Abstract
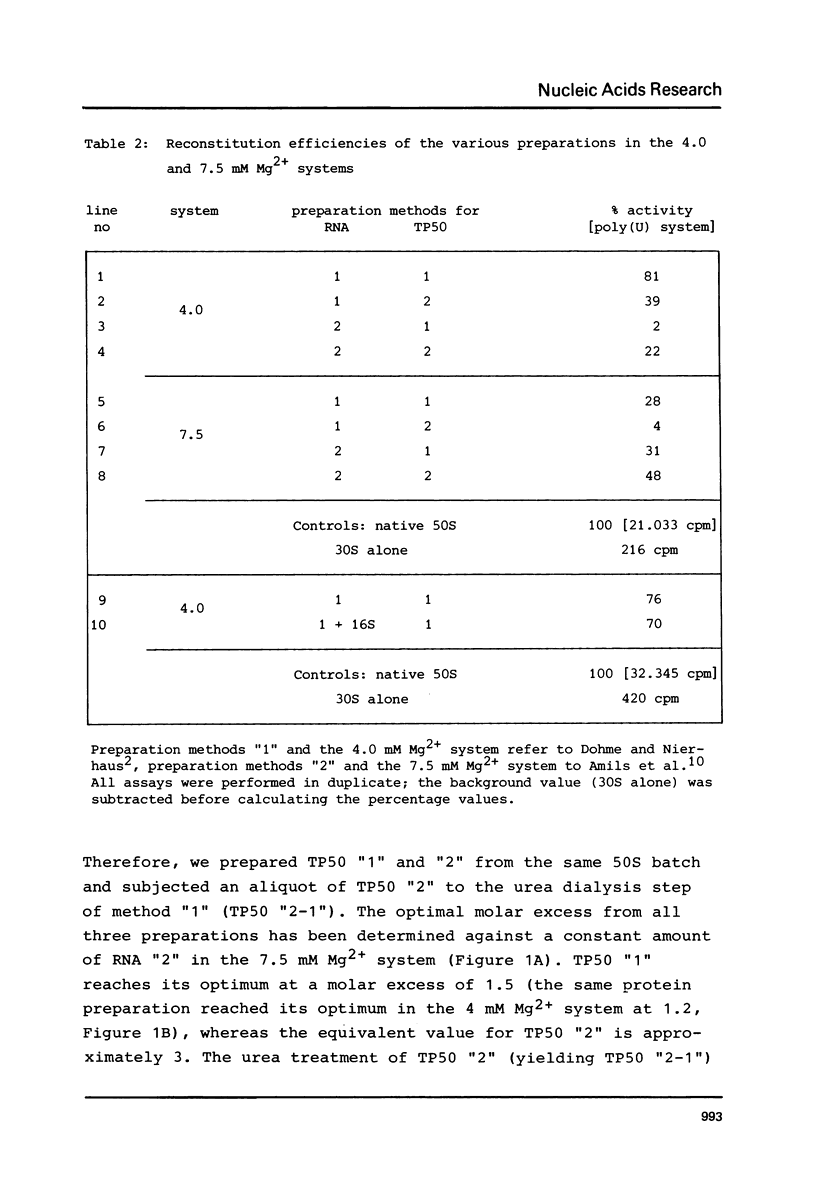
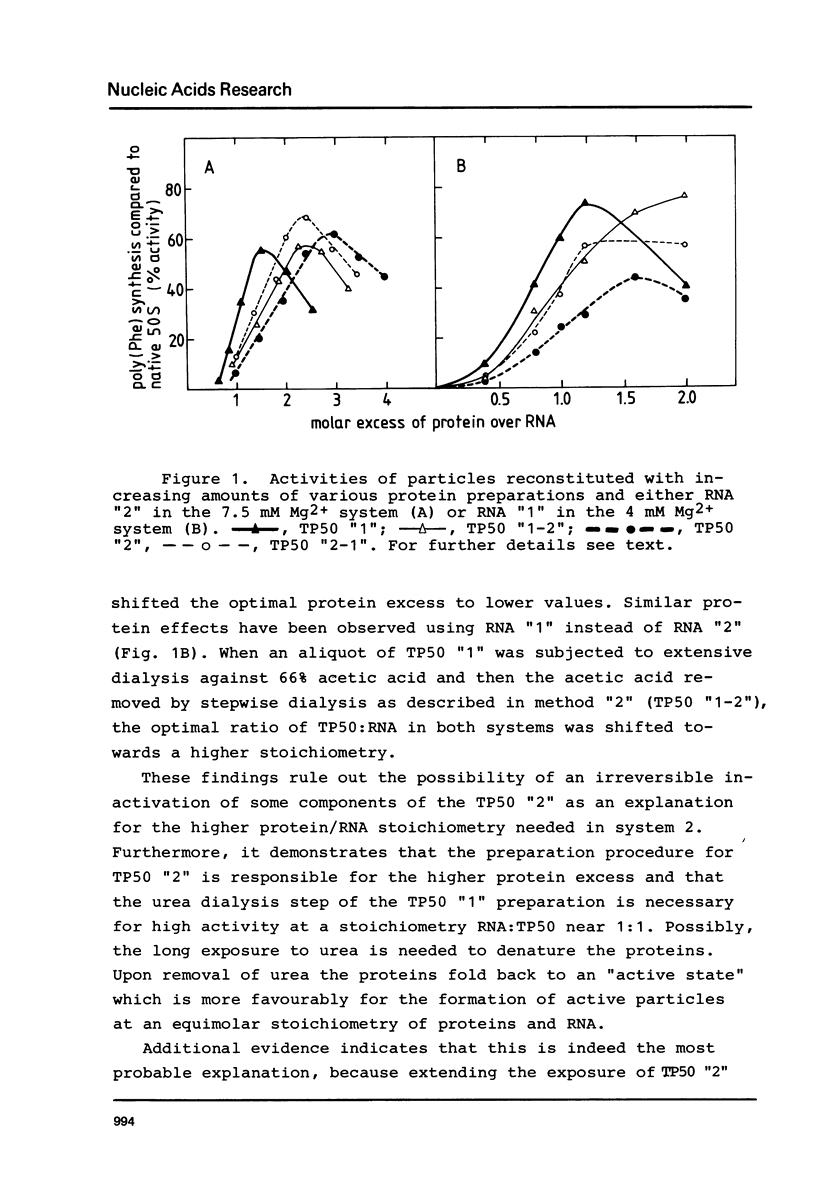
A two-step procedure has been described for the total reconstitution of 50S ribosomal subunit from E. coli. RNA and proteins are mixed with stoichiometry of 1:1.2 and incubated at 44 degrees C in 4.0 mM Mg2+ followed by a second incubation at 50 degrees C in 20 mM Mg2+ (Dohme and Nierhaus, J. Mol. Biol. 107, 585 (1976)). A modified method recently reported makes use of an altered preparation technique for the RNA and proteins and requires an RNA to protein stoichiometry of 1:2.5 and 7.5 mM Mg2+ in the first incubation (Amils et al., Nucl. Acid Res. 5, 2455 (1978)). The latter requirements are not compatible with the findings obtained with the first procedure. A comparison of the various RNA and protein fractions from the different groups revealed that the Mg2+ dependence of reconstitution is a function of the RNA preparation, whereas the stoichiometry depends upon the technique used for isolation of the protein fraction. The different RNA preparations were compared in the electron microscope.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Amils R., Matthews E. A., Cantor C. R. An efficient in vitro total reconstitution of the Escherichia coli 50S ribosomal subunit. Nucleic Acids Res. 1978 Jul;5(7):2455–2470. doi: 10.1093/nar/5.7.2455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dohme F., Nierhaus K. H. Role of 5S RNA in assembly and function of the 50S subunit from Escherichia coli. Proc Natl Acad Sci U S A. 1976 Jul;73(7):2221–2225. doi: 10.1073/pnas.73.7.2221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dohme F., Nierhaus K. H. Total reconstitution and assembly of 50 S subunits from Escherichia coli Ribosomes in vitro. J Mol Biol. 1976 Nov 15;107(4):585–599. doi: 10.1016/s0022-2836(76)80085-x. [DOI] [PubMed] [Google Scholar]
