Abstract
This study is aimed to assess the long-term health-related quality of life (HRQL) of patients after esophagectomy for esophageal cancer in comparison with es-tablished norms, and to evaluate changes in HRQL during the different stages of follow-up after esophageal resection. A systematic review was performed by searching medical databases (Medline, Embase and the Cochrane Library) for potentially relevant studies that appeared between January 1975 and March 2011. Studies were included if they addressed the question of HRQL after esophageal resection for esophageal cancer. Two researchers independently performed the study selection, data extraction and analysis processes. Twenty-one observational studies were included with a total of 1282 (12-355) patients. Five studies were performed with short form-36 (SF-36) and 16 with European Organization for Research and Treatment of Cancer (EORTC) QLQ C30 (14 of them also utilized the disease-specific OES18 or its previous version OES24). The analysis of long-term generic HRQL with SF-36 showed pooled scores for physical, role and social function after esophagectomy similar to United States norms, but lower pooled scores for physical function, vitality and general health perception. The analysis of HRQL conducted using the Global EORTC C30 global scale during a 6-mo follow-up showed that global scale and physical function were better at the baseline. The symptom scales indicated worsened fatigue, dyspnea and diarrhea 6 mo after esophagectomy. In contrast, however, emotional function had significantly improved after 6 mo. In conclusion, short- and long-term HRQL is deeply affected after esophagectomy for cancer. The impairment of physical function may be a long-term consequence of esophagectomy involving either the respiratory system or the alimentary tract. The short- and long-term improvement in the emotional function of patients who have undergone successful operations may be attributed to the impression that they have survived a near-death experience.
Keywords: Health-related quality of life, Esophageal cancer, Esophagectomy, Short form 36, European Organization for Research and Treatment of Cancer QLQ C30, European Organization for Research and Treatment of Cancer OES18
INTRODUCTION
Esophageal cancer is an increasingly common cancer with a poor prognosis. During recent decades, the incidence of esophageal cancer has risen steadily, and it is now the fastest rising solid tumor in most Western countries. Moreover, moderate to high incidence rates have been reported in other areas, including parts of China, Central Asia, South and East Africa, South America, Northern France, and the United States among African-Americans[1]. Despite recent improvements in diagnosis, surgical treatment and (neo)-adjuvant therapy, the prognosis of patients with esophageal cancer remains poor, with overall 5-year survival rates of only 5%-15%[1,2]. Esophagectomy is the standard treatment for those patients who present with resectable esophageal cancer[3-5], but it offers a limited (25%-35%) chance of cure[5,6] and is associated with a considerable risk of serious complications[4,5,7]. Therefore, the use of chemotherapy or radiotherapy in combination with surgery has been tested. Nevertheless, meta-analyses of randomized trials of neoadjuvant chemotherapy and chemoradiation followed by surgery and surgery alone for patients with esophageal carcinoma showed only minor survival advantages[8,9]. Only patients with a complete pathologic response to neoadjuvant therapy enjoy a significantly better chance of survival, whereas non-responders have a worse prognosis than patients undergoing surgery alone[10,11].
For a long time, morbidity and mortality represented the main (and often the only) outcome measure that could be used to evaluate esophagectomy for esophageal cancer. The morbidity and mortality rates associated with the procedure and the poor patient survival rate left almost no space for further consideration. However, in recent years, along with the increase in the success of the therapy, health-related quality of life (HRQL) has generally become accepted as an important outcome parameter, along with long-term survival, mortality, and complication rates. In fact, knowledge of risk factors for poor postoperative HRQL may be relevant to clinical decision making. Moreover, these findings may be used to inform patients of the long-term consequences of surgery. On this basis, the aim of this systematic review was to analyze quality of life after curative surgery for esophageal cancer.
INCLUSION AND EXCLUSION CRITERIA
Since we expected to find only observational studies, the checklist proposed by Meta-analysis of Observational Studies in Epidemiology group 38 was used as a guideline to perform this systematic review[12]. We defined observational studies as reports that used data from existing databases, cross-sectional studies, case series, case-control studies, or studies with a historical control or a cohort design.
Studies were eligible for inclusion if they reported on a series of patients who underwent esophagectomy because of esophageal cancer and if post-operative quality of life was described and analyzed in the “material and methods” and “results” sections. Studies reporting on a mix of esophageal adenocarcinoma and squamous cell cancer patients were included. In contrast, those reporting on malignancies other than esophageal adenocarcinoma or squamous cell cancer were excluded. All studies eligible for inclusion in this systematic review also had to present detailed information on the methods used to assess quality of life and on when the questionnaire was administered. Studies that analyzed HRQL using questionnaires other than short form-36 (SF-36), European Organization for Research and Treatment of Cancer (EORTC) QLQ C30 and OES18/24 and those that only presented their results graphically were excluded. When studies were discovered to report (partially) similar patient data, only the most recent and complete data sets were considered.
SEARCH STRATEGY
Four medical databases were used in this research: Med-line (January 1978 to March 2011), the Cochrane Database of Systematic Reviews, the Cochrane Central Register of Controlled Trials and Embase. These databases were searched with the help of a clinical librarian. The keywords and medical subject headings used were “esophageal cancer”, “esophagectomy” and “quality of life”, as indicated in Figure 1. Only clinical studies written in English were selected. A manual cross-reference search of the eligible papers was performed to identify additional relevant articles. Based on the initial search results, two researchers (Scarpa M and Valente S) independently selected the studies that matched the inclusion criteria. Data quoted as unpublished and data from abstracts were not used. Any disagreements between the two researchers regarding which studies should be included were resolved through discussion.
Figure 1.
Key words. HRQL: Health related quality of life; QoL: Quality of life.
DATA EXTRACTION
Data were extracted only from original articles using a preformatted sheet with a set of pre-defined parameters: demographic data, histologic type, cancer stage, cancer site, type of surgery (two-way or three-way esophagectomy), type of reconstruction (esophagogastroplasty or esophagocoloplasty), neoadjuvant or adjuvant therapy, timing of follow-up and HRQL data gathering, type of questionnaire used, item and total results.
OUTCOME MEASURE: QUALITY OF LIFE INSTRUMENTS
Studies were included if at least one of the following validated quality of life instruments was used: the EORTC-QLQ-C30, the EORTC-QLQ-OES18 or 24, or the SF-36. A summary of these questionnaires appears below.
The EORTC-QLQ-C30 questionnaire was developed by the Quality of Life division of EORTC. This 30-item questionnaire explores the generic quality of life of patients affected by oncologic diseases. It is a self-report instrument that includes five functional scales (physical, role, emotional, social and cognitive), three symptom scales (fatigue, nausea and vomiting, and pain), a global health status scale and six single items (dyspnea, insomnia, appetite loss, constipation, diarrhea, and financial difficulties)[13]. The EORTC-QLQ-OES-18 and 24 are two extra modules that are used specifically for esophageal cancer. These questionnaires consist of 18 questions (or 24 in the previous version) assessing dysphagia, deglutition, abdominal/gastrointestinal symptoms, eating difficulties, pain, and emotional problems related to esophageal cancer and to the side effects of chemotherapy/radiotherapy[14]. The SF-36 consists of 36 items within 8 dimensions: psychological functioning, role limitations due to physical problems, pain, general health perceptions, energy/vitality, social functioning, and role limitations due to emotional problems and mental health[15].
STATISTICS
A clinical statistician was consulted to assess the accuracy of our analysis. The Review Manager 4.2 software (The Cochrane Collaboration, Copenhagen: Nordic Cochrane Centre, 2003) was used to process the data and conduct the analysis. For studies presenting HRQL results obtained from patients who had undergone the same treatment regimen and been presented with the same questionnaire at the same point in time with respect to their surgery, a meta-analysis of the HRQL scores was attempted. The results were presented as weighted mean differences [95% confidence interval (CI)]. Based on previous research, a difference of at least 10 mean score points between time points or comparison groups was considered clinically relevant, whereas a difference of 5-10 was considered weak[16]. P < 0.05 was considered to indicate that the results were significant.
STUDY SELECTION
The studies selected are shown in Figure 2. The initial search yielded 380 articles, of which 349 did not meet the criteria for inclusion. Based on their titles, 286 papers were excluded because they clearly covered a variety of unsuitable topics. Forty-four articles were then excluded on the basis of their abstracts; these were case reports and review articles without original data or articles on forms of neoplasm other than esophageal carcinoma. Of the 50 candidate papers, an additional 29 were then excluded, of which 10 were not focused on quality of life after esophagectomy for esophageal carcinoma[14,17-25], 10 presented their results only in a graphical format[26-35], 7 used quality of life questionnaires other than the SF36, QLQ C30 or OES24/18[36-42] and 2 reported data from the same dataset as other studies[43,44]. Although they reported the same dataset, three studies from the Karolinska Institute of Stockholm were included in the review because they analyzed different aspects of HRQL after esophagectomy[45,46]. However, the patients included in these studies were counted only once in all totals.
