Abstract
Background
Over the last 15 years, visceral leishmaniasis (VL) has emerged as a public health concern in Tbilisi, the capital of Georgia.
Methodology/Principal Findings
Seroepidemiological surveys were conducted to determine the prevalence and incidence of infection in children and dogs within the main focus of VL, and to identify risk factors associated with human infection. Of 4,250 children investigated, 7.3% were positive by direct agglutination test in a baseline survey; an apparent incidence rate of 6.0% was estimated by one year follow-up. None of the seropositive children progressed to VL during the survey. Increased seropositivity at one year was predicted by presence at baseline of clustered flying insects (OR = 1.49; P = 0.001), perceived satisfactory sanitation (OR = 1.65; P<0.001), stray dogs (OR = 1.33; P = 0.023), and by persistent fever during the 6 months prior to baseline survey (OR = 14.2; P<0.001). Overall, 18.2% (107/588) of domestic and 15.3% (110/718) of stray dogs were seropositive by the rk39 dipstick test. Clinical VL signs were found in 1.3% of domestic and 2.9% of stray, seropositive dogs. Parasites isolated from human and dog samples were identified by PCR and phylogenetic analysis of the Leishmania 70 kDa heat-shock protein (HSP70) gene as Leishmania infantum.
Conclusions/Significance
There is an active focus of L. infantum transmission in Tbilisi with a high prevalence of human and canine infections.
Author Summary
Visceral leishmaniasis (VL) has emerged as a public health problem in Tbilisi, the capital of Georgia. Dogs are the main infection reservoirs for transmission by sand flies of Leishmania infantum to humans, many of whom may become infected without developing disease. Since majority of cases are in children we were interested to know the rate of infection in children and in dogs living within the area where cases of VL have been found, and what factors may affect the risk of infection. Using a test that detects the presence of antibodies in blood as a marker of infection, 7.3% of 4,250 children examined were positive at the baseline survey, and 6% became positive after one year. Overall, 18.2% of domestic and 15.3% of stray dogs were seropositive. The infected children were more apt to live in areas where clustered flying insects and stray dogs were observed, and were far more likely to have experienced a persistent fever in the 6 months before the survey. We conclude that there is very active transmission of L. infantum to both humans and dogs in Tbilisi, and that children remain at high risk of developing clinical disease and sub-clinical infection.
Introduction
Visceral leishmaniasis (VL) caused by parasites of the Leishmania donovani complex (L. donovani, L. infantum/L. chagasi) is a severe disease with a fatal outcome if left untreated. It is clinically characterized by low-grade fever, enlarged spleen and liver, and weight loss. Anthroponotic VL caused by L. donovani is endemic in East African countries, Northeast India, Nepal, and Bangladesh [1], [2]. In Latin America VL is zoonotic and the causative agent is L. chagasi [3]. In the Mediterranean basin zoonotic VL is caused by L. infantum, with incidence ranging from 0.02/100,000 to 8.53/100,000 [4]. Dogs are recognized as the primary reservoirs of zoonotic VL [5] and the prevalence of canine VL in Mediterranean countries varies from 1.1% to 48.4% [4], [6].
Zoonotic VL is widely distributed in countries of the former Soviet Union. Human cases are registered in Middle Asia and the Caucasus, including the Republic of Georgia [7]. Historically, leishmaniasis in Georgia has been sporadic and confined mainly to the eastern part of the country [8]. Current active foci are located in Tbilisi, the capital of Georgia, and in the region Shida Kartli (Figure 1A) [9], [10]. Since 1990, the number of VL cases recorded annually has increased substantially, from 10–12 cases in the early 90's, to 171 cases in 2008. Out of 1535 patients registered in Georgia during 1995–2008, 917 (60%) were from Tbilisi, with 17 fatal cases (official statistical records, National Center for Disease Control (NCDC), Tbilisi, Georgia).
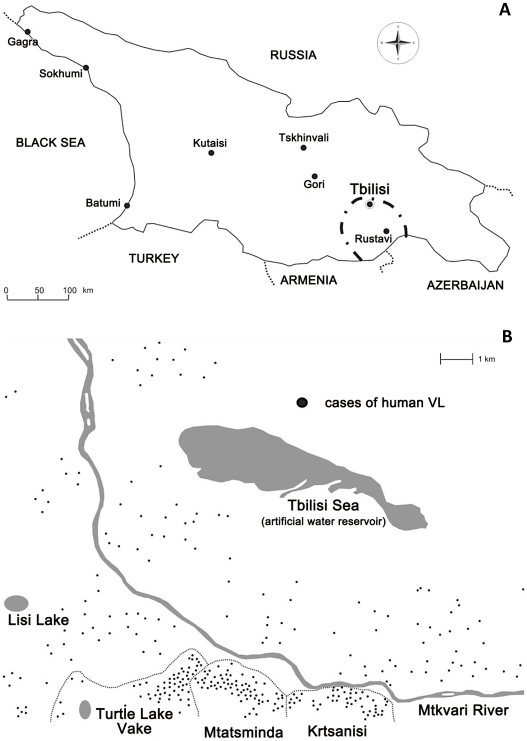
Figure 1. Maps showing distribution of human cases of visceral leishmaniasis in Georgia.
Map of Georgia (A) showing the principal active foci of VL in Tbilisi and Shida Kartli region (outlined by —·—). Map of Tbilisi (B) showing distribution of VL cases and location of Krtsanisi, Mtatsminda, and Vake districts surveyed in this study.
Due to the lack of VL surveillance system in Georgia, the prevalence and incidence of human and canine infection with Leishmania has remained unknown. Consequently, an appropriate control strategy has not been formulated. In this paper we present the results of a 3-year prospective study carried out during 2006–2008 in Tbilisi with the aims to determine the prevalence and incidence of Leishmania infection in children, as well as its prevalence in domestic and stray dogs, and to confirm the identity of the Leishmania parasite responsible for the disease.
Materials and Methods
Study area
Tbilisi is located at 500–800 m above sea level and is divided into northern and southern parts by the Mtkvari River. The southern part is mostly hilly with canyons and ravines; the northern part is terraced. Of the 10 urban districts, most of the VL cases (∼70%) registered during 1997–2004 originated from Krtsanisi, Mtatsminda and Vake located in the southern part of the city along the foot of the Mtatsminda Mountain. These 3 districts, representing active foci of VL, were selected as study sites (Figure 1B). The populations of the districts were: Krtsanisi – 27,047 (5,155 children 1–14 yrs of age); Mtatsminda – 43,133 (8,914); Vake – 27,053 (6,855).