- Nierhaus K. H., Dohme F. Total reconstitution of functionally active 50S ribosomal subunits from Escherichia coli. Proc Natl Acad Sci U S A. 1974 Dec;71(12):4713–4717. doi: 10.1073/pnas.71.12.4713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nierhaus K. H., Montejo V. A protein involved in the peptidyltransferase activity of Escherichia coli ribosomes. Proc Natl Acad Sci U S A. 1973 Jul;70(7):1931–1935. doi: 10.1073/pnas.70.7.1931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schreiner G., Nierhaus K. H. Protein involved in the binding of dihydrostreptomycin to ribosomes of Escherichia coli. J Mol Biol. 1973 Nov 25;81(1):71–82. doi: 10.1016/0022-2836(73)90248-9. [DOI] [PubMed] [Google Scholar]
- Sieber G., Nierhaus K. H. Kinetic and thermodynamic parameters of the assembly in vitro of the large subunit from Escherichia coli ribosomes. Biochemistry. 1978 Aug 22;17(17):3505–3511. doi: 10.1021/bi00610a013. [DOI] [PubMed] [Google Scholar]
- Spillmann S., Dohme F., Nierhaus K. H. Assembly in vitro of the 50 S subunit from Escherichia coli ribosomes: proteins essential for the first heat-dependent conformational change. J Mol Biol. 1977 Sep 25;115(3):513–523. doi: 10.1016/0022-2836(77)90168-1. [DOI] [PubMed] [Google Scholar]
- Spillmann S., Nierhaus K. H. The ribosomal protein L24 of Escherichia coli is an assembly protein. J Biol Chem. 1978 Oct 10;253(19):7047–7050. [PubMed] [Google Scholar]
- Teraoka H., Nierhaus K. H. Proteins fro Escherichia coli ribosomes involved in the binding of erythromycin. J Mol Biol. 1978 Dec 5;126(2):185–193. doi: 10.1016/0022-2836(78)90358-3. [DOI] [PubMed] [Google Scholar]
- Yamada T., Mizugichi Y., Nierhaus K. H., Wittmann H. G. Resistance to viomycin conferred by RNA of either ribosomal subunit. Nature. 1978 Oct 5;275(5679):460–461. doi: 10.1038/275460a0. [DOI] [PubMed] [Google Scholar]




