Abstract
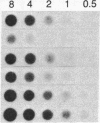
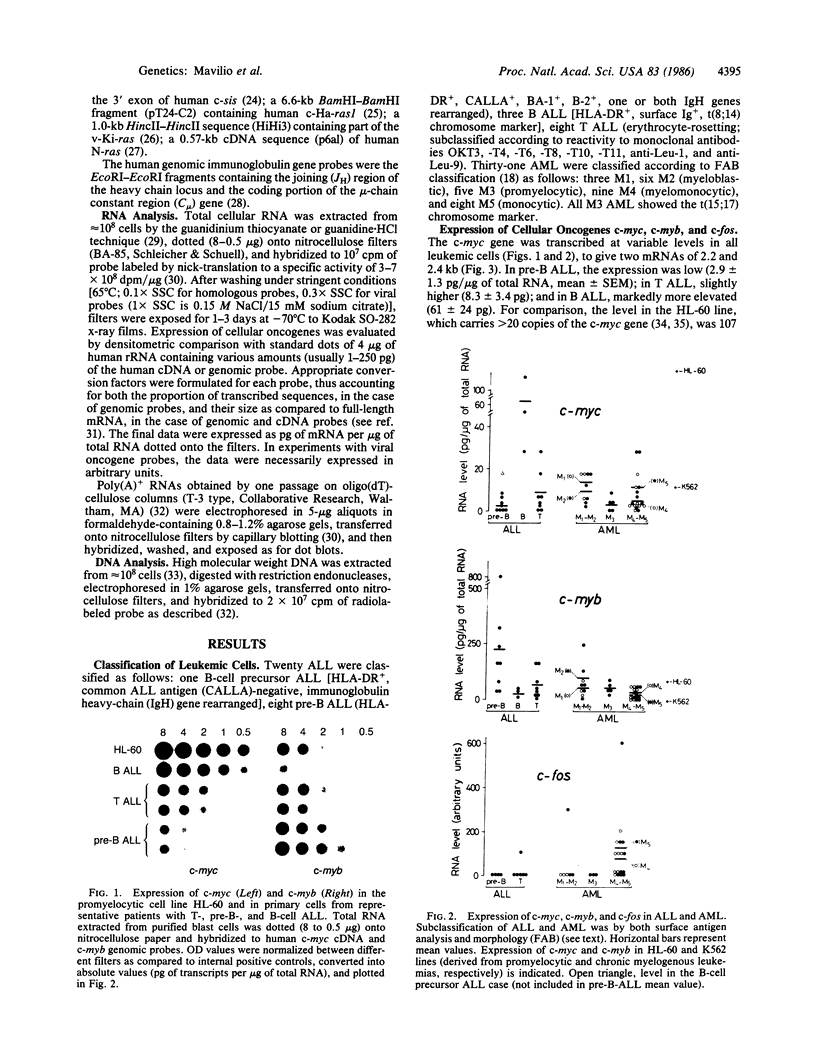
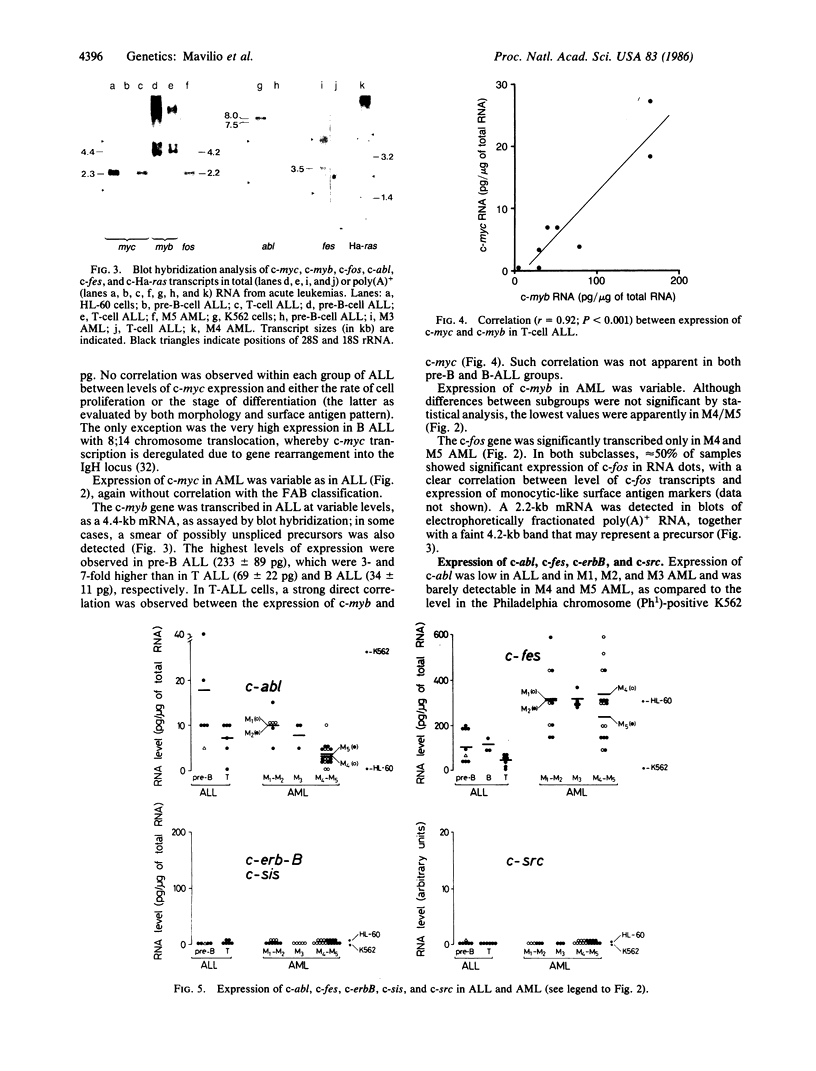
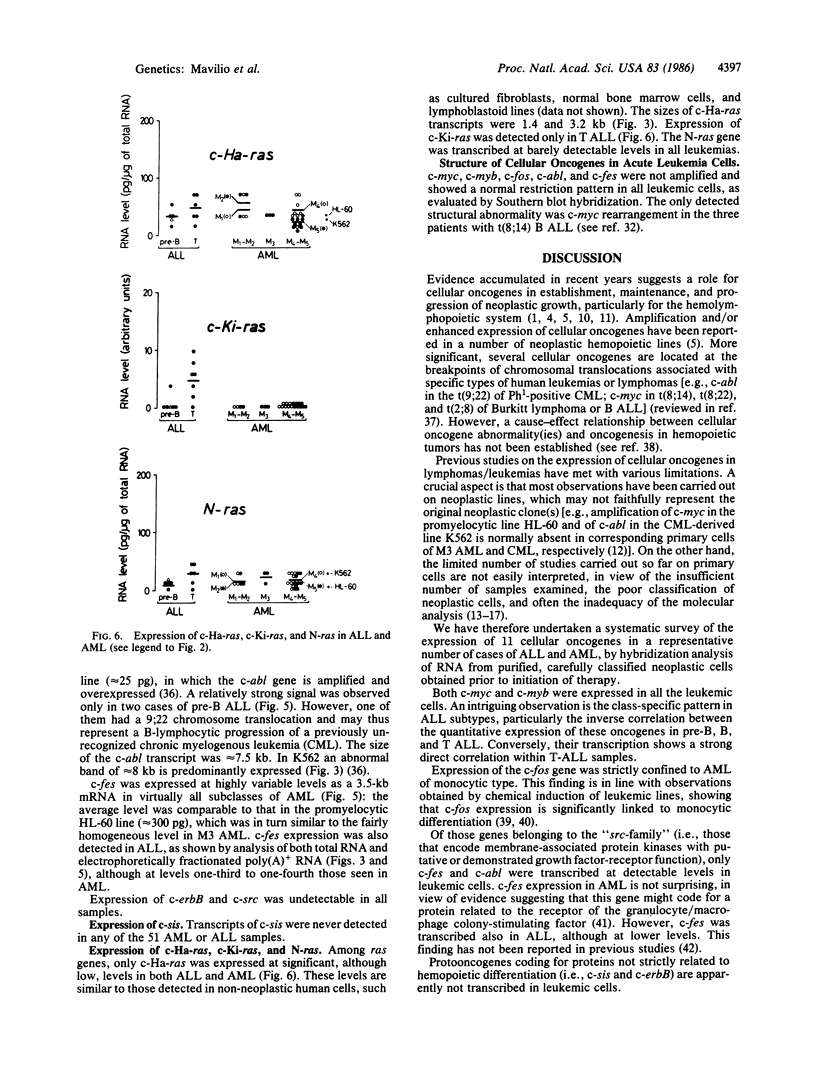
The structure and the expression of 11 cellular oncogenes (protooncogenes) were analyzed in primary cells from 20 acute lymphocytic (ALL) and 31 acute myelogenous (AML) leukemia patients. Neoplastic cells, obtained prior to initiation of therapy, were purified and classified, on the basis of both surface antigen pattern and morphology, into pre-B, B, and T ALL and M1-M5 AML. RNA was extracted and analyzed for expression of cellular oncogenes coding for nuclear proteins (c-myc, c-myb, c-fos), the beta-chain of platelet-derived growth factor (c-sis), growth factor receptors or related proteins (c-src, c-abl, c-fes, c-erbB), or putative intermediate transducers of mitogenic signals (c-Ha-ras, c-Ki-ras, c-N-ras). Quantitative analysis of total RNA was carried out by dot blot hybridization to specific cDNA or genomic probes. Number and size of transcripts were evaluated by blot hybridization of electrophoretically fractionated poly(A)+ RNA. Expression of c-myc and c-myb was detected in all leukemic cells at variable levels and was characterized by well-defined patterns within ALL subtypes. Conversely, significant levels of c-fos transcripts were detected only in myelomonocytic (M4) and monocytic (M5) leukemias. Among the "src-family," c-fes was expressed more in AML than ALL, and c-abl was expressed at variable but not elevated levels in all leukemia types. c-Ha-ras was uniformly expressed at low levels, as in non-neoplastic cells. c-Ki-ras transcription was detected only in T ALL; N-ras expression was barely demonstrable. The structure of these protooncogenes was not grossly modified, as evaluated by Southern analysis, except for c-myc rearrangement in B ALL. These studies indicate that cellular oncogene expression in specific subtypes of leukemic cells may relate to either the proliferative activity (c-myc, c-myb) or the differentiation state (c-fos) of the cells, or possibly to expression of receptors for putative hemopoiesis-related growth factors (c-fes, c-abl). Our data provide a basis for in-depth analysis of protooncogene expression in normal and neoplastic hemopoiesis.