Study design and sample collection
a. Human
Since approximately 70% of VL cases have been registered in children less than 14 years old, children 1–14 years of age were selected by randomized probability-proportional-to-size cluster sampling using 2002 census areas as clusters. All 1–14 years old children in each household (n = 4250) were surveyed at baseline and one year follow-up. The survey evaluated the medical history of each child, demographic characteristics of the household, and possible risk-factors associated with infection. After written consent from their parents or guardians, children underwent a physical examination for detection of clinical symptoms consistent with VL. A finger-prick blood sample was collected on filter paper (Whatman No. 3). Parents or guardians of serologically positive children were warned about possible development of the disease and asked to notify NCDC in the event clinical signs of VL occurred. The protocols of the study were approved by the Ethics Committee of NCDC.
b. Dogs
A cross-sectional study of domestic and stray dogs was performed to determine seroprevalence rate in canine populations. A total of 588 domestic dogs were selected from households in the human study areas and 718 stray dogs were also chosen from the same streets and neighboring suburbs. Once examined and sampled, a collar was placed on each stray dog to avoid repeated involvement of the same dogs in the survey. Owners provided consent for sample collection involving domestic dogs. Prior to sample collection, all dogs (domestic and stray) were examined for clinical signs of canine VL (alopecia, local or generalized lymphadenopathy, ocular and periocular lesions, epistaxis, onychogrypyhosis, emaciation, splenomegaly). Dogs were anesthetized intramuscularly with 2% acepromazine. Peripheral blood was taken from a femur or foreleg vein in Eppendorf® tubes (≥2 ml) and centrifuged for serum collection. Bone marrow aspirates were obtained from the iliac crest only from seropositive domestic dogs and used for smears, parasite culture and PCR. This animal protocol was approved by the Ethics Committee of the National Center for Disease Control (N05/04-02-492) according to and in compliance with the Georgian legislation and international norms in biological research.
Parasitological diagnosis
Bone marrow aspirates were smeared onto microscopic slides, dried, fixed with methanol, stained with Giemsa, and examined microscopically under a 100× oil immersion objective for presence of Leishmania amastigotes.
Direct Agglutination Test (DAT)
Standard procedures for human serodiagnosis by DAT [11], [12] were performed using the freeze-dried Leishmania antigen (Royal Tropical Institute, Amsterdam, Netherlands) to measure the antibody titer in finger-prick blood. Briefly, blood samples were eluted from filter papers overnight in serum diluent and serially diluted in twofold dilutions from 1∶100 to 1∶51,200 in 96-well plates. The DAT antigen was added to each dilution and the results were read after 18 h of incubation at room temperature.
To determine the cut off point for the DAT, blood samples from 100 children of the same age group living in the non-endemic Adjara region, in the city of Batumi (western Georgia) were tested at dilutions ranging from 1∶200 to 1∶25,600. None of the Batumi samples was positive at or above 1∶6,400 dilutions, thus samples giving titers of ≥1∶6,400 were considered seropositive. This cutoff is higher than is typically used for serodiagnosis of active VL [11], [12], therefore the specificity of the DAT in our survey should be at least as high as seen in the previous studies (119/124 = 96% in a mostly endemic population [11], 434/435 = 99.8% in a population with various other diseases [12]). Using the 1∶6,400 cutoff for detection of active cases in an endemic population, Oskam et al. [13] observed perfect specificity of the DAT (130/130 = 100%).
Samples that produced borderline agglutination at ≥1∶6,400 were scored as suspected positive, although the prevalence and incidence rates were calculated from only ‘confirmed’ seropositives. The suspected positives were included in the denominators of these calculations. Blood samples from patients with confirmed diagnosis of VL provided by the Research Institute of Medical Parasitology and Tropical Medicine (RIMPTM, Tbilisi, Georgia) were used as positive controls, and blood samples from DAT negative healthy individuals without signs or symptoms of leishmaniasis were used as negative controls.
rK39 dipstick test
The Kalazar Detect rapid test was performed according to the manufacturer's protocol (InBios International, Inc. Seattle, WA, USA) using 20 µl for each canine serum sample. The test was considered positive when a control line and test line appeared in the test area within 10 minutes, negative if only a single control line appeared, and invalid if none of the lines appeared.
Leishmania species identification
PCR analysis was performed to identify Leishmania parasites isolated from dogs and humans. DNA was extracted using the DNeasy Blood & Tissue Mini Kit (QIAGEN) from bone marrow cultures isolated from serologically positive domestic dogs and a culture established from the bone marrow of a patient with clinically confirmed VL diagnosis at the RIMPTM.
The primers Uni21 (5′ GGG GTT GGT GTA AAA TAG GCC 3′) and Lmj4 (5′ CTA GTT TCC CGC TCC GAG 3′) were used for initial PCR analysis [14]. The PCR was performed in a 25 µl reaction mixture, containing 12.5 µl of 2× FastStart PCR Master (Roche), 1 µl of each primer (10 µM), and 5 µl of template DNA (∼20 ng). Conditions for the reaction were as follows: initial denaturation at 94°C for 5 min, amplification for 35 cycles at 94°C–1 min, 60°C–1 min, 72°C–1.5 min, elongation at 72°C–10 min, and holding at 4°C. The PCR products were separated in 1.4% agarose gel, stained with ethidium bromide and visualized by ultraviolet light. L. infantum (MHOM/ES/00/UCM-1) and L. major MHOM/IL/80/Friedlin reference strains were included for comparison.
Further Leishmania identification was achieved by amplification of the 70 kDa heat-shock protein (HSP70) gene from dog and human samples using primers HSP70sen (5′-GACGGTGCCTGCCTACTTCAA-3′) and HSP70ant (5′-CCGCCCATGCTCTGGTACATC-3′) [15]. The PCR was completed in a volume of 50 µl contained 5 µl of 10× PCR Buffer II, 1 µl of each primer (10 µM), 50 ng of template DNA, 1 µl of Taq DNA polymerase (AccuPrime™ Taq DNA Polymerase System; Invitrogen) and 16 µl of H2O. The samples were incubated at 94°C for 2 minutes followed by 35 cycles of 94°C for 30 seconds, 60°C for 30 seconds and 68°C for 1.5 minutes. The amplified 1422 bp products were separated by electrophoresis through 1.2% agarose gel and visualized using SYBR Safe DNA gel stain (Invitrogen). The excised bands were extracted with Ultrafree-DA DNA Extraction (Millipore) and cleaned with three washes of ultrapure H2O through an YM-30 Microcon filter (Millipore). The cleaned products were ligated into pCR 4-TOPO (Invitrogen) and the plasmid transformed into One Shot TOP10 competent Escherichia coli (Invitrogen). Colonies were screened by PCR with HSP70sen and HSP70ant primers. Positive colonies were grown overnight and the plasmids purified with the PureLink Quick Plasmid Miniprep Kit (Invitrogen). The plasmids were sequenced bidirectionally with M13R (5′-CAGGAAACAGCTATGACC-3′) and M13F (5′-TGTAAAACGACGGCCAGT-3′).
