Abstract
Objective
Respiratory complications account for a major cause of morbidity and mortality in subjects with spinal cord injury (SCI) due to paralysis of the expiratory muscles and the consequent inability to generate effective cough. We demonstrated previously that effective cough can be restored in SCI via spinal cord stimulation (SCS) with disc leads positioned on the lower thoracic and upper lumbar spinal cord via laminotomy incisions. In this study, the effectiveness of wire leads, which can be placed using minimally invasive techniques, to activate the expiratory muscles was evaluated.
Design
Animal study.
Setting
Research laboratory.
Animals
Dogs (n = 8).
Interventions
In separate trials, disc and wire leads were inserted onto the dorsal epidural space at the T9, T11, and L1 spinal cord levels. Effects of electrical stimulation with disc, single wire, and two wire leads placed in parallel were compared.
Outcome measures
Airway pressure generation following stimulation with disc and various configurations of wire leads were compared.
Results
Several different configurations of wire leads resulted in airway pressures that were similar to those generated with monopolar stimulation with disc leads (MSDLs). For example, combined monopolar stimulation with parallel wire leads at the T9 + T11 and T9 + L1 levels resulted in airway pressures that were 103.5 ± 6.4 and 101.9 ± 7.0%, respectively, of those achieved with MSDL. Bipolar stimulation with parallel wire leads at T9–T11 and T9–L1 resulted in airway pressures that were 94.2 ± 3.4 and 96.8 ± 5.0%, respectively, of the pressures achieved with MSDL. Other wire configurations were also evaluated, but were generally less effective.
Conclusion
These results suggest that specific configurations of wire leads, which can be placed via minimally invasive techniques, result in comparable activation of the expiratory muscles compared to disc leads and may be a useful technique to restore cough in persons with SCI.
Keywords: Spinal cord injuries, Cough reflex, Electrical stimulation, Respiration, Respiratory complication, Pulmonary function
Introduction
In patients with spinal cord injury (SCI), respiratory complications account for a major cause of morbidity and mortality due to paralysis of the expiratory muscles and the consequent inability to generate an effective cough.1–19 As a result, most of these patients suffer from a markedly reduced ability to clear airway secretions resulting in the development of atelectasis and recurrent respiratory tract infections. In recent human studies, we demonstrated that an effective cough can be restored in subjects with SCI via stimulation of electrodes positioned on the lower thoracic and upper lumbar spinal cord.20–22 This procedure involves the placement of disc leads at the T9, T11, and L1 spinal cord levels on the dorsal epidural surface via laminotomy incisions, and a ground electrode positioned in the subcutaneous tissue. This method is successful in achieving an effective means of secretion clearance, as demonstrated by the generation of large airway pressures and peak expiratory airflow rates, which approximate values observed during maximum cough efforts in normal people.20,21 Importantly, this technique significantly facilitates airway clearance, reduces the difficulty in raising secretions, reduces the need for caregiver support for secretion management, reduces the incidence of respiratory tract infections, and improves life quality.20,22
While successful in restoring an effective cough in each subject, this technique requires an invasive procedure for disc lead placement (typically, 4–6 hours of operating room time) and 2–3-day hospital admission. As a result, this method entails the potential for significant risk, including bleeding, infection, and discomfort, in an already compromised patient population. The hypothesis of the present investigation is that comparable activation of the expiratory muscles can be achieved with wire leads, which can be inserted via minimally invasive techniques, eliminating the need for the much more invasive laminotomy procedure. The other potential advantage of this procedure is that it can be performed on an out-patient basis resulting in significant reductions in cost and inconvenience.
In the present animal study, airway pressure generation was measured following stimulation utilizing commercially available wire leads, positioned at the T9, T11, and L1 spinal levels, and compared, in separate trials, to pressures generated during spinal cord stimulation (SCS) with disc leads at the same levels. We demonstrated that comparable levels of expiratory muscle activation can be generated with specific configurations of wire leads, which may provide a minimally invasive method of cough restoration in patients with SCI.
Methods
All experiments were performed following the approval of the Institutional Animal Care and Use Committee of Case Western Reserve University.
