore-mediated iron acquisition. Mol. Cell 10: 1033–1043 [DOI] [PubMed] [Google Scholar]
- 29. Hagen T. A., Cornelissen C. N. 2006. Neisseria gonorrhoeae requires expression of TonB and the putative transporter TdfF to replicate within cervical epithelial cells. Mol. Microbiol. 62: 1144–1157 [DOI] [PubMed] [Google Scholar]
- 30. Halliwell B. 1978. Superoxide-dependent formation of hydroxyl radicals in the presence of iron salts. Its role in degradation of hyaluronic acid by a superoxide-generating system. FEBS Lett. 96: 238–242 [DOI] [PubMed] [Google Scholar]
- 31. Hantke K. 1990. Dihydroxybenzoylserine—a siderophore for E. coli. FEMS Microbiol. Lett. 55: 5–8 [DOI] [PubMed] [Google Scholar]
- 32. Heinrichs D. E., Poole K. 1993. Cloning and sequence analysis of a gene (pchR) encoding an AraC family activator of pyochelin and ferripyochelin receptor synthesis in Pseudomonas aeruginosa. J. Bacteriol. 175: 5882–5889 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Ison C. A., Dillon J. A., Tapsall J. W. 1998. The epidemiology of global antibiotic resistance among Neisseria gonorrhoeae and Haemophilus ducreyi. Lancet 351 (Suppl. 3): 8–11 [DOI] [PubMed] [Google Scholar]
- 34. Jackson L. A., et al. 2010. Transcriptional and functional analysis of the Neisseria gonorrhoeae Fur regulon. J. Bacteriol. 192: 77–85 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Kellogg D. S., Jr., Peacock W. L., Jr., Deacon W. E., Brown L., Pirkle D. I. 1963. Neisseria gonorrhoeae. I. Virulence genetically linked to clonal variation. J. Bacteriol. 85: 1274–1279 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Laemmli U. K. 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685 [DOI] [PubMed] [Google Scholar]
- 37. Lee B. C., Schryvers A. B. 1988. Specificity of the lactoferrin and transferrin receptors in Neisseria gonorrhoeae. Mol. Microbiol. 2: 827–829 [DOI] [PubMed] [Google Scholar]
- 38. Lee E. H., Rouquette-Loughlin C., Folster J. P., Shafer W. M. 2003. FarR regulates the farAB-encoded efflux pump of Neisseria gonorrhoeae via an MtrR regulatory mechanism. J. Bacteriol. 185: 7145–7152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Lewis D. A. 2010. The gonococcus fights back: is this time a knock out? Sex. Transm. Infect. 86: 415–421 [DOI] [PubMed] [Google Scholar]
- 40. Li M., et al. 2009. Evolution of virulence in epidemic community-associated methicillin-resistant Staphylococcus aureus. Proc. Natl. Acad. Sci. U. S. A. 106: 5883–5888 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Livak K. J., Schmittgen T. D. 2001. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25: 402–408 [DOI] [PubMed] [Google Scholar]
- 42. Marri P. R., et al. 2010. Genome sequencing reveals widespread virulence gene exchange among human Neisseria species. PLoS One 5: e11835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. McAllister C. F., Stephens D. S. 1993. Analysis in Neisseria meningitidis and other Neisseria species of genes homologous to the FKBP immunophilin family. Mol. Microbiol. 10: 13–23 [DOI] [PubMed] [Google Scholar]
- 44. McClelland R. S., et al. 2001. Treatment of cervicitis is associated with decreased cervical shedding of HIV-1. AIDS 15: 105–110 [DOI] [PubMed] [Google Scholar]
- 45. McCormack W. M., Stumacher R. J., Johnson K., Donner A. 1977. Clinical spectrum of gonococcal infection in women. Lancet i: 1182– 1185 [DOI] [PubMed] [Google Scholar]
- 46. Muller S. I., Valdebenito M., Hantke K. 2009. Salmochelin, the long-overlooked catecholate siderophore of Salmonella. Biometals 22: 691–695 [DOI] [PubMed] [Google Scholar]
- 47. Munday P. E. 1997. Clinical aspects of pelvic inflammatory disease. Hum. Reprod. 12: 121–126 [PubMed] [Google Scholar]
- 48. O'Brien I. G., Cox G. B., Gibson F. 1971. Enterochelin hydrolysis and iron metabolism in Escherichia coli. Biochim. Biophys. Acta 237: 537–549 [DOI] [PubMed] [Google Scholar]
- 49. O'Brien I. G., Gibson F. 1970. The structure of enterochelin and related 2,3-dihydroxy-N-benzoylserine conjugates from Escherichia coli. Biochim. Biophys. Acta 215: 393–402 [DOI] [PubMed] [Google Scholar]
