Abstract
We describe three males with X-linked SCID (X-SCID) who were successfully treated by reduced-intensity SCT from unrelated cord blood (CB). Mean age at transplant was 5.7 months (range, 3–9 months). Pre-transplant conditioning for all patients consisted of fludarabine (FLU) (30 mg/m2 per day) from day −7 to day −2 (total dose 180 mg/m2) and BU 4 mg/kg per day from day −3 to day −2 (total dose 8 mg/kg). All CB units were serologically matched at HLA-A, B and DR loci. Although two patients had suffered from fungal or bacterial pneumonia before transplantation, there were no other infectious complications during transplantation. All patients engrafted and achieved 100% donor chimerism. We also confirmed full donor chimerism of both T and B cells. Only one patient developed acute GVHD grade III, which was resolved by increasing the dose of oral corticosteroid. None of the patients has developed chronic GVHD during follow up for 21–77 months. None of the patient received i.v. Ig replacement post transplant, or showed delay in psychomotor development. Reduced-intensity conditioning consisting of FLU and BU and transplantation from unrelated CB was an effective and safe treatment for these patients with X-SCID.
Keywords: SCT, X-SCID, reduced-intensity SCT, cord blood, fludarabine/busulfan
Introduction
SCID is the most severe form of primary immunodeficiency. About half of all cases are X-linked SCID (X-SCID; T-B+NK-SCID), caused by deficits of cytokine receptor common gamma chain.1 Haematopoietic SCT is the only curative treatment for these high-risk patients.2, 3 In the early series, HLA-identical sibling BMT led to complete immunological reconstitution with no conditioning regimen.4, 5 Thereafter, transplants from closely matched unrelated volunteer donor (MUD) have been performed with better outcomes in terms of both survival and immunological reconstitution than following those using haploidentical donors; the majority of patients with myeloablative conditioning no longer required i.v. Ig replacement after MUD transplantation.2, 5, 6 However, MUD transplantation can require a lengthy search for a suitable donor, often over 3 months, and is associated with both high frequency and intensity of GVHD. Moreover, immunological reconstitution is often incomplete especially with X-SCID after MUD transplant with no conditioning.4 Most of these problems are resolvable by transplanting with umbilical cord blood (CB) already typed with a known number of CD34 cells supplied from CB bank, when a lower incidence of GVHD is seen.7, 8, 9 Following the first report of CB SCT (CBT) for primary immunodeficiency patients using their sibling donors,10 successful unrelated CBTs have been described.11, 12, 13, 14, 15, 16 Because the OS rates are over 70%, CB could be a promising source of stem cells when an HLA-identical sibling donor is not available.
X-SCID is fatal and requires SCT in the first year of life despite possible late complications such as mental and physical retardation after myeloablative transplantation received in infancy.17, 18, 19 On the other hand, most have already suffered from bacterial and/or fungal infection at diagnosis of X-SCID. Recently developed reduced-intensity conditioning (RIC) regimens have been used in unrelated SCT for primary immunodeficiency patients, because of their intense immune suppressive qualities and reduced myelotoxicity.20 Thus, reduced-intensity SCT (RIST) from CB could be a choice for patients with X-SCID but is not fully established to date. We report our single centre experience of successful allogeneic RIST from unrelated CB for treatment of X-SCID.
Patients and methods
Patients
Three patients with X-SCID received unrelated CBT because they had no HLA-matched sibling donors. As shown in Table 1, mutations in the common gamma chain gene were detected in all patients. Patients 1 and 2 had suffered from pneumonia caused by aspergillus and bacteria, respectively, at the diagnosis of X-SCID. Patient 3 was diagnosed as having X-SCID at birth because his brother had the same disease (Table 1).