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bennett J. M., Catovsky D., Daniel M. T., Flandrin G., Galton D. A., Gralnick H. R., Sultan C. Proposals for the classification of the acute leukaemias. French-American-British (FAB) co-operative group. Br J Haematol. 1976 Aug;33(4):451–458. doi: 10.1111/j.1365-2141.1976.tb03563.x. [DOI] [PubMed] [Google Scholar]
- Bishop J. M. Cellular oncogenes and retroviruses. Annu Rev Biochem. 1983;52:301–354. doi: 10.1146/annurev.bi.52.070183.001505. [DOI] [PubMed] [Google Scholar]
- Blick M., Westin E., Gutterman J., Wong-Staal F., Gallo R., McCredie K., Keating M., Murphy E. Oncogene expression in human leukemia. Blood. 1984 Dec;64(6):1234–1239. [PubMed] [Google Scholar]
- Bos J. L., Toksoz D., Marshall C. J., Verlaan-de Vries M., Veeneman G. H., van der Eb A. J., van Boom J. H., Janssen J. W., Steenvoorden A. C. Amino-acid substitutions at codon 13 of the N-ras oncogene in human acute myeloid leukaemia. 1985 Jun 27-Jul 3Nature. 315(6022):726–730. doi: 10.1038/315726a0. [DOI] [PubMed] [Google Scholar]
- Chirgwin J. M., Przybyla A. E., MacDonald R. J., Rutter W. J. Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry. 1979 Nov 27;18(24):5294–5299. doi: 10.1021/bi00591a005. [DOI] [PubMed] [Google Scholar]
- Collins S. J., Kubonishi I., Miyoshi I., Groudine M. T. Altered transcription of the c-abl oncogene in K-562 and other chronic myelogenous leukemia cells. Science. 1984 Jul 6;225(4657):72–74. doi: 10.1126/science.6587568. [DOI] [PubMed] [Google Scholar]
- Collins S., Groudine M. Amplification of endogenous myc-related DNA sequences in a human myeloid leukaemia cell line. Nature. 1982 Aug 12;298(5875):679–681. doi: 10.1038/298679a0. [DOI] [PubMed] [Google Scholar]
- Croce C. M., Nowell P. C. Molecular basis of human B cell neoplasia. Blood. 1985 Jan;65(1):1–7. [PubMed] [Google Scholar]