- 50. Payne S. M., Mey A. R. 2004. Pathogenic Escherichia coli, Shigella, and Salmonella p. 199–218 In Crosa A. R. M. J. H., Payne S. M. (ed.), Iron Transport in Bacteria ASM Press, Washington DC: [Google Scholar]
- 51. Pettersson A., et al. 1990. Monoclonal antibodies against the 70-kilodalton iron-regulated protein of Neisseria meningitidis are bactericidal and strain specific. Infect. Immun. 58: 3036–3041 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Pollack J., Ames B., Neilands J. 1970. Iron transport in Salmonella typhimurium: mutants blocked in the biosynthesis of enterobactin. J. Bacteriol. 104: 635–639 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Pradel E., Guiso N., Locht C. 1998. Identification of AlcR, an AraC-type regulator of alcaligin siderophore synthesis in Bordetella bronchiseptica and Bordetella pertussis. J. Bacteriol. 180: 871–880 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Prentki P., Krisch H. M. 1984. In vitro insertional mutagenesis with a selectable DNA fragment. Gene 29: 303–313 [DOI] [PubMed] [Google Scholar]
- 55. Price L. B., et al. 2010. The effects of circumcision on the penis microbiome. PLoS One 5: e8422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Raffatellu M., et al. 2009. Lipocalin-2 resistance confers an advantage to Salmonella enterica serotype Typhimurium for growth and survival in the inflamed intestine. Cell Host Microbe 5: 476–486 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Rouquette-Loughlin C. E., Balthazar J. T., Hill S. A., Shafer W. M. 2004. Modulation of the mtrCDE-encoded efflux pump gene complex of Neisseria meningitidis due to a Correia element insertion sequence. Mol. Microbiol. 54: 731–741 [DOI] [PubMed] [Google Scholar]
- 58. Sebastian S., Agarwal S., Murphy J. R., Genco C. A. 2002. The gonococcal fur regulon: identification of additional genes involved in major catabolic, recombination, and secretory pathways. J. Bacteriol. 184: 3965–3974 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Skaar E. P., Lazio M. P., Seifert H. S. 2002. Roles of the recJ and recN genes in homologous recombination and DNA repair pathways of Neisseria gonorrhoeae. J. Bacteriol. 184: 919–927 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Snyder L. A., Saunders N. J. 2006. The majority of genes in the pathogenic Neisseria species are present in non-pathogenic Neisseria lactamica, including those designated as ‘virulence genes’. BMC Genomics 7: 128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Soper D. E. 1991. Diagnosis and laparoscopic grading of acute salpingitis. Am. J. Obstet. Gynecol. 164: 1370–1376 [DOI] [PubMed] [Google Scholar]
- 62. Strange H. R., Zola T. A., Cornelissen C. N. 2011. The fbpABC operon is required for Ton-independent utilization of xenosiderophores by Neisseria gonorrhoeae strain FA19. Infect. Immun. 79: 268–278 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Thompson E. A., Feavers I. M., Maiden M. C. 2003. Antigenic diversity of meningococcal enterobactin receptor FetA, a vaccine component. Microbiology 149: 1849–1858 [DOI] [PubMed] [Google Scholar]
- 64. Towbin H., Staehelin T., Gordon J. 1979. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc. Natl. Acad. Sci. U. S. A. 76: 4350–4354 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. West S. E., Sparling P. F. 1987. Aerobactin utilization by Neisseria gonorrhoeae and cloning of a genomic DNA fragment that complements Escherichia coli fhuB mutations. J. Bacteriol. 169: 3414–3421 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. West S. E., Sparling P. F. 1985. Response of Neisseria gonorrhoeae to iron limitation: alterations in expression of membrane proteins without apparent siderophore production. Infect. Immun. 47: 388–394 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Zhu M., Valdebenito M., Winkelmann G., Hantke K. 2005. Functions of the siderophore esterases IroD and IroE in iron-salmochelin utilization. Microbiology 151: 2363–2372 [DOI] [PubMed] [Google Scholar]