Table 1. Patient characteristics.
| Patient | 1 | 2 | 3 |
|---|---|---|---|
| Age at diagnosis (months) | 8 | 4 | 0 |
| Age at CBT (months) | 9 | 5 | 3 |
| Mutations in common gamma chain | 682T>G in exon 5 | 9_10insA in exon 1 | 216G>A in exon 2 |
| HLA identity | 6/6 | 6/6 | 6/6 |
| Nucleated cell dose ( × 107/kg) | 11.2 | 20.4 | 15.6 |
| CD34+ cell dose ( × 105/kg) | 6.7 | 5.3 | 3.2 |
| Cytomegarovirus serology | Positive | Positive | Positive |
| Haematological recovery | |||
| Neutrophil>500/μL | day+19 | day+22 | Day+27 |
| Platelet>50 × 109/L | day+28 | day+43 | Day+31 |
| Complications at CBT | aspergillus pneumonia | bacterial pneumonia | None |
| Additional infections during CBT | None | None | None |
| GVHD | |||
| Acute(grade) | 0 | III | 0 |
| Chronic | — | — | — |
| Chimerism (donor%) | 100 | 100 | 100 |
| B-cell engrafted(donor % (day)) | 100 (day+120) | 100 (day+89) | 100 (day+83) |
| i.v.Ig replace at present | None | None | None |
| Performance Status (ECOG, Oken MM et al35) | PS-0 | PS-0 | PS-0 |
| Outcome | Alive +77mo | Alive +69mo | Alive +21 mo |
| Height | −0.53 s.d. | −0.49 s.d. | −2.4 s.d. |
| Body mass index | 15.0 | 16.1 | 15.6 |
| Mental status | Normal | Normal | Normal |
Abbreviations: CBT=cord blood transplantation; ECOG=Eastern Cooperative Oncology Group.
Conditioning regimen and GVHD prophylaxis
Pre-transplant conditioning for all patients consisted of fludarabine (FLU) (30 mg/m2 per day) from day −7 to day −2 (total dose 180 mg/m2) and BU 4 mg/kg per day (oral in patient 1 and 2; i.v. in patient 3) from day −3 to day −2 (total dose 8 mg/kg). Neither ATG nor Campath was included in the conditioning regimen.
Prophylaxis for aGVHD included cyclosporine A (3 mg/kg) from day −1 to day +180, and methylprednisolone 0.5 mg/kg per day (day+7 to +13), 1 mg/kg per day (day+14 to +28), 0.5 mg/kg per day (day+29 to +42), 0.3 mg/kg per day (day+43 to +56) and 0.2 mg/kg per day (day+57 to +72). Cyclosporine A was discontinued by day+180, after confirming the absence of clinical GVHD.
Graft characteristics
As shown in Table 1, all CBs were collected from female donors. All of these CB units were serologically well matched at 6/6 (A, B, DR) HLA loci. Infused nucleated cell doses were 11–20 × 107/kg (mean, 15 × 107/kg), which contained CD34+ stem cells, ranging from 3.2 to 6.7 × 105/kg (mean, 5.1 × 105/kg).
Supportive care
Supportive care for transplantation in our institution has been previously described.21 Briefly, all patients received a continuous infusion of low molecular weight heparin, 100 unit/kg per day, as prophylaxis against hepatic veno-occlusive disease (VOD), from day −7 to day +30. Oral polymixin B and amphotericin B, inhaled vancomycin, tobramycin and amphtericin B were given as antibacterial and antifungal prophylaxis. Antiviral prophylaxis consisted of oral acyclovir (600 mg/m2 per day) from day −7 to day +35 and weekly i.v. γ-globulin (200 mg/kg per 2 weeks) from day −6 to day +90. G-CSF was given i.v. at 5 μg/kg from day +5 until the ANC reached 500/μL or more for 3 consecutive days. Oral mucositis was treated with i.v. pentazocine and parental nutrition.
aGVHD was diagnosed and graded according to the Seattle criteria,22 and treated with prednisolone. CMV infection was diagnosed on the basis of CMV antigenemia, and treated with ganciclovir in combination with anti-CMV high titer γ-globulin.
