Abstract
BACKGROUND
Defects in extravillous trophoblast (EVT) function could contribute to placental dysfunction resulting in adverse obstetrical outcomes. Adverse obstetrical outcomes have been highly correlated with intrauterine infection; however, the mechanisms linking infection to placental dysfunction remain unclear. We investigated the effects of inflammation on EVT cytokine production and invasion early in pregnancy and determined the cell signaling pathways mediating this response.
METHODS AND RESULTS
In our model of inflammation, EVT cells, isolated following first trimester pregnancy terminations (n= 6) were stimulated with lipopolysaccharide (LPS). LPS induced a dose-dependent increase in interleukin (IL)-8 and IL-6 protein production (P < 0.01) and decreased EVT invasion (P = 0.01) versus control. The LPS-mediated changes in cytokine production (P < 0.001) and invasion (P < 0.001) were reversed by dexamethasone (DEX). Exposure to LPS resulted in an increase in mitogen-activated protein kinase (MAPK) signaling pathway phosphorylation, including p44/42 MAPK (P < 0.01), p38 MAPK (P < 0.05), MAPK extracellular signal-regulated kinase 1/2 (MEK1/2) (P< 0.01) and stress-activated protein kinase/c-Jun N-terminal kinase (JNK; P < 0.001), which was reversed by DEX (P < 0.05) for all MAPKs except p38. MAPK-specific inhibitors to MEK1/2 (U0126), p38 MAPK (SB 202190) and JNK (SP 600125) significantly reversed the LPS-mediated increase in IL-6 (P < 0.001) and IL-8 (P < 0.001) production. While U0126 reversed the LPS-induced decrease in EVT invasion (P < 0.001), SB 202190 (P < 0.001) and SP 600125 (P< 0.001) decreased EVT invasion, further indicating that MEK1/2 phosphorylation may be inflammation dependent while p38 MAPK and JNK phosphorylation occurs independently of an inflammatory stimulus.
CONCLUSIONS
LPS increased IL-8 and IL-6 and decreased EVT invasion through activation of MAPK signaling. MEK1/2 activation may contribute to placental dysfunction, in the setting of inflammation-associated adverse obstetrical outcomes.
Keywords: extravillous trophoblast, lipopolysaccharide, interleukin-6, interleukin-8, mitogen-activated protein kinase
Introduction
A successful pregnancy is directly dependent on normal development of the placenta and adequate function of the trophoblast cells. The extravillous trophoblast cells (EVTs) are a highly proliferative and migratory cell population that invades the uterine spiral arteries allowing for an increase in maternal blood flow to the placenta (Brosens et al., 1967; Kurman, 1992; Graham and Lala, 1992; Genbacev et al., 1993; Kaufmann and Castellucci, 1997). Adverse obstetrical outcomes, including pre-eclampsia, intrauterine growth restriction (IUGR) and preterm birth, are often diagnosed during the later stages of pregnancy; however, there is a growing body of research which suggests that these adverse obstetrical outcomes may all stem from a common abnormality in placental function (Ness and Sibai, 2006).
Defects in EVT cellular function involving inflammatory factors within the placental environment could be contributing to the placental dysfunction seen in IUGR, pre-eclampsia (Arechavaleta-Velasco et al., 2002; Dadelszen and Magee, 2002; Dadelszen et al., 2003) and preterm birth (Goldenberg et al., 2000; Gonçalves et al., 2002; Lamont, 2003). Although signs of systemic illness or inflammation are not seen in most women with adverse pregnancy outcomes, evidence of localized inflammation (histological chorioamnionitis) is observed in >85% of spontaneous preterm births delivered at <28 weeks gestation (Yoon et al., 2000) suggesting that intrauterine infection may occur early in pregnancy, preceding any observed pregnancy complications (Gonçalves et al., 2002). Thus, infection and/or inflammation occurring at the maternal–fetal interface during early pregnancy could contribute to the abnormal placental function associated with adverse obstetrical outcomes. However, the specific mechanisms regulating this process are unknown.
The innate immune system comprises cells that express a series of receptors known as the toll-like receptors (TLRs) (Janeway and Medzhitov, 2002; Medzhitov and Janeway, 2002). TLRs have the ability to recognize uniquely expressed, pathogen-associated molecular patterns, such as bacterial lipopolysaccharide (LPS). LPS is recognized by the TLR4 receptor (Poltorak et al., 1998; Hoshino et al., 1999), which most commonly acts through a MyD88-dependent intracellular signaling pathway, activates nuclear factor (NF)-κB and consequently initiates an immune response, including an increase in cytokines and co-stimulatory molecules (Medzhitov and Janeway, 2002, Takeda et al., 2003; Abrahams et al., 2005). While transcription factors such as NF-κB are common to several cell types and play a general role in the regulation of inflammatory genes, other intracellular signaling pathways, such as the mitogen-activated protein kinase (MAPK) pathway, have also been shown to be activated during an inflammatory state. Previous studies have shown that LPS activates the MAPK pathway including extracellular signal-regulated kinase (ERK) (ERK1/2; 44 and 42 kDa, respectively), stress-activated protein kinase/c-Jun N-terminal kinase (SAPK/JNK), MAPK/ERK Kinase [MAPK extracellular signal-regulated kinase (MEK)1/2] and p38 MAPK in many cell types, specifically in term syncytiotrophoblasts (Lucchi and Moore, 2007), trophoblasts (Jiang et al., 2007) and choriodecidua explants (Shoji et al., 2007), providing evidence that the MAPKs are involved in the placental immune response. Additionally, MAPKs have been shown to regulate trophoblast motility and migration (Chakraborty et al., 2003; Qiu et al., 2004a,b; LaMarca et al., 2008; Prast et al., 2008). Collectively, these data suggest that MAPK pathways may provide a mechanistic link between inflammation and trophoblast invasion in placental tissues. There are no studies to date that have investigated the effects of LPS on first trimester EVT MAPK activity.
