Abstract
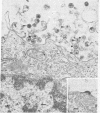
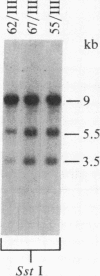
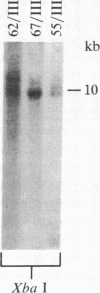
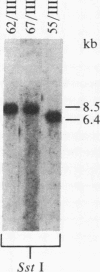
We isolated six human T-cell lymphotropic virus type I (HTLV-I)-transformed T-cell clones carrying the phenotypic markers of helper and suppressor T cells. Five of the transformed T-cell clones produced infectious HTLV-I, but one (clone 55) contained a defective provirus and was therefore not competent for viral replication. To test whether there is interference between HTLV-I and the cytopathic virus HTLV-III in infection and/or their biological effects, we superinfected these T-cell clones with HTLV-III. The recipient cells that we used displayed either the OKT4 or the OKT8 membrane antigens (helper or suppressor phenotype, respectively). The superinfection was successful in all cases, regardless of phenotype of the recipient cells and status of viral production. Both HTLV-III and HTLV-I were expressed by the infected cell lines containing complete HTLV-I proviruses, as demonstrated by electron microscopy and immunofluorescence. However, only HTLV-III in the virus mixture obtained from the culture supernatants was transmitted to the human neoplastic T-cell line H9. The nonproducer clone 55 did not express HTLV-I upon superinfection with HTLV-III. HTLV-III exerted its cytopathic effect on all but one of the superinfected T-cell clones 15-20 days after infection. The exception, clone 67, is also the only cell clone that expresses the phenotypic marker of suppressor T lymphocytes (OKT8); the other clones carry the OKT4 antigen, correlated with helper functions. The virus released from the superinfected clone 67 is cytopathic for fresh peripheral and umbilical-cord blood lymphocytes, suggesting that cellular factors, rather than a genetic change in the virus, may be responsible for the lack of cytopathic effect of HTLV-III on the suppressor T-cell clone 67.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aldovini A., De Rossi A., Feinberg M. B., Wong-Staal F., Franchini G. Molecular analysis of a deletion mutant provirus of type I human T-cell lymphotropic virus: evidence for a doubly spliced x-lor mRNA. Proc Natl Acad Sci U S A. 1986 Jan;83(1):38–42. doi: 10.1073/pnas.83.1.38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allan J. S., Coligan J. E., Lee T. H., McLane M. F., Kanki P. J., Groopman J. E., Essex M. A new HTLV-III/LAV encoded antigen detected by antibodies from AIDS patients. Science. 1985 Nov 15;230(4727):810–813. doi: 10.1126/science.2997921. [DOI] [PubMed] [Google Scholar]
- Arya S. K., Guo C., Josephs S. F., Wong-Staal F. Trans-activator gene of human T-lymphotropic virus type III (HTLV-III). Science. 1985 Jul 5;229(4708):69–73. doi: 10.1126/science.2990040. [DOI] [PubMed] [Google Scholar]
- Barré-Sinoussi F., Chermann J. C., Rey F., Nugeyre M. T., Chamaret S., Gruest J., Dauguet C., Axler-Blin C., Vézinet-Brun F., Rouzioux C. Isolation of a T-lymphotropic retrovirus from a patient at risk for acquired immune deficiency syndrome (AIDS). Science. 1983 May 20;220(4599):868–871. doi: 10.1126/science.6189183. [DOI] [PubMed] [Google Scholar]
- Clapham P., Nagy K., Cheingsong-Popov R., Exley M., Weiss R. A. Productive infection and cell-free transmission of human T-cell leukemia virus in a nonlymphoid cell line. Science. 1983 Dec 9;222(4628):1125–1127. doi: 10.1126/science.6316502. [DOI] [PubMed] [Google Scholar]