Studies were performed on eight mongrel dogs (weight: 13.7 ± 0.6 kg). All animals were anesthetized with pentobarbital sodium (25 mg/kg) given intravenously. Anesthetic level was maintained with supplemental doses of pentobarbital sodium (1–2 mg/kg), as needed. A large bore cuffed endotracheal tube was placed directly into the trachea in the mid-cervical region and sutured in place. A small catheter was positioned in the femoral vein to provide supplemental anesthesia and administer fluids. A separate catheter was placed in the femoral artery to monitor blood pressure. A heating blanket (Harvard Apparatus, Cambridge, MA, USA) was used to maintain body temperature at 38 ± 0.5°C. End-tidal PCO2 was monitored continuously at the tracheal opening with a rapidly responding CO2 analyzer (DRE Inc., Louisville, KY, USA). A differential pressure transducer (Validyne, MP45, Northridge, CA, USA) was used to measure airway pressure at the airway opening. Airway pressure was always measured under conditions of airway occlusion at functional residual capacity (FRC).
Laminectomies were performed at the T8 and L2 levels through which leads were inserted onto the dorsal epidural space in the midline and advanced to the T9, T11, and L1 spinal cord levels according to previously described techniques.23–26 The disc lead was an eight-electrode assembly with 4-mm-diameter platinum electrode contacts embedded in polyurethane plastic with 10 mm contact spacing (disposable subdural eight contact strip electrode (1 × 8), Ad-Tech Medical Instrument Corp, Racine, WI, USA). In separate trials, wire leads with four platinum–iridium (90%/10%) 6 mm electrode contacts were inserted onto the dorsal epidural space in the midline. The wire electrode span (66 mm) allows for combined stimulation at the T9 + T11 levels. Use of two longitudinally placed leads was necessary for combined T9 and L1 stimulation. The effects of two wire leads placed in parallel were also evaluated. In this instance, each wire lead was positioned just lateral to the midline. Exact placement of the leads was determined by direct visualization. The effects of monopolar SCS with two parallel wire leads in which each of the electrode contacts at the T9, T11, and L1 levels were connected together and functioned as a common cathode (Y connection) were also evaluated. A remote ground electrode was positioned in the back musculature. Since we have previously demonstrated, both in animal24,25 and human20,21 studies, that any combination of two of the three spinal cord levels tested (T9, T11, and L1) resulted in maximum pressure generation, we focused our evaluation to stimulation of each site individually and the simultaneous stimulation of various combinations of two sites. The effects of both monopolar and bipolar stimulation were also evaluated.
Two grass square-wave pulse stimulators (Model S88, Grass Instruments, West Warwick, RI, USA) equipped with stimulus isolation units (SIU5, Grass Instruments) were used to provide electrical stimulation. Similar to the SCS clinical trials, stimulation was applied over a range of stimulus amplitudes (10–40 V) at 50 Hz with a pulse width of 0.2 millisecond. The upper stimulus amplitude limit was set at 40 V since this amplitude resulted in near maximal activation of the expiratory muscles in our prior studies and higher amplitudes generally resulted in unwanted lower-extremity movement. Stimulations were applied in random sequence at each stimulus site. In instances requiring a remote ground electrode, a 1-cm disc electrode was implanted in the back musculature. Since cough is a ballistic maneuver requiring high airway pressure development, our focus was on the measurement of maximum airway pressure development, which was used as an index of maximum expiratory muscle force development.
All recordings were monitored and stored on a computer utilizing a data acquisition and analyzing system (Spike 2 with 1401 interface, Cambridge Electronic Design, Cambridge, UK).
Data and statistical analyses
Airway pressure generation over a wide range of stimulus amplitudes was compared for each electrode contact at each of the three spinal cord levels and in various combinations. Since monopolar SCS with the disc leads, when applied in clinical trials,20,21 resulted in airway pressure generation that approximated pressures generated with a normal maximum cough, airway pressure generation achieved during SCS with these leads served as our gold standard to which all comparisons were made. Data are reported as mean ± SE. Statistical analysis was performed using a repeated measures analysis of variance and paired t-test. A P value <0.05 was taken as indicative of statistical significance.
Results
SCS was well tolerated in each animal and not associated with significant changes in blood pressure or pulse rate throughout the stimulation trials.
The effects of SCS with the disc and each of the wire lead arrangements on airway pressure generation are shown below in separate figures. At the top of each figure, the specific configuration is shown schematically.
With the disc and each of the various wire lead configurations, there were progressive increases in pressure development with increasing stimulus amplitude. In addition, there was a tendency toward a plateau in pressure development between 30 and 40 V. Airway pressure generation with maximal stimulus intensity is shown for all stimulus configurations in Table 1.
Table 1.