We hypothesize that local infection and/or inflammation at the level of the uterus/decidua are key contributors to the pathogenesis of certain pregnancy complications. We theorize that micro-organisms, specifically gram-negative bacteria which express LPS, can activate TLR4 (Lien et al., 2000; Tapping et al., 2000) in trophoblasts, resulting in production of the pro-inflammatory cytokines, interleukin (IL)-8 and IL-6 and altered trophoblast invasion. While it has been posited that placental dysfunction, resulting from activation of the innate immune response, is involved in the development of many adverse obstetrical outcomes (Kaga et al., 1996; Elovitz et al., 2003; Adams Waldorf et al., 2008), there is a paucity of research focused on the effects of infection and inflammation on first trimester placental EVT function. Therefore, the present studies were performed to help to elucidate the effects of inflammation on placental EVT function, including the production of pro-inflammatory cytokines and trophoblast invasion, and to determine if the MAPK signaling pathway regulates this response. Our studies, in a model system, could help to identify novel mechanisms involved in the regulation of cytokine expression and trophoblast invasion during an inflammatory state and thus identify new potential treatment targets or biomarkers early in pregnancy, before the onset of clinical symptoms, for those women with adverse obstetrical outcomes.
Materials and Methods
Cell culture
The collection of first trimester EVTs was approved by the Institutional Review Board at the University of Pennsylvania (IRB# 700943). Patient consents were not required.
Primary EVTs were isolated from first trimester villous tissue using a modified protocol originally described by Graham et al. (1992) and Getsios et al. (1998). Briefly, finely minced chorionic villi, collected from de-identified elective first trimester pregnancy termination tissues (n= 6), were cultured at 37°C in RPMI 1640 medium containing 20% fetal bovine serum (FBS). EVTs, which outgrow from attached villous fragments, were separated from villous tissue on the 10–12th day of culture. The isolated EVTs were cultured and propagated in RPMI 1640 medium containing 20% FBS. The EVTs used in our experiments were previously characterized by immunostaining for the trophoblast cell markers cytokeratin-8 and -18 and human leukocyte antigen-G (HLA-G) (Neudeck et al., 1997). In previous preparations in our laboratory, >95% of isolated trophoblast cells stained positive for cytokeratin-8 and -18 and HLA-G (Koi et al., 2002). In the current study, staining for cytokeratin-7 and integrin alpha-1 provided further evidence of the purity of the EVT cell population, as shown by other investigators using similar primary cultures, and confirming the purity of our EVT cell preparations (Graham et al., 1992; Getsios et al., 1998).
Our preliminary experiments investigating the response of the isolated primary EVTs to LPS treatment by measuring IL-8 released into the media showed a wide range of cytokine production (data not shown). We hypothesize that this is most likely a result of the genetic variation in TLR responsiveness observed in human studies (Kline et al., 1999; Schwartz, 2001). While all six placentas tested showed a response to LPS, we used only those EVTs isolated from the three placentas that showed a more robust response (greater than 15-fold increase in IL-8 expression) to perform the following experiments because we were most interested in determining the function of trophoblast cells in the presence of an inflammatory response.
Immunocytochemistry
To verify the purity of the isolated EVT cells, they were stained for trophoblast-specific markers cytokeratin-7 and integrin alpha-1. EVTs were plated at 2 × 105 cells/ml onto glass chamber slides. After 24h, the slides were fixed with 10% buffered formalin. EVTs were stained using the avidin:biotin enzyme complex method following the protocol provided with the Vectastain Elite ABC Kit (Vector Laboratories, Burlingame, CA, USA). The primary antibodies used were a mouse monoclonal antibody to cytokeratin-7, clone OV-TL 12/20 (Dako, Carpinteria, CA, USA) at a 1:50 dilution and a mouse monoclonal antibody to integrin alpha-1, clone FB12 (Millipore, Billerica, MA, USA) at a dilution of 1:100. The secondary antibody was a biotinylated horse anti-mouse immunoglobulin G followed by the addition of the avidin:biotin enzyme complex (ABC). Sections were stained brown with 3,3′-diaminobenzidine (Sigma Chemical Co., St. Louis, MI, USA) and counterstained with hematoxylin (Dako, Carpinteria, CA, USA) before being dehydrated and mounted. To validate the staining procedure, EVTs were incubated without the primary antibody. Staining for each antibody was performed on duplicate slides with each slide containing a section incubated with and without (negative control) the primary antibody. Stained cells were photographed at ×10 magnification.