Nucleotide sequences of the HSP70 gene of Leishmania parasites from dog and human samples were aligned against published sequences for L. infantum, L. donovani, L. tropica, L. major and L. aethiopica strains [16] using Clustal X, version 1.83 [17]. Phylogenetic analysis was conducted on the alignments using MEGA, version 3.1, generating trees by neighbor-joining and testing phylogeny with 1,000 replications to calculate node support [18].
Statistical analysis
Prevalence and incidence rates with confidence intervals were estimated using logistic models with generalized estimating equations (GEE) with a working independence assumption [19] to account for the within-family correlation. The responses for the logistic models were either the baseline DAT tests (for prevalence) or the one-year DAT tests only from those who were negative at baseline (for incidence). These estimates make no adjustment for the fact that the DAT is an imperfect diagnostic tool. However, because we expect the specificity to be near 100% (see DAT section above), and because imperfect sensitivity will only cause the apparent prevalence to be higher than the true prevalence [20], our prevalence estimates should be interpreted as estimating the lower bounds on the true prevalence. Our incidence estimates should also be interpreted cautiously as apparent incidences, since the adjustments needed to estimate true incidence [20] require sensitivity estimates. While the DAT sensitivity studies in the literature [21] typically refer to clinical disease for which the DAT results can be readily validated by parasite detection, our interest was subclinical infection for which there is so far no proven way to confirm the presence of parasites in the blood or other tissues.
We predicted seropositivity after one year using baseline disease status, region, and answers to survey questions at baseline using GEE cumulative logit models on the ordered responses: negative, suspected positive, and confirmed positive. We eliminated several survey questions that essentially duplicated other questions, and with the remaining questions we built one overall GEE model. Additionally, we built separate GEE models for each survey question using only that question plus baseline disease status and region. Odds ratios (ORs) are ratios of either the odds of confirmed seropositivity or the odds of suspected seropositivity [22]. ORs for yes/no questions estimate the ratio of the odds of disease for a subject who answered “yes” over the odds for a different hypothetical subject who answered “no” but is matched on all other variables in the model. Analyses were done in SAS version 9.1 with a p-value of <0.05 considered statistically significant.
Results
Seroprevalence and incidence of VL in children
We obtained baseline DAT responses on 99.6% of the subjects for a total of 4,250 children from 2,968 households; 1,459 children (34%) were 1–4 years and 2,791 (66%) were 5–14 years; 2,096 (49%) were girls and 2,150 (51%) were boys. The overall baseline prevalence was 7.3% with an incidence rate, estimated by a one year follow-up study, of 6.0%. The baseline seropositivity was highest in Mtatsminda (10.6%) followed by Vake (6.3%) and Krtsanisi (0.5%), while the incidence rate was highest in Vake (7.9%) followed by Mtatsminda (5.2%) and Krtasanisi (4.5%) (Table 1).
Table 1. Baseline prevalence and annual incidence (one year follow-up) of Leishmania seropositivity in surveyed children.
| Study area | Prevalencea | Incidence | ||
| N positive at baseline/N total at baseline (%) | 95% CIb | N positive at follow up and negative at baseline/N negative at baseline (%) | 95% CIb | |
| Vake | 95/1520 (6.25) | (5.06–7.70) | 110/1388 (7.93) | (6.26–9.99) |
| Mtatsminda | 211/1995 (10.58) | (9.17–12.17) | 92/1777 (5.18) | (4.02–6.64) |
| Krtsanisi | 4/735 (0.54) | (0.21–1.44) | 33/731 (4.51) | (3.11–6.52) |
| Total (all districts) | 310/4250 (7.29) | (6.48–8.20) | 235/3896 (6.03) | (5.15–7.05) |
Prevalence estimates do not include two children with active form of VL detected during the study.
95% confidence interval calculated using GEE methods to account for within family correlation.
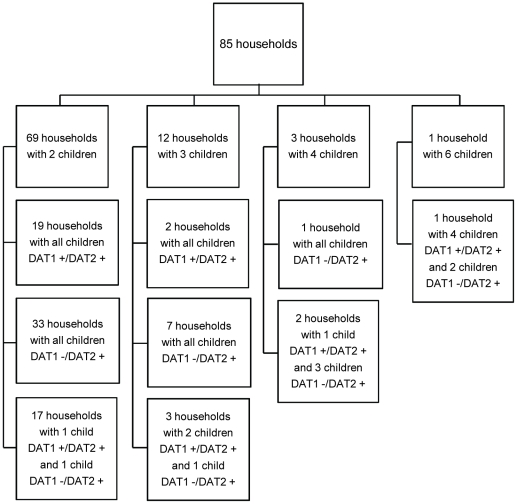
None of the seropositive children who were asymptomatic at baseline developed clinical signs of VL during the follow-up period, although 7% (296 children) of all investigated children were seropositive during both the baseline survey and one year follow-up. Fourteen of 310 children (4.5%) who were positive at baseline became negative in the follow-up survey. In 85 households with 2 or more children, all children were positive either in both baseline and follow up surveys, or seroconverted during the follow up period (Figure 2).
Figure 2. Households with all children tested seropositive for Leishmania at the baseline and/or follow-up surveys.
Eighty five households with 2 or more children where all children were DAT positive either in both baseline and follow up surveys, or seroconverted during the follow up period: DAT1− children negative in baseline survey, DAT1+ children positive in baseline survey, DAT2+ children positive in follow-up survey.
Hepatosplenomegaly, jointly with other clinical signs of active disease (weight loss, fatigue, anorexia, fever of unknown etiology), was detected in 2 children during clinical examination in the baseline survey. These children were excluded from the study and sent to the RIMPTM where the diagnosis of VL was parasitologically confirmed. Twenty two children with a previous history of VL were identified during the baseline study. Of these children, 11 (50%) were positive by DAT both in the baseline and follow-up surveys.
We predicted seropositivity at follow-up from only baseline disease status, region, and each baseline variable alone (Table 2, unadjusted OR) or by a complete model with all baseline variables included (Table 2, adjusted OR). We found that by both unadjusted and adjusted methods the odds of seropositivity at follow-up were increased by clustered flying insects at sunset/sunrise, perceived satisfactory sanitary conditions, and stray dogs; odds were decreased by the presence of nearby woodlands. The leading risk factor positively associated with seropositivity, with an OR of 14.2 (unadjusted) or 13.6 (adjusted) (p<0.001 for both methods), was fever lasting more than 2 weeks for which antibiotic therapy was not effective, and occurring during the 6 months prior to the day of the baseline interview (Table 2). No association of seropositivity by either method was found in relation to age or gender, or the use of nets on doors and windows. The use of repellents and the nearby existence of facilities for domestic animals were significant only after adjusting for the other variables. A history of lack of appetite at baseline was predictive of disease one year later but this effect disappears after adjusting for the other variables. Importantly, there was no association of seropositivity with having a pet dog(s) at home or in the yard by either method (Table 2).