Maximum airway pressure generation during electrical stimulation with disc and different configurations of wire leads
| Monopolar disc lead | Monopolar single wire lead | Bipolar single wire lead | Monopolar parallel wire lead | Bipolar parallel wire lead | Monopolar parallel wire lead: Y connection | |
|---|---|---|---|---|---|---|
| Paw (cmH2O) | Paw (cmH2O) | Paw (cmH2O) | Paw (cmH2O) | Paw (cmH2O) | Paw (cmH2O) | |
| T9 | 58.4 ± 1.7 | 42.4 ± 2.4* | 56.3 ± 4.3 | 50.3 ± 8.6 | ||
| T11 | 41.4 ± 3.3 | 28.6 ± 2.3* | 36.0 ± 3.1 | 35.3 ± 7.5 | ||
| L1 | 14.6 ± 1.5 | 11.0 ± 1.2 | 12.1 ± 1.6 | 13.7 ± 4.3 | ||
| T9 + T11 | 78.8 ± 2.4 | 64.0 ± 4.8* | 48.5 ± 2.7* | 82.0 ± 6.9 | 74.0 ± 3.0 | 66.3 ± 5.4* |
| T9 + L1 | 82.9 ± 3.1 | 67.5 ± 6.5* | 54.3 ± 5.0* | 84.6 ± 6.9 | 79.6 ± 3.3 | 66.7 ± 6.2* |
*P < 0.05 compared to monopolar disc electrode.
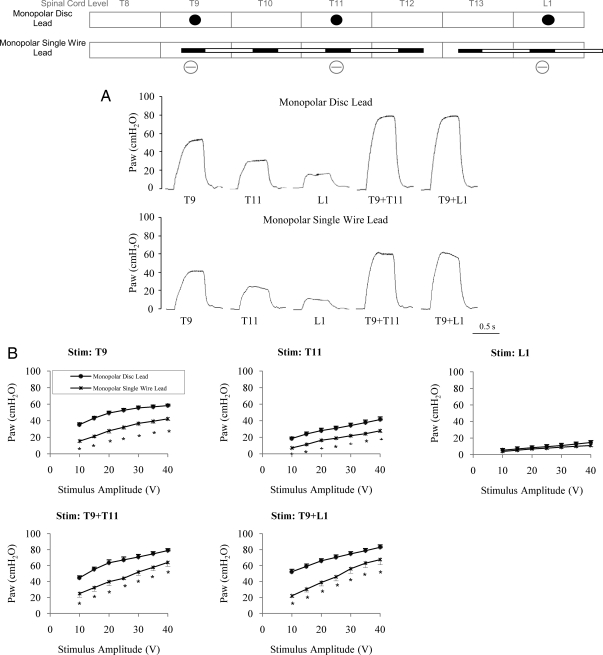
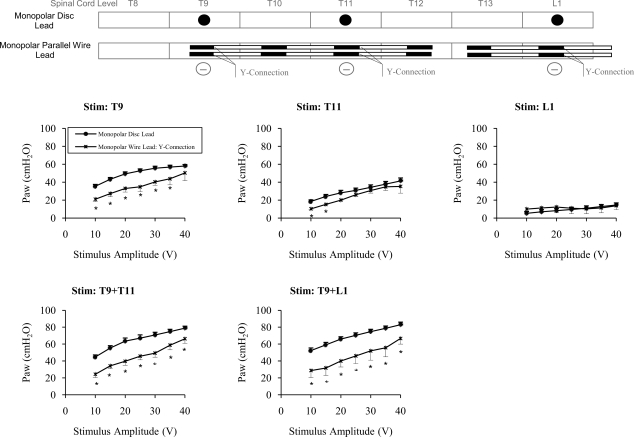
Evaluation of monopolar SCS with a single-wire lead (Fig. 1)
Figure 1.
Effects of monopolar SCS (40 V, 50 Hz, 0.2 millisecond) at the T9, T11, and L1 spinal levels alone and combined T9 + T11 and T9 + L1 stimulation on airway pressure development with the disc and wire leads. A schematic of this configuration is placed at the top of this and all subsequent figures. The effect of SCS is shown for one animal in Panel A; mean results are presented in Panel B. P < 0.05 compared to monopolar disc. See text for further explanation.