Chimerism studies
Hematological recovery was defined as achievement of both an ANC>500/μL for 3 consecutive days and a platelet count >50 × 109/L for 7 consecutive days without the need for transfusion. Chimerism was tested by FISH of peripheral mononuclear cells using X and Y chromosome probes. Chimerism of T or B cell lineage was assessed by flow cytometry using intracytoplasmic staining of common gamma chain together with fluorescein-labelled antibodies against CD3 and CD19.
Immunological reconstitution studies
Immunological reconstitution status after transplantation was monitored by serum immunoglobulin levels (IgG, IgA and IgM) and flow cytometry analyses of peripheral mononuclear cells for CD3, CD4, CD8, CD16, CD19 and CD56.
Results
Engraftment and chimerism
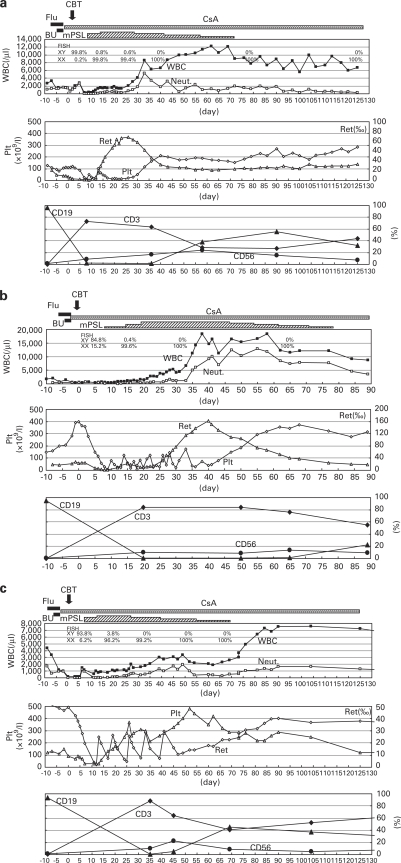
As shown in Figures 1a–c, all patients achieved engraftment of ANC>500/μL at a mean of 22 days (range, 19–27 days). Mean time to platelet engraftment (platelets >50 × 109/L) was 34 days (range, 28–43 days). Donor chimerism (100%) was demonstrated by FISH with X and Y chromosome probes at 1-year post transplantation. We also confirmed full donor chimerism of both T and B cells. Chimerism has been stable to date with a mean follow-up of 53 months (range, 21–77 months). Frequencies of transfusion were similar to the other patients with non-malignant conditions, such as congenital metabolic disorders, receiving RIST in our institute.
Figure 1.
Clinical course of the three patients with X-SCID receiving RIST from CB. a, Patient 1; b, Patient 2; c, Patient 3; ▪, WBC; □, Neutrophils (Neut); ▵, Reticulocyte (Ret); ◊, Platelet (Plt); ⧫, CD3; ▴, CD19; •, CD56; respectively. Note that all patients have engrafted and reached >500/μL neutrophils with day+19, +22, +27, respectively (mean day+22). Platelet engraftment (platelets >50 × 109/L) was day+28, +43, +31, respectively (mean day+34).
Regimen related toxicity
Patient 1 received anti-fungal treatment with i.v. micafungin 3 mg/kg before conditioning until day+90 for pre-existing lung aspergillosis, which showed no exacerbation during CBT. Clinical and radiological improvement were achieved after CB engraftment. Patient 2 was already suffering from bacterial pneumonia on admission; he received appropriate antibiotic treatment and recovered at the beginning of conditioning. He had no additional severe infectious complications up to engraftment. Patient 3 received CBT at 3 months of age with no previous infections. He also had no severe infectious complications up to engraftment. Although mild mucositis and increase of serum transaminase occurred, none of the patients experienced complicated severe regimen related toxicities such as VOD.
GVHD disease
Only patient 2 developed acute GVHD grade III, consisting of mild skin rash and diarrhea that resolved by increasing the oral corticosteroid dose. None of the patients have developed chronic GVHD or associated complications during follow-up.