Trophoblast cell treatments
EVTs were cultured as described above, plated at 1 × 105 cells/ml and grown until 90% confluent. These cells were exposed to LPS (Escherichia coli 055:B5) (0–100 μg/ml) (Sigma, St. Louis, MO, USA) for 24h. Primary EVT cells, cultured in 1640 RPMI medium containing 20% charcoal-stripped (steroid-free) FBS, were pretreated with dexamethasone (100 nM) (DEX; Sigma) for 2h prior to LPS (1 μg/ml) treatment. DEX vehicle control (ethanol)-treated EVTs were included in these experiments. Experiments investigating the involvement of the MAPK pathway in trophoblast function were performed by pretreating EVTs with MAPK-specific inhibitors (50μM), including U0126 (MEK1/2 inhibitor), SB 202190 (p38 MAP Kinase inhibitor) and SP 600125 (JNK inhibitor) (Calbiochem, Gibbstown, NJ, USA), for 1h prior to LPS treatment (1 μg/ml) for 24h. MAPK inhibitor vehicle control [dimethylsulfoxide (DMSO)]-treated EVTs were included in these experiments. The medium was collected and used for the assays described below.
Cytokine enzyme-linked immunosorbent assay
Previous studies have shown that gram-negative bacteria (LPS)-induced activation of TLR4 results in up-regulation of cytokines in term trophoblasts (Shimoya et al., 1992; Ma et al., 2006), therefore, we measured IL-8 and IL-6 protein in cell culture media from EVT (n = 4) cells after 24h of treatment with LPS (0–100 μg/ml) with or without DEX (100 nM) or MAPK inhibitor (50μM) pretreatment. IL-8 and IL-6 were measured by ligand specific commercially available enzyme-linked immunosorbent assay (ELISA) kits using reagents from R&D Systems (Minneapolis, MN, USA). Data from experiments measuring IL-8 and IL-6 after DEX or MAPK inhibitor pretreatment are expressed as a percentage of LPS-treated EVTs at baseline. IL-8 and IL-6 ELISA results are representative of four independent experiments.
Matrigel invasion assay
The invasiveness of primary EVTs (n = 3) through an extracellular matrix (ECM) was measured using a commercially available cell invasion assay kit (Chemicon, Temecula, CA, USA). Briefly, EVTs (1 × 105 cells/ml) were plated onto ECMatrix gel-coated cell culture inserts in 24-well plates and exposed to LPS (0.1 μg/ml) with or without DEX (100 nM) or MAPK inhibitor (50 μM) pretreatment. An LPS (0–1 μg/ml) dose–response experiment was performed (data not shown) and the 0.1 μg/ml dose of LPS was the lowest effective dose resulting in a robust change in trophoblast invasion. After 72h, the non-invading cells and the ECMatrix gel from the upper surface of the inserts was removed using a cotton-tipped swab. Invasive cells on the lower surface of the membrane were stained with 0.2% crystal violet. The membranes were mounted on microscope slides. Stained cells from three random microscope fields at ×20 magnification were photographed, counted and analyzed. Data from experiments measuring EVT invasion after DEX or MAPK inhibitor pretreatment are expressed as a percentage of control (non-treated cells). Invasion assay results are representative of EVTs isolated from three different placentas run in duplicate wells.
Lactate dehydrogenase cytotoxicity assay
The viability of EVTs after treatment with LPS (1 μg/ml) or MAPK inhibitors (50μM) was determined by measuring lactate dehydrogenase (LDH) leakage into the medium using the CytoTox 96 Non-Radioactive Cytotoxicity Assay (Promega Corp., Madison, WI, USA). After collecting cell culture medium, the trophoblast cells were lysed with RIPA Buffer containing protease (Complete Protease Inhibitor Cocktail Tablets, Roche Diagnostics, Mannheim, Germany) and phosphatase (Phosphatase Inhibitor Cocktail 2 and 3, Sigma) inhibitors. Released LDH (an indirect measure of loss of cell membrane integrity) was measured with a coupled enzymatic assay that results in the conversion of a tetrazolium salt into a red formazan product. The amount of color formed is proportional to the number of lysed cells. The enzymatic reaction was measured spectrophotometrically by absorbance readings at 490 nm. The percentage of cytotoxicity was calculated as experimental LDH (cell culture media)/maximum release of LDH (cell lysates). Results from the LDH cytotoxicity assay are representative of LPS-treated cells from seven independent experiments and MAPK-treated cells from three independent experiments.
Trophoblast cell lysates
Isolated EVTs were plated at 1 × 105 cells/ml until 90% confluent. These cells were exposed to LPS (1 μg/ml) (Sigma) for 0, 5, 10, 20, 40 and 80min. Experiments with DEX (100 nM) pretreatment (2h) followed by LPS were also performed. At each time point, the cells were lysed with cell lysis buffer (Cell Signaling Technology, Danvers, MA, USA) containing protease (Roche, Indianapolis, IN, USA) and phosphatase inhibitors (Sigma). Cell lysates were centrifuged and the supernatant was used for the MAPK multi-target ELISA described below.
MAPK multi-target ELISA
Activation of the MAPK signaling pathway occurs through protein phosphorylation. Phospho-p44/42 MAPK (Thr202/Tyr204) (ERK1/2), phospho-p38 MAPK (Thr180/Tyr182), total MEK1, phospho-MEK1/2 (Ser217/221), total SAPK/JNK and phospho-SAPK/JNK (Thr183/Tyr185) was measured in EVT cell lysates after LPS treatment (n = 5) with or without DEX pretreatment (n = 4). Phosphorylation of MAPK pathway members was measured from 0 to 80min in order to identify the time of peak activation. Phosphorylated MAPKs were measured by a multi-target commercially available solid phase sandwich ELISA (Cell Signaling Technology). Data from experiments measuring phosphorylated MAPK signaling proteins in EVTs are expressed as a percentage of control and are representative of four or five independent experiments.