During monopolar stimulation, the spinal cord electrode contact functioned as a cathode with a remote ground. The effects of monopolar SCS at the T9, T11, L1, T9 + T11, and T9 + L1 spinal levels on airway pressure development with the disc and wire lead systems are shown for one animal in Fig. 1A. SCS with the wire lead system resulted in substantial airway pressure development, but in each instance was smaller than that achieved with the disc lead. The mean relationships between stimulus amplitude and airway pressure generation for each lead type and at each spinal cord level is shown in Fig. 1B. During stimulation of the wire lead contacts located at the T9 and T11 spinal levels, each alone, airway pressure generation was significantly smaller at all stimulus amplitudes when compared with the disc lead contacts at the same levels. However, there were no significant differences between each of the lead types during L1 stimulation. During combined stimulation at T9 + T11 and T9 + L1, airway pressures were significantly smaller during stimulation with the wire lead at all stimulus amplitudes (Table 1). Nonetheless, airway pressures generated during stimulation with the wire lead contacts were substantial, ranging between 68.9 ± 4.3 and 81.2 ± 6.0% of those generated during maximal monopolar stimulation with disc leads (MSDLs).
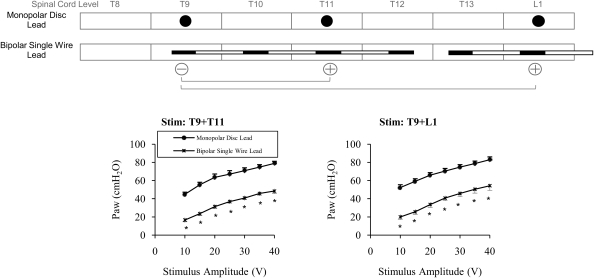
Evaluation of bipolar SCS with a single-wire lead (Fig. 2)
Figure 2.
Mean relationships between stimulus amplitude and airway pressure generation for disc leads during monopolar stimulation (T9 + T11 and T9 + L1) and wire leads during bipolar stimulation (T9–T11 and T9–L1). *P < 0.05 compared to monopolar disc. See text for further explanation.
In this arrangement, the T9 contact served as the cathode and either the T11 or L1 contact served as the anode, in separate trials. During T9–T11 and T9–L1 stimulation, pressure generation with this configuration resulted in substantially smaller airway pressure generation at each level of current (P < 0.05). During maximum stimulation, airway pressures generated with the bipolar wire lead were 61.7 ± 3.2 and 65.3 ± 5.0%, respectively, of those generated with MSDL (Table 1).
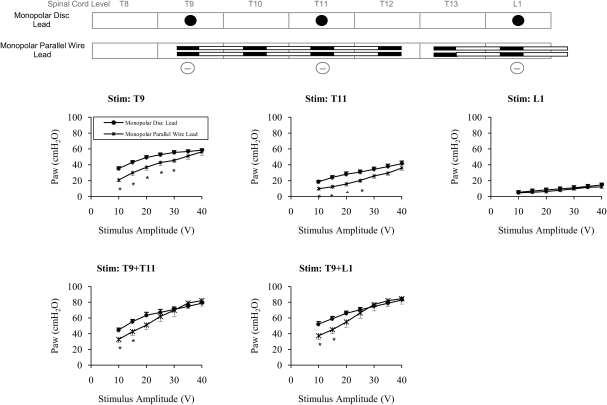
Evaluation of monopolar SCS with two parallel wire leads (Fig. 3)
Figure 3.
Mean relationships between stimulus amplitude and airway pressure generation during monopolar stimulation with disc and parallel wire leads. *P < 0.05 compared to monopolar disc. See text for further explanation.
In this arrangement, two wire leads are placed in parallel with each contact serving as a separate cathode with a remote ground. During T9 and T11 stimulation, each alone, airway pressure generation was significantly smaller with the wire leads compared to MSDL during stimulation at 10–30 V, but not significantly different at higher voltages. There were no significant differences during L1 stimulation alone. During T9 + T11 and T9 + L1 stimulation, airway pressure generation was significantly smaller with the wire leads at 10 and 15 V. However, there were no significant differences in airway pressure generation at higher voltages. Maximum airway pressures ranged between 82.8 ± 5.4 and 103.5 ± 6.4% of those achieved with MSDL (NS).
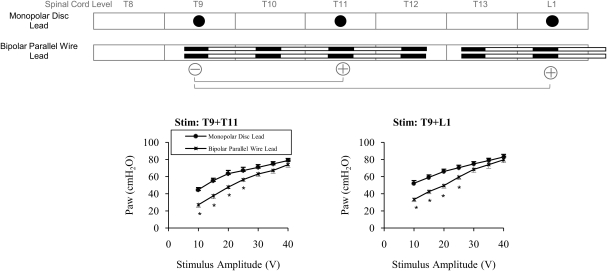
Evaluation of bipolar SCS with two parallel wire leads (Fig. 4)
Figure 4.