Immunological reconstitution
PBL sub-populations gradually reconstituted in all patients after UCB transplantations. As shown in the figures, peripheral blood CD3-, CD19-, CD56-positive cells gradually increased up to almost normal age-related levels by 1-year post transplantation. We confirmed common gamma chain expression in all of these lymphocyte sub-populations. None of the patient had received further i.v. Ig replacement by 5–6 months after transplantation. Moreover, in all the patients, specific antibodies were produced against influenza, pertussis and measles following vaccination.
Growth and psychomotor development
As shown in Table 1, all three patients have shown normal psychomotor development and performance status to date. Only patient 3 had short stature of −2.4 s.d. at 21 months follow-up at post transplant, although the other two patients had normal growth development.
Discussion
We describe three patients with X-SCID successfully treated by CBT with a RIC regimen using FLU/BU. Both FLU/melphalan (LPAM) and FLU/BU regimens with or without modifications such as additional serotherapy are widely used for RIC regimen.23, 24 Although the FLU/LPAM regimen results in satisfactory engraftment,20 LPAM is toxic to both primitive and committed stem cells resulting in early onset and prolonged duration of neutropenia.25 In addition, LPAM-containing conditioning has been identified as a risk factor for VOD,26 possibly because LPAM induces more severe mucosal injury in the oral cavity and gastrointestinal tract compared with other agents.27, 28, 29 On the other hand, BU is preferentially toxic to committed stem cells.25 Although conditioning regimens including standard-dose BU are associated with a high rate of treatment-related complications due to organ toxicity, reduced-dose BU in combination with FLU is less myelosuppressive and toxic than FLU/LPAM or the standard-dose of BU regimen.30 Our patients achieved full donor T- and B-cell chimerism and clinical cure with minimum complications related to myelotoxicity. To date, none of our patients has shown mental or growth delay with the exception of patient 3, who had short stature of −2.4 s.d. at 21 months after CBT. Growth and endocrine function are commonly affected by standard myeloablative conditioning but this has not been reported with BU-based RIC for SCT during infancy and childhood.31 Patient 3 possibly has growth retardation following the BU-based RIC regimen because he underwent SCT at 3 months old. Further studies are necessary to determine the appropriate time for SCT to prevent late complications such as growth retardation.
Although CBT is more tolerant to HLA disparity, HLA incompatibility increases the incidence and intensity of GVHD and transplantation-related mortality.16, 32 The low incidence of severe GVHD in our series of patients possibly reflects the good match of the CB units. As unrelated CB is immediately available from CB banks, it is advantageous for patients with X-SCID who need urgent transplantation.
Pre-existing infections are the principal risk factors for a poor outcome after SCT. Patients 1 and 2 overcame pre-existing fungal and bacterial pneumonia, respectively, and recovered after transplantation. This suggests that, in addition to the appropriate antimicrobial therapy, early immunological reconstitution with minimum immune suppression after SCT using a RIC regimen contributes to the freedom from infection. Detection of fungal Ags or viral nucleotides by PCR-based techniques is critical for the early diagnosis and appropriate treatment of infections. On the other hand, early diagnosis of X-SCID before any infectious episodes, as shown in patient 3, is necessary for safe SCT, particularly in the patients with SCID. Recently developed neonatal mass-screening by quantitative assay for T-cell receptor excision circles might be useful for early diagnosis of patients with a wide variety of SCID genotypes.33, 34
In conclusion, CBT is a suitable alternative to BMT in X-SCID patients requiring SCT who have no matched sibling donors. RIC consisting of FLU and BU is an effective and safe treatment for such cases. Further studies are necessary to determine the appropriate time for SCT to minimize late complications such as growth retardation.
Acknowledgments
We appreciate the willingness of patients and parents to participate in this clinical study. This work was supported by grants from the Japanese Ministry of Health, Labor and Welfare.
The authors declare no conflict of interest.