- Curran T., Peters G., Van Beveren C., Teich N. M., Verma I. M. FBJ murine osteosarcoma virus: identification and molecular cloning of biologically active proviral DNA. J Virol. 1982 Nov;44(2):674–682. doi: 10.1128/jvi.44.2.674-682.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dalla-Favera R., Gelmann E. P., Gallo R. C., Wong-Staal F. A human onc gene homologous to the transforming gene (v-sis) of simian sarcoma virus. Nature. 1981 Jul 2;292(5818):31–35. doi: 10.1038/292031a0. [DOI] [PubMed] [Google Scholar]
- Dalla-Favera R., Martinotti S., Gallo R. C., Erikson J., Croce C. M. Translocation and rearrangements of the c-myc oncogene locus in human undifferentiated B-cell lymphomas. Science. 1983 Feb 25;219(4587):963–967. doi: 10.1126/science.6401867. [DOI] [PubMed] [Google Scholar]
- Dalla-Favera R., Wong-Staal F., Gallo R. C. Onc gene amplification in promyelocytic leukaemia cell line HL-60 and primary leukaemic cells of the same patient. Nature. 1982 Sep 2;299(5878):61–63. doi: 10.1038/299061a0. [DOI] [PubMed] [Google Scholar]
- Doolittle R. F., Hunkapiller M. W., Hood L. E., Devare S. G., Robbins K. C., Aaronson S. A., Antoniades H. N. Simian sarcoma virus onc gene, v-sis, is derived from the gene (or genes) encoding a platelet-derived growth factor. Science. 1983 Jul 15;221(4607):275–277. doi: 10.1126/science.6304883. [DOI] [PubMed] [Google Scholar]
- Duesberg P. H. Activated proto-onc genes: sufficient or necessary for cancer? Science. 1985 May 10;228(4700):669–677. doi: 10.1126/science.3992240. [DOI] [PubMed] [Google Scholar]
- Duesberg P. H. Retroviral transforming genes in normal cells? Nature. 1983 Jul 21;304(5923):219–226. doi: 10.1038/304219a0. [DOI] [PubMed] [Google Scholar]
- Ellis R. W., Defeo D., Shih T. Y., Gonda M. A., Young H. A., Tsuchida N., Lowy D. R., Scolnick E. M. The p21 src genes of Harvey and Kirsten sarcoma viruses originate from divergent members of a family of normal vertebrate genes. Nature. 1981 Aug 6;292(5823):506–511. doi: 10.1038/292506a0. [DOI] [PubMed] [Google Scholar]
- Erikson J., ar-Rushdi A., Drwinga H. L., Nowell P. C., Croce C. M. Transcriptional activation of the translocated c-myc oncogene in burkitt lymphoma. Proc Natl Acad Sci U S A. 1983 Feb;80(3):820–824. doi: 10.1073/pnas.80.3.820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feldman R. A., Gabrilove J. L., Tam J. P., Moore M. A., Hanafusa H. Specific expression