Figure 2.
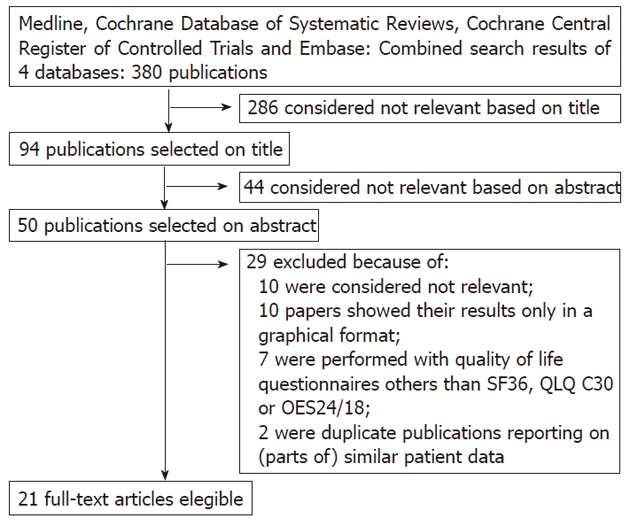
Study selection. SF-36: Short form-36.
The 21 studies included in the analysis were all published between 1995 and 2010 and are listed according their aim and then chronologically in Tables 1 and 2. Five studies were performed using SF36 and 16 using EORTC QLQ C30 (14 of them also utilized the disease-specific OES18 or the previous version, the OES24). Nine studies were observational cross-sectional studies and twelve were prospective ones. Patients were enrolled consecutively in 17 of them. Generic and disease-specific questionnaires were both used in 18 studies, and the HRQL was the primary focus for 15 of them. Five were population-based studies.
Table 1.
Studies characteristics: Aims of the studies and timing of health-related quality of life recording
| Sudy | Study aim | Timing post op HRQL measures |
| McLarty et al[54] | Analysis of HRQL in long term survivors after surgery alone | A single assessment > 60 mo |
| De Boer et al[53] | Analysis of HRQL in long term survivors after transhiatal esophagectomy | A single assessment 3.5 (2.1-5.4) yr |
| Headrick et al[49] | Analysis of HRQL in long term survivors after esophagectomy for HGD or adenocarcinoma | A single assessment 5.3 (0.5-9) yr |
| Cense et al[52] | Analysis of HRQL in long term survivors after esophagocolonplasty | A single assessment 35 (7-97) mo |
| Moraca et al[50] | Analysis of HRQL in long term survivors after esophagectomy for HGD or Tis | A single assessment 4.9 (0.5-12) yr |
| Reynolds et al[55] | Comparison between HRQL after neoadjuvant CT-RT+ surgery and after surgery alone | Baseline, after CT-RT, 3, 6, 9, 12 mo po |
| Avery et al[56] | Comparison between HRQL after neoadjuvant CT-RT+ surgery and after definitive CT-RT | 1, 5, 3, 6, 9 mo |
| van Meerten et al[57] | Analysis of HRQL after neoadjuvant CT-RT + surgery | Baseline, after CT-RT, 3, 6, 9, 12 mo po |
| Wang et al[59] | Comparison between open surgery and minimally invasive esophagectomy | 2, 4, 12, 24 wk |
| Parameswaran et al[58] | Analysis of HRQL after minimally invasive esophagectomy | 6, 12 mo |
| Viklund et al[45] | Analysis of HRQL predictors after esophagectomy for cancer (type of recstruction) | A single assessment 6 mo |
| Rutegard et al[46] | Analysis of HRQL predictors after esophagectomy for cancer (type of recstruction) | A single assessment 6 mo |
| Rutegard et al[47] | Analysis of HRQL predictors after esophagectomy for cancer (type of recstruction) | A single assessment 6 mo |
| Olsen et al[60] | Analysis of HRQL in long term survivors (surgery alone or neoadjuvant CT-RT + surgery) | A single assessmentat 24 mo po |
| Lagergren et al[61] | Analysis of HRQL in long term survivors (surgery alone or neoadjuvant CT-RT + surgery) | Baseline and 36 mo |
| Djarv et al[62] | Analysis of HRQL in long term survivors (surgery alone or neoadjuvant CT-RT + surgery) | 6, 36 mo |
| Courrech Staal et al[63] | Analysis of HRQL in long term survivors (surgery alone or neoadjuvant CT-RT + surgery) | A single assessment at 54 (16–162) mo |
| Blazeby et al[64] | Comparison between HRQL after surgery alone and after palliative RT | A single assessment 16 (10-24) wk |
| Ariga et al[65] | Comparison between surgery alone and definitive CT-RT + salvage surgery | A single assessment 24 mo |
| Schneider et al[48] | Comparison between HRQL after emergency and elective esophagectomy | 1 wk and 9 mo |
| Rosmolen et al[51] | Comparison between HRQL after endoscopical ablation and esophagectomy for early Barrett’s neoplasms | A single assessment at 24 (17–35) mo |
The 21 studies included in the analysis are listed according their aim and then chronologically. HRQL: Health-related quality of life; HGD: High grade dysplasia; CT-RT: Chemotherapy-radiotherapy; op: Operative; po: Post-operative.
Table 2.
Studies characteristics: Studies setting and feature
| Study | Year | Country | Center | Prospective | Consecutive | HRQL as primaryendpoint | PreoperativeHRQLassessment | SF36 | OES18 /OES24 | QLQ C30 | Populationbased study |
| McLarty et al[54] | 1997 | United States | Mayo Clinic, Rochester MI | No | Yes | No | No | Yes | No | No | No |
| De Boer et al[53] | 2000 | Netherlands | Academic Medical Centre, Amsterdam | No | No | Yes | No | Yes | No | No | No |
| Headrick et al[49] | 2002 | United States | Mayo Clinic, Rochester MI | No | Yes | No | No | Yes | No | No | No |
| Cense et al[52] | 2004 | Netherlands | Academic Medical Centre, Amsterdam | No | No | Yes | No | Yes | No | No | No |
| Moraca et al[50] | 2006 | United States | Virginia Mason Medical Centre, Seattle | No | Yes | No | No | Yes | No | No | No |
| Reynolds et al[55] | 2006 | Ireland | St James's Hospital, Dublin | Yes | Yes | Yes | Yes | No | No | Yes | No |
| Avery et al[56] | 2007 | United Kingdom | University of Bristol, Bristol | Yes | Yes | Yes | Yes | No | Yes | Yes | No |
| van Meerten et al[57] | 2008 | Netherlands | Erasmus University, Rotterdam | Yes | Yes | Yes | Yes | No | Yes | Yes | No |
| Wang et al[59] | 2009 | China | Fudan University, Shanghai | Yes | Yes | Yes | Yes | No | Yes | Yes | No |
| Parameswaran et al[58] | 2010 | United Kingdom | Royal Devon and Exeter NHS Trust, Exeter | Yes | Yes | Yes | Yes | No | Yes | Yes | No |
| Viklund et al[45] | 2005 | Sweden | Karolinska Institute, Stockholm | Yes | Yes | Yes | No | No | Yes | Yes | Yes |
| Rutegard et al[46] | 2008 | Sweden | Karolinska Institute, Stockholm | Yes | No | Yes | No | No | Yes | Yes | Yes |
| Rutegard et al[47] | 2008 | Sweden | Karolinska Institute, Stockholm | Yes | No | Yes | No | No | Yes | Yes | Yes |
| Olsen et al[60] | 2005 | Sweden | Sahlgrenska University Hospital, Goteborg | No | Yes | No | No | No | Yes | Yes | No |
| Lagergren et al[61] | 2007 | United Kingdom | University of Bristol, Bristol | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes |
| Djarv et al[62] | 2008 | Sweden | Karolinska Institute, Stockholm | Yes | Yes | Yes | No | No | Yes | Yes | Yes |
| Courrech Staal et al[63] | 2010 | Netherlands | Netherlands Cancer Institute, Amsterdam | Yes | Yes | Yes | No | No | Yes | Yes | No |
| Blazeby et al[64] | 1995 | United Kingdom | University of Bristol, Bristol | No | Yes | Yes | No | No | No | Yes | No |
| Ariga et al[65] | 2009 | Japan | University of Yamaga, Yamaga | Yes | Yes | No | No | No | Yes | Yes | No |
| Schneider et al[48] | 2010 | Germany | University of Heidelberg, Heidelberg | Yes | Yes | Yes | Yes | No | Yes | Yes | No |
| Rosmolen et al[51] | 2010 | Netherlands | Academic Medical Center, Amsterdam | No | Yes | Yes | No | Yes | Yes | Yes | No |
The 21 studies included in the analysis are listed according their aim and then chronologically. HRQL: Health-related quality of life; SF36: Short form-36.
