Abstract
Background
Diabetes is a leading cause of chronic kidney disease (CKD). Whether reclassification of CKD stages based on glomerular filtration rate estimated using the CKD Epidemiology Collaboration (CKD-EPI) equation versus the Modification of Diet in Renal Disease (MDRD) Study equation modifies estimates of prevalent risk factors across stages is unknown.
Methods
This is a cross-sectional analysis of data from the Kidney Early Evaluation Program (KEEP), a community-based health screening program targeting individuals 18 years and older with diabetes, hypertension, or a family history of diabetes, hypertension, or kidney disease. Of 109,055 participants, 68.2% were women and 31.8% were African American. Mean age was 55.3 ± 0.05 years. Clinical, demographic, and laboratory data were collected from August 2000 through December 2009. Glomerular filtration rate was estimated using the CKD-EPI and MDRD Study equations.
Results
CKD was present in 25.6% and 23.5% of the study population using the MDRD Study and CKD-EPI equations, respectively. Diabetes was present in 42.4% and 43.8% of participants with CKD, respectively. Prevalent risk factors for diabetes included obesity (body mass index >30 kg/m2), 44.0%; hypertension, 80.5%; cardiovascular disease, 23.2%; family history of diabetes, 55.9%; and dyslipidemia, 43.0%. In a logistic regression model after adjusting for age and other risk factors, odds for diabetes increased significantly compared with no CKD with each CKD stage based on the CKD-EPI equation and similarly with stages based on the MDRD Study equation. Using a CKD-EPI–adjusted model, ORs were: stage 1, 2.08 (95% CI, 1.90–2.27); stage 2, 1.86 (95% CI, 1.72–2.02); stage 3, 1.23 (95% CI, 1.17–1.30); stage 4, 1.69 (95% CI, 1.42–2.03); and stage 5, 2.46 (95% CI, 1.46–4.14).
Conclusions
Using the CKD-EPI equation led to a lower prevalence of CKD but to similar diabetes prevalence rates associated with CKD across all stages compared with the MDRD Study equation. Diabetes and other CKD risk factor prevalence was increased compared with the non-CKD population.
INDEX WORDS: Chronic kidney disease, diabetes mellitus, estimated glomerular filtration rate
Diabetes mellitus is a major public health problem that is reaching pandemic proportions in the United States1 and worldwide.2 Based on data from the National Health and Nutrition Examination Survey (NHANES) 2003–2006, diabetes prevalence is estimated to be approximately 11% (23.6 million people), with another 57 million people in the prediabetic state.1 Worldwide, diabetes is predicted to affect 439 million adults by 2030, with a 69% increase between 2010 and 2030 in developing countries and a 20% increase in developed countries.2
Diabetes is the most common cause of end-stage renal disease in the United States, accounting for up to 46% of patients starting on renal replacement therapy each year.3,4 Although both diabetes and chronic kidney disease (CKD) are strong cardiovascular disease (CVD) risk factors,5–8 the presence of diabetes in end-stage renal disease and CKD populations confers even higher CVD morbidity and mortality risk compared with the non-CKD population.9,10 Risk factors for type 2 diabetes include advanced age, obesity, physical inactivity, male sex, nonwhite race, family history of diabetes, and dyslipidemia, including high triglyceride and low high-density lipoprotein cholesterol levels and other components of the metabolic syndrome.11,12 Some of these risk factors, such as obesity and physical inactivity, are largely modifiable; early intervention has been shown in large trials to prevent or delay progression to type 2 diabetes in up to 60% of people in the prediabetic stage.13,14
Although diabetes is a well-established risk factor for CKD, accumulating evidence also indicates that CKD, particularly end-stage renal disease, is associated with a higher incidence of newly diagnosed diabetes, including after kidney transplant and after hemodialysis therapy initiation.9,15–19 The prevalence of and risk factors for diabetes in patients with earlier CKD stages (estimated glomerular filtration rate [eGFR] >60 mL/min/1.73 m2) have not been evaluated previously in large studies. The most commonly used equation to estimate GFR is the Modification of Diet in Renal Disease (MDRD) Study equation.20 It has been shown to be valid, but to underestimate measured GFR at levels >60 mL/min/1.73 m2, leading to overdiagnosis of prevalent CKD. The CKD Epidemiology Collaboration (CKD-EPI) equation21 was developed in people with and without kidney disease. It has been shown to be a better estimate of measured GFR at higher levels than the MDRD Study equation.
Therefore, in this analysis of data from the Kidney Early Evaluation Program (KEEP), we sought to assess the prevalence of diabetes mellitus in participants at various stages of CKD based on GFR estimated using the MDRD Study and the CKD-EPI equations. We also sought to examine and characterize the various risk factors, including waist circumference, for diabetes in patients with CKD.
METHODS
Study Participants
KEEP is a free community-based health screening program targeting individuals 18 years and older with diabetes, hypertension, or a family history of diabetes, hypertension, or kidney disease. We included 123,704 eligible KEEP participants from August 2000 through December 31, 2009, from 48 National Kidney Foundation affiliates and 2,634 screening programs in 50 states and the District of Columbia. We excluded participants who previously had undergone dialysis or kidney transplant or whose diabetes status was missing in the data, leaving a study population of 109,055.
Definitions
GFR was estimated using both the 4-variable MDRD Study equation20 and the CKD-EPI equation.21 Albumin-creatinine ratio was calculated from urine samples and categorized as <30, 30–300, or >300 mg/g. CKD stages were defined as follows: stage 1, eGFR ≥90 mL/min/1.73 m2 and albumin-creatinine ratio ≥30 mg/g; stage 2, eGFR of 60–89 mL/min/1.73 m2 and albumin-creatinine ratio ≥30 mg/g; stage 3, eGFR of 30–59 mL/min/1.73 m2; stage 4, eGFR of 15–29 mL/min/1.73 m2; and stage 5, eGFR <15 mL/min/1.73 m2.