Table 2. Odds ratios from unadjusted and adjusted cumulative logistic modelsa predicting seropositivity of children at follow-up.
| Parameters | Unadjusted ORb (95% CI) | P value | Adjusted ORc (95% CI) | P value |
| Fever lasting more than 2 weeks during the last 6 months for which antibiotic therapy was not effective | 14.2 (6.1–32.7) | <0.001 | 13.6 (4.5–40.7) | <0.001 |
| Woodlands with deciduous trees | 0.50 (0.38–0.67) | <0.001 | 0.66 (0.46–0.95) | 0.024 |
| Perceived satisfactory sanitary conditions | 1.65 (1.28–2.14) | <0.001 | 1.68 (1.25–2.28) | <0.001 |
| Clustered flying insects during the sunset/sunrise | 1.49(1.17–1.90) | 0.001 | 1.58 (1.16–2.15) | 0.004 |
| Stray dogs | 1.33 (1.04–1.70) | 0.023 | 1.72 (1.28–2.32) | <0.001 |
| Anorexia and/or sudden weight loss during the last 6 months | 8.2 (2.4–28.6) | 0.001 | 0.89 (0.18–4.38) | 0.888 |
| Pet dog at home | 1.30 (0.75–2.26) | 0.349 | 1.24 (0.63–2.44) | 0.525 |
| Dog in the yard | 0.50 (0.24–1.02) | 0.056 | 0.35 (0.11–1.06) | 0.062 |
| Facilities for poultry or animals in the yard or neighboring area | 0.69 (0.42–1.12) | 0.133 | 0.50 (0.26–0.96) | 0.036 |
| Rodents or their burrows in nearby area | 0.99 (0.79–1.24) | 0.929 | 0.83 (0.62–1.11) | 0.208 |
| Use of repellents for protection from insects | 0.86 (0.65–1.14) | 0.304 | 0.66 (0.47–0.94) | 0.020 |
| Use of nets on doors and windows | 1.02 (0.82–1.28) | 0.843 | 1.00 (0.78–1.30) | 0.993 |
Logistic models with generalized estimating equations (GEE) were used to predict disease status in 1–14 year old children in one year after baseline survey.
Model includes only baseline disease status and region plus the variable listed in the first row.
Model includes variable listed in first column plus: baseline disease status, previous diagnosis of Leishmaniasis, whether children visit parks in the evening, region, age (0–4, 5–9, 10+), sex, type of dwelling (isolated apartment, municipal apartment, private house), and floor.
Seroprevalence of VL in dogs
Overall, 107 of 588 domestic dogs (18.2%) and 110 of 718 stray dogs (15.3%) were found seropositive by the rK39 dipstick test (Table 3). Among the domestic dogs surveyed, the highest seroprevalence rate was in Vake – 31.2% (81 of 260 dogs), followed by Krtsanisi – 10.1% (14 of 138), and Mtatsminda – 6.3% (12 of 190). In the stray population, however, the highest seroprevalence rate was observed in Krtsanisi – 19.9% (36 of 181), followed by 14.5% (48 of 331) in Vake, and 12.6% (26 of 206) in Mtatsminda (Table 3). Clinical signs of canine VL were found only in 1.9% (2 of 107) and 2.7% (3 of 110) of seropositive domestic and stray dogs, respectively. However, the presence of Leishmania amastigotes was confirmed by microscopy in 49 of 75 (65%) bone marrow aspirates taken from seropositive domestic dogs. No statistically significant difference was observed between seropositivity and gender or age of investigated dogs. A difference in seroprevalence according to breed was observed among the 33 breeds and mongrels included in this study. A higher seropositivity was observed in Doberman Pinchers – 3 of 3, hounds – 4 of 5, pit bulls – 7 of 16, European shepherds – 2 of 6, German shepherds – 23 of 70, Drathaars – 1 of 3; compared to Caucasus shepherds – 1 of 31, Rottweilers – 1 of 15, poodles – 3 of 31, and mongrels – 31 of 232.
Table 3. Prevalence of Leishmania seropositivity in domestic and stray dogs from the study area.
| Study area | Domestic dogs | Stray dogs | ||
| N positive/N total (%) | 95% CI | N positive/N total (%) | 95% CI | |
| Vake | 81/260 (31.2) | (25.6–37.2) | 48/331 (14.5) | (10.9–18.8) |
| Mtatsminda | 12/190 (6.3) | (3.3–10.8) | 26/206 (12.6) | (8.4–17.9) |
| Krtsanisi | 14/138 (10.1) | (5.7–16.4) | 36/181 (19.9) | (14.3–26.5) |
| Total (all districts) | 107/588 (18.2) | (15.2–21.6) | 110/718 (15.3) | (12.8–18.2) |
Leishmania species identification
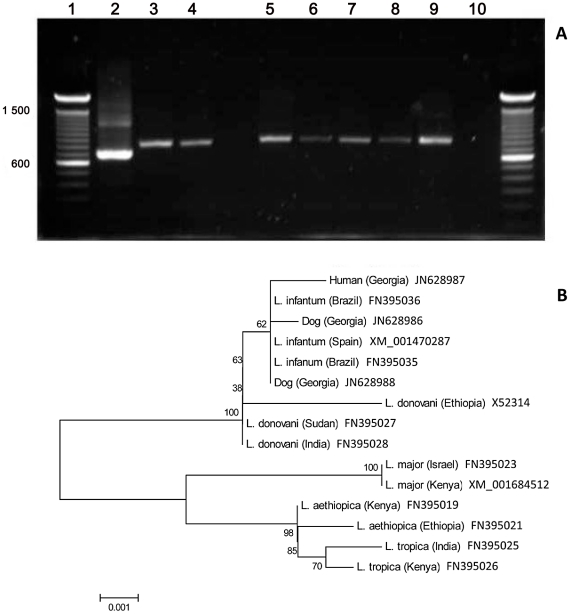
Amplification of DNA extracted from bone marrow of five seropositive dogs and one infected child with parasitologically confirmed VL produced single bands of approximately 800 bp identical in size to the L. infantum reference strain (Figure 3A). No amplification was observed in the negative control without template DNA.
Figure 3. Identification of Leishmania species isolated from a human and dogs.
(A) kDNA-PCR performed on DNA extracted from the bone marrow of a sick child and bone marrow cultures of five representative dogs. Lane 1, DNA size marker (100 bp ladder); lane 2, L. major (MHOM/IL/80/Friedlin); lane 3, L. infantum (MHOM/ES/00/UCM-1); lane 4, bone marrow, child, Tbilisi; lanes 5–9, bone marrow, dogs, Tbilisi; lane 10, negative control. (B) Phylogenetic analysis of Leishmania 70 kDa heat-shock protein (HSP70) genes. The sequences are represented by the Leishmania sp., country of origin in parentheses and GenBank nucleotide accession number. Node values indicate branch support.