Study characteristics
There were 21 observational studies analyzed, and these studies included data for a total of 1282 patients. The number of patients ranged from 12 to 355 per study. The follow-up duration after esophagectomy, as reported in the articles, was between 4 and 63 mo. The range of mean ages reported by the different papers was 59 to 69 years. The indication for surgery was esophageal adenocarcinoma in 835 patients and squamous cell carcinoma in 395. However, in the series by Schneider et al[48], 5 out of 17 patients were operated on for esophageal perforation. Hendrick et al[49], Moraca et al[50] and Rosmolen et al[51] included 35, 24 and 7 patients, respectively, who underwent esophagectomy because of high-grade dysplasia. The characteristics of the patients included in each study are described in Table 3.
Table 3.
Patient characteristics
| Study | Patients | Median/1mean age (range) | Recruitment period | Adenocarcinoma | Squamous cell carcinoma | Tis/ HGD/ pCR | Stage I | Stage II | Stage III | Stage IV | Lower esophagus or cardias | Medium or upper esophagus | Mean/1median follow up (mo) | Esophago-gastroplasty | Esophago-colonplasty | Esophago-jejeunoplasty | Neoadjuvant treatment | Adjuvant RT/CT/CT+RT |
| McLarty et al[54] | 107 | 62 (30-81) | 1972-1990 | 72 | 28 | 0 | 34 | 73 | 0 | 62 | 45 | > 60 | 99 | 3 | 4 | 0 | 9 | |
| De Boer et al[53] | 35 | 66.41 (42-87) | 1993-1996 | 27 | 6 | 2 | 14 | 1 | 18 | 32 | 3 | > 24 | 35 | 0 | 0 | |||
| Headrick et al[49] | 54 | 64 (36-83) | 1991-1997 | 53 | 1 | 5/35 | 7 | 2 | 5 | 54 | 0 | 63.61 (6-108) | ||||||
| Cense et al[52] | 14 | 69.9 (51-81) ECP, 66.4 (42-87) EGP | 1993-2002 | 6 | 7 | 1 | 8 | 3 | 2 | 10 | 3 | 35 (7-97) | 0 | 14 | ||||
| Moraca et al[50] | 36 | 661 (43-88) | 1991-2003 | 35 | 0 | 12/24 | 12 | 58.8 (6-144) | 36 | 0 | 0 | 0 | 0 | |||||
| Subtotal | 246 (14-107) | 193 | 42 | 3 | 75 | 79 | 25 | 0 | 158 | 51 | 0 | 170 | 17 | 4 | 0 | 9 | ||
| Reynolds et al[55] | 107 | 61 (29-79) | 1999-2004 | 34 | 68 | 25 | 63 | 83 | 3 | 3 | 87 | 87 | ||||||
| Avery et al[56] | 94 | 62.41 | 2000-2004 | 64 | 17 | 14 | 67 | 1.5 | 69 | 69 | ||||||||
| van Meerten et al[57] | 54 | 59 (40-75) | 2001-2004 | 41 | 12 | 13 | 12 | 6 | 16 | 5 | 49 | 5 | 3 | 54 | 54 | |||
| Subtotal | 255 (54-107) | 239 | 97 | 13 | 37 | 83 | 166 | 8 | 49 | 5 | 7.5 | 0 | 0 | 0 | 210 | 210 | ||
| Wang et al[59] | 56 | 60.7 ± 9.3 VATS 58.2 ± 11.5 open | 2007-2008 | 3 | 52 | 11 | 37 | 8 | 10 | 46 | 6 | 56 | 0 | |||||
| Parameswaran et al[58] | 62 | 67 (49-80) | 2005-2007 | 57 | 5 | 3 | 7 | 21 | 27 | 4 | 12 ± 0.5 | 62 | 48 | |||||
| Subtotal | 118 (56-62) | 60 | 57 | 3 | 18 | 58 | 35 | 4 | 10 | 46 | 6 | 118 | 0 | 0 | 48 | 0 | ||
| Viklund et al[45] | 64 (34-84)1 | 2001-2003 | 6 | 16 | ||||||||||||||
| Rutegard et al[46] | 91 pts < 60, 127 pts 60-70, 137 pts >70 | 2001-2005 | 6 | 37 | ||||||||||||||
| Rutegard et al[47] | 355 | 91 pts < 60, 127 pts 60-70, 137 pts > 70 | 2001-2005 | 271 | 84 | 81 | 120 | 133 | 18 | 302 | 53 | 6 | 37 | |||||
| Subtotal | 355 | |||||||||||||||||
| Olsen et al[60] | 18 | 62.5 (20-76) | 1997-2001 | 3 | 5 | 1 | 9 | 24 | 13 | 4 | 1 | 8 | 8 | |||||
| Lagergren et al[61] | 47 | 63 (44-79) | 2000-2003 | 35 | 9 | 8 | 10 | 23 | 6 | 23 | 24 | > 36 | 29 | |||||
| Djarv et al[62] | 87 | 27 pts < 60, 28 pts 60-70, 32 pts > 70 | 2001-2007 | 68 | 19 | 34 | 36 | 13 | 3 | 75 | 12 | > 36 | 69 | 2 | 16 | |||
| Courrech Staal et al[63] | 36 | 2009 | 22 | 14 | 0 | 4 | 12 | 12 | 8 | 28 | 8 | 54 (range, 16–162) | 21 | |||||
| Subtotal | 188 (18-87) | 125 | 42 | 8 | 53 | 72 | 40 | 11 | 126 | 44 | 82 | 6 | 17 | 58 | 8 | |||
| Blazeby et al[64] | 33 | 64 (62-76) | 1993-1994 | 19 | 14 | 4 (2.5-6) | 0 | |||||||||||
| Ariga et al[65] | 48 | 65.5 (46-78) | 2001-2004 | 0 | 48 | 10 | 15 | 23 | 20 | 28 | 41.2 (95% CI: 35.1-47.2) | |||||||
| Schneider et al[48] | 12 | 59 | 2001-2005 | 1 | 11 | > 9 | 10 | 2 | ||||||||||
| Rosmolen et al[51] | 27 | 63.0 (9.5) | 2001-2005 | 27 | 0 | 2 | 19 | 6 | 24 (IQR 17 – 35) | 23 | 4 | 0 | 0 | |||||
| Subtotal | 120 (12-48) | 47 | 73 | 2 | 29 | 21 | 23 | 0 | 20 | 28 | 33 | 6 | 0 | 0 | 0 | |||
The 21 studies included in the analysis are listed according their aim and then chronologically. pts: Patients; ECP: Esophagocolonplasty; EGP: Esophagogastroplasty; VATS: Video assisted thoracoscopy; CI: Confidence interval; HGD: High grade dysplasia; pCR: Pathological complete response; RT: Radiotherapy; CT: Chemotherapy; IQR: Interquartile range.
Long term generic HRQL after esophagectomy vs healthy subjects
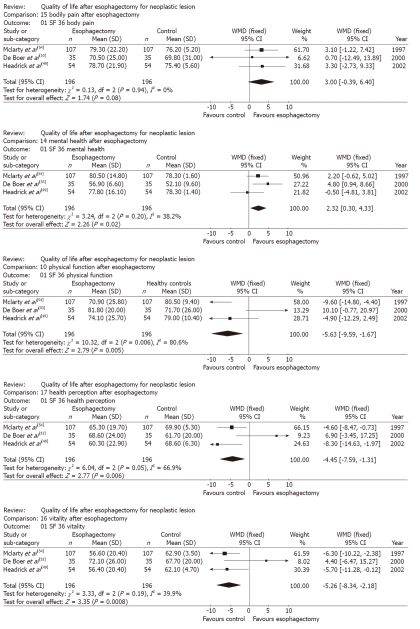
Five studies analyzed the long-term generic HRQL of 246 patients using the SF-36 questionnaire (median follow-up range: 36-64 mo)[49,50,52-54]. The studies by Moraca et al[50] and Cense et al[52] were not used for the meta-analysis because the SF-36 scores in the first were not reported in a standard, comparable manner and because all patients reported on in the second underwent esophagocoloplasty. The studies by Hendrick et al[49], De Boer et al[53] and McLarty et al[54] were sufficiently homogenous, and thus, a meta-analysis of their results was attempted. The pooled scores for physical function, physical role, and social function after esophagectomy were similar to sex- and age-matched United States norms, whereas the pooled scores for physical function, vitality and general health perception were lower than the relevant norms (P = 0.005, P < 0.001 and P = 0.006, respectively). In contrast, scores for bodily pain and mental health in long-term survivors after esophagectomy were higher than the relevant norms (P = 0.08 and P = 0.02, respectively). The significant weighted mean differences that emerged based on the comparison between the long-term generic HRQL of patients who had undergone esophagectomy and that of healthy subjects are shown in Figure 3.