- Dalgleish A. G., Beverley P. C., Clapham P. R., Crawford D. H., Greaves M. F., Weiss R. A. The CD4 (T4) antigen is an essential component of the receptor for the AIDS retrovirus. Nature. 1984 Dec 20;312(5996):763–767. doi: 10.1038/312763a0. [DOI] [PubMed] [Google Scholar]
- Fisher A. G., Collalti E., Ratner L., Gallo R. C., Wong-Staal F. A molecular clone of HTLV-III with biological activity. Nature. 1985 Jul 18;316(6025):262–265. doi: 10.1038/316262a0. [DOI] [PubMed] [Google Scholar]
- Franchini G., Mann D. L., Popovic M., Zicht R. R., Gallo R. C., Wong-Staal F. HTLV-I infection of T and B cells of a patient with adult T-cell leukemia-lymphoma (ATLL) and transmission of HTLV-I from B cells to normal T cells. Leuk Res. 1985;9(11):1305–1314. doi: 10.1016/0145-2126(85)90116-x. [DOI] [PubMed] [Google Scholar]
- Gallo R. C., Salahuddin S. Z., Popovic M., Shearer G. M., Kaplan M., Haynes B. F., Palker T. J., Redfield R., Oleske J., Safai B. Frequent detection and isolation of cytopathic retroviruses (HTLV-III) from patients with AIDS and at risk for AIDS. Science. 1984 May 4;224(4648):500–503. doi: 10.1126/science.6200936. [DOI] [PubMed] [Google Scholar]
- Hahn B. H., Shaw G. M., Arya S. K., Popovic M., Gallo R. C., Wong-Staal F. Molecular cloning and characterization of the HTLV-III virus associated with AIDS. Nature. 1984 Nov 8;312(5990):166–169. doi: 10.1038/312166a0. [DOI] [PubMed] [Google Scholar]
- Harada S., Koyanagi Y., Yamamoto N. Infection of HTLV-III/LAV in HTLV-I-carrying cells MT-2 and MT-4 and application in a plaque assay. Science. 1985 Aug 9;229(4713):563–566. doi: 10.1126/science.2992081. [DOI] [PubMed] [Google Scholar]
- Kalyanaraman V. S., Sarngadharan M. G., Robert-Guroff M., Miyoshi I., Golde D., Gallo R. C. A new subtype of human T-cell leukemia virus (HTLV-II) associated with a T-cell variant of hairy cell leukemia. Science. 1982 Nov 5;218(4572):571–573. doi: 10.1126/science.6981847. [DOI] [PubMed] [Google Scholar]
- Kan N. C., Franchini G., Wong-Staal F., DuBois G. C., Robey W. G., Lautenberger J. A., Papas T. S. Identification of HTLV-III/LAV sor gene product and detection of antibodies in human sera. Science. 1986 Mar 28;231(4745):1553–1555. doi: 10.1126/science.3006245. [DOI] [PubMed] [Google Scholar]
- Klatzmann D., Champagne E., Chamaret S., Gruest J., Guetard D., Hercend T., Gluckman J. C., Montagnier L. T-lymphocyte T4 molecule behaves as the receptor for human retrovirus LAV. Nature. 1984 Dec 20;312(5996):767–768. doi: 10.1038/312767a0. [DOI] [PubMed] [Google Scholar]
- Kramarsky B., Sarkar N. H., Moore D. H. Ultrastructural comparison of a virus from a Rhesus-monkey mammary carcinoma with four oncogenic RNA viruses. Proc Natl Acad Sci U S A. 1971 Jul;68(7):1603–1607. doi: 10.1073/pnas.68.7.1603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee T. H., Coligan J. E., Sodroski J. G., Haseltine W. A., Salahuddin S. Z., Wong-Staal F., Gallo R. C., Essex M. Antigens encoded by the 3'-terminal region of human T-cell leukemia virus: evidence for a functional gene. Science. 1984 Oct 5;226(4670):57–61. doi: 10.1126/science.6089350. [DOI] [PubMed] [Google Scholar]
- Longo D. L., Gelmann E. P., Cossman J., Young R. A., Gallo R. C., O'Brien S. J., Matis L. A. Isolation of HTLV-transformed B-lymphocyte clone from a patient with HTLV-associated adult T-cell leukaemia. Nature. 1984 Aug 9;310(5977):505–506. doi: 10.1038/310505a0. [DOI] [PubMed] [Google Scholar]