Mean relationships between stimulus amplitude and airway pressure generation during monopolar stimulation with the disc leads (T9 + T11 and T9 + L1) and bipolar stimulation with the parallel wire configuration (T9–T11 and T9–L1). *P < 0.05 compared to monopolar disc. See text for further explanation.
The configuration in this trial is the same as that presented in Fig. 3 except that simultaneous bipolar stimulation was applied with each of the contacts at the T9 and T11 levels, and in separate trials at the T9 and L1 levels. As in Fig. 2, the T9 contact served as the cathode and the T11 or the L1 contact served at the anode, in separate trials. During bipolar stimulation of the wire lead at T9–T11 and T9–L1, airway pressures were significantly smaller than those achieved with MSDL at low stimulus amplitudes (10–25 V). However, there were no significant differences in pressure generation at higher stimulus amplitudes (Table 1).
Evaluation of monopolar SCS with two parallel wire leads: Y connection (Fig. 5)
Figure 5.
Mean relationships between stimulus amplitude and airway pressure generation during MSDL and the parallel wire Y-connection configuration. *P < 0.05 compared to monopolar disc. See text for further explanation.
In this instance, each contact at the T9 level and, in turn, at the T11 and L1 levels, functioned as a single cathode, in separate trials, with a remote ground (Fig. 5). During T9 stimulation alone, airway pressures were significantly smaller at most stimulus amplitudes (10–35 V) but not significantly different at 40 V. During T11 stimulation alone, airway pressure generation was significantly smaller at 10 and 15 V but not significantly different at higher stimulus amplitudes. There were no differences during L1 stimulation. During combined stimulation at the T9 + T11 and T9 + L1 levels, airway pressure generation was significantly smaller with the wire lead at all stimulus amplitudes (Fig. 5, Table 1).
Discussion
In previous animal studies performed in our laboratory, we demonstrated that a major portion of the expiratory muscles could be activated reproducibly and in concert by the application of electrical stimulation in the region of the lower thoracic and upper lumbar spinal cord with disc leads.23–26 Importantly, the results of these studies translated well into clinical trials in which SCS resulted in near maximum expiratory muscle activation and was successful in restoring an effective cough mechanism in subjects with SCI.20–22 Airway pressures generated with the disc leads, therefore, represented our gold standard with which other leads could be compared. The results of the present study demonstrate that the application of electrical stimulation with wire leads at the same spinal cord levels also results in substantial positive airway pressures. Moreover, airway pressures generated with electrical stimulation applied with monopolar or bipolar wire lead configurations, in a parallel arrangement, are not significantly different than those generated with MSDL, implying a similar degree of expiratory muscle activation. These results suggest that the use of wire leads, which can be placed using minimally invasive techniques, is a potential alternative method of restoring an effective cough mechanism.
Previous animal studies
In previous experiments in dogs, we evaluated the mechanisms by which the expiratory muscles are activated via SCS. Using 4 mm disc electrode contacts, we found that maximum positive airway pressures occurred with stimulation applied at the T9–T10 dorsal epidural spinal cord level.23–26 Stimulation above and below this region resulted in progressive reductions in airway pressure generation. Employing a stimulus frequency of 50 Hz, airway pressure generation rose progressively with increasing stimulus amplitude until a plateau was reached at ∼15 mA.25,26 With SCS at maximum amplitude (15 mA), increases in stimulus frequency also resulted in progressive increases in pressure generation until a plateau was reached between 30 and 40 Hz.26 Pressure generation was highly reproducible as the coefficient of variance was found to be less than 10% in each animal. Concerning the mechanism of expiratory muscle activation by this method, we found that the ventral roots were activated both by direct stimulation in the vicinity of the stimulating electrode and also more distally via spinal cord pathways.23 Optimal activation was achieved with stimulation of two electrode contacts, as the addition of a third electrode contact did not result in further increases in pressure generation.24,25 Based upon the magnitude of pressure generation, we concluded that near maximum activation of the expiratory muscles can be achieved with the application of modest stimulus currents.