Phylogenetic analysis of the 70 kDa heat-shock protein (HSP70) nucleotide sequences from DNA extracted from the dog and human bone marrow confirmed the identity of the parasite as L. infantum (Figure 3B).
Discussion
We report results of the first large seroepidemiological study of an important, emerging focus of human and canine VL in Tbilisi. Overall, 310 (7.3%) of 4,250 investigated children were found seropositive by DAT at the baseline survey, with 235 out of 3,896 seronegative children (6.0%) converting to positive within a one year follow-up. The choice of DAT as the serological test was justified by its prior application in a large number of field-based epidemiologic surveys of VL [23]–[25]. Selection of the cut-off titer of ≥1∶6,400 was based on results obtained from an extensive survey of children living in a non-endemic region of Georgia. For comparison, DAT titers of ≥1∶3,200 have been used by different investigators as a cut-off for the assessment of L. donovani/L. infantum seropositivity [23], [25]–[28], although Harith et al. [23] suggested a single serum dilution of 1∶6,400 for use in mass screenings. Moreover, they compared filter paper eluates with the corresponding serum samples and found no significant difference in DAT titers [23].
The true prevalence is likely higher than 7.3% because of the imperfect sensitivity of the DAT at the 1∶6,400 cut-off. With imperfect sensitivity, some of those that have VL will not be detected. By using probability laws, and assuming the sensitivity and specificity are known, we can get an estimate of the true prevalence [20] (see Text S1): True Prevalence = (Apparent Prevalence+Specificity−1)/(Sensitivity+Specificity−1). Using data on clinical VL from Oskam et al. (1999), we estimated the specificity as 100% (130/130) and the sensitivity as 71.7% (147/205), so that the corrected estimate of true prevalence using the above formula is 10.2%. These calculations depend on the estimates of sensitivity and specificity, and if the specificity is less than 100% then the True Prevalence estimate would lower than 10.2%, while if the sensitivity is lower (which we might expect for detecting subclinical disease) then the estimated true prevalence would be higher than 10.2%.
Different prevalence rates of asymptomatic seropositive humans have been found in Greece (0.5%) [6], Turkey (2.6%) [29], France (10%–28%) [30], Israel (3.0%) [31], Iran (1.6%) [32], and Azerbaijan (8.0%) (neighboring country of Georgia) [33]. Our data indicate the existence of a high frequency of asymptomatic human carriers in Tbilisi that conforms with results of studies reporting circulation of L. infantum in peripheral blood of asymptomatic healthy individuals [34]–[36]. All of the 310 children who were seropositive at baseline remained asymptomatic during the one year follow-up, and only 14 (4.5%) reverted to seronegative. Reversion of seropositive asymptomatic individuals to seronegative is well described [37], for example, in Kenya, where 75% of seropositive asymptomatic individuals became seronegative within 12–36 months without developing clinical signs of VL [38], or in Spain, where only 50% of the L. infantum asymptomatic seropositive blood donors remained positive after one year [39]. The question of how such changes in serological titers reflect changes in parasitemia remains unresolved.
Interestingly, we found clusters of seropositive children in 85 households with more than one child where all children living in the same household were either positive for anti-Leishmania antibody in both baseline and follow-up surveys, or converted to positive in the follow-up survey. None of these children developed clinical signs of VL during our study. Of these households, 33 (39%) were located in Vake district and half of them (17 households) on the same street; 48 households (56%) were in Mtatsminda district, and only 4 households (5%) in Krtsanisi district. Existence of these clusters, along with the absence of an association of seropositivity with domestic dogs, is consistent with the possibility that asymptomatic human cases serve as infection reservoirs.
One important objective of our survey was identification of the potential risk factors associated with seropositivity. We found that children who had persistent fever within the 6 months prior to the interview had a much higher probability (OR = 14.2 unadjusted; p<0.001) of becoming seropositive than children without this symptom. Some investigators have classified seropositive patients without any symptoms of VL as asymptomatic and seropositive patients having one or a combination of mild symptoms as subclinical. Subclinical patients can progress to the classic form of VL or resolve their symptoms over different periods of time [32], [37], [40], [41]. The seropositive children in our study with no other symptoms but fever can be considered as having a subclinical form of VL, and represent a significant and under reported morbid condition associated with L. infantum infection in this population.
Children living in the households that were perceived to have unsatisfactory sanitary conditions were at significantly lower risk of infection (p<0.001, both adjusted and unadjusted), as this perception may be associated with more attention to sanitary issues. Children living where clusters of blood-sucking insects were reported had higher risk of infection (p = 0.001 unadjusted, p = 0.004 adjusted). The results of the analysis indicated no significant association of seropositivity with sex or age of the investigated population, which conforms to the data from similar studies [6], [42], [43]. The presence of domestic livestock in the yard or the use of screens on the doors and windows or repellents against insects also did not have a statistically significant association with seropositive children. Decreased association with seropositivity in the presence of woodlands (p<0.001, unadjusted; p = 0.024, adjusted) suggests that woodlands act as a barrier to sand fly-human contact, likely due to the presence of wild animals as a preferred blood meal source.
Although Gavgani et al. [44] suggested that domestic dogs represent a significant risk factor for human VL, our findings did not show any association of seropositive children with domestic dogs, similar to other studies [35], [43]. By contrast, our analysis showed significant correlation with the nearby presence of stray dogs (, p = 0.024, unadjusted; p<0.001, adjusted). The increased risk of infection is associated with a high overall seroprevalence rate in our survey detected by the rK39 dipstick test for stray dogs (15.3%). This conforms well with the range of average seroprevalence rates of canine leishmaniasis reported from different Mediterranean countries: 20% in Portugal, 8.5% in Spain, 4–20% in France, 2–15% in Italy, 25% in Greece, 20% in Cyprus, 15.7% in Turkey [4]. It is curious that there was no association with seropositivity and the presence of domestic dogs despite the fact that the seroprevalence rate in domestic dogs was even higher (18.2%).
A high prevalence of infected asymptomatic dogs within L. infantum endemic areas of Spain, Italy, Brazil, Iran has been a consistent finding [5], [45]–[47]; lower rates were reported from Greece, Azerbaijan and Turkey [6], [33], [48]. While some authors reported that sand flies were not able to acquire infections from asymptomatic dogs [5], others proved that transmission to the vector from asymptomatic, seropositive dogs was possible [49]–[51]. Importantly, our own studies revealed that 49 of 75 bone marrow aspirates taken from seropositive domestic dogs were positive for amastigotes by microscopy, confirming their potential as infection reservoirs.