Figure 3.
Long term generic health-related quality of life in patients after esophagectomy for esophageal cancer. WMD: Weighted mean difference; SF: Short form.
Generic and disease specific HRQL after neoadjuvant therapy and esophagectomy
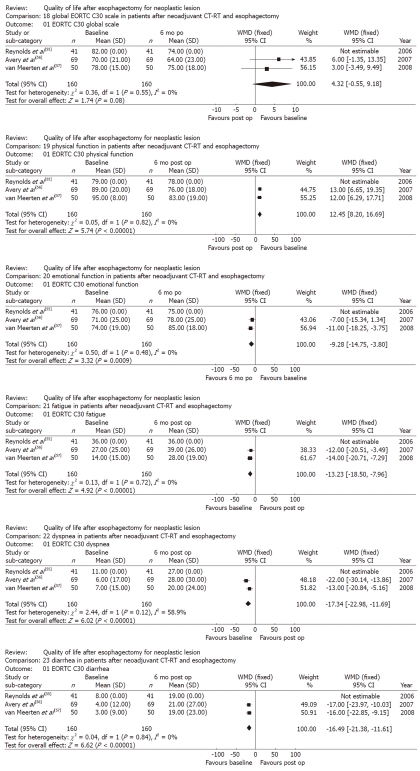
Three studies analyzed generic and disease-specific HRQL for 255 patients using the QLC-30 and OES-18 questionnaires at different stages in the follow-up timeline (baseline, 3, 6, 9 and 12 mo) after neoadjuvant therapy and esophagectomy[55-57]. Reynolds et al[55] and Avery et al[56] compared the HRQL of patients who had undergone neoadjuvant therapy with that of patients who had undergone esophagectomy alone. Based on these studies, data for the patients who had undergone neoadjuvant therapy and esophagectomy were identified. Data from the study by Reynolds et al[55] were uploaded for meta-analysis, but could not be used because the standard de-viation values were missing. The baseline scores were compared to scores obtained after a 6-mo follow-up. The EORTC- QLQ-C30 global scale results tended to be better at the baseline than at the 6-mo follow-up (P = 0.08), and physical function was also better at the baseline (P < 0.001). Likewise, the symptom scales showed worsened fatigue, dyspnea and diarrhea 6 mo after the procedure (P < 0.001, P < 0.001 and P < 0.001, respectively). In contrast, emotional function had significantly improved after 6 mo (P < 0.001). The significant weighted mean difference results associated with the comparison between the baseline HRQL figures and the figures achieved 6 mo after neoadjuvant therapy and esophagectomy are shown in Figure 4.
Figure 4.
Generic and disease specific health-related quality of life in patients after neoadjuvant chemotherapy-radiotherapy and esophagectomy for esophageal cancer. WMD: Weighted mean difference; CT-RT: Chemotherapy-radiotherapy; EORTC: European Organization for Research and Treatment of Cancer; op: Operative; po: Post-operative.
Generic and disease specific HRQL after minimally invasive esophagectomy
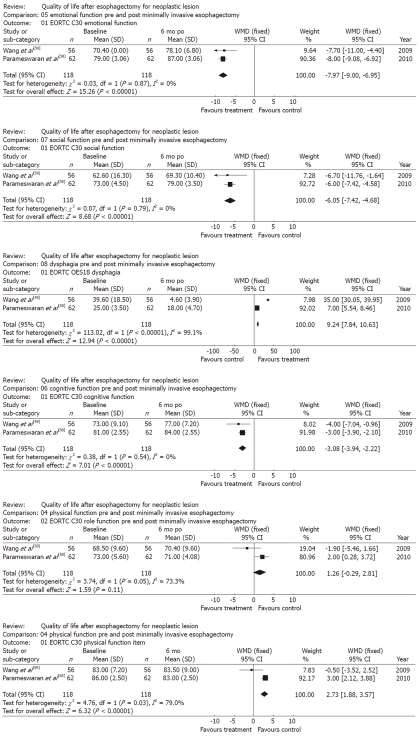
Two studies prospectively analyzed the generic and disease-specific HRQL of 255 patients using the EORTC-QLQ-C-30 and OES-18 questionnaires at different points on the follow-up timeline (baseline, 3, 6, 9 and 12 mo) after minimally invasive esophagectomy[58,59]. The surgical techniques used in the two studies were similar and included thoracoscopic esophageal mobilization and mediastinal lymphadenectomy, followed by laparoscopic gastric mobilization and resection, and abdominal lymphadenectomy. Wang et al[59] compared the HRQL results achieved after minimally invasive esophagectomy with those achieved after open esophagectomy, and the study by Parameswaran et al[58] was an uncontrolled prospective study. Therefore, the baseline scores were compared to scores obtained during a 6-mo follow-up. Social function, cognitive function, emotional function and dysphagia proved to be significantly improved during the 6-mo follow-up (P < 0.001, P < 0.001, P < 0.001 and P < 0.001, respectively). In contrast, physical function was better at the baseline (P < 0.001), and role function tended to appear worse at the 6-mo follow-up point. The significant weighted mean difference results associated with the comparison between the baseline HRQL figures and the figures from 6 mo after neoadjuvant therapy and esophagectomy are shown in Figure 5.
Figure 5.
Generic and disease specific health-related quality of life after minimally invasive surgery for esophageal cancer. WMD: Weighted mean difference; EORTC: European Organization for Research and Treatment of Cancer; po: Post-operative.
Predictors of generic and disease specific HRQL after esophagectomy
Three articles from the Karolinska Institute, Stockholm specifically investigated predictors of generic and disease-specific HRQL after esophagectomy for cancer by analyzing the data from the Swedish Esophageal and Cardia Cancer register, which had conducted a nationwide, prospective, population-based study of how esophageal surgery-related factors had influenced quality of life 6 mo after surgery[45-47]. Although they reported information from the same dataset, these studies were all included in the review because they analyzed different aspects of HRQL after esophagectomy. The first study, by Viklund et al[45], included 100 patients and indicated that surgery-related complications were the main predictor of reduced global quality of life 6 mo after surgery. Except for anastomotic strictures, each of the predefined complications (e.g., anastomotic leakage, infections, cardiopulmonary complications, and operative technical complications) contributed to a decrease in quality-of-life scores. Rutegard et al[46], using a larger study population, showed that extensive surgery (characterized as using the transthoracic approach, extensive lymphadenectomy, and wider resection margins and as being of a longer duration) was not associated with worse HRQL measures than less extensive operations. Moreover, they observed that the severity of dysphagia was similar in patients who had handsewn and stapled anastomoses. Technical surgical complications were confirmed to have a deleterious effect on several aspects of HRQL. Finally, the same group concluded that no clinically relevant differences in terms of generic and disease-specific HRQL were found to be correlated with differences in the volume of surgeries conducted at hospitals (low volume: 0-9 operations/year; high volume: > 9 operations/year) or by particular surgeons (low volume: 0-6 operations/year; high-volume: > 6 operations/year)[47].
Long-term generic and disease-specific HRQL after esophagectomy
Data from the articles that analyzed long-term generic and disease-specific HRQL after esophagectomy were collected, but their clinical heterogeneity was so high that it was impossible to pool them. Four studies analyzed the generic HRQL of 152 (18-87) patients using the QLC-30 questionnaire and evaluated disease-specific HRQL using the OES-18 questionnaire in conducting long-term follow-up[60-63]. Fagevik et al[60] evaluated 18 patients 2 years after thoraco-abdominal esophageal resection and observed that, 2 years after surgery, respiratory function was significantly lower than it was prior to surgery, as was physical performance. In contrast, HQRL was comparable to age- and sex-matched population norms for most other functions. After 2 years, diarrhea, dyspnea, appetite loss and fatigue were still clinically significant. Similarly, Lagergren et al[61] analyzed 47 patients who had survived for at least 3 years after esophagectomy for a malignant disease. In these long-term survivors, most HRQL items had returned to preoperative levels by the 3-year assessment; however, their scores for physical function, breathlessness, diarrhea, and reflux remained significantly worse than at the baseline. Nevertheless, patients reported significantly better emotional function 3 years after surgery than before treatment. Djärv et al[62] reported the results for a cohort of 87 patients who had survived for at least 3 years after esophagectomy for cancer, using data from the prospective Swedish nationwide population study. As in other studies, these patients reported significantly more problems with fatigue, diarrhea, appetite loss, nausea and vomiting than did those in the reference population, and they also reported significantly poorer role and social function. Finally, Courrech Staal et al[63] compared the generic and disease-specific HRQL of 36 patients with esophagectomy (who had or had not received neoadjuvant therapy) after a median follow-up of 54 mo with the data for a reference sample of patients with esophageal cancer (1031 patients) and with that of the general population (7802 subjects). These long-term survivors reported better health-related quality of life than the reference sample of patients with esophageal cancer, even if their HRQL appeared lower than that of the reference sample of individuals from the general population[63].
