Abstract
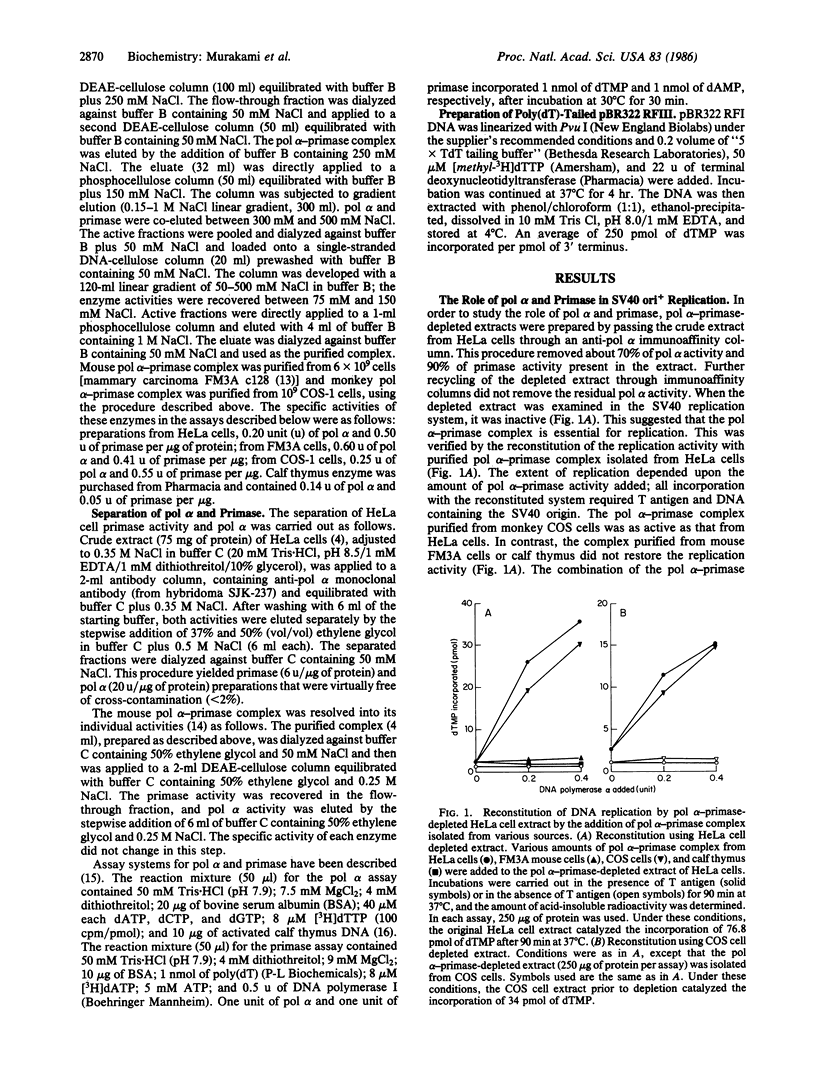
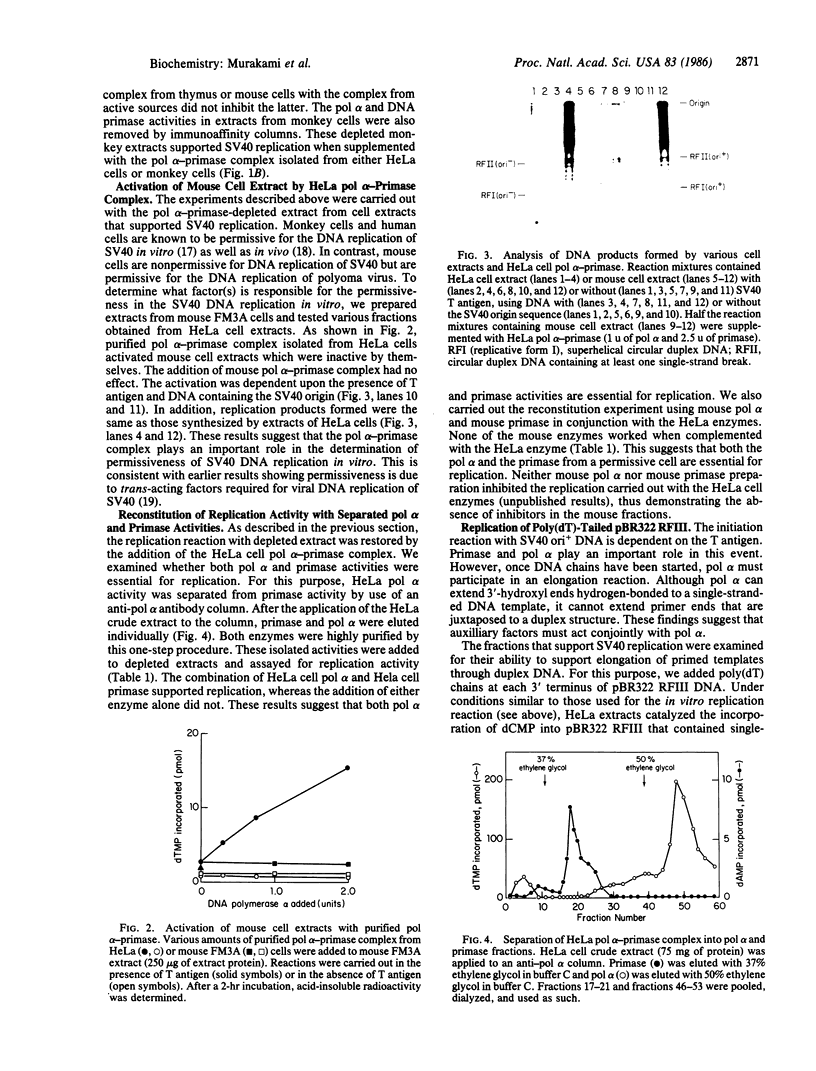
The role of DNA polymerase alpha (pol alpha) and DNA primase has been investigated in the simian virus 40 (SV40) DNA replication system in vitro. Removal of pol alpha and primase activities from crude extracts of HeLa cells or monkey cells by use of an anti-pol alpha immunoaffinity column resulted in the loss of replication activity. The addition of purified pol alpha-primase complex isolated from HeLa cells or monkey cells restored the replication activity of depleted extracts. In contrast, the pol alpha-primase complex isolated from either mouse cells or calf thumus did not. Extracts prepared from mouse cells (a source that does not support replication of SV40) did not replicate SV40 DNA. However, the addition of purified pol alpha-primase complex isolated from HeLa cells activated mouse cell extracts. pol alpha and primase from HeLa cells were extensively purified and separated by a one-step immunoaffinity adsorption and elution procedure. Both activities were required to restore DNA synthesis; the addition of pol alpha or primase alone supported replication poorly. Crude extracts of HeLa cells that were active in SV40 replication catalyzed the synthesis of full-length linear double-stranded (RFIII) DNA in reaction mixtures containing poly(dT)-tailed pBR322 RFIII. Maximal activity was dependent on the addition of oligo(dA), ATP, and creatine phosphate and was totally inhibited by aphidicolin. Since pol alpha alone could not replicate this substrate and since there was no degradation of input DNA, we propose that other enzymatic activities associate with pol alpha, displace the non-template strand, and allow the enzyme to replicate through duplex regions.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- APOSHIAN H. V., KORNBERG A. Enzymatic synthesis of deoxyribonucleic acid. IX. The polymerase formed after T2 bacteriophage infection of Escherichia coli: a new enzyme. J Biol Chem. 1962 Feb;237:519–525. [PubMed] [Google Scholar]
- Ariga H., Sugano S. Initiation of simian virus 40 DNA replication in vitro. J Virol. 1983 Nov;48(2):481–491. doi: 10.1128/jvi.48.2.481-491.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Botchan M., Topp W., Sambrook J. Studies on simian virus 40 excision from cellular chromosomes. Cold Spring Harb Symp Quant Biol. 1979;43(Pt 2):709–719. doi: 10.1101/sqb.1979.043.01.079. [DOI] [PubMed] [Google Scholar]