Statistical analysis
Statistical analysis was performed for all experiments with the Sigma Stat Software (Statistical Package for the Social Sciences Inc., Chicago, IL, USA). For data that were normally distributed, one-way analysis of variance (ANOVA) or repeated measures ANOVA was used. If statistical significance was reached (P < 0.05), then pair-wise comparison by Student–Newman–Keuls test or comparison to control by Dunn's method was performed. When the treatment groups were unequal and/or the data were non-parametric, ANOVA on ranks followed by the Dunn's method for pair-wise comparisons were utilized. For results requiring comparisons of multiple groups, two-way repeated measures ANOVA was used, followed by comparison to control by the Holm–Sidak method. A P < 0.05 was considered statistically different. All arithmetic means are presented ± SEM. All experiments were performed in duplicate and repeated at least three times.
Results
Characterization of isolated EVTs
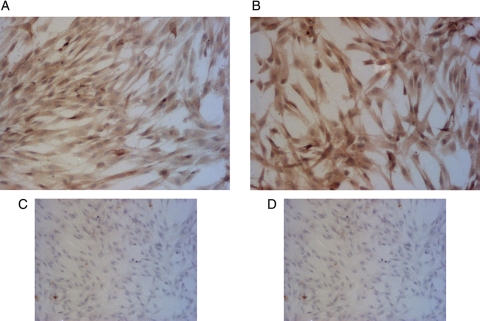
To ensure that our EVT isolation procedure produced a pure population of EVT cells, we stained the isolated cells with the trophoblast cell markers, cytokeratin-7 and integrin alpha-1. As shown by the brown staining in Fig. 1, the isolated EVTs expressed both cytokeratin-7 (Fig. 1A) and integrin alpha-1 (Fig. 1B). Additionally, staining done in the absence of the primary antibodies for cytokeratin-7 (Fig. 1C) and integrin alpha-1 (Fig. 1D) showed very little brown color, validating the specificity of the primary antibodies.
Figure 1.
Immunocytochemical staining of cytokeratin-7 (A) and integrin alpha-1 (B) in primary human EVT cells. Formalin fixed EVTs were stained using the avidin:biotin method. The cells were counterstained with hematoxylin. Positive cells are stained brown and the nuclei are blue. To validate the staining procedure, EVTs were incubated without cytokeratin-7 (C) or integrin alpha-1 (D) primary antibody. Pictures shown were taken with a bright-field microscope at ×10 magnification.
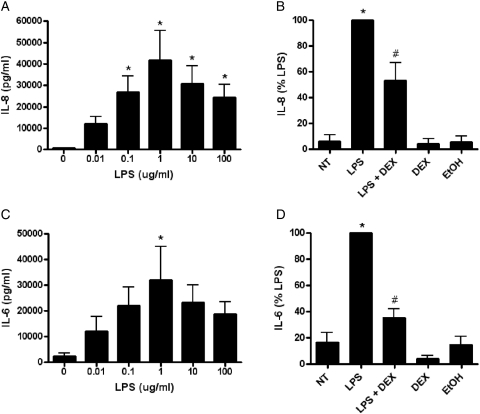
Effect of LPS on EVT IL-8 and IL-6 cytokine production
After observing TLR4 protein and mRNA in EVTs (data not shown), we wanted to investigate if these receptors were functional by determining IL-8 and IL-6 cytokine production after LPS treatment. LPS treatment (for 24 h) caused a dose-dependent increase in IL-8 (P < 0.01) and IL-6 (P < 0.05) production in EVTs when compared with non-treated cells (Fig. 2A and C, respectively). Additionally, pretreatment with DEX significantly reversed the LPS (1 μg/ml)-mediated increase in IL-8 (P < 0.001) and IL-6 (P < 0.001) in EVTs when compared with non-treated cells (Fig. 2B and D, respectively). Treatments with DEX alone and the DEX vehicle control (EtOH) were unchanged compared with non-treated cells.
Figure 2.
The effect of LPS on IL-8 and IL-6 protein expression with or without dexamethasone pretreatment in human EVTs. IL-8 (A) and IL-6 (C) protein expression was measured in EVT cell culture supernatants by ELISA after 24h of incubation with LPS. Pretreatment (2h) with DEX (100 nM) reversed the LPS (1 μg/ml) mediated increase in IL-8 (B) and IL-6 (D) protein expression. DEX alone and ethanol (EtOH) (DEX vehicle control) treatments were also included as negative controls. Each bar represents mean ± SEM. Data in Fig. 2B and D are presented as a percentage of LPS-treated EVT cells. Three independent experiments were performed with cells isolated from three different placentas. *P < 0.05 versus non-treated control, one-way repeated measures analysis of variance (RM ANOVA), #P < 0.001 versus LPS, one-way RM ANOVA, Student–Newman–Keuls (SNK).
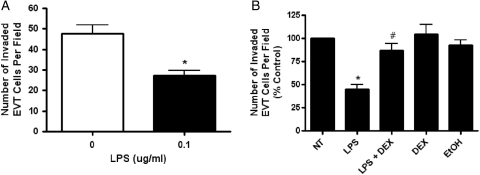
Effect of LPS on EVT invasion
The LPS-mediated increase in cytokine response in EVTs led us to investigate the effects of LPS on trophoblast invasion. LPS treatment resulted in a decrease in EVT (P= 0.01) trophoblast cell invasion through the ECM (Fig. 3A). Additional experiments examining the effects of DEX pretreatment on EVT invasion showed a significant reversal of the LPS-mediated decrease in invasion of EVTs (P < 0.001; Fig. 3B). Treatment with DEX alone or vehicle control (EtOH) did not alter EVT trophoblast invasion when compared with non-treated cells. Additionally, treatment with LPS had no effect on levels of LDH released into the medium from EVTs (0.344 % cytotoxicity non-treated versus 0.355 % cytotoxicity 1 μg/ml LPS, P = 0.70). EVT proliferation was also unchanged after LPS treatment, as maximal LDH readings from the cell lysates did not differ (2.58 absorbance of non-treated versus 2.52 absorbance of 1 μg/ml LPS, P = 0.96) (data not shown). These results indicate that the decreased invasion observed in EVTs was not a result of cell death caused by LPS but is instead a regulated response to an inflammatory stimulus.