- Mann D. L., Clark J., Clarke M., Reitz M., Popovic M., Franchini G., Trainor C. D., Strong D. M., Blattner W. A., Gallo R. C. Identification of the human T cell lymphoma virus in B cell lines established from patients with adult T cell leukemia. J Clin Invest. 1984 Jul;74(1):56–62. doi: 10.1172/JCI111418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mann D. L., Popovic M., Murray C., Neuland C., Strong D. M., Sarin P., Gallo R. C., Blattner W. A. Cell surface antigen expression in newborn cord blood lymphocytes infected with HTLV. J Immunol. 1983 Oct;131(4):2021–2024. [PubMed] [Google Scholar]
- Manzari V., Wong-Staal F., Franchini G., Colombini S., Gelmann E. P., Oroszlan S., Staal S., Gallo R. C. Human T-cell leukemia-lymphoma virus (HTLV): cloning of an integrated defective provirus and flanking cellular sequences. Proc Natl Acad Sci U S A. 1983 Mar;80(6):1574–1578. doi: 10.1073/pnas.80.6.1574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyoshi I., Yoshimoto S., Kubonishi I., Taguchi H., Shiraishi Y., Ohtsuki Y., Akagi T. Transformation of normal human cord lymphocytes by co-cultivation with a lethally irradiated human T-cell line carrying type C virus particles. Gan. 1981 Dec;72(6):997–998. [PubMed] [Google Scholar]
- Poiesz B. J., Ruscetti F. W., Gazdar A. F., Bunn P. A., Minna J. D., Gallo R. C. Detection and isolation of type C retrovirus particles from fresh and cultured lymphocytes of a patient with cutaneous T-cell lymphoma. Proc Natl Acad Sci U S A. 1980 Dec;77(12):7415–7419. doi: 10.1073/pnas.77.12.7415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poiesz B. J., Ruscetti F. W., Reitz M. S., Kalyanaraman V. S., Gallo R. C. Isolation of a new type C retrovirus (HTLV) in primary uncultured cells of a patient with Sézary T-cell leukaemia. Nature. 1981 Nov 19;294(5838):268–271. doi: 10.1038/294268a0. [DOI] [PubMed] [Google Scholar]
- Popovic M., Flomenberg N., Volkman D. J., Mann D., Fauci A. S., Dupont B., Gallo R. C. Alteration of T-cell functions by infection with HTLV-I or HTLV-II. Science. 1984 Oct 26;226(4673):459–462. doi: 10.1126/science.6093248. [DOI] [PubMed] [Google Scholar]
- Popovic M., Lange-Wantzin G., Sarin P. S., Mann D., Gallo R. C. Transformation of human umbilical cord blood T cells by human T-cell leukemia/lymphoma virus. Proc Natl Acad Sci U S A. 1983 Sep;80(17):5402–5406. doi: 10.1073/pnas.80.17.5402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Popovic M., Sarin P. S., Robert-Gurroff M., Kalyanaraman V. S., Mann D., Minowada J., Gallo R. C. Isolation and transmission of human retrovirus (human t-cell leukemia virus). Science. 1983 Feb 18;219(4586):856–859. doi: 10.1126/science.6600519. [DOI] [PubMed] [Google Scholar]
- Popovic M., Sarngadharan M. G., Read E., Gallo R. C. Detection, isolation, and continuous production of cytopathic retroviruses (HTLV-III) from patients with AIDS and pre-AIDS. Science. 1984 May 4;224(4648):497–500. doi: 10.1126/science.6200935. [DOI] [PubMed] [Google Scholar]
- Rho H. M., Gallo R. C. Biochemical and immunological properties of the DNA polymerase and RNAase H activities of purified feline leukemia virus reverse transcriptase. Cancer Lett. 1980 Sep;10(3):207–221. doi: 10.1016/0304-3835(80)90073-7. [DOI] [PubMed] [Google Scholar]
- Robert-Guroff M., Blayney D. W., Safai B., Lange M., Gelmann E. P., Gutterman J. W., Mansell P. W., Goedert J. L., Groopman J. E., Steigbigel N. H. HTLV-I-specific antibody in AIDS patients and others at risk. Lancet. 1984 Jul 21;2(8395):128–131. doi: 10.1016/s0140-6736(84)91047-x. [DOI] [PubMed] [Google Scholar]