Diabetes was defined as a history of diabetes (self-report or retinopathy), use of medications to treat diabetes, fasting blood glucose level ≥126 mg/dL, or nonfasting blood glucose level ≥200 mg/dL in the absence of self-report or medication use. Hypertension was defined as self-report, use of medication to treat hypertension, or systolic blood pressure ≥130 mm Hg or diastolic blood pressure ≥80 mm Hg. CVD history was defined as self-reported history of heart attack, heart angioplasty, bypass surgery, heart failure, abnormal heart rhythm, or stroke. Dyslipidemia was defined as total cholesterol level >200 mg/dL or triglyceride level >150 mg/dL. Waist circumference was measured as described by NHANES22 in a subgroup of the KEEP population (n = 20,135).
Statistical Analysis
To test differences in characteristics between participants with and without diabetes, χ2 statistics were used. One-way analysis of variance was used to test differences in continuous variables between participants with and without diabetes. Mean ± standard error are reported for continuous variables. We used logistic regression, expressed as odds ratio (OR) and 95% confidence interval (CI), to describe the association of CKD stages and other clinical characteristics with diabetes (dependent variable). Data for cholesterol and triglycerides were available from 2005; therefore, data used in the logistic regression included participants screened from 2005–2009. Separate models were constructed using MDRD Study eGFR and CKD-EPI eGFR. Both models were adjusted for risk factors for diabetes, including age, sex, race, education, obesity, dyslipidemia, hypertension, CVD, and family history of diabetes. In the subgroup analysis for participants screened from 2008–2009, we further adjusted for waist circumference, for which data were available from 2008.
RESULTS
Demographics of the Study Population
For the included 109,055 eligible KEEP participants, demographic, clinical, and laboratory data were collected between August 2000 and December 31, 2009. Mean age was 55.3 ± 0.05 years. Mean body mass index (BMI) was 30.2 ± 0.02 kg/m2. Of the total cohort, 68.2% were women; 31.8% were African American, 49.7% were white, and 11.9% were Hispanic.
Demographics and Prevalence of and Risk Factors for Diabetes
Of 109,055 participants, 34,144 (31.3%) had diabetes. Mean fasting blood glucose level was 94.7 ± 0.1 mg/dL for nondiabetic and 138.5 ± 0.71 mg/dL for diabetic participants (P < 0.001). Diabetes prevalence increased significantly with each age category, from 9.9% for age 18–30 years to 39.4% for age >75 years (P < 0.001; Table 1), with the highest prevalence (41.7%) for age 61–75 years. Diabetes prevalence was higher in men (32.8%) than women (30.6%; P < 0.001). The prevalence was slightly higher in whites (32.4%) than African Americans (29.6%) and members of other races (31.3%; P <0.001) and slightly lower in Hispanics (30.0%) than non-Hispanics (31.5%; P <0.001).
Table 1.
Participant Characteristics
| Characteristics | No. | % Diabetesa | Column Percents
|
P | ||
|---|---|---|---|---|---|---|
| No Diabetes | Diabetesa | |||||
| No. | 109,055 | 31.1 | 74,911 | 34,144 | ||
| Age category | <0.001 | |||||
| 18–30 y | 7,558 | 9.9 | 9.1 | 2.2 | ||
| 31–45 y | 21,198 | 19.5 | 22.8 | 12.1 | ||
| 46–60 y | 37,964 | 31.3 | 34.8 | 34.8 | ||
| 61–75 y | 31,584 | 41.7 | 24.6 | 38.5 | ||
| >75 y | 10,751 | 39.4 | 8.7 | 12.4 | ||
| Sex | <0.001 | |||||
| Men | 34,643 | 32.8 | 31.1 | 33.3 | ||
| Women | 74,412 | 30.6 | 68.9 | 66.7 | ||
| Race | <0.001 | |||||
| White | 54,258 | 32.4 | 48.9 | 51.6 | ||
| African American | 34,700 | 29.6 | 32.6 | 30.0 | ||
| Other | 20,097 | 31.3 | 18.4 | 18.4 | ||
| Ethnicity | <0.001 | |||||
| Hispanic | 12,981 | 30.0 | 12.1 | 11.4 | ||
| Non-Hispanic | 96,074 | 31.5 | 87.9 | 88.6 | ||
| Education | <0.001 | |||||
| <High school | 15,918 | 39.0 | 13.1 | 18.5 | ||
| ≥High school | 91,667 | 29.9 | 86.9 | 81.5 | ||
| Risk factors | ||||||
| Current smoker | 11,270 | 27.2 | 11.5 | 9.4 |
|
<0.001 |
| Non-/ex-smoker | 92,595 | 31.8 | 88.5 | 90.6 | ||
| Obesityb | 47,941 | 39.5 | 39.2 | 56.0 |
|
<0.001 |
| No obesity | 59,881 | 24.8 | 60.8 | 44.0 | ||
| Hypertension | 87,752 | 34.5 | 76.8 | 88.5 |
|
<0.001 |
| No hypertension | 21,294 | 18.4 | 23.2 | 11.5 | ||
| Cardiovascular disease | 25,326 | 43.4 | 19.1 | 32.2 |
|
<0.001 |
| No cardiovascular disease | 83,729 | 27.7 | 80.9 | 67.8 | ||
| Dyslipidemiac | 46,914 | 33.5 | 61.9 | 62.4 |
|
0.2 |
| No dyslipidemia | 28,649 | 33.0 | 38.1 | 37.6 | ||
| Family history diabetes | 61,054 | 36.6 | 54.1 | 69.6 |
|
<0.001 |
| No family history diabetes | 42,676 | 22.9 | 45.9 | 30.4 | ||
Diabetes defined as self-reported, using medication, fasting glucose level ≥126 mg/dL, or nonfasting glucose level ≥200 mg/dL.