Figure 3.
The effect of LPS on first trimester primary EVT invasion with or without DEX pretreatment. (A) EVT cell invasion through a matrigel-coated membrane was measured after treatment with LPS (0.1 μg/ml). (B) Pretreatment (2h) with DEX (100 nM) reversed the LPS mediated decrease in EVT invasion. DEX alone and EtOH (DEX vehicle control) treatments were included as negative controls. Each bar represents mean ± SEM. Three independent experiments were performed with cells isolated from three different placentas. The data in Fig. 3B are presented as a percentage of the control non-treated EVTs. *P < 0.01 versus non-treated control, t-test, #P < 0.01 versus LPS, one-way ANOVA, SNK.
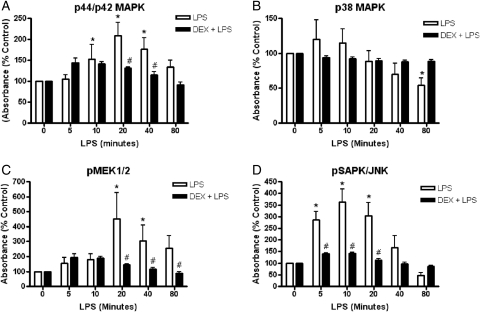
Effect of LPS on the EVT MAPK signaling pathway
We sought to investigate the molecular signaling pathways responsible for the observed LPS effect on EVTs, including an increase in cytokine release and a decrease in invasion. There was a significant interaction between LPS (1 μg/ml) treatment and the time of phosphorylation of p44/42 MAPK (P = 0.006) (Fig. 4A). LPS-stimulated phosphorylation of p44/42 MAPK increased over time reaching a peak at 20min (P< 0.0001). DEX significantly reversed the LPS-mediated increase in p44/42 MAPK activation at the peak of phosphorylation, 20min (P = 0.008) and at 40min (P = 0.031). There was a significant interaction between LPS treatment and time of p38 MAPK phosphorylation (P = 0.025) (Fig. 4B). However, there were no significant differences detected in p38 MAPK phosphorylation between LPS and LPS + DEX at any of the time points measured. Additionally, there was a significant interaction between LPS treatment and time of MEK1/2 phosphorylation (P = 0.008) (Fig. 4C). The LPS-mediated activation of phospho-MEK1/2 increased over time reaching a peak at 20min (P = 0.0001). DEX significantly reversed the LPS-mediated increase in phospho-MEK1/2 at 20min (P = 0.0001), 40min (P = 0.013) and 80min (P= 0.031). Similarly, a significant interaction between LPS treatment and time was seen in the phosphorylation of SAPK/JNK (P < 0.001) (Fig. 4D). LPS-stimulated activation of phospho-SAPK (pSAPK)/JNK increased over time reaching a peak at 10min (P = 0.0001). DEX significantly reversed the LPS-mediated increase in phospho-MEK1/2 at 5min (P = 0.005), 10min (P = 0.0001) and 20min (P = 0.001). Total MEK1 (P = 0.403; data not shown) and total SAPK/JNK (P = 0.488; data not shown), which acted as protein controls, were not significantly different at any of the time points measured. Additionally, no differences were found between ethanol-treated cells (DEX vehicle control) when compared with non-treated cells (P = 0.556; data not shown) or in cells treated with DEX alone when compared with non-treated cells for all MAPK family members measured (data not shown).
Figure 4.
The effect of LPS (1 μg/ml) on MAPK phosphorylation with or without DEX (100 nM) pretreatment (2h) in first trimester primary EVTs. EVTs were treated with LPS for the specified amount of time. Phosphorylation of p44/42 MAPK (A), p38 MAPK (B), pMEK1/2 (C) and pSAPK/JNK (D) was measured in cell lysates using a PathScan Multi-Target ELISA. Total MEK1 and total SAPK/JNK (not shown) were unchanged with LPS or LPS + DEX treatment and acted as positive controls for protein levels. Each bar represents mean ± SEM. *P < 0.05 versus non-treated control, two-way RM ANOVA, Holm–Sidak (HS) method, #P < 0.05 versus LPS, two-way RM ANOVA, HS.
