Abstract
We present the 16th reported case of Acute Lymphoblastic Leukemia (ALL) with involvement in the bladder. Our patient was a 22 year-old man with T-cell ALL with a mediastinal mass. He received hyperfractionated cyclophos-phamide, vincristine, doxorubicin, dexamethasone (HyperCVAD) with mediastinal radiation. Prior to starting maintenance, he relapsed in the bladder and marrow. He received a nelarabine-based induction regimen and achieved remission. This was followed by an unrelated 11/12 HLA-matched myeloablative allogeneic stem cell transplant. He is in complete remission for the past 409 days.
Key words: precursor t-cell acute lymphoblastic leukemia/lymphoma, bladder involvement, allogeneic stem cell transplantation.
Introduction
Bladder involvement in acute lymphoblastic lymphoma/leukemia is a rare entity. Here we present the 16th reported case of such a phenomenon. Our patient relapsed in the bladder and is now doing very well, one and a half years out from a myeloablative allogeneic transplant.
Case Report
Our patient was a 22 year-old white male with no significant past medical history. He was studying abroad in Hong Kong when he began experiencing several days of back pain. He visited a physician and a chest x-ray was ordered. It revealed a mediastinal mass. The patient returned to California. Soon after returning he began experiencing dyspnea, particularly when lying flat. He was seen by his local physician who ordered an excisional biopsy of a palpable left supraclavicular lymph node. Pathology revealed precursor T-cell acute lymphoblastic lymphoma. A PET-CT revealed a large anterior mediastinal mass with left supraclavicular adenopathy and right pleural soft tissue masses, all with elevated FDG activity. There was also a large right pleural effusion. The patient had a normal CBC at presentation to our hospital. His WBC count was 5.5×103/UL with a 73.2% neutrophils and 17.1% lymphocytes, hemoglobin was 14.3 G/DL, and platelets were 227×109/UL. A bone marrow biopsy was negative for disease with a normal male karyotype. The patient was then started on the HyperCVAD regimen. He completed all 8 cycles without complications and was in remission based on PET-CT findings. He then received radiation therapy to the mediastinal mass. The patient was getting ready to start maintenance chemotherapy when he began complaining of gross hematuria. A repeat PET-CT was performed which revealed increased uptake in the bladder and the bone marrow. His CBC at this time showed a WBC count of 2.0×103/UL with 51.3% neutrophils and 34.6% lymphocytes, a hemoglobin of 13.4, and a platelet count of 144,000×109/UL. This CBC was not appreciably different from his last month’s CBC at completion of his HyperCVAD. A urinalysis around this time revealed only a hazy clarity to the urine, 2 RBC/high power field (HPF), 1 WBC/HPF, and 2+ mucous but was otherwise normal. He was seen by urology and a cystoscopy noted a bladder mass. Bone marrow biopsy revealed 5–10% involvement of blasts consistent recurrent precursor T-cell acute lymphoblastic leukemia (Figures 1, 2). The flow cytometry identified these cells as expressing CD2, CD5 (weak), CD7, Cd10, and TdT with very weak/absent CD8 which was consistent with precursor T-cell lymphoblasts. The karyotype was 46XY. The patient returned to his urologist who performed a biopsy on the bladder mass that was also morphologically consistent with T-cell acute lymphoblastic leukemia although immunohistologic staining could not be performed (Figure 3).
Figure 1.

Hematoxylin and Eosin stain of bone marrow biopsy showing leukemic infiltration.
Figure 2.

Terminal deoxynucleotidyl transferase stain of bone marrow biopsy.
Figure 3.
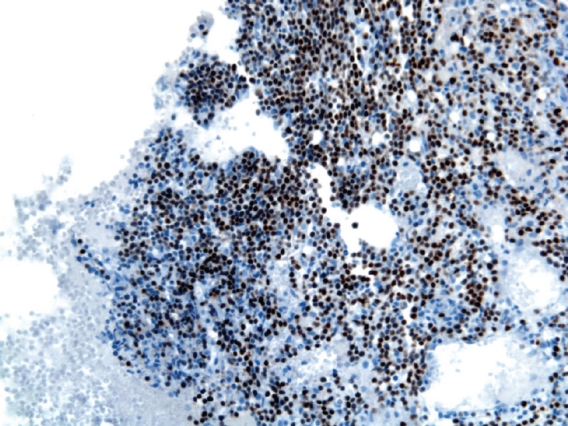
Hematoxylin and Eosin of bladder biopsy showing leukemic infiltration.
The patient received reinduction chemotherapy. His regimen matched a Children’s Oncology Group regimen incorporating nelarabine (patient was off protocol due to his age). He no longer experienced gross hematuria after the initiation of reinduction chemotherapy. He received therapy under this regimen for approximately 2 months and then proceeded to myeloablative allogeneic hematopoietic stem cell transplantation using an 11/12 unrelated donor. The conditioning regimen included total body irradiation with a cranial boost, cyclophosphamide, and etopo-side. The patient tolerated the regimen well with no GVHD or readmissions after his engraftment and discharge. He is now 409 days out from transplantation and is without relapse or complaints. He subsequently returned to college where he completed his last semester of college and received his degree.