Body mass index ≥30 kg/m2.
Dyslipidemia defined as cholesterol level >200 mg/dL or triglyceride level >150 mg/dL.
High school education or higher was reported for 84.0% of the total cohort. Diabetes prevalence was significantly higher in participants with less than high school education (39.0%) than those with higher education levels (29.9%; P < 0.001; Table 1). Current smoking was reported for 10.3% of the cohort. Diabetes prevalence was lower in current smokers (27.2%) than nonsmokers/ex-smokers (31.8%; P <0.001).
The presence of hypertension, reported in 80.5% of the total cohort, was associated with a high prevalence of diabetes (34.5%) compared with absence of hypertension (18.4%; P < 0.001; Table 1). Mean systolic blood pressure was higher, 136.5 ± 0.11 mm Hg for diabetic participants and 131.9 ± 0.07 mm Hg for nondiabetic participants (P < 0.001), and corresponding values for mean diastolic blood pressure were lower, 78.6 ± 0.06 and 79.7 ± 0.04 mm Hg (P <0.001).
Obesity, defined as BMI ≥30 kg/m2, was found in 47,941 participants (44.0%) and was associated with a higher prevalence of diabetes (39.5%) compared with lack of obesity (24.8%; P < 0.001). In a subgroup analysis of 20,135 participants with measured waist circumference, waist circumference was significantly higher for diabetic participants (41.8 ± 0.07 inches) than nondiabetic participants (38.6 ± 0.05; P <0.001).
CVD prevalence was 23.2% for the total cohort. The rate of diabetes was higher in participants with (43.4%) than without CVD (27.7%; P <0.001; Table 1). Dyslipidemia, defined as total cholesterol level >200 mg/dL or triglyceride level >150 mg/dL, was present in 43.0% of participants, but was not associated with higher rates of diabetes (33.5% vs 33.0%; P = 0.2, for dyslipidemic and nondyslipidemic participants, respectively). A family history of diabetes was reported for 55.9% of the KEEP cohort. Diabetes was reported in 36.6% of participants with a family history of diabetes and 22.9% of those without a family history (P <0.001).
Determining the Prevalence of Diabetes in CKD
Using the CKD-EPI and MDRD Study equations to define CKD stages, prevalent rates for diabetes were similar in participants identified with CKD and across stages (Table 2); prevalent rates for participants without CKD were identical. For participants identified as having CKD, diabetes prevalence markedly increased (43.9%, CKD-EPI equation; 42.5%, MDRD Study equation). There was a graded trend for increasing diabetes prevalence with advancing CKD stage, except that rates decreased at stage 3.
Table 2.
Prevalence of Diabetes in CKD Using MDRD Study and CKD-EPI GFR Estimating Equations
| Non-CKD | All CKD | Stage 1 | Stage 2 | Stage 3 | Stage 4–5 | |
|---|---|---|---|---|---|---|
| MDRD Study Equation | ||||||
| Total | 81,068 | 27,987 | 3,217 | 5,443 | 18,275 | 1,052 |
| Total diabetes | 22,264 | 11,880 | 1,439 | 2,497 | 7,383 | 561 |
| Diabetes (%) | 27.5 | 42.5 | 44.7 | 45.9 | 40.4 | 53.3 |
| CKD-EPI Equation | ||||||
| Total | 83,475 | 25,580 | 4,145 | 4,814 | 15,500 | 1,121 |
| Total diabetes | 22,921 | 11,223 | 1,753 | 2,326 | 6,550 | 594 |
| Diabetes (%) | 27.5 | 43.9 | 42.3 | 48.3 | 42.3 | 53.0 |
Note: Values are number or percentage.
Abbreviation: CKD, chronic kidney disease; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; MDRD, Modification of Diet in Renal Disease.
Using logistic regression, we assessed the association of diabetes with CKD in 2 separate models (one for each equation), adjusting for diabetes risk factors, including age, sex, race, education, obesity, dyslipidemia, hypertension, CVD, and family history of diabetes. Adjusted model 1 used the MDRD Study equation to define CKD and adjusted model 2 used the CKD-EPI equation (Table 3).
Table 3.