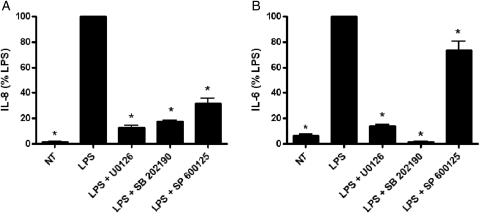
Effect of MAPK inhibitors on IL-8 and IL-6 cytokine production
In order to determine if activation of the MAPK signaling pathway is contributing to the LPS-induced increase in IL-8 and IL-6 cytokine production, we investigated the effect of specific MAPK pathway inhibitors on cytokine expression. Pretreatment with inhibitors specific to three different MAPK pathways including MEK1/2 (U0126), p38 MAPK (SB 202190) and JNK (SP 600125), significantly reversed the LPS-induced increase in both IL-8 (P< 0.001) (Fig. 5A) and IL-6 (P< 0.001) (Fig. 5B). Treatment of EVTs with U0126, SB 202190 or SP 600125 alone had no effect on baseline IL-8 (P = 0.42, 0.45 and 0.60, respectively) or IL-6 (P = 0.11, 0.08 and 0.97, respectively) levels (data not shown). Additionally, treatment with DMSO (MAPK inhibitor vehicle control) had no effect on IL-8 (P = 0.77) or IL-6 (P = 0.47) cytokine expression when compared with non-treated cells (data not shown). Measurements of LDH released into the media after treatment with all three MAPK inhibitors were not different in EVTs (U0126: 0.22 non-treated versus 0.26 50μM U0126, P = 0.2, SB 202190: 0.22 non-treated versus 0.22 50μM SB 202190, P = 1.0, SP 600125: 0.22 non-treated versus 0.28 50μM SP 600125, P = 0.7) (data not shown) when compared with non-treated cells. As such, these results indicate that the decreased cytokine production observed in EVTs in the presence of MAPK inhibitors is not secondary to cell death.
Figure 5.
The effect of MAPK inhibitor pretreatment (50μM) on the LPS (1 μg/ml)-mediated increase in IL-8 (A) and IL-6 (B) cytokine production in first trimester primary EVTs. IL-8 and IL-6 protein expression was measured by ELISA in EVT cell culture supernatants following pretreatment with U0126 (MEK1/2 inhibitor), SB 202190 (p38 MAPK inhibitor) and SP 600125 (JNK inhibitor) for 1h prior to the addition of LPS. Treatments with U0126, SB 202190, SP 600125 or DMSO (MAPK inhibitor vehicle control) alone had no effect on IL-8 or IL-6 expression (not shown). Each bar represents mean ± SEM. Data are presented as a percentage of LPS-treated EVTs. Three independent experiments were performed with cells isolated from three different placentas. *P < 0.001 versus LPS, one-way ANOVA, SNK.
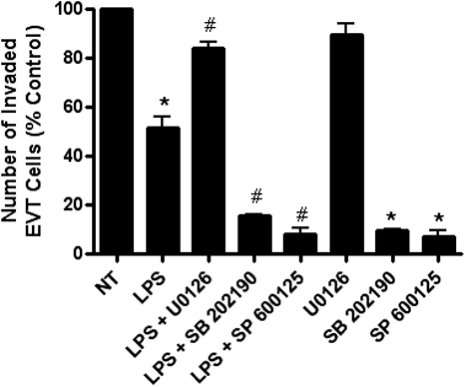
Effect of MAPK inhibitors on EVT invasion
In our effort to determine the mechanism involved in the LPS-mediated decrease in trophoblast invasion, we investigated the effects of inhibitors targeted to specific MAPK signaling pathways on EVT invasion (Fig. 6). EVTs pretreated with a MEK1/2-specific inhibitor (U0126) followed by LPS exposure significantly reversed the LPS-mediated decrease in invasion (P < 0.001). Pretreatment with inhibitors specific to p38 MAPK (SB 202190) and JNK (SP 600125) prior to LPS exposure did not reverse the LPS-induced decrease in invasion and instead resulted in an even greater decrease in EVT invasion than those cells treated with LPS alone (SB 202190: P < 0.001, SP 600125: P < 0.001). U0126 treatment alone had no effect on EVT invasion when compared with non-treated cells. Treatment with SB 202190 (P < 0.001) and SP 600125 (P < 0.001) alone resulted in significantly decreased EVT invasion, independent of LPS treatment. Additionally, treatment with DMSO (MAPK inhibitor vehicle control) had no effect on EVT invasion (P = 0.77) when compared with non-treated cells (data not shown).
Figure 6.
The effect of MAPK inhibitor pretreatment (50μM) on the LPS (1 μg/ml)-mediated decrease in first trimester primary EVT cell invasion. EVT cell invasion through a matrigel-coated membrane was measured after pretreatment (1 h) with U0126 (MEK1/2 inhibitor), SB 202190 (p38 MAPK inhibitor) and SP 600125 (JNK inhibitor) with or without additional treatment with LPS. Each bar represents mean ± SEM. Data are presented as a percentage of control non-treated EVTs. Three independent experiments were performed with cells isolated from three different placentas. *P < 0.001 versus non-treated control, one-way ANOVA, SNK, #P < 0.001 versus LPS, one-way ANOVA, SNK.