of the human cellular fps/fes-encoded protein NCP92 in normal and leukemic myeloid cells. Proc Natl Acad Sci U S A. 1985 Apr;82(8):2379–2383. doi: 10.1073/pnas.82.8.2379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feramisco J. R., Gross M., Kamata T., Rosenberg M., Sweet R. W. Microinjection of the oncogene form of the human H-ras (T-24) protein results in rapid proliferation of quiescent cells. Cell. 1984 Aug;38(1):109–117. doi: 10.1016/0092-8674(84)90531-2. [DOI] [PubMed] [Google Scholar]
- Ferrari S., Torelli U., Selleri L., Donelli A., Venturelli D., Moretti L., Torelli G. Expression of human c-fes onc-gene occurs at detectable levels in myeloid but not in lymphoid cell populations. Br J Haematol. 1985 Jan;59(1):21–25. doi: 10.1111/j.1365-2141.1985.tb02959.x. [DOI] [PubMed] [Google Scholar]
- Franchini G., Gelmann E. P., Dalla-Favera R., Gallo R. C., Wong-Staal F. Human gene (c-fes) related to the onc sequences of Snyder-Theilen feline sarcoma virus. Mol Cell Biol. 1982 Aug;2(8):1014–1019. doi: 10.1128/mcb.2.8.1014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franchini G., Wong-Staal F., Baluda M. A., Lengel C., Tronick S. R. Structural organization and expression of human DNA sequences related to the transforming gene of avian myeloblastosis virus. Proc Natl Acad Sci U S A. 1983 Dec;80(24):7385–7389. doi: 10.1073/pnas.80.24.7385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heldin C. H., Westermark B. Growth factors: mechanism of action and relation to oncogenes. Cell. 1984 May;37(1):9–20. doi: 10.1016/0092-8674(84)90296-4. [DOI] [PubMed] [Google Scholar]
- Land H., Parada L. F., Weinberg R. A. Cellular oncogenes and multistep carcinogenesis. Science. 1983 Nov 18;222(4625):771–778. doi: 10.1126/science.6356358. [DOI] [PubMed] [Google Scholar]
- Leder P., Battey J., Lenoir G., Moulding C., Murphy W., Potter H., Stewart T., Taub R. Translocations among antibody genes in human cancer. Science. 1983 Nov 18;222(4625):765–771. doi: 10.1126/science.6356357. [DOI] [PubMed] [Google Scholar]
- McClain K. L. Expression of oncogenes in human leukemias. Cancer Res. 1984 Nov;44(11):5382–5389. [PubMed] [Google Scholar]