As for the risk factors associated with dogs becoming seropositive, there was no association with sex or age of dogs, or as mentioned, with domestic versus stray dogs. In contrast to our results, Gavgani et al. [44] showed significantly higher seroprevalence of L. infantum in stray (26.6%) in comparison with domestic (12.7%) dogs. Several studies showed no specific correlation between seropositivity and sex of dogs [47], [52], [53], although age-related differences supporting higher rates in older dogs have been reported [6], [47]. The influence of breed of dogs on seroprevalence is also inconclusive [6], [52], [53], though in general, higher rates of canine VL were reported in German shepherds, Dobermans, boxers and hounds, which favorably support the results of our survey.
Numerous PCR-based methods have been used for detection and identification of Leishmania parasites, including amplification of the kinetoplast DNA (kDNA) [14], [54]–[58]. Using the Uni21 and Lmj4 primer pair [14], the 800 bp amplicon was obtained from bone marrow DNA of five serologically positive dogs and one child with parasitologically confirmed VL. Though Leishmania-specific, this PCR product size is similar in L. infantum, L. donovani and L. tropica. Therefore, the identity of the Leishmania species from our study area was confirmed as L. infantum by sequence analysis of the 70 kDa heat-shock protein (HSP70), demonstrating that this species is responsible for human and canine VL in Tbilisi.
Taken together, our results confirm the presence of an extremely active focus of VL in Tbilisi with a high prevalence of human and canine infections. Importantly, recent data, demonstrating the spread of human VL cases to other parts of Tbilisi and to non-endemic territories of Georgia, indicate an urgent need to formulate and implement effective control measures.
Supporting Information
Appendix: Derivation of prevalence adjustment formula.
(DOC)
STROBE Checklist.
(DOC)
Acknowledgments
The authors would like to thank Hamide Aslan Suau for laboratory assistance.
Footnotes
The authors have declared that no competing interests exist.
This study was supported by the US Department of Health and Human Services (DHHS) Biotechnology Engagement Program through the International Science and Technology Center (grant ISTC G-1081/BTEP N89). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.World Health Organization (WHO) Manual of Visceral Leishmaniasis Control. Division of Control of Tropical Diseases. Geneva, Switzerland: WHO; 1996. [Google Scholar]
- 2.Dereure J, El-Safi SH, Bucheton B, Boni M, Kheir MM, et al. Visceral leishmaniasis in eastern Sudan: parasite identification in humans and dogs; host-parasite relationships. Microbes Infect. 2003;5:1103–1108. doi: 10.1016/j.micinf.2003.07.003. [DOI] [PubMed] [Google Scholar]
- 3.Romero GAS, Boelaert M. Control of Visceral Leishmaniasis in Latin America – A Systematic Review. PLoS Negl Trop Dis. 2010;4:e584. doi: 10.1371/journal.pntd.0000584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dujardin J-C, Campino L, Cañavate C, Dedet J-P, Gradoni L, et al. Spread of Vector-borne Diseases and Neglect of Leishmaniasis, Europe. Emerg Infect Dis. 2008;14:1013–1018. doi: 10.3201/eid1407.071589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Solano-Gallego L, Morell P, Arboix M, Alberola J, Ferrer L. Prevalence of Leishmania infantum Infection in Dogs Living in an Area of Canine Leishmaniasis Endemicity Using PCR on Several Tissues and Serology. J Clin Microbiol. 2001;39:560–563. doi: 10.1128/JCM.39.2.560-563.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Papadopoulou C, Kostoula A, Dimitriou D, Panagiou A, Bobojianni C, et al. Human and canine leishmaniasis in asymptomatic and symptomatic population in Northwestern Greece. J Infection. 2005;50:53–60. doi: 10.1016/j.jinf.2004.05.004. [DOI] [PubMed] [Google Scholar]
- 7.Maroli M, Krasnonos L, Gafurov I. Epidemiological and entomological survey in a focus of visceral leishmaniasis in Pap district (Fergana Valley) of Namangan region, Uzbekistan. Acta Trop. 2001;80:223–228. doi: 10.1016/s0001-706x(01)00179-6. [DOI] [PubMed] [Google Scholar]
- 8.Maruashvili GM. About types of visceral leishmaniasis foci. (Russian) Medical Parasitology (Moscow) 1961;30:188–190. [PubMed] [Google Scholar]
- 9.Bardjadze BG. Some questions about visceral leishmaniasis in Georgia. (Georgian) Sabchota Medicina (Tbilisi) 1966;2:28–32. [Google Scholar]
- 10.Chubabria G, Zenaishvili O. Modern concepts of visceral leishmaniasis in Georgia. (Russian) Medical Parasitology (Moscow) 2003;2:27–30. [Google Scholar]
- 11.Meredith SEO, Kroon NCM, Sondorp E, Seaman J, Goris MGA, et al. Leish-KIT, a Stable Direct Agglutination Test Based on Freeze-Dried Antigen for Serodiagnosis of Visceral Leishmaniasis. J Clin Microbiol. 1995;33:1742–1745. doi: 10.1128/jcm.33.7.1742-1745.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Abdallah KAA, Nour BYM, Schallig HDFH, Mergani A, Hamid Z, et al. Evaluation of the direct agglutination test based on freeze-dried Leishmania donovani promastigotes for the serodiagnosis of visceral leishmaniasis in Sudanese patients. Trop Med Int Health. 2004;9:1127–1131. doi: 10.1111/j.1365-3156.2004.01308.x. [DOI] [PubMed] [Google Scholar]
- 13.Oskam L, Nieuwenhuijs JL, Hailu A. Evaluation of the direct agglutination test (DAT) using freeze-dried antigen for the detection of anti-Leishmania antibodies in stored sera from various patient groups in Ethiopia. T Roy Soc Trop Med H. 1999;93:275–277. doi: 10.1016/s0035-9203(99)90021-4. [DOI] [PubMed] [Google Scholar]
- 14.Anders G, Eisenberger C, Jonas F, Greenblatt C. Distinguishing Leishmania tropica and Leishmania major in the Middle East using the polymerase chain reaction with kinetoplast DNA-specific primers. T Roy Soc Trop Med H. 2002;96:S1/87-S1192. doi: 10.1016/s0035-9203(02)90057-x. [DOI] [PubMed] [Google Scholar]