References
- Fischer A, Le Deist F, Hacein-Bey-Abina S, André-Schmutz I, Basile Gde S, de Villartay JP, et al. Severe combined immunodeficiency. A model disease for molecular immunology and therapy. Immunol Rev. 2005;203:98–109. doi: 10.1111/j.0105-2896.2005.00223.x. [DOI] [PubMed] [Google Scholar]
- Grunebaum E, Mazzolari E, Porta F, Dallera D, Atkinson A, Reid B, et al. Bone marrow transplantation for severe combined immune deficiency. JAMA. 2006;295:508–518. doi: 10.1001/jama.295.5.508. [DOI] [PubMed] [Google Scholar]
- Filipovich AH. Hematopoietic cell transplantation for correction of primary immunodeficiencies. Bone Marrow Transplant. 2008;42:S49–S52. doi: 10.1038/bmt.2008.121. [DOI] [PubMed] [Google Scholar]
- Buckley RH, Schiff SE, Schiff RI, Market ML, Williams LW, Roberts JL, et al. Hematopoietic stem cell transplantation for the treatment of severe combined immunodeficiency. N Engl J Med. 1999;340:508–516. doi: 10.1056/NEJM199902183400703. [DOI] [PubMed] [Google Scholar]
- Antoine C, Muller S, Cant A, Cavazzano-Calvo M, Veys P, Vosen J, et al. Long-term survival and transplantation of haemopoietic stem cells for immunodeficiencies: report of the European experience 1968–1999. Lancet. 2003;361:553–560. doi: 10.1016/s0140-6736(03)12513-5. [DOI] [PubMed] [Google Scholar]
- Haddad E, Landais P, Friedrich N, Gerritsen B, CavazzanaCalvo M, Morgan G, et al. Long-term immune reconstitution and outcome after HLA non-identical T-cell depleted bone marrow transplantation for severe combined immunodeficiency: an European retrospective study of 116 patients. Blood. 1998;91:3646–3653. [PubMed] [Google Scholar]
- Gluckman E, Rocha V, Boyer-Chammard A, Locatelli F, Arcese W, Pasquini R, et al. Outcome of cord blood transplantation from related and unrelated donors. N Eng J Med. 1997;337:373–382. doi: 10.1056/NEJM199708073370602. [DOI] [PubMed] [Google Scholar]
- Barker JN, Davies SM, de For T, Ramsay NKC, Weisdof DJ, Wagner JE. Survival after transplantation of unrelated umbilical cord blood is comparable to that of leukocyte antigen-matched unrelated donor bone marrow: results of a matched-pair analysis. Blood. 2001;97:2957–2961. doi: 10.1182/blood.v97.10.2957. [DOI] [PubMed] [Google Scholar]
- Rocha V, Cornish J, Sievers EL, Filipovich A, Locatelli F, Peters C, et al. Comparison of outcomes of unrelated bone marrow and umbilical cord blood transplants in children with acute leukemia. Blood. 2001;97:2962–2971. doi: 10.1182/blood.v97.10.2962. [DOI] [PubMed] [Google Scholar]
- Wagner JE, Rosenthal J, Sweetman R, Shu XO, Davies S, Ramsay NKC, et al. Successful transplantation of HLA matched and HLA-mismatched umbilical cord blood from unrelated donor; analysis of engraftment and acute graft-versus host disease. Blood. 1996;88:795–802. [PubMed] [Google Scholar]
- Ziegner UHM, Ochs HD, Schanen C, Feig SA, Seyama K, Futatoni T, et al. Unrelated umbilical cord stem cell transplantation for X-linked immunodeficiencies. J Pediatr. 2001;138:570–573. doi: 10.1067/mpd.2001.112511. [DOI] [PubMed] [Google Scholar]
- Fagioli F, Biaxin E, Berger M, Nesi F, Saroglia EH, Minero R, et al. Successful unrelated cord blood transplantation in two children with severe combined immunodeficiency. Bone Marrow Transplant. 2003;31:133–136. doi: 10.1038/sj.bmt.1703800. [DOI] [PubMed] [Google Scholar]