Clinical trial results with use of disc leads
Based upon the data garnered from these previous animal studies,23–26 a clinical trial of this technique was performed in patients with spinal cord injury.20–22 A fully implantable, radiofrequency-activated, electrical stimulation system was surgically placed in each subject. Partial hemi-laminotomies were required to place leads with single disc electrodes in the dorsal epidural space at the T9, T11, and L1 spinal levels. Lead wires were tunneled under the skin and connected to a radiofrequency receiver which was placed in a subcutaneous pocket over the anterior chest or abdominal wall. SCS was performed by depressing a button on an external control box which was connected by a rubberized cable to an external antenna, which was taped to the skin over the implanted receiver. In our initial group of 16 subjects, SCS resulted in the generation of large positive airway pressures and high peak expiratory airflow rates during stimulation at each electrode contact. Changes in airway pressure correlated closely with changes in peak expiratory airflow rate, indicating that airway pressure generation was predictive of peak expiratory airflow rate development, another parameter of cough efficacy. As with the animal studies, combined stimulation of only two electrode contacts was necessary to achieve maximum airway pressure generation. SCS was performed at home by either the subjects themselves or caregivers 2–3 times/day on a chronic basis, and also as needed for secretion management. The degree of difficulty in raising secretions improved markedly, and the need for alternative methods of secretion removal was virtually eliminated. Life quality related to respiratory care also improved significantly with subjects reporting greater control of their breathing problems. Importantly, the incidence of respiratory tract infections fell significantly, and the level of trained caregiver support related to secretion management also decreased significantly. Side effects were minimal. Three subjects developed mild hemodynamic effects associated with the initial application of SCS which abated completely with chronic stimulation over a 2–3 week period. Some subjects experienced some leg jerks during SCS, which were painless and well tolerated. There was no incidence of bowel or bladder leakage.
Electrode characteristics and safe electrical limit
While monopolar stimulation with a single wire lead generally resulted in significant expiratory muscle activation, as evidenced by the large positive airway pressures, these values were significantly lower than those achieved with the disc leads at any given stimulus amplitude. The reason for these differences is not clear. However, the diameter of the disc electrode contact is larger than the wire lead electrode contact (4 vs. 1.27 mm, respectively). Given midline placement, therefore, the disc electrode contact is physically closer to the ventral roots bilaterally. In addition, since the disc electrode is insulated on one side, the entire surface area is in contact with the spinal cord. In contrast, only a portion of the wire lead electrode contact, which is not insulated, makes direct contact with the spinal cord. The parallel wire lead arrangement, which results in electrode contacts positioned closer to the ventral roots and twice the surface area, results in expiratory muscle activation that is comparable to the disc lead system.
To avoid potential tissue damage and corrosion of stimulating electrodes, the generally accepted safe limit of the charge density for implanted electrodes is a maximum of 200 µC/cm2.27–29 Utilizing electrical stimulation with maximum stimulus parameters (40 V), in previous animal studies (unpublished observations), we have evaluated the total resistance of the system (603 Ω). Given the surface area of the electrode (0.2394 cm2), the maximum charge density per pulse for bipolar and monopolar wire lead electrodes is 55.14 µC/cm2, which is almost four times less than the maximum limit. A further safety factor is that the stimulus train is applied for only 0.4–0.6 seconds in duration to initiate the cough response, which further limits the amount of accumulated charge applied to the tissue.
An additional important finding was that bipolar stimulation with the parallel wire leads at the T9–T11 and T9–L1 levels also resulted in airway pressure generation that was comparable to that achieved with monopolar stimulation with the disc leads. Compared to monopolar stimulation, bipolar stimulation results in a more localized distribution of current and, therefore, may result in fewer side effects, such as leg movement.
Clinical implications
Ideally, implantation of a device, such as the cough system, for purposes of prophylaxis against potential complications, should provide a high likelihood of clinical benefit with minimal or no risk. While the current technology utilizing disc leads is relatively safe, it does require an invasive procedure with its associated risks. For this reason, patient and physician acceptance of this method in an already compromised patient population is suboptimal. In fact, several patients refused to participate in our initial trial due to the invasive nature of the procedure. Hopefully, use of wire leads, which can be placed utilizing minimally invasive techniques on an out-patient basis, will provide the same degree of expiratory muscle activation and clinical benefits. This technique would be less costly and entail less risk and discomfort. It should be mentioned that placement of wire leads has been in clinical use for several decades for control of spasticity and pain, including failed back surgery syndrome and radicular pain syndrome.30–36 A clinical trial of this technology will be necessary, however, to establish the safety and efficacy of this method in persons with SCI.
Conclusion
The results of these studies support the hypothesis that use of wire leads, which can be placed via minimally invasive techniques, results in substantial activation of the expiratory muscles and the consequent generation of large airway pressures. Moreover, the degree of expiratory muscle activation with wire leads was comparable to that generated with the disc leads, which has been shown to restore an effective cough in persons with SCI.20–22 Use of wire lead technology, therefore, may provide a less invasive and less costly method of restoring an effective cough mechanism in this patient population.