PECULIAR ASPECTS OF HRQL AFTER ESOPHAGECTOMY
Data from the four articles that analyzed peculiar aspects of HRQL after esophagectomy were collected, but although the EORTC questionnaires were used, the level of clinical heterogeneity was so high that it was impossible to pool them[48,51,64,65]. In 1995, in one of the first studies focused on HRQL, Blazeby et al[64] observed that patients treated using esophagectomy reported significantly better physical, emotional, cognitive, and global health scores than did those who had received palliative treatment. These patients also had significantly worse pain, fatigue, appetite loss, constipation, and dysphagia. Ariga et al[65] performed a prospective direct comparison of outcomes after treatment in patients with resectable esophageal cancer who had received definitive chemoradiation and those who had undergone surgery alone. They surveyed HRQL in patients who had survived more than 2 years using a cross-sectional approach, and concluded that the HRQL of patients who had definitive chemoradiation and that of patients who had undergone surgery alone were similar. Diarrhea, appetite loss and eating problem scores were worse in patients who had undergone surgery alone than in those who had undergone chemoradiation[65]. Moreover, Schneider et al[48] compared the HRQL of those patients who had undergone elective and emergency esophagectomy with those who had undergone collar reconstruction. They observed a temporary decrease in postoperative HRQL in both groups and a return to preoperative values during the follow-up except for physical functioning, which remained decreased in patients who had undergone elective esophagectomy for cancer. Finally, Rosmolen et al[51] compared HRQL and fear of cancer among 81 patients who had undergone endoscopic treatment and 27 patients who had undergone surgery for early Barrett’s neoplasia. They observed that patients in the surgery group reported significantly more eating problems and reflux symptoms on the EORTC-OES18 questionnaire, whereas endoscopy patients reported more fear of recurrence than surgery patients. They concluded that preserving the esophagus after endoscopy, which is preferable from a clinical perspective, may induce fear of cancer recurrence[51].
CONCLUSION
Esophageal cancer is an increasingly common cancer with a poor prognosis. Esophagectomy is the standard treatment for those patients who present with resectable esophageal cancer[3-5], but it still offers a limited (25%-35%) chance of cure[5,6] and is associated with considerable risk of serious complications[4,5,7]. For a long time, morbidity and mortality represented the main, and often the only, outcome measures that could be used to evaluate esophagectomy for esophageal cancer. The morbidity and mortality rates associated with the procedure and the patients’ poor survival rates due to the aggressive nature of the disease left almost no space for further analysis. However, in recent decades, along with the increased success of the therapy, HRQL has become an important outcome parameter in addition to survival, mortality, and complication rates[67]. In fact, postoperative HRQL can yield information that is relevant for clinical decision-making and help to inform patients about the long-term consequences of surgery. With that in mind, this systematic review was designed to collect and analyze data reflecting patterns in HRQL after curative surgery for esophageal cancer.
Twenty-one studies published between 1995 and 2011 were included in this analysis. One limitation of this review is the clinical heterogeneity of the studies included. To increase homogeneity, only studies performed using SF36 or with EORTC-QLQ-C30 and OES18 (or the previous version, OES24) were included. Nevertheless, virtually every study used a different surgical approach and a different means of comparison, and they also did not present exact data. Furthermore, data for the recruited patients were collected during a range of intervals after surgery. Therefore, it was very difficult and sometimes impossible to obtain sufficiently homogeneous data to recalculate the statistical analyses or perform a meaningful meta-analysis. For example, 5 studies analyzed long-term generic HRQL after esophagectomy in comparison with that of healthy subjects using SF-36 questionnaires distributed at roughly comparable intervals after esophagectomy[49,50,52-54] but two studies could not be used for the meta-analysis, one because the SF-36 scores were not reported in a standard or comparable way and the other because all patients underwent esophagocoloplasty[50,52].
On the other hand, the remaining three studies were sufficiently homogenous, and thus, a meta-analysis of their results was attempted[49,53,54]. In these three studies, the pooled scores for physical, role, and social function after esophagectomy were similar to sex- and age-matched United States norms. In contrast, in a group of patients with similar follow-up, Djärv et al[62] and Courrech et al[63] observed significantly poorer role and social function. Differences between the HRQL measurement tools (SF36 and QLQ C30) may have created this difference in the results, and cross-cultural differences among the different groups of patients may also have been a factor. Health-related quality of life may vary from one population to another according to differences in cultural heritage, value systems, family structure, medical systems, values and norms related to illness-related communication, and other factors[67].
In the three studies that analyzed long-term generic HRQL after esophagectomy vs in healthy subjects, the pooled physical function, vitality and general health perception scores were lower than the sex- and age-matched norms[49,53,54]. Similarly, in patients alive at 3 years analyzed using the EORTC-QLQ-C30 and OES18, Djärv et al[62] and Courrech et al[63] reported encountering significantly more problems with fatigue, diarrhea, appetite loss, nausea and vomiting than in the reference population. Moreover, Lagergren et al[61] and Fagevik et al[60] observed that scores for physical function, breathlessness, diarrhea, and reflux were significantly worse than at the baseline. Finally, also in the three studies that prospectively analyzed generic and disease-specific HRQL using QLC-30 and OES-18 questionnaires after neoadjuvant therapy and esophagectomy, the EORTC C30 global scale results tended to be worse 6 mo after esophagectomy, as were physical function, fatigue, dyspnea and diarrhea[55-57]. Physical function impairment is a long-term consequence of esophagectomy that can involve either the respiratory system (which can be impaired by the thoracotomy sequelae) or the alimentary tract (which can be affected by accelerated transit and functional sequelae).
In contrast, in the three studies that compared HRQL after esophagectomy to that of healthy subjects using SF-36, bodily pain and mental health in long-term survivors after esophagectomy was higher than normal[49,53,54]. Similarly, in the three studies that prospectively analyzed generic and disease-specific HRQL using QLC-30 and OES-18 questionnaires after neoadjuvant therapy and eso-phagectomy, emotional function was significantly better at the time of the 6-mo follow up[55-57]. Moreover, Lagergren et al[61] observed that long-term survivors reported significantly better emotional function even 3 years after surgery. The short- and long-term improvement in emotional function in patients successfully operated on for esophageal cancer may be attributed to their sensation of having been quite close to death and having survived. In our opinion, this experience is different to that of having survived a car accident because of the duration, which can give patients sufficient time to experience the challenge in a positive way. On the other hand, it should be noted that this improved emotional function was observed in survivors.
Several studies analyzed a specific aspect of HRQL after esophagectomy. Two studies prospectively analyzed generic and disease-specific HRQL after minimally invasive esophagectomy[58,59]. The surgical techniques reported on in both studies were similar, and thus, it was possible to compare the results. The results were similar to those report by larger studies performed with group of patients who had undergone open esophagectomy, in that these patients experienced significantly improved social function, cognitive function, emotional function and dysphagia. However, physical function worsened 6 mo after esophagectomy[58,59]. In their direct comparison, Wang et al[59] concluded that global quality of life and physical functioning were better in the minimally invasive group than in the open surgery group. Additional larger studies should explore the exact benefit of minimally invasive esophagectomy in terms of HRQL.
Data from a Swedish nationwide, prospective, population-based study were used in three studies by the Karolinska Institute, Stockholm that investigated the most important predictors of HRQL after surgery for esophageal cancer using data from 6 mo after surgery[45-47]. Unexpectedly, extensive surgery, as used in the transthoracic approach, was not associated with lower HRQL than less invasive operations[46]. No clinically relevant differences in generic or disease-specific HRQL were observed based on the volume of procedures done at hospitals[47]. Age, sex, and body mass index showed no association with HRQL 6 mo after surgery, but patients with comorbidity, tumors in a more advanced stage (III to IV), or tumors located in the middle or upper esophagus exhibited an increased risk of poor HRQL. Moreover, the occurrence of surgery-related complications was the main predictor of reduced postoperative HRQL[45,47]. Except for anastomotic strictures, each of the predefined complications (e.g., anastomotic leakage, infections, cardiopulmonary complications, and operative technical complications) decreased the patients’ HRQL scores. In patients with non-neoplastic diseases such as Crohn’s disease or ulcerative colitis, postoperative complications did not seem to have a long-term effect on HRQL[68,69], so the reasons why patients submitted to esophagectomy can experience post-operative complications that can heavily affect HRQL must be different. Firstly, a 6-mo follow-up period may be not sufficiently long enough, as the problems caused by the complications may be still ongoing. Secondly, the different type of surgery that implies usually has thoracotomy play a direct role. In fact, the complications of a thoracotomy may have direct implications for dyspnea, fatigue and pain.