Associations of CKD Stage and Other Participant Characteristics With Diabetes, 2005–2009
| Characteristics | Unadjusted | Adjusted Model 1a | Adjusted Model 2b |
|---|---|---|---|
| MDRD Study equation | |||
| No CKD | 1.00 (reference) | 1.00 (reference) | — |
| CKD stage 1 | 2.06 (1.88–2.26) | 2.26 (2.05–2.49) | — |
| CKD stage 2 | 2.20 (2.05–2.36) | 1.78 (1.65–1.92) | — |
| CKD stage 3 | 1.78 (1.70–1.86) | 1.20 (1.15–1.26) | — |
| CKD stage 4 | 2.83 (2.37–3.38) | 1.71 (1.42–2.07) | — |
| CKD stage 5 | 3.26 (1.96–5.41) | 2.64 (1.55–4.51) | — |
| CKD-EPI equation | |||
| No CKD | 1.00 (reference) | — | 1.00 (reference) |
| CKD stage 1 | 1.85 (1.71–2.01) | — | 2.08 (1.90–2.27) |
| CKD stage 2 | 2.47 (2.29–2.66) | — | 1.86 (1.72–2.02) |
| CKD stage 3 | 1.93 (1.84–2.02) | — | 1.23 (1.17–1.30) |
| CKD stage 4 | 2.82 (2.38–3.34) | — | 1.69 (1.42–2.03) |
| CKD stage 5 | 3.11 (1.90–5.10) | — | 2.46 (1.46–4.14) |
| Age (/decade) | 1.33 (1.32–1.35) | 1.33 (1.31–1.35) | 1.33 (1.31–1.35) |
| Women (vs men) | 0.93 (0.90–0.97) | 0.87 (0.84–0.90) | 0.87 (0.84–0.91) |
| African American (vs white) | 0.88 (0.85–0.91) | 0.87 (0.83–0.90) | 0.86 (0.83–0.90) |
| Other race (vs white) | 0.94 (0.89–0.98) | 1.19 (1.13–1.25) | 1.19 (1.13–1.25) |
| High school graduate (vs less) | 0.69 (0.66–0.72) | 0.80 (0.77–0.84) | 0.80 (0.77–0.84) |
| Current smoking | 0.84 (0.79–0.88) | 1.04 (0.98–1.10) | 1.04 (0.98–1.10) |
| Obesityc | 1.99 (1.93–2.06) | 2.07 (1.99–2.14) | 2.07 (1.99–2.14) |
| Hypertension | 2.20 (2.10–2.31) | 1.42 (1.35–1.50) | 1.42 (1.35–1.50) |
| Cardiovascular disease | 1.80 (1.73–1.86) | 1.42 (1.36–1.47) | 1.42 (1.36–1.47) |
| Dyslipidemiad | 1.03 (0.996–1.07) | 0.87 (0.84–0.90) | 0.87 (0.84–0.90) |
| Family history diabetes | 2.08 (2.01–2.15) | 2.20 (2.12–2.28) | 2.20 (2.12–2.28) |
Note: Values shown are odds ratio (95% confidence interval). Logistic regression, dependent variable diabetes/no diabetes; data for cholesterol and triglycerides were available from 2005; n = 66,799 after excluding missing values in covariates; diabetes defined as self-reported, using medication, fasting glucose level ≥126 mg/dL, or nonfasting glucose level ≥200 mg/dL.
Abbreviations: CKD, chronic kidney disease; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; MDRD, Modification of Diet in Renal Disease.
MDRD Study equation.
CKD-EPI equation.
Body mass index ≥30 kg/m2.
Dyslipidemia defined as cholesterol level >200 mg/dL or triglyceride level >150 mg/dL.
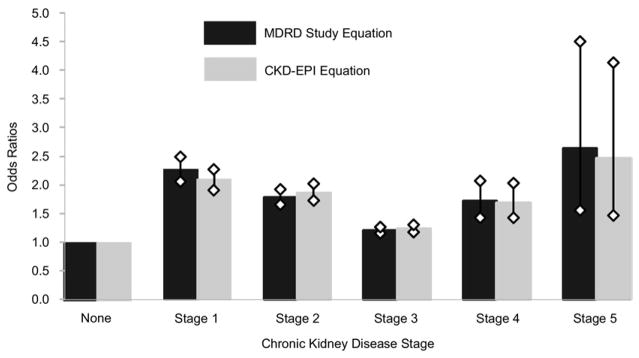
Using model 1 with the non-CKD population as the reference, ORs for diabetes in CKD were: stage 1, 2.26 (95% CI, 2.05–2.49); stage 2, 1.78 (95% CI, 1.65–1.92); stage 3, 1.20 (95% CI, 1.15–1.26); stage 4, 1.71 (95% CI, 1.42–2.07); and stage 5, 2.64 (95% CI, 1.55–4.51; Table 3; Fig 1).
Figure 1.
Odds ratios and 95% confidence intervals for diabetes in chronic kidney disease by stage, Modification of Diet in Renal Disease (MDRD) Study and Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equations.
Using model 2, results were similar using CKD-EPI staging. Compared with no CKD, ORs for diabetes in CKD were: stage 1, 2.08 (95% CI, 1.90–2.27); stage 2, 1.86 (95% CI, 1.72–2.02); stage 3, 1.23 (95% CI, 1.17–1.30); stage 4, 1.69 (95% CI, 1.42–2.03); and stage 5, 2.46 (95% CI, 1.46–4.14; Table 3; Fig 1).
We assessed the association of waist circumference and diabetes in a subgroup of the KEEP population (n = 20,135). The OR for diabetes per 10-inch increase in waist circumference was 2.06 (95% CI, 1.92–2.21) in both adjusted regression models 1 and 2. This OR was similar to that for family history of diabetes (2.08 [95% CI, 1.95–2.22]) and higher than ORs for obesity measured using BMI (1.14 [95% CI, 1.05–1.25]), hypertension (1.40 [95% CI, 1.27–1.54]), CVD (1.29 [95% CI, 1.20–1.39]), and age per decade (1.35 [95% CI, 1.32–1.39]; Table 4).
Table 4.