- Robert-Guroff M., Ruscetti F. W., Posner L. E., Poiesz B. J., Gallo R. C. Detection of the human T cell lymphoma virus p19 in cells of some patients with cutaneous T cell lymphoma and leukemia using a monoclonal antibody. J Exp Med. 1981 Dec 1;154(6):1957–1964. doi: 10.1084/jem.154.6.1957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaw G. M., Harper M. E., Hahn B. H., Epstein L. G., Gajdusek D. C., Price R. W., Navia B. A., Petito C. K., O'Hara C. J., Groopman J. E. HTLV-III infection in brains of children and adults with AIDS encephalopathy. Science. 1985 Jan 11;227(4683):177–182. doi: 10.1126/science.2981429. [DOI] [PubMed] [Google Scholar]
- Slamon D. J., Press M. F., Souza L. M., Murdock D. C., Cline M. J., Golde D. W., Gasson J. C., Chen I. S. Studies of the putative transforming protein of the type I human T-cell leukemia virus. Science. 1985 Jun 21;228(4706):1427–1430. doi: 10.1126/science.2990027. [DOI] [PubMed] [Google Scholar]
- Sodroski J., Patarca R., Rosen C., Wong-Staal F., Haseltine W. Location of the trans-activating region on the genome of human T-cell lymphotropic virus type III. Science. 1985 Jul 5;229(4708):74–77. doi: 10.1126/science.2990041. [DOI] [PubMed] [Google Scholar]
- Sodroski J., Rosen C., Goh W. C., Haseltine W. A transcriptional activator protein encoded by the x-lor region of the human T-cell leukemia virus. Science. 1985 Jun 21;228(4706):1430–1434. doi: 10.1126/science.2990028. [DOI] [PubMed] [Google Scholar]
- Southern E. M. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol. 1975 Nov 5;98(3):503–517. doi: 10.1016/s0022-2836(75)80083-0. [DOI] [PubMed] [Google Scholar]
- Staal S. P., Hartley J. W., Rowe W. P. Isolation of transforming murine leukemia viruses from mice with a high incidence of spontaneous lymphoma. Proc Natl Acad Sci U S A. 1977 Jul;74(7):3065–3067. doi: 10.1073/pnas.74.7.3065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tedder R. S., Shanson D. C., Jeffries D. J., Cheingsong-Popov R., Clapham P., Dalgleish A., Nagy K., Weiss R. A. Low prevalence in the UK of HTLV-I and HTLV-II infection in subjects with AIDS, with extended lymphadenopathy, and at risk of AIDS. Lancet. 1984 Jul 21;2(8395):125–128. doi: 10.1016/s0140-6736(84)91046-8. [DOI] [PubMed] [Google Scholar]
- Wahl G. M., Stern M., Stark G. R. Efficient transfer of large DNA fragments from agarose gels to diazobenzyloxymethyl-paper and rapid hybridization by using dextran sulfate. Proc Natl Acad Sci U S A. 1979 Aug;76(8):3683–3687. doi: 10.1073/pnas.76.8.3683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weller S. K., Joy A. E., Temin H. M. Correlation between cell killing and massive second-round superinfection by members of some subgroups of avian leukosis virus. J Virol. 1980 Jan;33(1):494–506. doi: 10.1128/jvi.33.1.494-506.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong-Staal F., Gallo R. C. The family of human T-lymphotropic leukemia viruses: HTLV-I as the cause of adult T cell leukemia and HTLV-III as the cause of acquired immunodeficiency syndrome. Blood. 1985 Feb;65(2):253–263. [PubMed] [Google Scholar]
- de Rossi A., Aldovini A., Franchini G., Mann D., Gallo R. C., Wong-Staal F. Clonal selection of T lymphocytes infected by cell-free human T-cell leukemia/lymphoma virus type I: parameters of virus integration and expression. Virology. 1985 Jun;143(2):640–645. doi: 10.1016/0042-6822(85)90405-2. [DOI] [PubMed] [Google Scholar]
- di Marzo Veronese F., Sarngadharan M. G., Rahman R., Markham P. D., Popovic M., Bodner A. J., Gallo R. C. Monoclonal antibodies specific for p24, the major core protein of human T-cell leukemia virus type III. Proc Natl Acad Sci U S A. 1985 Aug;82(15):5199–5202. doi: 10.1073/pnas.82.15.5199. [DOI] [PMC free article] [PubMed] [Google Scholar]