- Mitchell R. L., Zokas L., Schreiber R. D., Verma I. M. Rapid induction of the expression of proto-oncogene fos during human monocytic differentiation. Cell. 1985 Jan;40(1):209–217. doi: 10.1016/0092-8674(85)90324-1. [DOI] [PubMed] [Google Scholar]
- Müller R., Curran T., Müller D., Guilbert L. Induction of c-fos during myelomonocytic differentiation and macrophage proliferation. Nature. 1985 Apr 11;314(6011):546–548. doi: 10.1038/314546a0. [DOI] [PubMed] [Google Scholar]
- Müller R., Verma I. M. Expression of cellular oncogenes. Curr Top Microbiol Immunol. 1984;112:73–115. doi: 10.1007/978-3-642-69677-0_4. [DOI] [PubMed] [Google Scholar]
- Peschle C., Mavilio F., Sposi N. M., Giampaolo A., Caré A., Bottero L., Bruno M., Mastroberardino G., Gastaldi R., Testa M. G. Translocation and rearrangement of c-myc into immunoglobulin alpha heavy chain locus in primary cells from acute lymphocytic leukemia. Proc Natl Acad Sci U S A. 1984 Sep;81(17):5514–5518. doi: 10.1073/pnas.81.17.5514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfeifer-Ohlsson S., Goustin A. S., Rydnert J., Wahlström T., Bjersing L., Stehelin D., Ohlsson R. Spatial and temporal pattern of cellular myc oncogene expression in developing human placenta: implications for embryonic cell proliferation. Cell. 1984 Sep;38(2):585–596. doi: 10.1016/0092-8674(84)90513-0. [DOI] [PubMed] [Google Scholar]
- Rosson D., Tereba A. Transcription of hematopoietic-associated oncogenes in childhood leukemia. Cancer Res. 1983 Aug;43(8):3912–3918. [PubMed] [Google Scholar]
- Rowley J. D. Biological implications of consistent chromosome rearrangements in leukemia and lymphoma. Cancer Res. 1984 Aug;44(8):3159–3168. [PubMed] [Google Scholar]
- Roy-Burman P., Devi B. G., Parker J. W. Differential expression of c-erbB, c-myc and c-myb oncogene loci in human lymphomas and leukemias. Int J Cancer. 1983 Aug 15;32(2):185–191. doi: 10.1002/ijc.2910320208. [DOI] [PubMed] [Google Scholar]
- Santos E., Tronick S. R., Aaronson S. A., Pulciani S., Barbacid M. T24 human bladder carcinoma oncogene is an activated form of the normal human homologue of BALB- and Harvey-MSV transforming genes. Nature. 1982 Jul 22;298(5872):343–347. doi: 10.1038/298343a0. [DOI] [PubMed] [Google Scholar]