Some articles analyzed more peculiar aspects of HRQL after esophagectomy[48,64,65]. The observation that patients who received palliative treatment had significantly worse pain, fatigue, appetite loss, constipation, and dysphagia might be expected[64]. In contrast, the results presented by Ariga et al[65] are much less expected. They observed that patients with squamous cell carcinoma who underwent definitive chemoradiation had similar general HRQL scores and lower diarrhea, appetite loss and eating problem scores than those who had undergone surgery alone[65]. These results could be mainly attributed to the effect of the loss of function of the stomach transposed in the thorax. Schneider et al[48] observed the persistence of decreased physical functioning in patients who had undergone elective esophagectomy for cancer, as compared to patients who had undergone emergency esophagectomy for benign conditions. The conclusions of this study may suggest that the long-term impairment of physical functioning could be due more to the cancer itself or to radiation/chemotherapy than to esophagectomy. Finally, the analysis of the HRQL after endoscopic treatment and surgery for early Barrett’s neoplasia showed that conservative, non-definitive treatments such as endoscopic ablation may cause more fear of recurrence than more invasive but definitive treatments, such as esophagectomy[51]. Fear of cancer recurrence may negatively impact HRQL, and proper counseling may be advisable when patients elect such options[51].
In conclusion, short- and long-term generic and disease-specific HRQL is deeply affected by esophagectomy for cancer. The impairment of physical function may be a long-term consequence of esophagectomy and can involve either the respiratory system (which can be impaired by the thoracotomy sequelae) or the alimentary tract (which can be affected by accelerated transit and functional sequelae). The short- and long-term improvement in the emotional function of patients who have been successfully operated on may be attributed to their impression of having survived a near-death experience.
ACKNOWLEDGMENTS
The authors are particularly grateful to Dr. Mauro Apostolico, clinical librarian at the Veneto Institute of Oncology, Padova, for his kind help in retrieving the studies of this systematic review; and to Francesco Cavallin, clinical statistician at the Veneto Institute of Oncology, Padova, for his suggestions.
Footnotes
Peer reviewer: Guy Douglas Eslick, PhD, Program in Molecular and Genetic Epidemiology, Harvard School of Public Health, 677 Huntington Avenue, Building II, 2nd Floor, Boston, MA 02115, United States
S-Editor Sun H L-Editor Rutherford A E-Editor Li JY
References
- 1.Jemal A, Murray T, Samuels A, Ghafoor A, Ward E, Thun MJ. Cancer statistics, 2003. CA Cancer J Clin. 2003;53:5–26. doi: 10.3322/canjclin.53.1.5. [DOI] [PubMed] [Google Scholar]
- 2.Ruol A, Castoro C, Portale G, Cavallin F, Sileni VC, Cagol M, Alfieri R, Corti L, Boso C, Zaninotto G, et al. Trends in management and prognosis for esophageal cancer surgery: twenty-five years of experience at a single institution. Arch Surg. 2009;144:247–254; discussion 254. doi: 10.1001/archsurg.2008.574. [DOI] [PubMed] [Google Scholar]
- 3.De Vita F, Di Martino N, Orditura M, Cosenza A, Galizia G, Del Genio A, Catalano G. Preoperative chemoradiotherapy for squamous cell carcinoma and adenocarcinoma of the esophagus: a phase II study. Chest. 2002;122:1302–1308. doi: 10.1378/chest.122.4.1302. [DOI] [PubMed] [Google Scholar]
- 4.Enzinger PC, Mayer RJ. Esophageal cancer. N Engl J Med. 2003;349:2241–2252. doi: 10.1056/NEJMra035010. [DOI] [PubMed] [Google Scholar]
- 5.Wu PC, Posner MC. The role of surgery in the management of oesophageal cancer. Lancet Oncol. 2003;4:481–488. doi: 10.1016/s1470-2045(03)01167-7. [DOI] [PubMed] [Google Scholar]
- 6.Jamieson GG, Mathew G, Ludemann R, Wayman J, Myers JC, Devitt PG. Postoperative mortality following oesophagectomy and problems in reporting its rate. Br J Surg. 2004;91:943–947. doi: 10.1002/bjs.4596. [DOI] [PubMed] [Google Scholar]
- 7.Viklund P, Lindblad M, Lu M, Ye W, Johansson J, Lagerg-ren J. Risk factors for complications after esophageal cancer resection: a prospective population-based study in Sweden. Ann Surg. 2006;243:204–211. doi: 10.1097/01.sla.0000197698.17794.eb. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gebski V, Burmeister B, Smithers BM, Foo K, Zalcberg J, Simes J. Survival benefits from neoadjuvant chemoradiotherapy or chemotherapy in oesophageal carcinoma: a meta-analysis. Lancet Oncol. 2007;8:226–234. doi: 10.1016/S1470-2045(07)70039-6. [DOI] [PubMed] [Google Scholar]
- 9.Greer SE, Goodney PP, Sutton JE, Birkmeyer JD. Neoadjuvant chemoradiotherapy for esophageal carcinoma: a meta-analysis. Surgery. 2005;137:172–177. doi: 10.1016/j.surg.2004.06.033. [DOI] [PubMed] [Google Scholar]
- 10.Zacherl J, Sendler A, Stein HJ, Ott K, Feith M, Jakesz R, Siewert JR, Fink U. Current status of neoadjuvant therapy for adenocarcinoma of the distal esophagus. World J Surg. 2003;27:1067–1074. doi: 10.1007/s00268-003-7063-z. [DOI] [PubMed] [Google Scholar]
- 11.Brücher BL, Stein HJ, Zimmermann F, Werner M, Sarbia M, Busch R, Dittler HJ, Molls M, Fink U, Siewert JR. Responders benefit from neoadjuvant radiochemotherapy in esophageal squamous cell carcinoma: results of a prospective phase-II trial. Eur J Surg Oncol. 2004;30:963–971. doi: 10.1016/j.ejso.2004.06.008. [DOI] [PubMed] [Google Scholar]
- 12.Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, Moher D, Becker BJ, Sipe TA, Thacker SB. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA. 2000;283:2008–2012. doi: 10.1001/jama.283.15.2008. [DOI] [PubMed] [Google Scholar]
- 13.Aaronson NK, Ahmedzai S, Bergman B, Bullinger M, Cull A, Duez NJ, Filiberti A, Flechtner H, Fleishman SB, de Haes JC. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst. 1993;85:365–376. doi: 10.1093/jnci/85.5.365. [DOI] [PubMed] [Google Scholar]
- 14.Blazeby JM, Alderson D, Winstone K, Steyn R, Hammerlid E, Arraras J, Farndon JR. Development of an EORTC questionnaire module to be used in quality of life assessment for patients with oesophageal cancer. The EORTC Quality of Life Study Group. Eur J Cancer. 1996;32A:1912–1917. doi: 10.1016/0959-8049(96)00199-2. [DOI] [PubMed] [Google Scholar]
- 15.Aaronson NK, Muller M, Cohen PD, Essink-Bot ML, Fekkes M, Sanderman R, Sprangers MA, te Velde A, Verrips E. Translation, validation, and norming of the Dutch language version of the SF-36 Health Survey in community and chronic disease populations. J Clin Epidemiol. 1998;51:1055–1068. doi: 10.1016/s0895-4356(98)00097-3. [DOI] [PubMed] [Google Scholar]
- 16.Osoba D, Rodrigues G, Myles J, Zee B, Pater J. Interpreting the significance of changes in health-related quality-of-life scores. J Clin Oncol. 1998;16:139–144. doi: 10.1200/JCO.1998.16.1.139. [DOI] [PubMed] [Google Scholar]
- 17.Chang AC, Ji H, Birkmeyer NJ, Orringer MB, Birkmeyer JD. Outcomes after transhiatal and transthoracic esophagectomy for cancer. Ann Thorac Surg. 2008;85:424–429. doi: 10.1016/j.athoracsur.2007.10.007. [DOI] [PubMed] [Google Scholar]
- 18.Martin L, Lagergren J, Lindblad M, Rouvelas I, Lagergren P. Malnutrition after oesophageal cancer surgery in Sweden. Br J Surg. 2007;94:1496–1500. doi: 10.1002/bjs.5881. [DOI] [PubMed] [Google Scholar]