Association of CKD Stage, Waist Circumference, and Other Participant Characteristics With Diabetes, June 2008–2009
| Characteristics | Adjusted Model 1a | Adjusted Model 2b |
|---|---|---|
| MDRD Study equation | ||
| No CKD | 1.00 (reference) | — |
| CKD stage 1 | 2.44 (2.00–2.98) | — |
| CKD stage 2 | 1.57 (1.37–1.79) | — |
| CKD stage 3 | 1.13 (1.03–1.23) | — |
| CKD stage 4 | 1.79 (1.27–2.52) | — |
| CKD stage 5 | 2.65 (0.94–7.47) | — |
| CKD-EPI equation | ||
| No CKD | — | 1.00 (reference) |
| CKD stage 1 | — | 2.05 (1.73–2.43) |
| CKD stage 2 | — | 1.61 (1.40–1.86) |
| CKD stage 3 | — | 1.14 (1.04–1.25) |
| CKD stage 4 | — | 1.61 (1.16–2.24) |
| CKD stage 5 | — | 2.89 (1.05–7.95) |
| Age (/decade) | 1.35 (1.32–1.39) | 1.35 (1.31–1.39) |
| Women (vs men) | 1.05 (0.97–1.12) | 1.04 (0.97–1.12) |
| African American (vs white) | 0.91 (0.84–0.98) | 0.90 (0.84–0.97) |
| Other race (vs white) | 1.30 (1.19–1.43) | 1.30 (1.19–1.43) |
| High school graduate (vs less) | 0.78 (0.71–0.85) | 0.78 (0.71–0.85) |
| Current smoking | 1.05 (0.94–1.17) | 1.05 (0.93–1.17) |
| Waist (/10 inches) | 2.06 (1.92–2.21) | 2.06 (1.92–2.21) |
| Obesityc | 1.14 (1.05–1.25) | 1.14 (1.05–1.25) |
| Hypertension | 1.40 (1.27–1.54) | 1.40 (1.27–1.54) |
| Cardiovascular disease | 1.29 (1.20–1.39) | 1.29 (1.20–1.39) |
| Dyslipidemiad | 0.92 (0.86–0.98) | 0.92 (0.86–0.98) |
| Family history diabetes | 2.08 (1.95–2.22) | 2.08 (1.95–2.22) |
Note: Values shown are odds ratio (95% confidence interval). Logistic regression, dependent variable diabetes/no diabetes; data for waist circumference were available from 2008; n = 20,135 after excluding missing values in covariates and further excluding missing values for waist circumference; diabetes defined as self-reported, using medication, fasting glucose level ≥126 mg/dL, or nonfasting glucose level ≥200 mg/dL.
Abbreviations: CKD, chronic kidney disease; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; MDRD, Modification of Diet in Renal Disease.
MDRD Study equation.
CKD-EPI equation.
Body mass index ≥30 kg/m2.
Dyslipidemia defined as cholesterol level >200 mg/dL or triglyceride level >150 mg/dL.
DISCUSSION
Although several studies show a high prevalence of diabetes in patients with advanced CKD (eGFR <60 mL/min/1.73 m2),5,9,15 this analysis is among the first to describe a high prevalence of diabetes in earlier stages of CKD using the CKD-EPI equation to estimate GFR. Our data suggest that increases in diabetes prevalence were similar in participants identified with CKD and across stages using the CKD-EPI and MDRD Study equations. Our data further suggest that using the CDK-EPI equation to assess prevalent rates of comorbid conditions (eg, diabetes in this study), even at higher levels of GFR, is similar to using the MDRD Study equation. This is important in the context of potentially significant reclassification of individuals at higher eGFRs using the CKD-EPI equation. Why this occurs warrants further investigation.
We report a significant increase in diabetes prevalence compared with the non-CKD population, beginning with stage 1 CKD and increasing with advancing stage, except for stage 3. Diabetes prevalence exceeded half the population in CKD stages 4 and 5. ORs showed a similar pattern of diabetes prevalence in CKD stages based on the MDRD Study20 and CKD-EPI21 equations. The finding of high odds of diabetes in patients with CKD was independent of traditional diabetes risk factors and continued after adjustment for age, sex, race, hypertension, obesity, and CVD. The implications of these findings are significant because the presence of hyperglycemia in patients with CKD is associated with a worse prognosis regarding CVD morbidity and mortality.6,9,10 Furthermore, early diagnosis of diabetes and aggressive control of CVD risk factors, such as hypertension, dyslipidemia, and other components of metabolic syndrome, are of paramount importance in decreasing the CVD burden and slowing the progression of CKD.23 Early recognition of diabetes and tight control of hyperglycemia can prevent or halt the progression of CKD and retinopathy.24,25 The United Kingdom Prospective Diabetes Study (UKPDS) showed that a decrease in hemoglobin A1c level from 8% to 7% decreased the rate of nephropathy by 32% and the rate of retinopathy by 21%.24,25 Similar findings have been shown in other populations.26
Regarding risk factors for type 2 diabetes, our study followed a pattern seen in previous reports11–14 in which age, sex, hypertension, obesity, CVD, and family history significantly increased the odds of diabetes. However, several additional observations were made in our study. The odds of diabetes were increased in whites compared with African Americans and other racial and ethnic groups. This may reflect increased diabetes self-reporting, early diagnosis, and treatment with antidiabetic medications in white populations. Furthermore, the rate of undiagnosed diabetes in African Americans and other racial and ethnic minorities might have been higher. This raises a limitation of our study: neither hemoglobin A1c nor glucose tolerance tests were used to confirm the diagnosis of diabetes.