- Schimke R. T. Gene amplification in cultured animal cells. Cell. 1984 Jul;37(3):705–713. doi: 10.1016/0092-8674(84)90406-9. [DOI] [PubMed] [Google Scholar]
- Slamon D. J., deKernion J. B., Verma I. M., Cline M. J. Expression of cellular oncogenes in human malignancies. Science. 1984 Apr 20;224(4646):256–262. doi: 10.1126/science.6538699. [DOI] [PubMed] [Google Scholar]
- Taparowsky E., Shimizu K., Goldfarb M., Wigler M. Structure and activation of the human N-ras gene. Cell. 1983 Sep;34(2):581–586. doi: 10.1016/0092-8674(83)90390-2. [DOI] [PubMed] [Google Scholar]
- Thomas P. S. Hybridization of denatured RNA and small DNA fragments transferred to nitrocellulose. Proc Natl Acad Sci U S A. 1980 Sep;77(9):5201–5205. doi: 10.1073/pnas.77.9.5201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ullrich A., Coussens L., Hayflick J. S., Dull T. J., Gray A., Tam A. W., Lee J., Yarden Y., Libermann T. A., Schlessinger J. Human epidermal growth factor receptor cDNA sequence and aberrant expression of the amplified gene in A431 epidermoid carcinoma cells. 1984 May 31-Jun 6Nature. 309(5967):418–425. doi: 10.1038/309418a0. [DOI] [PubMed] [Google Scholar]
- Waterfield M. D., Scrace G. T., Whittle N., Stroobant P., Johnsson A., Wasteson A., Westermark B., Heldin C. H., Huang J. S., Deuel T. F. Platelet-derived growth factor is structurally related to the putative transforming protein p28sis of simian sarcoma virus. Nature. 1983 Jul 7;304(5921):35–39. doi: 10.1038/304035a0. [DOI] [PubMed] [Google Scholar]
- Weinberg R. A. ras Oncogenes and the molecular mechanisms of carcinogenesis. Blood. 1984 Dec;64(6):1143–1145. [PubMed] [Google Scholar]
- Xu Y. H., Ishii S., Clark A. J., Sullivan M., Wilson R. K., Ma D. P., Roe B. A., Merlino G. T., Pastan I. Human epidermal growth factor receptor cDNA is homologous to a variety of RNAs overproduced in A431 carcinoma cells. 1984 Jun 28-Jul 4Nature. 309(5971):806–810. doi: 10.1038/309806a0. [DOI] [PubMed] [Google Scholar]
- Yunis J. J. The chromosomal basis of human neoplasia. Science. 1983 Jul 15;221(4607):227–236. doi: 10.1126/science.6336310. [DOI] [PubMed] [Google Scholar]