References
- 1.Bellamy R, Pitts FW, Stauffer ES. Respiratory complications in traumatic quadriplegia: analysis of 20 years' experience. J Neurosurg 1973;39(5):596–600 [DOI] [PubMed] [Google Scholar]
- 2.Bromley I. Tetraplegia and paraplegia: a guide for physiotherapists. 3rd ed. New York: Churchill Livingstone; 1985 [Google Scholar]
- 3.Brown R, DiMarco AF, Hoit JD, Garshick E. Respiratory dysfunction and management in spinal cord injury. Respir Care 2006;51(8):853–70 [PMC free article] [PubMed] [Google Scholar]
- 4.Carter RE. Respiratory aspects of spinal cord injury management. Paraplegia 1987;25(3):262–6 [DOI] [PubMed] [Google Scholar]
- 5.Cheshire DJ. Respiratory management in acute traumatic tetraplegia. Paraplegia 1964;1(4):252–61 [DOI] [PubMed] [Google Scholar]
- 6.DeTroyer A, Estenne M, Heilporn A. Mechanism of active expiration in tetraplegic subjects. N Eng J Med 1986;314(12):740–4 [DOI] [PubMed] [Google Scholar]
- 7.DeTroyer A, Pride NB. The respiratory system in neuromuscular disorders. In: Roussos C, Macklem PT. (eds.) The Thorax. New York: Marcel Dekker; 1985. p. 1089–1121 [Google Scholar]
- 8.DeVivo MJ, Krause JS, Lammertse DP. Recent trends in mortality and causes of death among persons with spinal cord injury. Arch Phys Med Rehabil 1999;80(11):1411–9 [DOI] [PubMed] [Google Scholar]
- 9.Dryden DM, Saunders LD, Rowe BH, May LA, Yiannakoulias N, Svenson LW, et al. Utilization of health services following spinal cord injury: a 6-year follow-up study. Spinal Cord 2004;42(9):513–25 [DOI] [PubMed] [Google Scholar]
- 10.Estenne M, DeTroyer A. Cough in tetraplegic subjects: an active process. Ann Intern Med 1990;112(1):22–8 [DOI] [PubMed] [Google Scholar]
- 11.Frankel HL, Coll JR, Charlifue SW, Whiteneck GG, Gardner BP, Jamous MA, et al. Long-term survival in spinal cord injury: a fifty year investigation. Spinal Cord 1998;36(4):266–74 [DOI] [PubMed] [Google Scholar]
- 12.Hagen EM, Lie SA, Rekand T, Gilhus NE, Gronning M. Mortality after traumatic spinal cord injury: 50 years of follow-up. J Neurol Neurosurg Psychiatry 2010;81(4):368–73 [DOI] [PubMed] [Google Scholar]
- 13.Jackson AB, Groomes TE. Incidence of respiratory complications following spinal cord injury. Arch Phys Med Rehabil 1994;75(3):270–5 [DOI] [PubMed] [Google Scholar]
- 14.Kelly BJ, Luce JM. The diagnosis and management of neuromuscular diseases causing respiratory failure. Chest 1991;99(6):1485–94 [DOI] [PubMed] [Google Scholar]
- 15.Mansel JK, Norman JR. Respiratory complications and management of spinal cord injuries. Chest 1990;97(6):1446–52 [DOI] [PubMed] [Google Scholar]
- 16.McMichan JC, Michel L, Westbrook PR. Pulmonary dysfunction following traumatic quadriplegia: recognition, prevention and treatment. J Am Med Assoc 1980;243(6):528–31 [PubMed] [Google Scholar]
- 17.National SCI Statistical Center Spinal cord injury facts and figures at a glance. J Spinal Cord Med 2010;33(4):439–40 [PubMed] [Google Scholar]
- 18.Polatty RC, McElaney MA, Marcelino V. Pulmonary complication in the spinal cord injury patient. Phys Med Rehab: State of the Art Reviews 1987;1(3):353–73 [Google Scholar]
- 19.Soden RJ, Walsh J, Middleton JW, Craven ML, Rutkowski SB, Yeo JD. Causes of death after spinal cord injury. Spinal Cord 2000;38(10):604–10 [DOI] [PubMed] [Google Scholar]