Discussion
Acute lymphoblastic leukemia (ALL) is composed of a heterogenous group of malignancies and encompasses some of the most complex treatments in cancer. Precursor T-cell ALL is a subtype of ALL noted for its more successful long term outcome in adults when compared to B-cell ALL.1
The backbone of ALL induction regimens is comprised of combinations of vincristine, corticosteroids, and anthracyclines. Dexame-thasone is generally the steroid of choice due to its high levels of penetration into the cerebrospinal fluid.2 Complete remission rates have been reported at 72–92% with a median remission duration of 18 months.3 In our patient, prior to the discovery of bladder involvement, a hyper-CVAD regimen (hyperfractionated cyclophosphamide, vincristine, doxorubicin, dexamethasone), which included radiation for consolidation in patients with a mediastinal mass, was used. This protocol was reported by Thomas et al. as achieving a complete remission (CR) in 91% of patients studied with lymphoblastic lymphoma. At three years, progression free survival was 66% and overall survival was 70%.4 Our patient underwent eight cycles of hyper-CVAD and consolidation with radiation and was scheduled to undergo maintenance therapy prior to his relapse. An alternative initial strategy that could have been pursued was autologous stem cell transplantation which has also demonstrated favorable long-term outcomes in T-cell ALL. In a study by Vancouver General Hospital, patients were given primary therapy utilizing hematopoietic stem cell transplant. This strategy incorporated intensive therapy, which combined features of non-Hodgkin’s lymphoma and ALL treatment.
This was then followed by high dose consolidation therapy. In their study, the four year event free survival was 69% in those patients who received autografts. Some patients with bone marrow involvement and/or younger age proceeded to allogeneic transplant and did not demonstrate disease relapse at 38–141 months since time of diagnosis.5
Current therapies for T-cell ALL produce high responses, but not approximately one half of patients will relapse within 2 years.6 Our patient unfortunately relapsed just prior to his maintenance therapy. His initial salvage chemotherapy incorporated nelarabine in his regimen. This regimen was based on the Children’s Oncology Group (COG) protocol AALL0434. In this protocol, the Berlin-Frankfurt-Munster (BFM) chemotherapy combination is used as the backbone for intense combination chemotherapy.7 Patients may then be randomized to nelarabine in the consolidation cycles. Nelarabine has been shown to have antineoplastic activity in patients with relapsed/refractory T-cell ALL.8–9 As a result, nelarabine was approved by the Food and Drug Administration specifically for T-cell leukemias.10 The mechanism of action includes incorporation of ara-G, which is a deoxyguanosine analogue and the active metabolite of nelarabine, into the deoxyribonucleic acid (DNA) of leukemic blasts. This accumulation leads to inhibition of DNA synthesis and cell death. In the study by DeAngelo et al. from the Cancer and Leukemia Group B, nelarabine demonstrated complete remission rates of 31% with minimal toxicities in relapsed/refractory ALL patients even as a single agent.11 Current ongoing clinical trials are now evaluating nelarabine as a frontline agent in newly diagnosed ALL such as the above referenced COG study.12
Our patient had a rare presentation of relapse appearing in the bladder. In our review of the literature, only 15 such cases had been previously reported.13 Interestingly, gross hematuria as a presentation of acute leukemia is also quite rare as noted by the paucity of cases in the literature.14,15 Our patient’s PET findings revealed uptake suspicious in the bladder whereas the kidneys’ appearance on PET was more in line with physiologic uptake. However, ALL is known to more commonly relapse in the kidneys, and we propose that seeding into the bladder was a possible mechanism.16,17 The kidneys may act as a reservoir for leukemic cells even in a patient thought to be in complete remission.18 In an autopsy study performed at the National Institute of Health, it was noted that patients with leukemia thought to be in remission in the marrow actually had occult involvement in the kidneys.19 The authors noted that leukemic cell infiltrates were found at autopsy in the tissues of 10 of 15 acute leukemia patients dying during complete bone marrow remission and noted the kidneys as the most common site of leukemic infiltrate in deceased leukemia patients. In such cases of bladder involvement, tsre did not appear to be a standard of therapy.13 Our patient’s T cell ALL was clearly behaving aggressively, having relapsed in the marrow and bladder subsequent to chemotherapy and the decision was made to treat him as aggressively as possible with a myeloablative allogeneic stem cell transplant. He is now doing well over one year out from transplantation.