Discussion
It has been hypothesized that adverse obstetrical outcomes, including pre-eclampsia, IUGR and preterm birth, develop as a result of placental dysfunction during the first trimester of pregnancy. However, despite some progress in this research field, the underlying mechanisms that cause placental dysfunction remain unclear. Therefore, this current study was designed to investigate the specific mechanisms associated with inflammation-mediated first trimester placental dysfunction that may precede adverse obstetrical outcomes, and to identify possible therapeutic targets of intervention early in pregnancy. In our in vitro model of inflammation, the results clearly show that LPS induces an increase in the inflammatory cytokines IL-8 and IL-6, and a decrease in invasion of EVTs that cannot be explained by increased rates of cell death. The reversal of these effects by DEX, an anti-inflammatory glucocorticoid (Ray and Prefontaine, 1994; Auphan et al., 1995) indicates that these results are associated with the activation of established inflammatory signaling pathways. The observed LPS-mediated increase in phosphorylation of several MAPK family members suggests that the MAPK pathway is contributing significantly to LPS-mediated trophoblast dysfunction. Additionally, we found that direct inhibition of the MAPK pathway reversed the LPS-induced increase in IL-8 and IL-6 production by EVTs, suggesting a clear link between inflammation-associated MAPK phosphorylation and enhanced cytokine expression. Perhaps even more interesting is the observation that MEK1/2 phosphorylation, specifically, seems to contribute to the LPS-induced decrease in EVT invasion while p38 MAPK and JNK seem to mediate EVT invasion independent of an inflammatory stimulus. These results indicate that activation of the MEK/ERK pathway signaling may be intimately involved in regulating trophoblast invasion in an inflammatory state. The findings of this study provide evidence for inflammation-induced placental trophoblast dysfunction which may ultimately contribute to the development of adverse obstetrical outcomes.
Recent studies investigating abnormal placental function have clearly implicated the production and/or release of cytokines as contributing to the pathophysiology of pre-eclampsia and IUGR (Tjoa et al., 2004). Previous studies have demonstrated that LPS treatment enhances cytokine expression in whole placental tissue (Shimoya et al., 1999), term syncytiotrophoblasts (Ma et al., 2006) and first trimester trophoblasts (Abrahams et al., 2005). Our findings of increased pro-inflammatory cytokines, IL-8 and IL-6, in first trimester EVTs are in agreement with these previous studies even though different placental cell types and stages of gestation were investigated. Abrahams et al. (2005) utilized first trimester trophoblasts for their experiments investigating the effects of LPS on cytokine production (by cytokine array) and found that IL-8 among other cytokines was significantly increased. Our results agree with those of others who have found increased IL-6 production in response to LPS treatment in a first trimester cytotrophoblast cell line (Abrahams et al., 2004) and term syncytiotrophoblasts (Ma et al., 2006). Additionally, the LPS-mediated increase in IL-8 and IL-6 observed in our study was reversed by DEX pretreatment indicating that the increase in these cytokines is regulated by established inflammatory signaling pathways. Trophoblast cells, which express the necessary TLRs (Abrahams et al., 2004), play a pivotal role in initiation of the normal innate immune response, including cytokine production, which is critical for implantation and to facilitate leukocyte recruitment for host defense (Wood et al., 1997). While acute inflammation is needed for a successful pregnancy, a chronic immune response caused by a pathological condition, such as an intrauterine infection, can result in excessive production of these same cytokines leading to placental dysfunction. Therefore, the ability of LPS to induce cytokine expression indicates that first trimester EVTs have the ability to regulate the innate immune response occurring within the placenta. Additionally, these results lead to the hypothesis that the primary function of EVTs, invasion of the uterine spiral arteries, could be compromised during an inflammatory state.
Very few studies have been performed to investigate the effects of LPS on EVT invasion. Previous studies which utilized a co-culture model of HTR-8/SVneo cells and LPS-activated macrophages to mimic the activation of the immune response in the placenta resulted in a decrease in trophoblast cell invasiveness (Renaud et al., 2005; Renaud et al., 2007) indicating that an activated immune response would cause abnormalities in trophoblast function. Additional studies found that LPS treatment significantly decreased HTR-8/SVneo migration (Kim et al., 2005). Similarly, our studies demonstrate that activation of the innate immune response by LPS results in decreased first trimester EVT invasion. The LPS-mediated decrease in invasion was reversed after treatment with DEX, indicating that the effects of LPS on trophoblast invasion are specifically related to alterations within the inflammatory pathway and not to other mechanisms.
It is well known that glucocorticoids, such as DEX, exert their anti-inflammatory effects by increasing the expression of IκB (Auphan et al., 1995; Scheinman et al., 1995), which is known to inhibit the NF-κB-mediated increases in expression of genes associated with inflammation. As TLR4 signaling pathways are mediated through NF-κB (Pålsson-McDermott and O'Neill, 2004), it is easy to hypothesize that DEX treatment reversed the LPS-mediated alterations in trophoblast function in part through antagonism of the action of NF-κB transcription factor. The ability of DEX to block both the increase in IL-8 and IL-6 cytokines and the decrease in trophoblast invasion suggests that treatment with DEX early in pregnancy could potentially prevent placental dysfunction caused by inflammation. However, additional experiments investigating the effects of DEX treatment on trophoblast function after an inflammatory exposure are warranted. It is noted that identifying patients in the first or early second trimester who are at risk for exposure to intrauterine inflammation remains problematic.
The ability of LPS to both increase cytokine production and decrease trophoblast invasion would indicate that cytokine expression is intimately involved in the regulation of EVT function and thus invasion. Contrary to our results, several studies have found that exogenous IL-6 and IL-8 treatment of HTR8/SVneo cells increased trophoblast invasion (Jovanovic and Vicovac, 2009; Jovanovic et al., 2010). While this discrepancy in results may be explained by the different model systems used, we believe that the effects of IL-6 and IL-8 alone on trophoblast invasion may not accurately represent the response of trophoblast cells during an inflammatory state. While alterations in cytokine levels by an activated immune response play a role in regulating trophoblast function, the connection between elevated cytokines and decreased trophoblast invasion is complex and most likely involves a balance of pro and anti-invasive factors, including other cytokines/chemokines, integrins, adhesion and proteolytic molecules.