- 20.DiMarco AF, Kowalski KE, Geertman RT, Hromyak DR. Spinal cord stimulation: a new method to produce cough in patients with spinal cord injury. Am J Respir Crit Care Med 2006;173(12):1386–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.DiMarco AF, Kowalski KE, Geertman RT, Hromyak DR. Lower thoracic spinal cord stimulation to restore cough in patients with spinal cord injury: results of a National Institutes of Health-sponsored clinical trial. Part I: methodology and effectiveness of expiratory muscle activation. Arch Phys Med Rehabil 2009;90(5):717–25 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.DiMarco AF, Kowalski KE, Geertman RT, Hromyak DR, Frost FS, Creasey GH, et al. Lower thoracic spinal cord stimulation to restore cough in patients with spinal cord injury: results of a National Institutes of Health-sponsored clinical trial. Part II: clinical outcomes. Arch Phys Med Rehabil 2009;90(5):726–32 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.DiMarco AF, Kowalski KE, Supinski GS, Romaniuk JR. Mechanism of expiratory muscle activation during lower thoracic spinal cord stimulation. J Appl Physiol 2002;92(6):2341–6 [DOI] [PubMed] [Google Scholar]
- 24.DiMarco AF, Romaniuk JR, Kowalski KE, Supinski G. Mechanical contribution of expiratory muscles to pressure generation during spinal cord stimulation. J Appl Physiol 1999;87(4):1433–9 [DOI] [PubMed] [Google Scholar]
- 25.DiMarco AF, Romaniuk JR, Kowalski KE, Supinski G. Pattern of expiratory muscle activation during lower thoracic spinal cord stimulation. J Appl Physiol 1999;86(6):1881–9 [DOI] [PubMed] [Google Scholar]
- 26.DiMarco AF, Romaniuk JR, Supinski GS. Electrical activation of the expiratory muscles to restore cough. Am J Respir Crit Care Med 1995;151(5):1466–71 [DOI] [PubMed] [Google Scholar]
- 27.Brummer SB, Turner MJ. Electrical stimulation with Pt electrodes: II-estimation of maximum surface redox (theoretical non-gassing) limits. IEEE Trans Biomed Eng 1977;24(5):440–3 [DOI] [PubMed] [Google Scholar]
- 28.Agnew WF, Yuen TG, McCreery DB, Bullara LA. Histopathological evaluation of prolonged intracortical electrical stimulation. Exp Neurol 1986;92(1):162–85 [DOI] [PubMed] [Google Scholar]
- 29.Grill WM. Signal considerations for chronically implanted electrodes for brain interfacing. In: Reichert WM. (ed.) Indwelling neural implants: strategies for contending with the in vivo environment. Boca Raton, FL: CRC Press; 2008 [PubMed] [Google Scholar]
- 30.Bennett DS, Alo KM, Oakley J, Feler CA. Spinal cord stimulation for complex regional pain syndrome I [RSD]: a retrospective multicenter experience from 1995 to 1998 of 101 patients. Neuromodulation 1999;2(3):202–10 [DOI] [PubMed] [Google Scholar]
- 31.Burchiel KJ, Anderson VC, Brown FD, Fessler RG, Friedman WA, Pelofsky S, et al. Prospective, multicenter study of spinal cord stimulation for relief of chronic back and extremity pain. Spine 1996;21(23):2786–94 [DOI] [PubMed] [Google Scholar]
- 32.Cameron T. Safety and efficacy of spinal cord stimulation for the treatment of chronic pain: a 20-year literature review. J Neurosurg 2004;1003 Suppl Spine:254–67 [DOI] [PubMed] [Google Scholar]
- 33.Kumar K, Hunter G, Demeria D. Spinal cord stimulation in treatment of chronic benign pain: challenges in treatment planning and present status, a 22-year experience. Neurosurgery 2006;58(3):481–96 [DOI] [PubMed] [Google Scholar]
- 34.North RB, Kidd DH, Petrucci L, Dorsi MJ. Spinal cord stimulation electrode design: a prospective, randomized, controlled trial comparing percutaneous with laminectomy electrodes: part II-clinical outcomes. Neurosurgery 2005;57(5):990–6 [DOI] [PubMed] [Google Scholar]
- 35.Shealy CN, Mortimer JT, Reswick JB. Electrical inhibition of pain by stimulation of the dorsal columns: preliminary clinical report. Anesth Analg 1967;46(4):489–91 [PubMed] [Google Scholar]
- 36.Taylor RS, Taylor RJ, Van Buyten JP, Buchser E, North R, Bayliss S. The cost effectiveness of spinal cord stimulation in the treatment of pain: a systematic review of the literature. J Pain Symptom Manage 2004;27(4):370–8 [DOI] [PubMed] [Google Scholar]