Dyslipidemia, including low levels of high-density lipoprotein cholesterol and increased triglyceride levels, is a core component of metabolic syndrome and is associated with insulin resistance and increased risk of type 2 diabetes and CVD.7,27 In our study, dyslipidemia was defined as total cholesterol level >200 mg/dL or triglyceride level >150 mg/dL. It occurred in 43% of participants, but was not associated with a higher rate of diabetes (33.5% vs 33%; P = 0.2). This perhaps is due to lack of control for glycemic levels, lack of high-density lipoprotein measurement, and increased treatment for dyslipidemia in diabetic patients, as dictated by the standard of care.12
The relationship between diabetes and cigarette smoking is complex. Although studies have established smoking as a diabetes risk factor in both men and women,28,29 with the risk attributed to alteration in fat distribution, increased insulin resistance, and direct β cell toxicity,30,31 recent analyses from the Atherosclerosis Risk in Communities (ARIC) Study31 suggest a high short-term risk of diabetes with smoking cessation that was attributed to weight gain.31 In this analysis of the KEEP cohort, diabetes was less prevalent in smokers compared with ex-/nonsmokers; however, the adjusted OR for diabetes was not statistically significant. This lower prevalence of diabetes in smokers likely reflects smoking cessation efforts provided to people with diabetes and the tendency of patients with diabetes to quit smoking with longer diabetes duration.12,32 Another possible mechanism, in light of the ARIC Study, is possible weight gain associated with smoking cessation,31 which could explain the nonsignificant odds of diabetes for smokers versus ex-/nonsmokers. Of note, the overall rate of smoking in our KEEP cohort was 9.4% in diabetic and 11.5% in nondiabetic participants, almost half the rate of 20.6% reported in 2008 for the US population.33
Regarding waist circumference and risk of diabetes, obesity is a well-established risk factor for diabetes and CVD, and the rapid rise of the diabetes epidemic generally is attributed in large part to the obesity epidemic.34 BMI is used widely to define obesity. However, accumulating evidence indicates that waist circumference is a better predictor of diabetes and CVD risk than BMI in that it provides a better estimate of fat distribution that cannot be obtained by measuring BMI.35–38 In our analysis, participants with obesity were twice as likely to have diabetes as participants without obesity. However, in the subanalysis of 20,135 KEEP participants for whom waist circumference was obtained, the OR associated with BMI was attenuated from 2.06 to 1.14 when waist circumference was introduced in the model. Our data support the evidence that central obesity measured using waist circumference is a better predictor of diabetes than BMI.35–38
Finally, the increased risk of diabetes seen in all CKD stages after adjustment for traditional risk factors, such as age, sex, race, hypertension, CVD, obesity, and family history of diabetes, likely is a reflection of increased nontraditional risk factors for CVD in our predominantly obese cohort, including expected increases in levels of adipokines, cytokines, and acute-phase reactants; oxidative stress; microalbuminuria; insulin resistance; and endothelial dysfunction.7 These risk factors are common in patients with diabetes and those with CKD, and they significantly contribute to the increased risk of CVD in these patients.39–41 Albuminuria is the hallmark of stage 1 CKD, for which the odds of diabetes more than doubled compared with KEEP participants without CKD. These findings suggest a possible role of albuminuria and associated expected increases in level of adipokines, cytokines, and acute-phase reactants and endothelial dysfunction42,43 as the common-soil hypothesis in the development of diabetes and CKD observed in our KEEP cohort.
The U-shaped risk of diabetes in patients with CKD stages 1–5 may be explained by the combination of CKD (eGFR) and non-CKD (conventional) risk factors. It also could reflect the sample-size variation for each stage of CKD. Tighter CIs and lower ORs for stage 3 reflect the larger sample of about 18,000 participants; samples for the other stages were smaller and susceptible to confounding factors, such as microalbuminuria. This should be addressed in future studies. Furthermore, although our study indicated that diabetes is present in 53% of the CKD population at stages 4–5, data from US Renal Data System show the incidence of diabetes in patients with CKD to be <50%. This likely is explained because many patients with CKD die before initiating dialysis therapy.9
Our study is limited by the cross-sectional nature of the analysis and lack of measurement of possible confounders that could at least partially explain the increased prevalence of diabetes in patients with CKD. These include microalbuminuria and inflammatory and oxidative stress markers. Additional limitations include ascertainment of diabetes using blood glucose level and the absence of hemoglobin A1c measurement that currently is recommended for diabetes diagnosis.12
Acknowledgments
The KEEP Steering Committee comprises: George Bakris, MD, FACP, FASN; Peter McCullough, MD, MPH; Andrew Bomback, MD; Claudine Jurkovitz, MD, MPH; Bryan Kestenbaum, MD; Louis Kuritzky, MD; Samy McFarlane, MD, MPH, FACP; Rajendra H. Mehta, MD; Keith Norris, MD; Michael Shlipak, MD, MPH; James Sowers, MD; Manjula Kurella Tamura, MD, MPH; Lesley Stevens, MD, MS; Adam Whaley-Connell, DO, MSPH; Ex-Officio: Bryan Becker, MD; Allan Collins, MD, FACP; Andrew Narva, MD, FACP; Nilka Rios Burrows, MPH; Joseph Vassalotti, MD; Emeritus: Wendy Brown, MD, MPH, FACP, FAHA; Michael Klag, MD, MPH; Data Coordinating Center: Shu-Cheng Chen, MS; Corporate Partner Representatives: Dennis L. Andress, MD; Reshma Kewalramani, MD; Jose A. Menoyo, MD.
The authors thank Shane Nygaard, BA, and Nan Booth, MSW, MPH, ELS, of the Chronic Disease Group for manuscript preparation and manuscript editing, respectively.
Support: The KEEP is a program of the National Kidney Foundation Inc and is supported by Amgen, Abbott, Siemens, Astellas, Fresenius Medical Care, Genzyme, LifeScan, Nephroceuticals, and Pfizer. Dr Stevens receives grant support from Gilead Inc. Dr Whaley-Connell is supported by the Veteran’s Affairs Career Development Award-2. Dr Norris is supported by National Institutes of Health grants MD00182 and RR026138.
Footnotes
Financial Disclosure: Dr Norris has consulted with Amgen, King Pharmaceuticals, and Abbott. Dr Collins has consulted with Amgen. The remaining authors declare that they have no relevant financial interests.