- Knutsen AP, Wall DA. Umbilical cord blood transplantation in severe T cell immunodeficiency disorders: two-year experience. J Clin Immunol. 2000;20:466–476. doi: 10.1023/a:1026463900925. [DOI] [PubMed] [Google Scholar]
- Tsuji Y, Imai K, Kajiwara A, Aoki Y, Isoda T, Tomizawa D, et al. Hematopoieticstem cell transplantation for 30 patients with primary immunodeficiency diseases: 20 years' experience of a single team. Bone Marrow Transplant. 2006;37:469–477. doi: 10.1038/sj.bmt.1705273. [DOI] [PubMed] [Google Scholar]
- Bhattacharya A, Slatter MA, Chapman CE, Barge D, Jackson A, Flood TJ, et al. Single centre experience of umbilical cord stem cell transplantation for primary immunodeficiency. Bone Marrow Transplant. 2005;36:295–299. doi: 10.1038/sj.bmt.1705054. [DOI] [PubMed] [Google Scholar]
- Heredia CD, Ortega JJ, Diaz MA, Olive T, Badell I, Gonzalez-Vicent M, et al. Unrelated cord blood transplantation for severe combined immunodeficiency and other primary immunodeficiencies. Bone Marrow Transplant. 2008;41:627–633. doi: 10.1038/sj.bmt.1705946. [DOI] [PubMed] [Google Scholar]
- Cohen A, Uderzo C, van Lint MT, Esperou H, Rovelli A, Bakker B, et al. Final height of patients who underwent bone marrow transplantation for hematological disorders during childhood: a study by working party for late effects EBMT. Blood. 1999;93:4109–4115. [PubMed] [Google Scholar]
- Leung W, Hudson M, Zhu Y, Rivera GK, Ribeiro RC, Sandlund JT, et al. Late effects in survivors of infant leukemia. Leukemia. 2000;14:1185–1190. doi: 10.1038/sj.leu.2401818. [DOI] [PubMed] [Google Scholar]
- Leung W, Hudson MM, Strickland DK, Phipps S, Srivastava DK, Ribeiro RC, et al. Late effects of treatment in survivors of childhood acute myeloid leukemia. J Clin Oncol. 2000;18:3273–3279. doi: 10.1200/JCO.2000.18.18.3273. [DOI] [PubMed] [Google Scholar]
- Rao K, Amrolia PJ, Jones A, Cale CM, Naik P, King D, et al. Improved survival after unrelated donor bone marrow transplantation in children with primary immunodeficiency using a reduced-intensity conditioning regimen. Blood. 2005;105:879–885. doi: 10.1182/blood-2004-03-0960. [DOI] [PubMed] [Google Scholar]
- Iguchi A, Kobayashi R, Yoshida M, Kobayashi K, Matsuo K, Kitajima I, et al. Vascular endothelial growth factor (VEGF) is one of the causative and predictive cytokines of hepatic veno-occlusive disease (VOD) in stem cell transplantation. Bone Marrow Transplant. 2001;27:1173–1180. doi: 10.1038/sj.bmt.1703061. [DOI] [PubMed] [Google Scholar]
- Storb R, Deeg HJ, Farewell V, Doney K, Appelbaum F, Beatty P, et al. Marrow transplantation for severe aplastic anemia: Methotrexate and cyclosporine for prevention of acute graft-versus-host disease. Blood. 1986;68:119–125. [PubMed] [Google Scholar]
- Giralt S, Thall PF, Khouri I, Wang X, Braunschweig I, Ippolitti C, et al. Melphalan and purine analog-containing preparative regimens: reduced-intensity conditioning for patients with hematologic malignancies undergoing allogeneic progenitor cell transplantation. Blood. 2001;97:631–637. doi: 10.1182/blood.v97.3.631. [DOI] [PubMed] [Google Scholar]
- Slavin S, Nagler A, Naparstek E, Kapelushnik Y, Aker M, Cividalli G, et al. Nonmyeloablative stem cell transplantation and cell therapy as an alternative to conventional bone marrow transplantation with lethal cytoreduction for the treatment of malignant and nonmalignant hematologic diseases. Blood. 1998;91:756–763. [PubMed] [Google Scholar]