- 15.Garcia L, Kindt A, Bermudez H, Llanos-Cuentas A, De Doncker S, et al. Culture-independent species typing of neotropical Leishmania for clinical validation of a PCR-based assay targeting heat shock protein 70 genes. J Clin Microbiol. 2004;42:2294–2297. doi: 10.1128/JCM.42.5.2294-2297.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Montalvo AM, Fraga J, Monzote L, Montano I, De Doncker S, et al. Heat-shock protein 70 PCR-RFLP: a universal simple tool for Leishmania species discrimination in the New and Old World. Parasitology. 2010;137:1159–1168. doi: 10.1017/S0031182010000089. [DOI] [PubMed] [Google Scholar]
- 17.Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG. The CLUSTAL_X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res. 1997;25:4876–4882. doi: 10.1093/nar/25.24.4876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kumar S, Tamura K, Nei M. MEGA3: Integrated software for Molecular Evolutionary Genetics Analysis and sequence alignment. Brief Bioinform. 2004;5:150–163. doi: 10.1093/bib/5.2.150. [DOI] [PubMed] [Google Scholar]
- 19.Liang KY, Zeger SL. Longitudinal Data Analysis Using Generalized Linear Models Biometrika. 1986;73:13–22. [Google Scholar]
- 20.Yanagawa T, Gladen BC. Estimating disease rates from a diagnostic test. Am J Epidemiol. 1984;119:1015–1023. doi: 10.1093/oxfordjournals.aje.a113806. [DOI] [PubMed] [Google Scholar]
- 21.Chappuis F, Rijal S, Soto A, Menten J, Boelaert M. A meta-analysis of the diagnostic performance of the direct agglutination test and rK39 dipstick for visceral leishmaniasis. BMJ. 2006;333:723. doi: 10.1136/bmj.38917.503056.7C. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Agresti A. Categorical Data Analysis. Wiley: New York; 1990. [Google Scholar]
- 23.Harith AE, Kolk AHJ, Leeuwenburg J, Muigai R, Huigen E. Improvement of a Direct Agglutination Test for Field Studies of Visceral Leishmaniasis. J Clin Microbiol. 1988;26:1321–1325. doi: 10.1128/jcm.26.7.1321-1325.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bimal S, Das VNR, Sinha PK, Gupta AK, Verma N, et al. Usefulness of the direct agglutination test in the early detection of subclinical Leishmania donovani infection: a community-based study. Ann Trop Med Parasit. 2005;99:743–749. doi: 10.1179/136485905X65107. [DOI] [PubMed] [Google Scholar]
- 25.Schenkel K, Rijal S, Koirala S, Koirala S, Vanlerberghe V, et al. Visceral leishmaniasis in southeastern Nepal: A cross-sectional survey on Leishmania donovani infection and its risk factors. Trop Med Int Health. 2006;11:1792–1799. doi: 10.1111/j.1365-3156.2006.01735.x. [DOI] [PubMed] [Google Scholar]
- 26.Harith AE, Slappendel RJ, Reiter I, Van Knapen F, De Korte P. Application of a Direct Agglutination Test for Detection of Specific Anti-Leishmania Antibodies in the Canine Reservoir. J Clin Microbiol. 1989;27:2252–2257. doi: 10.1128/jcm.27.10.2252-2257.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.El-Masum MA, Evans DA, Minter DM, El-Harith A. Visceral leishmaniasis in Bangladesh: the value of DAT as a diagnostic tool. T Roy Soc Trop Med H. 1995;89:185–186. doi: 10.1016/0035-9203(95)90487-5. [DOI] [PubMed] [Google Scholar]
- 28.Joshi AB, Singhasivanon P, Khusmith S, Fungladda W, Nandy A. Evaluation of direct agglutination test (DAT) as an immunodiagnostic tool for diagnosis of visceral leishmaniasis in Nepal. The Southeast Asian J Trop Med Pub Health. 1999;30:583–585. [PubMed] [Google Scholar]
- 29.Doğan N, Bör O, Dinleyici EC, Töz SO, Ozbel Y. Investigation of anti-Leishmania seroprevalence by different serologic assays in children inhabiting in the northwestern part of Turkey. Mikrobiyol Bul. 2008;42:103–11. [PubMed] [Google Scholar]
- 30.Marty P, Izri A, Ozon C, Haas P, Rosenthal E, et al. A century of leishmaniasis in Alpes-Maritimes, France. Ann Trop Med Parasit. 2007;101:563–74. doi: 10.1179/136485907X229121. [DOI] [PubMed] [Google Scholar]
- 31.Adini I, Ephros M, Chen J, Jaffe CL. Asymptomatic Visceral Leishmaniasis, Northern Israel. Emerg Infect Dis. 2003;9:397–398. doi: 10.3201/eid0903.020297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Fakhar M, Motazedian MH, Hatam GR, Asgari Q, Kalantari M, et al. Asymptomatic human carriers of Leishmania infantum: possible reservoirs for Mediterranean visceral leishmaniasis in southern Iran. Ann Trop Med Parasit. 2008;102:577–583. doi: 10.1179/136485908X337526. [DOI] [PubMed] [Google Scholar]
- 33.Tagi-zade TA, Gasanida GB, Safianova VM, Shalmiev GB, Gadzhibekova EA. Visceral leishmaniasis in Ordubad District, Nakhichevan ASSR. Medical Parasitology (Moscow) 1989;3:22–27. [PubMed] [Google Scholar]
- 34.Le Fichoux Y, Quaranta J-F, Aufeuvre J-P, Lelievre A, Marty P, et al. Occurrence of Leishmania infantum Parasitemia in Asymptomatic Blood Donors Living in an Area of Endemicity in Southern France. J Clin Microbiol. 1999;37:1953–1957. doi: 10.1128/jcm.37.6.1953-1957.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Costa CHN, Srewart JM, Gomes RBB. Asymptomatic human carriers of Leishmania chagasi. Am J Trop Med Hyg. 2002;66:334–337. doi: 10.4269/ajtmh.2002.66.334. [DOI] [PubMed] [Google Scholar]
- 36.Martín-Sánchez J, Pineda JA, Morillas-Márquez F, García-García JA, Acedo C, et al. Detection of Leishmania infantum kinetoplast DNA in peripheral blood from asymptomatic individuals at risk for parenterally transmitted infections: relationship between PCR results and other Leishmania infection markers. Am J Trop Med Hyg. 2004;70:545–548. [PubMed] [Google Scholar]
- 37.Diza E, Kansouzidou A, Gerou S, Vezyri E, Metallidis S, et al. Leishmaniases in Northern Greece: seroprevalence of the infection and incidence of the disease during the period 2001–2006. Eur J Clin Microbiol Infect Dis. 2008;27:997–1003. doi: 10.1007/s10096-008-0538-y. [DOI] [PubMed] [Google Scholar]