References
- 1.Danaei G, Friedman AB, Oza S, Murray CJ, Ezzati M. Diabetes prevalence and diagnosis in US states: analysis of health surveys. Popul Health Metr. 2009;7:16. doi: 10.1186/1478-7954-7-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Shaw JE, Sicree RA, Zimmet PZ. Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res Clin Pract. 2010;87:4–14. doi: 10.1016/j.diabres.2009.10.007. [DOI] [PubMed] [Google Scholar]
- 3.Collins AJ, Foley RN, Gilbertson DT, Chen SC. The state of chronic kidney disease, ESRD, and morbidity and mortality in the first year of dialysis. Clin J Am Soc Nephrol. 2009;4(suppl 1):S5–S11. doi: 10.2215/CJN.05980809. [DOI] [PubMed] [Google Scholar]
- 4.Foley RN. Temporal trends in the burden of chronic kidney disease in the United States. Curr Opin Nephrol Hypertens. 2010;19:273–277. doi: 10.1097/MNH.0b013e328337bba7. [DOI] [PubMed] [Google Scholar]
- 5.Grundy SM, Benjamin IJ, Burke GL, et al. Diabetes and cardiovascular disease: a statement for healthcare professionals from the American Heart Association. Circulation. 1999;100:1134–1146. doi: 10.1161/01.cir.100.10.1134. [DOI] [PubMed] [Google Scholar]
- 6.McCullough PA, Li S, Jurkovitz CT, et al. CKD and cardiovascular disease in screened high-risk volunteer and general populations: the Kidney Early Evaluation Program (KEEP) and National Health and Nutrition Examination Survey (NHANES) 1999–2004. Am J Kidney Dis. 2008;51(suppl 2):S38–S45. doi: 10.1053/j.ajkd.2007.12.017. [DOI] [PubMed] [Google Scholar]
- 7.McFarlane SI, Banerji M, Sowers JR. Insulin resistance and cardiovascular disease. J Clin Endocrinol Metab. 2001;86:713–718. doi: 10.1210/jcem.86.2.7202. [DOI] [PubMed] [Google Scholar]
- 8.Sowers JR, Lester MA. Diabetes and cardiovascular disease. Diabetes Care. 1999;22(suppl 3):C14–C20. [PubMed] [Google Scholar]
- 9.Salifu MO, Abbott KC, Aytug S, et al. New-onset diabetes after hemodialysis initiation: impact on survival. Am J Nephrol. 2010;31:239–246. doi: 10.1159/000276542. [DOI] [PubMed] [Google Scholar]
- 10.Whaley-Connell A, Pavey BS, McCullough PA, et al. Dysglycemia predicts cardiovascular and kidney disease in the Kidney Early Evaluation Program. J Clin Hypertens (Greenwich) 2010;12:51–58. doi: 10.1111/j.1751-7176.2009.00190.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.American Diabetes Association. Executive summary: American Diabetes Association. standards of medical care in diabetes—2010. Diabetes Care. 2010;33(suppl 1):S4–S10. [Google Scholar]
- 12.American Diabetes Association. Standards of medical care in diabetes—2010. Diabetes Care. 2010;33(suppl 1):S11–S61. doi: 10.2337/dc10-S011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.The Diabetes Prevention Program Research Group. Strategies to identify adults at high risk for type 2 diabetes: the Diabetes Prevention Program. Diabetes Care. 2005;28:138–144. doi: 10.2337/diacare.28.1.138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Farag A, Karam J, Nicasio J, McFarlane SI. Prevention of type 2 diabetes: an update. Curr Diabetes Rep. 2007;7:200–207. doi: 10.1007/s11892-007-0032-4. [DOI] [PubMed] [Google Scholar]
- 15.Chadban S. New-onset diabetes after transplantation—should it be a factor in choosing an immunosuppressant regimen for kidney transplant recipients. Nephrol Dial Transplant. 2008;23:1816–1818. doi: 10.1093/ndt/gfn052. [DOI] [PubMed] [Google Scholar]
- 16.Iida S, Ishida H, Tokumoto T, et al. New-onset diabetes after transplantation in tacrolimus-treated, living kidney transplantation: long-term impact and utility of the pre-transplant OGTT. Int Urol Nephrol. 2010;42:935–945. doi: 10.1007/s11255-010-9712-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Luan FL, Langewisch E, Ojo A. Metabolic syndrome and new onset diabetes after transplantation in kidney transplant recipients. Clin Transplant. 2010;24:778–783. doi: 10.1111/j.1399-0012.2009.01194.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Marrero D, Hernandez D, Tamajon LP, et al. Pre-transplant weight but not weight gain is associated with new-onset diabetes after transplantation: a multi-centre cohort Spanish study. NDT Plus. 2010;3:ii15–ii20. doi: 10.1093/ndtplus/sfq065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Porrini E, Delgado P, Alvarez A, et al. The combined effect of pre-transplant triglyceride levels and the type of calcineurin inhibitor in predicting the risk of new onset diabetes after renal transplantation. Nephrol Dial Transplant. 2008;23:1436–1441. doi: 10.1093/ndt/gfm762. [DOI] [PubMed] [Google Scholar]
- 20.Levey AS, Coresh J, Greene T, et al. Using standardized serum creatinine values in the Modification of Diet in Renal Disease Study equation for estimating glomerular filtration rate. Ann Intern Med. 2006;145:247–254. doi: 10.7326/0003-4819-145-4-200608150-00004. [DOI] [PubMed] [Google Scholar]
- 21.Levey AS, Stevens LA, Schmid CH, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150:604–612. doi: 10.7326/0003-4819-150-9-200905050-00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.NHANES. [Accessed August 24, 2010];National Health and Nutrition Examination Survey III: body measurements (authropometry) 2008 http://www.cdc.gov/nchs/data/nhanes/nhanes3/cdrom/nchs/manuals/anthro.pdf.