- Westerhof GR, Ploemacher RE, Boudewijin A, Blokland I, Dillingh JH, McGown AT, et al. Comparison of different busulfan analogues for depletion of hematopoietic stem cells and promotion of donor-type chimerism in murine bone marrow transplant recipients. Cancer Res. 2000;60:5470–5478. [PubMed] [Google Scholar]
- Iguchi A, Kobayashi R, Kaneda M, Kobayashi K. Plasma protein C is a useful clinical marker for hepatic veno-occlusive disease (VOD) in stem cell transplantation. Pediatr Blood Cancer. 2010;54:437–443. doi: 10.1002/pbc.22314. [DOI] [PubMed] [Google Scholar]
- Rothbarth J, Woutersen RA, Sparidans RW, van der Velde CJ, Mulder GJ. Melphalan antitumor efficacy and hepatotoxicity: The effect of variable infusion duration in the hepatic artery. J Pharmacol Exp Ther. 2003;305:1098–1103. doi: 10.1124/jpet.103.049379. [DOI] [PubMed] [Google Scholar]
- Blijlevens N, Schwenkglenks M, Bacon P, D'Addio A, Einsele H, Maertens J, et al. Prospective oral mucositis audit: Oral mucositis in patients receiving high-dose melphalan or BEAM conditioning chemotherapy—European Blood and Marrow Transplantation Mucositis Advisory Group. J Clin Oncol. 2008;26:1519–1525. doi: 10.1200/JCO.2007.13.6028. [DOI] [PubMed] [Google Scholar]
- Costa LJ, Micallef IN, Inwards DJ, Johnston PB, Porrata LF, Litzow MR, et al. Effect of the dose per body weight of conditioning chemotherapy on severity of mucositis and risk of relapse after autologous haematopoietic stem cell transplantation in relapsed diffuse large B cell lymphoma. Br J Haematol. 2008;143:268–273. doi: 10.1111/j.1365-2141.2008.07342.x. [DOI] [PubMed] [Google Scholar]
- Shimoni A, Hardan I, Shem-Yov N, Rand A, Herscovici C, Yerushalmi R, et al. Comparison between two fludarabine-based reduced-intensity conditioning regimens before allogeneic hematopoietic stem-cell transplantation: fludarabine/melphalan is associated with higher incidence of acute graft-versus-host disease and non-relapse mortality and lower incidence of relapase than fludarabine/busulfan. Leukemia. 2007;21:2109–2116. doi: 10.1038/sj.leu.2404886. [DOI] [PubMed] [Google Scholar]
- Bakker B, Oostdijk W, Bresters D, Walenkamp MJE, Vossen JM, Wit JM. Disturbances of growth and endocrine function after busulfan-based condtioning for haematopoietic stem cell transplantation during infancy and childhood. Bone marrow Transplant. 2004;33:1049–1056. doi: 10.1038/sj.bmt.1704481. [DOI] [PubMed] [Google Scholar]
- Barker JN, Scaradavou A, Stevens CE, Rubinstein P.Analysis of 608 umbilical cord blood transplants: HLA is a critical determinant of transplant-related mortality in the post- engraftment period even in the absence of acute graft-vs-host disease Blood 200511(Suppl 1102(abstract no. 92). [Google Scholar]
- Routes JM, Grossman WJ, Verbsky J, Laessig RH, Hoffman GL, Brokopp CD, et al. Statewide newborn screening for severe T-cell lymphopenia. JAMA. 2009;302:2465–2470. doi: 10.1001/jama.2009.1806. [DOI] [PubMed] [Google Scholar]
- Baker MW, Grossman WJ, Laessig RH, Hoffman GL, Brokopp CD, Kurtycz DF, et al. Development of a routine newborn screening protocol for severe combined immunodeficiency. J Allergy Clin Immunol. 2009;124:522–527. doi: 10.1016/j.jaci.2009.04.007. [DOI] [PubMed] [Google Scholar]
- Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFadden ET, et al. Toxicity and response criteria of the EasternCooperative Oncology Group. Am J Clin Oncol. 1982;5:649–655. [PubMed] [Google Scholar]