- 38.Schaefer KU, Kurtzhals JA, Gachihi GS, Muller AS, Kager PA. A prospective sero–epidemiological study of visceral leishmaniasis in Baringo district, Rift Valley province, Kenya. T Roy Soc Trop Med H. 1995;89:471–475. doi: 10.1016/0035-9203(95)90070-5. [DOI] [PubMed] [Google Scholar]
- 39.Riera C, Fisa R, Udinab M, Gállegoa M, Portusa M. Detection of Leishmania infantum cryptic infection in asymptomatic blood donors living in an endemic area (Eivissa, Balearic Islands, Spain) by different diagnostic methods. T Roy Soc Trop Med H. 2004;98:102–110. doi: 10.1016/s0035-9203(03)00015-4. [DOI] [PubMed] [Google Scholar]
- 40.Badaro R, Jones TC, Carvalho EM, Sampaio D, Reed SG, et al. New perspectives on a subclinical form of visceral leishmaniasis. J Infect Dis. 1986;154:1003–11. doi: 10.1093/infdis/154.6.1003. [DOI] [PubMed] [Google Scholar]
- 41.Gama MEA, Costa JML, Gomes CMC, Corbett CEP. Subclinical Form of the American Visceral Leishmaniasis. Mem Inst Oswaldo Cruz, Rio de Janeiro. 2004;99:889–893. doi: 10.1590/s0074-02762004000800018. [DOI] [PubMed] [Google Scholar]
- 42.Crescente JAB, Silveira FT, Lainson R, Gomes CMC, Laurenti MD, et al. A cross-sectional study on the clinical and immunological spectrum of human Leishmania (L.) infantum chagasi infection in the Brazilian Amazon region. T Roy Soc Trop Med H. 2009;103:1250–1256. doi: 10.1016/j.trstmh.2009.06.010. [DOI] [PubMed] [Google Scholar]
- 43.Barão SC, Camargo-Neves VLF, Resende RM, Da Silva LJ. Human Asymptomatic Infection in Visceral Leishmaniasis: A Seroprevalence Study in an Urban Area of Low Endemicity. Preliminary Results. Am J Trop Med Hyg. 2007;77:1051–1053. [PubMed] [Google Scholar]
- 44.Gavgani ASM, Mohite H, Edrissian GH, Mohebali M, Davies CR. Domestic dog ownership in Iran is a risk factor for human infection with Leishmania infantum. Am J Trop Med Hyg. 2002;67:511–515. doi: 10.4269/ajtmh.2002.67.511. [DOI] [PubMed] [Google Scholar]
- 45.Otranto D, Paradies P, De Caprariis D, Stanneck D, Testini G, et al. Toward Diagnosing Leishmania infantum Infection in Asymptomatic Dogs in an Area Where Leishmaniasis Is Endemic. Clinical and Vaccine Immunology. 2009;16:337–343. doi: 10.1128/CVI.00268-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Dantas-Torres F, Felino de Brito EM, Brandão-Filho PS. Seroepidemiological survey on canine leishmaniasis among dogs from an urban area of Brazil. Vet Parasitol. 2006;140:54–60. doi: 10.1016/j.vetpar.2006.03.008. [DOI] [PubMed] [Google Scholar]
- 47.Mohebali M, Hajjaran H, Hamzavi Y, Mobedi I, Arshi S, et al. Epidemiological aspects of canine visceral leishmaniosis in the Islamic Republic of Iran. Vet Parasitol. 2005;129:243–251. doi: 10.1016/j.vetpar.2005.01.010. [DOI] [PubMed] [Google Scholar]
- 48.Ertabaklar H, Toz SO, Ozkan AT, Rastgeldi S, Balcioglu IC, et al. Serological and entomological survey in a zoonotic visceral leishmaniasis focus of North Central Anatolia, Turkey: Corum province. Acta Trop. 2005;93:239–246. doi: 10.1016/j.actatropica.2005.01.002. [DOI] [PubMed] [Google Scholar]
- 49.Molina R, Amela C, Nieto J, San-Andrés M, González F, et al. Infectivity of dogs naturally infected with Leishmania infantum to colonized Phlebotomus perniciosus. T Roy Soc Trop Med H. 1994;88:491–493. doi: 10.1016/0035-9203(94)90446-4. [DOI] [PubMed] [Google Scholar]
- 50.Michalsky EM, Rocha MF, Da Rocha Lima AC, França-Silva JC, Pires MQ, et al. Infectivity of seropositive dogs, showing different clinical forms of leishmaniasis, to Lutzomyia longipalpis phlebotomine sand flies. Vet Parasitol. 2007;147:67–76. doi: 10.1016/j.vetpar.2007.03.004. [DOI] [PubMed] [Google Scholar]
- 51.Alvar J, Molina R, San Andre ÂS, Tesouro M, Nieto J, et al. Canine leishmaniasis: clinical, parasitological and entomological follow-up after chemotherapy. Ann Trop Med Parasit. 1994;88:371–378. doi: 10.1080/00034983.1994.11812879. [DOI] [PubMed] [Google Scholar]
- 52.Morillas F, Sanchez Rabasco F, Ocaña J, Martin-Sanchez J, Ocaña-Wihelmi J, et al. Leishmaniosis in the focus of the Axarquía region, Malaga province, southern Spain: a survey of the human, dog, and vector. Parasitol Res. 1996;82:569–570. doi: 10.1007/s004360050164. [DOI] [PubMed] [Google Scholar]
- 53.Palatnik-de-Sousa CB, Dos Santos WR, França-Silva JC, da Costa RT, Reis AB, et al. Impact of canine control on the epidemiology of canine and human visceral leishmaniasis in Brazil. Am J Trop Med Hyg. 2001;65:510–517. doi: 10.4269/ajtmh.2001.65.510. [DOI] [PubMed] [Google Scholar]
- 54.Nasereddin A, Azmi K, Jaffe CL, Ereqat S, Amro A, et al. Kinetoplast DNA heterogeneity among Leishmania infantum strains in central Israel and Palestine. Vet Parasitol. 2009;161:126–130. doi: 10.1016/j.vetpar.2008.12.003. [DOI] [PubMed] [Google Scholar]
- 55.Abdeen ZA, Sawalha SS, Eisenberger CL, Khanfar HM, Greenblatt CL, et al. Epidemiology of visceral leishmaniasis in the Jenin District, West Bank: 1989–1998. Am J Trop Med Hyg. 2002;66:329–333. doi: 10.4269/ajtmh.2002.66.329. [DOI] [PubMed] [Google Scholar]
- 56.Manna L, Vitale F, Reale S, Caracappa S, Pavone LM, et al. Comparison of different tissue sampling for PCR-based diagnosis and follow-up of canine visceral leishmaniosis. Vet Parasitol. 2004;125:251–262. doi: 10.1016/j.vetpar.2004.07.019. [DOI] [PubMed] [Google Scholar]
- 57.Cascio A, Calattini S, Colomba C, Scalamogna C, Galazzi M, et al. Polymerase Chain Reaction in the Diagnosis and Prognosis of Mediterranean Visceral Leishmaniasis in Immunocompetent Children. Pediatrics. 2002;109:e27. doi: 10.1542/peds.109.2.e27. [DOI] [PubMed] [Google Scholar]
- 58.Noyes HA, Reyburn H, Bailey W, Smith D. A nested-PCR-schizodeme method for identifying Leishmania kinetoplast minicircle classes directly from clinical samples and its application to the study of epidemiology of Leishmania tropica in Pakistan. J Clin Microbiol. 1998;36:2877–2881. doi: 10.1128/jcm.36.10.2877-2881.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Appendix: Derivation of prevalence adjustment formula.
(DOC)
STROBE Checklist.
(DOC)