- Chang L. M., Rafter E., Augl C., Bollum F. J. Purification of a DNA polymerase-DNA primase complex from calf thymus glands. J Biol Chem. 1984 Dec 10;259(23):14679–14687. [PubMed] [Google Scholar]
- Conaway R. C., Lehman I. R. A DNA primase activity associated with DNA polymerase alpha from Drosophila melanogaster embryos. Proc Natl Acad Sci U S A. 1982 Apr;79(8):2523–2527. doi: 10.1073/pnas.79.8.2523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gronostajski R. M., Field J., Hurwitz J. Purification of a primase activity associated with DNA polymerase alpha from HeLa cells. J Biol Chem. 1984 Aug 10;259(15):9479–9486. [PubMed] [Google Scholar]
- Huberman J. A. New views of the biochemistry of eucaryotic DNA replication revealed by aphidicolin, an unusual inhibitor of DNA polymerase alpha. Cell. 1981 Mar;23(3):647–648. doi: 10.1016/0092-8674(81)90426-8. [DOI] [PubMed] [Google Scholar]
- Ikegami S., Taguchi T., Ohashi M., Oguro M., Nagano H., Mano Y. Aphidicolin prevents mitotic cell division by interfering with the activity of DNA polymerase-alpha. Nature. 1978 Oct 5;275(5679):458–460. doi: 10.1038/275458a0. [DOI] [PubMed] [Google Scholar]
- Kuhn B., Abdel-Monem M. DNA synthesis at a fork in the presence of DNA helicases. Eur J Biochem. 1982 Jun 15;125(1):63–68. doi: 10.1111/j.1432-1033.1982.tb06651.x. [DOI] [PubMed] [Google Scholar]
- Li J. J., Kelly T. J. Simian virus 40 DNA replication in vitro. Proc Natl Acad Sci U S A. 1984 Nov;81(22):6973–6977. doi: 10.1073/pnas.81.22.6973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li J. J., Kelly T. J. Simian virus 40 DNA replication in vitro: specificity of initiation and evidence for bidirectional replication. Mol Cell Biol. 1985 Jun;5(6):1238–1246. doi: 10.1128/mcb.5.6.1238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murakami Y., Eki T., Miyazawa H., Enomoto T., Hanaoka F., Yamada M. Further characterization of a murine temperature-sensitive mutant, tsFT20 strain, containing heat-labile DNA polymerase alpha-activity. Exp Cell Res. 1986 Mar;163(1):135–142. doi: 10.1016/0014-4827(86)90565-3. [DOI] [PubMed] [Google Scholar]