- 19.Branicki FJ, Law SY, Fok M, Poon RT, Chu KM, Wong J. Quality of life in patients with cancer of the esophagus and gastric cardia: a case for palliative resection. Arch Surg. 1998;133:316–322. doi: 10.1001/archsurg.133.3.316. [DOI] [PubMed] [Google Scholar]
- 20.Lagarde SM, Franssen SJ, van Werven JR, Smets EM, Tran TC, Tilanus HW, Plukker JT, de Haes JC, van Lanschot JJ. Patient preferences for the disclosure of prognosis after esophagectomy for cancer with curative intent. Ann Surg Oncol. 2008;15:3289–3298. doi: 10.1245/s10434-008-0068-y. [DOI] [PubMed] [Google Scholar]
- 21.Hulshof MC, Van Haaren PM, Van Lanschot JJ, Richel DJ, Fockens P, Oldenborg S, Geijsen ED, Van Berge Henegouwen MI, Crezee J. Preoperative chemoradiation combined with regional hyperthermia for patients with resectable esophageal cancer. Int J Hyperthermia. 2009;25:79–85. doi: 10.1080/02656730802464078. [DOI] [PubMed] [Google Scholar]
- 22.Nakajima M, Kato H, Miyazaki T, Fukuchi M, Manda R, Masuda N, Sohda M, Kamiyama Y, Nakabayashi T, Mochiki E, et al. Comprehensive investigations of quality of life after esophagectomy with special reference to the route of reconstruction. Hepatogastroenterology. 2007;54:104–110. [PubMed] [Google Scholar]
- 23.Barbour AP, Lagergren P, Hughes R, Alderson D, Barham CP, Blazeby JM. Health-related quality of life among patients with adenocarcinoma of the gastro-oesophageal junction treated by gastrectomy or oesophagectomy. Br J Surg. 2008;95:80–84. doi: 10.1002/bjs.5912. [DOI] [PubMed] [Google Scholar]
- 24.Cense HA, Hulscher JB, de Boer AG, Dongelmans DA, Tilanus HW, Obertop H, Sprangers MA, van Lanschot JJ. Effects of prolonged intensive care unit stay on quality of life and long-term survival after transthoracic esophageal resection. Crit Care Med. 2006;34:354–362. doi: 10.1097/01.ccm.0000195016.55516.3e. [DOI] [PubMed] [Google Scholar]
- 25.Blazeby JM, Kavadas V, Vickery CW, Greenwood R, Berrisford RG, Alderson D. A prospective comparison of quality of life measures for patients with esophageal cancer. Qual Life Res. 2005;14:387–393. doi: 10.1007/s11136-004-0622-4. [DOI] [PubMed] [Google Scholar]
- 26.Ribi K, Koeberle D, Schuller JC, Honegger H, Roth A, Hess V, Moosmann P, von Moos R, Borner M, Lombriser N, et al. Is a change in patient-reported dysphagia after induction chemotherapy in locally advanced esophageal cancer a predictive factor for pathological response to neoadjuvant chemoradiation? Support Care Cancer. 2009:Feb 7; Epub ahead of print. doi: 10.1007/s00520-008-0570-6. [DOI] [PubMed] [Google Scholar]
- 27.Safieddine N, Xu W, Quadri SM, Knox JJ, Hornby J, Sulman J, Wong R, Guindi M, Keshavjee S, Darling G. Health-related quality of life in esophageal cancer: effect of neoadjuvant chemoradiotherapy followed by surgical intervention. J Thorac Cardiovasc Surg. 2009;137:36–42. doi: 10.1016/j.jtcvs.2008.09.049. [DOI] [PubMed] [Google Scholar]
- 28.Egberts JH, Schniewind B, Bestmann B, Schafmayer C, Egberts F, Faendrich F, Kuechler T, Tepel J. Impact of the site of anastomosis after oncologic esophagectomy on quality of life--a prospective, longitudinal outcome study. Ann Surg Oncol. 2008;15:566–575. doi: 10.1245/s10434-007-9615-1. [DOI] [PubMed] [Google Scholar]
- 29.Chang LC, Oelschlager BK, Quiroga E, Parra JD, Mulligan M, Wood DE, Pellegrini CA. Long-term outcome of esophagectomy for high-grade dysplasia or cancer found during surveillance for Barrett’s esophagus. J Gastrointest Surg. 2006;10:341–346. doi: 10.1016/j.gassur.2005.12.007. [DOI] [PubMed] [Google Scholar]
- 30.Blazeby JM, Sanford E, Falk SJ, Alderson D, Donovan JL. Health-related quality of life during neoadjuvant treatment and surgery for localized esophageal carcinoma. Cancer. 2005;103:1791–1799. doi: 10.1002/cncr.20980. [DOI] [PubMed] [Google Scholar]
- 31.de Boer AG, van Lanschot JJ, van Sandick JW, Hulscher JB, Stalmeier PF, de Haes JC, Tilanus HW, Obertop H, Sprangers MA. Quality of life after transhiatal compared with extended transthoracic resection for adenocarcinoma of the esophagus. J Clin Oncol. 2004;22:4202–4208. doi: 10.1200/JCO.2004.11.102. [DOI] [PubMed] [Google Scholar]
- 32.Blazeby JM, Farndon JR, Donovan J, Alderson D. A prospective longitudinal study examining the quality of life of patients with esophageal carcinoma. Cancer. 2000;88:1781–1787. [PubMed] [Google Scholar]
- 33.Higuchi A, Minamide J, Ota Y, Takada K, Aoyama N. Evaluation of the quality of life after surgical treatment for thoracic esophageal cancer. Esophagus. 2006;3:53–59. [Google Scholar]
- 34.Leibman S, Smithers BM, Gotley DC, Martin I, Thomas J. Minimally invasive esophagectomy: short- and long-term outcomes. Surg Endosc. 2006;20:428–433. doi: 10.1007/s00464-005-0388-y. [DOI] [PubMed] [Google Scholar]
- 35.Teoh AY, Yan Chiu PW, Wong TC, Liu SY, Hung Wong SK, Ng EK. Functional performance and quality of life in patients with squamous esophageal carcinoma receiving surgery or chemoradiation: results from a randomized trial. Ann Surg. 2011;253:1–5. doi: 10.1097/SLA.0b013e3181fcd991. [DOI] [PubMed] [Google Scholar]
- 36.Bonnetain F, Bouché O, Michel P, Mariette C, Conroy T, Pezet D, Roullet B, Seitz JF, Paillot B, Arveux P, et al. A comparative longitudinal quality of life study using the Spitzer quality of life index in a randomized multicenter phase III trial (FFCD 9102): chemoradiation followed by surgery compared with chemoradiation alone in locally advanced squamous resectable thoracic esophageal cancer. Ann Oncol. 2006;17:827–834. doi: 10.1093/annonc/mdl033. [DOI] [PubMed] [Google Scholar]
- 37.de Boer AG, van Lanschot JJ, Stalmeier PF, van Sandick JW, Hulscher JB, de Haes JC, Sprangers MA. Is a single-item visual analogue scale as valid, reliable and responsive as multi-item scales in measuring quality of life? Qual Life Res. 2004;13:311–320. doi: 10.1023/B:QURE.0000018499.64574.1f. [DOI] [PubMed] [Google Scholar]
- 38.Brooks JA, Kesler KA, Johnson CS, Ciaccia D, Brown JW. Prospective analysis of quality of life after surgical resection for esophageal cancer: preliminary results. J Surg Oncol. 2002;81:185–194. doi: 10.1002/jso.10175. [DOI] [PubMed] [Google Scholar]
- 39.Baba M, Aikou T, Natsugoe S, Kusano C, Shimada M, Kimura S, Fukumoto T. Appraisal of ten-year survival following esophagectomy for carcinoma of the esophagus with emphasis on quality of life. World J Surg. 1997;21:282–285; discussion 286. doi: 10.1007/s002689900229. [DOI] [PubMed] [Google Scholar]
- 40.Tabira Y, Yasunaga M, Nagamoto N, Matsushita H, Fukunaga Y, Ihara T, Kawasuji M. Quality of life after esophagectomy for cancer: an assessment using the questionnaire with the face scale. Surg Today. 2002;32:213–219. doi: 10.1007/s005950200023. [DOI] [PubMed] [Google Scholar]
- 41.Barbier PA, Luder PJ, Schüpfer G, Becker CD, Wagner HE. Quality of life and patterns of recurrence following transhiatal esophagectomy for cancer: results of a prospective follow-up in 50 patients. World J Surg. 1988;12:270–276. doi: 10.1007/BF01658073. [DOI] [PubMed] [Google Scholar]
- 42.Collard JM, Otte JB, Reynaert M, Kestens PJ. Quality of life three years or more after esophagectomy for cancer. J Thorac Cardiovasc Surg. 1992;104:391–394. [PubMed] [Google Scholar]