- 23.McCullough PA, Verrill TA. Cardiorenal interaction: appropriate treatment of cardiovascular risk factors to improve outcomes in chronic kidney disease. Postgrad Med. 2010;122:25–34. doi: 10.3810/pgm.2010.03.2119. [DOI] [PubMed] [Google Scholar]
- 24.Adler AI, Stevens RJ, Manley SE, Bilous RW, Cull CA, Holman RR. Development and progression of nephropathy in type 2 diabetes: the United Kingdom Prospective Diabetes Study (UKPDS 64) Kidney Int. 2003;63:225–232. doi: 10.1046/j.1523-1755.2003.00712.x. [DOI] [PubMed] [Google Scholar]
- 25.UK Prospective Diabetes Study Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33) Lancet. 1998;352:837–853. [PubMed] [Google Scholar]
- 26.Shichiri M, Kishikawa H, Ohkubo Y, Wake N. Long-term results of the Kumamoto Study on optimal diabetes control in type 2 diabetic patients. Diabetes Care. 2000;23(suppl 2):B21–B29. [PubMed] [Google Scholar]
- 27.Eckel RH. Diabetic dyslipidemia and cardiovascular risk. Curr Diabetes Rep. 2008;8:421–423. doi: 10.1007/s11892-008-0072-4. [DOI] [PubMed] [Google Scholar]
- 28.Wannamethee SG, Shaper AG, Perry IJ. Smoking as a modifiable risk factor for type 2 diabetes in middle-aged men. Diabetes Care. 2001;24:1590–1595. doi: 10.2337/diacare.24.9.1590. [DOI] [PubMed] [Google Scholar]
- 29.Willi C, Bodenmann P, Ghali WA, Faris PD, Cornuz J. Active smoking and the risk of type 2 diabetes: a systematic review and meta-analysis. JAMA. 2007;298:2654–2664. doi: 10.1001/jama.298.22.2654. [DOI] [PubMed] [Google Scholar]
- 30.Rimm EB, Manson JE, Stampfer MJ, et al. Cigarette smoking and the risk of diabetes in women. Am J Public Health. 1993;83:211–214. doi: 10.2105/ajph.83.2.211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yeh HC, Duncan BB, Schmidt MI, Wang NY, Brancati FL. Smoking, smoking cessation, and risk for type 2 diabetes mellitus: a cohort study. Ann Intern Med. 2010;152:10–17. doi: 10.7326/0003-4819-152-1-201001050-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.McFarlane SI, Jacober SJ, Winer N, et al. Control of cardiovascular risk factors in patients with diabetes and hypertension at urban academic medical centers. Diabetes Care. 2002;25:718–723. doi: 10.2337/diacare.25.4.718. [DOI] [PubMed] [Google Scholar]
- 33.Centers for Disease Control and Prevention. Cigarette smoking among adults and trends in smoking cessation—United States, 2008. MMWR Morbid Mortal Wkly Rep. 2009;58:1227–1232. [PubMed] [Google Scholar]
- 34.Seidell JC. Obesity, insulin resistance and diabetes—a worldwide epidemic. Br J Nutr. 2000;83(suppl 1):S5–S8. doi: 10.1017/s000711450000088x. [DOI] [PubMed] [Google Scholar]
- 35.Feller S, Boeing H, Pischon T. Body mass index, waist circumference, and the risk of type 2 diabetes mellitus: implications for routine clinical practice. Dtsch Arztebl Int. 2010;107:470–476. doi: 10.3238/arztebl.2010.0470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Janiszewski PM, Janssen I, Ross R. Does waist circumference predict diabetes and cardiovascular disease beyond commonly evaluated cardiometabolic risk factors? Diabetes Care. 2007;30:3105–3109. doi: 10.2337/dc07-0945. [DOI] [PubMed] [Google Scholar]
- 37.Klein S, Allison DB, Heymsfield SB, et al. Waist circumference and cardiometabolic risk: a consensus statement from Shaping America’s Health: Association for Weight Management and Obesity Prevention; NAASO, the Obesity Society; the American Society for Nutrition; and the American Diabetes Association. Obesity (Silver Spring) 2007;15:1061–1067. doi: 10.1038/oby.2007.632. [DOI] [PubMed] [Google Scholar]
- 38.Lopatynski J, Mardarowicz G, Szczesniak G. A comparative evaluation of waist circumference, waist-to-hip ratio, waist-to-height ratio and body mass index as indicators of impaired glucose tolerance and as risk factors for type-2 diabetes mellitus. Ann Univ Mariae Curie Sklodowska Med. 2003;58:413–419. [PubMed] [Google Scholar]
- 39.Castro JP, El-Atat FA, McFarlane SI, Aneja A, Sowers JR. Cardiometabolic syndrome: pathophysiology and treatment. Curr Hypertens Rep. 2003;5:393–401. doi: 10.1007/s11906-003-0085-y. [DOI] [PubMed] [Google Scholar]
- 40.El-Atat FA, Stas SN, McFarlane SI, Sowers JR. The relationship between hyperinsulinemia, hypertension and progressive renal disease. J Am Soc Nephrol. 2004;15:2816–2827. doi: 10.1097/01.ASN.0000133698.80390.37. [DOI] [PubMed] [Google Scholar]
- 41.Shah S, Iqbal M, Karam J, Salifu M, McFarlane SI. Oxidative stress, glucose metabolism, and the prevention of type 2 diabetes: pathophysiological insights. Antioxid Redox Signal. 2007;9:911–929. doi: 10.1089/ars.2007.1629. [DOI] [PubMed] [Google Scholar]
- 42.Hanai K, Babazono T, Nyumura I, et al. Involvement of visceral fat in the pathogenesis of albuminuria in patients with type 2 diabetes with early stage of nephropathy. Clin Exp Nephrol. 2010;14:132–136. doi: 10.1007/s10157-009-0245-8. [DOI] [PubMed] [Google Scholar]
- 43.Suliman ME, Yilmaz MI, Carrero JJ, et al. Novel links between the long pentraxin 3, endothelial dysfunction, and albuminuria in early and advanced chronic kidney disease. Clin J Am Soc Nephrol. 2008;3:976–985. doi: 10.2215/CJN.03960907. [DOI] [PMC free article] [PubMed] [Google Scholar]