References
- 1.Jabbour EJ, Faderl S, Kantarjian HM. Adult acute lymphoblastic leukemia. Mayo Clin Proc. 2005;80:1517–27. doi: 10.4065/80.11.1517. [DOI] [PubMed] [Google Scholar]
- 2.Jones B, Freeman AI, Shuster JJ, et al. Lower incidence of meningeal leukemia when prednisone is replaced by dexamethasone in the treatment of acute lymphocytic leukemia. Med Pediatr Oncol. 1991;19:269–75. doi: 10.1002/mpo.2950190411. [DOI] [PubMed] [Google Scholar]
- 3.Stryckmans P, Debusscher L. Chemotherapy of adult acute lymphoblastic leukemia. Baillieres Clin Haematol. 1991;4:115–30. doi: 10.1016/s0950-3536(05)80287-2. [DOI] [PubMed] [Google Scholar]
- 4.Thomas DA, O'Brien S, Cortes J, et al. Outcome with the hyper-CVAD regimens in lymphoblastic lymphoma. Blood. 2004;104:1624–30. doi: 10.1182/blood-2003-12-4428. [DOI] [PubMed] [Google Scholar]
- 5.Song KW, Barnett MJ, Gascoyne RD, et al. Primary therapy for adults with T-cell lymphoblastic lymphoma with hematopoietic stem-cell transplantation results in favorable outcomes. Ann Oncol. 2007;18:535–40. doi: 10.1093/annonc/mdl426. [DOI] [PubMed] [Google Scholar]
- 6.Laport GF, Larson RA. Treatment of adult acute lymphoblastic leukemia. Semin Oncol. 1997;24:70–82. [PubMed] [Google Scholar]
- 7.Chaffee S. Intensified Methotrexate, Nelarabine (Compound 506U78; IND # 52611) and Augmented BFM therapy for children and young adults with newly diagnosed t-cell acute lymphoblastic lymphoma or t-cell lymphoblastic lymphoma. Available from: http://cancer.dartmouth.edu/pf/clinical_trial/AALL0434/
- 8.Tallen G, Ratei R, Mann G, et al. Long-term outcome in children with relapsed acute lymphoblastic leukemia after time-point and site-of-relapse stratification and intensified short-course multidrug chemotherapy: results of trial ALL-REZ BFM 90. J Clin Oncol. 2010;28:2339–47. doi: 10.1200/JCO.2009.25.1983. [DOI] [PubMed] [Google Scholar]
- 9.Berg SL, Blaney SM, Devidas M, et al. Phase II study of nelarabine (compound 506U78) in children and young adults with refractory T-cell malignancies: a report from the Children’s Oncology Group. J Clin Oncol. 2005;23:3376–82. doi: 10.1200/JCO.2005.03.426. [DOI] [PubMed] [Google Scholar]
- 10.Accelerated and Restricted Approvals Under Subpart H (drugs) and Subpart E (biologics) Available from: http://www.fda.gov/Drugs/DevelopmentApprovalProcess/HowDrugsareDevelopedandApproved/DrugandBiologicApprovalReports/AcceleratedandRestrictedApprovals/default.
- 11.DeAngelo DJ, Yu D, Johnson JL, et al. Nelarabine induces complete remissions in adults with relapsed or refractory T-lineage acute lymphoblastic leukemia or lymphoblastic lymphoma: Cancer and Leukemia Group B study 19801. Blood. 2007;109:5136–42. doi: 10.1182/blood-2006-11-056754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.ClinicalTrials.gov. Phase II study of hyper-CVAD plus nelarabine in previously untreated T-ALL and lymphoblastic lymphoma. Available from: http://clinicaltrials.gov/ct2/show/NCT00501826.
- 13.Martín Hernández M, Alonso y Gregorio S, Cansino Alcaide R, et al. Leukemic infiltration of the urinary bladder. A new case and literature review. Actas Urol Esp. 2008;32:563–6. doi: 10.1016/s0210-4806(08)73885-3. [DOI] [PubMed] [Google Scholar]
- 14.Suriya OM, Aleem A. Frank hematuria as the presentation feature of acute leukemia. Saudi J Kidney Dis Transpl. 2010;21:940–2. [PubMed] [Google Scholar]
- 15.Chang C, Chiou T, Hsieh Y, Cheng S. Leukemic infiltration of the urinary bladder presenting as uncontrollable gross hematuria in a child with acute lymphoblastic leukemia. J Pediatr Hematol Oncol. 2003;25:735–9. doi: 10.1097/00043426-200309000-00012. [DOI] [PubMed] [Google Scholar]
- 16.Hoelzer D. Acute lymphoblastic leukemia in adults. In: Hoffman R, editor. Hematology: Basic Principles and Practice. Churchill Livingstone; New York, NY: 1991. pp. 793–804. [Google Scholar]
- 17.Banday KA, Sirwal IA, Reshi AR, et al. Renal involvement in hematologic neoplasia. Indian J Nephrol. 2004;14:50–52. [Google Scholar]
- 18.Lundberg WB, Cadman ED, Finch SC, Capizzi RL. Renal failure secondary to leukemic infiltration of the kidneys. Am J Med. 1977;62:636–42. doi: 10.1016/0002-9343(77)90427-2. [DOI] [PubMed] [Google Scholar]
- 19.Nies BA, Bodey GP, Thomas LB, et al. The persistence of extramedullary leukemic infiltrates during bone marrow remission of acute leukemia. Blood. 1965;26:133–41. [PubMed] [Google Scholar]