- 43.Djärv T, Blazeby JM, Lagergren P. Predictors of postoperative quality of life after esophagectomy for cancer. J Clin Oncol. 2009;27:1963–1968. doi: 10.1200/JCO.2008.20.5864. [DOI] [PubMed] [Google Scholar]
- 44.Blazeby JM, Metcalfe C, Nicklin J, Barham CP, Donovan J, Alderson D. Association between quality of life scores and short-term outcome after surgery for cancer of the oesophagus or gastric cardia. Br J Surg. 2005;92:1502–1507. doi: 10.1002/bjs.5175. [DOI] [PubMed] [Google Scholar]
- 45.Viklund P, Lindblad M, Lagergren J. Influence of surgery-related factors on quality of life after esophageal or cardia cancer resection. World J Surg. 2005;29:841–848. doi: 10.1007/s00268-005-7887-9. [DOI] [PubMed] [Google Scholar]
- 46.Rutegård M, Lagergren J, Rouvelas I, Lindblad M, Blazeby JM, Lagergren P. Population-based study of surgical factors in relation to health-related quality of life after oesophageal cancer resection. Br J Surg. 2008;95:592–601. doi: 10.1002/bjs.6021. [DOI] [PubMed] [Google Scholar]
- 47.Rutegård M, Lagergren P. No influence of surgical volume on patients’ health-related quality of life after esophageal cancer resection. Ann Surg Oncol. 2008;15:2380–2387. doi: 10.1245/s10434-008-9964-4. [DOI] [PubMed] [Google Scholar]
- 48.Schneider L, Hartwig W, Aulmann S, Lenzen Ch, Strobel O, Fritz S, Hackert T, Keller M, Buchler MW, Werner J. Quality of life after emergency vs. elective esophagectomy with cervical reconstruction. Scand J Surg. 2010;99:3–8. doi: 10.1177/145749691009900102. [DOI] [PubMed] [Google Scholar]
- 49.Headrick JR, Nichols FC, Miller DL, Allen MS, Trastek VF, Deschamps C, Schleck CD, Thompson AM, Pairolero PC. High-grade esophageal dysplasia: long-term survival and quality of life after esophagectomy. Ann Thorac Surg. 2002;73:1697–1702; discussion 1702-1703. doi: 10.1016/s0003-4975(02)03496-3. [DOI] [PubMed] [Google Scholar]
- 50.Moraca RJ, Low DE. Outcomes and health-related quality of life after esophagectomy for high-grade dysplasia and intramucosal cancer. Arch Surg. 2006;141:545–559; discussion 545-559. doi: 10.1001/archsurg.141.6.545. [DOI] [PubMed] [Google Scholar]
- 51.Rosmolen WD, Boer KR, de Leeuw RJ, Gamel CJ, van Berge Henegouwen MI, Bergman JJ, Sprangers MA. Quality of life and fear of cancer recurrence after endoscopic and surgical treatment for early neoplasia in Barrett’s esophagus. Endoscopy. 2010;42:525–531. doi: 10.1055/s-0029-1244222. [DOI] [PubMed] [Google Scholar]
- 52.Cense HA, Visser MR, van Sandick JW, de Boer AG, Lamme B, Obertop H, van Lanschot JJ. Quality of life after colon interposition by necessity for esophageal cancer replacement. J Surg Oncol. 2004;88:32–38. doi: 10.1002/jso.20132. [DOI] [PubMed] [Google Scholar]
- 53.De Boer AG, Genovesi PI, Sprangers MA, Van Sandick JW, Obertop H, Van Lanschot JJ. Quality of life in long-term survivors after curative transhiatal oesophagectomy for oesophageal carcinoma. Br J Surg. 2000;87:1716–1721. doi: 10.1046/j.1365-2168.2000.01600.x. [DOI] [PubMed] [Google Scholar]
- 54.McLarty AJ, Deschamps C, Trastek VF, Allen MS, Pairolero PC, Harmsen WS. Esophageal resection for cancer of the esophagus: long-term function and quality of life. Ann Thorac Surg. 1997;63:1568–1572. doi: 10.1016/s0003-4975(97)00125-2. [DOI] [PubMed] [Google Scholar]
- 55.Reynolds JV, McLaughlin R, Moore J, Rowley S, Ravi N, Byrne PJ. Prospective evaluation of quality of life in patients with localized oesophageal cancer treated by multimodality therapy or surgery alone. Br J Surg. 2006;93:1084–1090. doi: 10.1002/bjs.5373. [DOI] [PubMed] [Google Scholar]
- 56.Avery KN, Metcalfe C, Barham CP, Alderson D, Falk SJ, Blazeby JM. Quality of life during potentially curative treatment for locally advanced oesophageal cancer. Br J Surg. 2007;94:1369–1376. doi: 10.1002/bjs.5888. [DOI] [PubMed] [Google Scholar]
- 57.van Meerten E, van der Gaast A, Looman CW, Tilanus HW, Muller K, Essink-Bot ML. Quality of life during neoadjuvant treatment and after surgery for resectable esophageal carcinoma. Int J Radiat Oncol Biol Phys. 2008;71:160–166. doi: 10.1016/j.ijrobp.2007.09.038. [DOI] [PubMed] [Google Scholar]
- 58.Parameswaran R, Blazeby JM, Hughes R, Mitchell K, Berrisford RG, Wajed SA. Health-related quality of life after minimally invasive oesophagectomy. Br J Surg. 2010;97:525–531. doi: 10.1002/bjs.6908. [DOI] [PubMed] [Google Scholar]
- 59.Wang H, Feng M, Tan L, Wang Q. Comparison of the short-term quality of life in patients with esophageal cancer after subtotal esophagectomy via video-assisted thoracoscopic or open surgery. Dis Esophagus. 2010;23:408–414. doi: 10.1111/j.1442-2050.2009.01025.x. [DOI] [PubMed] [Google Scholar]
- 60.Fagevik Olsén M, Larsson M, Hammerlid E, Lundell L. Physical function and quality of life after thoracoabdominal oesophageal resection. Results of a follow-up study. Dig Surg. 2005;22:63–68. doi: 10.1159/000085348. [DOI] [PubMed] [Google Scholar]
- 61.Lagergren P, Avery KN, Hughes R, Barham CP, Alderson D, Falk SJ, Blazeby JM. Health-related quality of life among patients cured by surgery for esophageal cancer. Cancer. 2007;110:686–693. doi: 10.1002/cncr.22833. [DOI] [PubMed] [Google Scholar]
- 62.Djärv T, Lagergren J, Blazeby JM, Lagergren P. Long-term health-related quality of life following surgery for oesophageal cancer. Br J Surg. 2008;95:1121–1126. doi: 10.1002/bjs.6293. [DOI] [PubMed] [Google Scholar]
- 63.Courrech Staal EF, van Sandick JW, van Tinteren H, Cats A, Aaronson NK. Health-related quality of life in long-term esophageal cancer survivors after potentially curative treatment. J Thorac Cardiovasc Surg. 2010;140:777–783. doi: 10.1016/j.jtcvs.2010.05.018. [DOI] [PubMed] [Google Scholar]
- 64.Blazeby JM, Williams MH, Brookes ST, Alderson D, Farndon JR. Quality of life measurement in patients with oesophageal cancer. Gut. 1995;37:505–508. doi: 10.1136/gut.37.4.505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Ariga H, Nemoto K, Miyazaki S, Yoshioka T, Ogawa Y, Sakayauchi T, Jingu K, Miyata G, Onodera K, Ichikawa H, et al. Prospective comparison of surgery alone and chemoradiotherapy with selective surgery in resectable squamous cell carcinoma of the esophagus. Int J Radiat Oncol Biol Phys. 2009;75:348–356. doi: 10.1016/j.ijrobp.2009.02.086. [DOI] [PubMed] [Google Scholar]
- 66.McLeod RS. Quality-of-life measurement in the assessment of surgical outcome. Adv Surg. 1999;33:293–309. [PubMed] [Google Scholar]
- 67.Levenstein S, Li Z, Almer S, Barbosa A, Marquis P, Moser G, Sperber A, Toner B, Drossman DA. Cross-cultural variation in disease-related concerns among patients with inflammatory bowel disease. Am J Gastroenterol. 2001;96:1822–1830. doi: 10.1111/j.1572-0241.2001.03878.x. [DOI] [PubMed] [Google Scholar]
- 68.Scarpa M, Ruffolo C, Bassi D, Boetto R, D’Incà R, Buda A, Sturniolo GC, Angriman I. Intestinal surgery for Crohn’s disease: predictors of recovery, quality of life, and costs. J Gastrointest Surg. 2009;13:2128–2135. doi: 10.1007/s11605-009-1044-y. [DOI] [PubMed] [Google Scholar]
- 69.Scarpa M, Angriman I, Ruffolo C, Ferronato A, Polese L, Barollo M, Martin A, Sturniolo GC, D’Amico DF. Health-related quality of life after restorative proctocolectomy for ulcerative colitis: long-term results. World J Surg. 2004;28:124–129. doi: 10.1007/s00268-003-7047-z. [DOI] [PubMed] [Google Scholar]