- Murakami Y., Yasuda H., Miyazawa H., Hanaoka F., Yamada M. Characterization of a temperature-sensitive mutant of mouse FM3A cells defective in DNA replication. Proc Natl Acad Sci U S A. 1985 Mar;82(6):1761–1765. doi: 10.1073/pnas.82.6.1761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakano N. Establishment of cell lines in vitro from a mammary ascites tumor of mouse and biological properties of the established lines in a serum containing medium. Tohoku J Exp Med. 1966 Jan 25;88(1):69–84. doi: 10.1620/tjem.88.69. [DOI] [PubMed] [Google Scholar]
- Ozer H. L., Slater M. L., Dermody J. J., Mandel M. Replication of simian virus 40 DNA in normal human fibroblasts and in fibroblasts from xeroderma pigmentosum. J Virol. 1981 Aug;39(2):481–489. doi: 10.1128/jvi.39.2.481-489.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simanis V., Lane D. P. An immunoaffinity purification procedure for SV40 large T antigen. Virology. 1985 Jul 15;144(1):88–100. doi: 10.1016/0042-6822(85)90308-3. [DOI] [PubMed] [Google Scholar]
- Stillman B. W., Gluzman Y. Replication and supercoiling of simian virus 40 DNA in cell extracts from human cells. Mol Cell Biol. 1985 Aug;5(8):2051–2060. doi: 10.1128/mcb.5.8.2051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugino A., Nakayama K. DNA polymerase alpha mutants from a Drosophila melanogaster cell line. Proc Natl Acad Sci U S A. 1980 Dec;77(12):7049–7053. doi: 10.1073/pnas.77.12.7049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki M., Enomoto T., Hanaoka F., Yamada M. Dissociation and reconstitution of a DNA polymerase alpha-primase complex. J Biochem. 1985 Aug;98(2):581–584. doi: 10.1093/oxfordjournals.jbchem.a135314. [DOI] [PubMed] [Google Scholar]
- Tanaka S., Hu S. Z., Wang T. S., Korn D. Preparation and preliminary characterization of monoclonal antibodies against human DNA polymerase alpha. J Biol Chem. 1982 Jul 25;257(14):8386–8390. [PubMed] [Google Scholar]
- Weaver D. T., DePamphilis M. L. Specific sequences in native DNA that arrest synthesis by DNA polymerase alpha. J Biol Chem. 1982 Feb 25;257(4):2075–2086. [PubMed] [Google Scholar]
- Wobbe C. R., Dean F., Weissbach L., Hurwitz J. In vitro replication of duplex circular DNA containing the simian virus 40 DNA origin site. Proc Natl Acad Sci U S A. 1985 Sep;82(17):5710–5714. doi: 10.1073/pnas.82.17.5710. [DOI] [PMC free article] [PubMed] [Google Scholar]