In order to fully understand the mechanisms involved in inflammation-induced trophoblast dysfunction, we further investigated the signaling pathways associated with the LPS-mediated effects on trophoblast function. We chose to focus on the MAPK pathway based on previous studies which have not only shown that this pathway is involved in regulating cytokine production during an inflammatory state (Lappas et al., 2007), but also that MAPK pathways contribute to altered trophoblast invasion (Qiu et al., 2004a,b; LaMarca et al., 2008; Prast et al., 2008). Our findings of LPS-mediated activation of p44/42 MAPK, p38 MAPK, pMEK1/2 and pSAPK/JNK indicate that these pathways are significantly involved in the inflammatory response within the placenta. The reversal of MAPK phophorylation with DEX pretreatment further provides evidence that these signaling molecules are activated during an inflammatory state. Additionally, pretreatment with inhibitors of MEK1/2, p38 MAPK and JNK reversed the LPS-mediated increase in IL-8 and IL-6 production, clearly indicating a direct link between phosphorylation of all three MAPK pathways and LPS-induced cytokine production. Several studies have previously shown that LPS activates MAPKs, including p44/42 MAPK, p38 MAPK and pJNK, in term choriodecidual explants (Shoji et al., 2007), term syncytiotrophoblasts (Lucchi and Moore, 2007) and term trophoblasts (Jiang et al., 2007). In agreement with these studies, our study is the first to show in first trimester EVTs that LPS mediates phophorylation of p44/42 MAPK, pMEK1/2 and pSAPK/JNK. While the statistical analysis of the LPS-mediated phosphorylation of p38 revealed an overall difference owing to changes in phospho-p38 over time, we found no change in phosphorylation at any of the time points with LPS treatment or with DEX pretreatment. However, pretreatment of EVTs with SB 202190 (p38 MAPK inhibitor) reversed the effect of LPS on IL-8 and IL-6 cytokine production indicating that p38 MAPK plays a significant role in the induction of cytokines by LPS. In agreement with our studies, activation of both p38 MAPK and pSAPK/JNK are known to initiate the production of cytokines necessary to elicit an inflammatory response (Kang et al., 2007). Our results would suggest that during early pregnancy the presence of LPS, as might occur with an infection, activates the MAPK pathway resulting in the production of pro-inflammatory cytokines, IL-6 and IL-8.
Since we have shown that MAPK pathway signaling contributes to the regulation of LPS-induced cytokine expression, we wanted to determine if these same MAPK signaling pathways contribute to the altered trophoblast invasion seen with LPS treatment. Interestingly, pretreatment with the MEK1/2 inhibitor (U0126) reversed the LPS-induced decrease in EVT cell invasion while inhibitors of p38 MAPK (SB 202190) and JNK (SP 600125), independent of LPS treatment, reduced EVT invasion even more than LPS-treated cells. This result strongly suggests that MEK1/2 phosphorylation is an inflammation-mediated event while the actions of p38 MAPK and JNK signaling appear to be unrelated to the presence of an inflammatory state. This would first suggest that the MAPK pathway is intimately associated with trophoblast invasion during normal pregnancies and second that the MEK/ERK signaling cascade plays a predominant role in regulating inflammation-associated trophoblast invasion. The MEK/ERK pathway has been previously associated with the regulation of gene expression, cellular proliferation, differentiation, angiogenesis, embryo development and tumor invasion. Interestingly, previous studies have shown that deletion of the ERK gene results in embryonic lethality early in mouse development, which is caused in part by defects in placental development, indicating that ERK plays an important role in placental function (Regan et al., 2002; Sohn et al., 2002; Saba-El-Leil et al., 2003; Yan et al., 2003). While there have been no studies to date which have investigated MAPK phosphorylation by LPS as a mechanism for decreased EVT invasion, several studies have implicated MEK/ERK phosphorylation in altered trophoblast function. Activation of insulin-like growth factor binding protein-1 and insulin-like growth factor-II by ERK1/2 has been shown to promote migration of HTR-8/SVneo cells (Gleeson et al., 2001; McKinnon et al., 2001). Similarly, other factors have been shown to stimulate HTR-8/SVneo cell migration and invasion via phosphorylation of ERK, including endothelin (Chakraborty et al., 2003), endothelial growth factor (Qiu et al., 2004a,b), prostaglandin estradiol (Nicola et al., 2008) and hCG (Prast et al., 2008). While many studies have provided evidence for a role of MEK/ERK activation in stimulating trophoblast invasion, a coordinated regulation of factors stimulating and inhibiting invasion is needed for a successful pregnancy. Consequently, if these factors are altered by an inflammatory state, the presence of inflammation, even one that remains at the level of the uterus, may shift this balance to cause abnormal trophoblast invasion.
The activation of proteolytic enzymes, particularly the matrix metalloprotinases, are essential to trophoblast invasion and movement through the ECM. Previous studies have pinpointed the up-regulation of tissue inhibitor of matrix metalloproteinases-1 (TIMP-1) as a critical factor in the inhibition of trophoblast invasion (Graham and Lala, 1991). Additionally, ERK phosphorylation has been shown to be responsible for an increase in TIMP-1 in HTR-8/SVneo cells (Qiu et al., 2004a,b). These studies, along with our results of an LPS-mediated decrease in EVT invasion through MEK/ERK phosphorylation, would suggest that LPS may be mediating trophoblast invasion through alterations in ECM proteases and their inhibitors. Therefore, it is interesting to hypothesize that LPS could be causing a decrease in trophoblast invasion through an increase in TIMP-1 associated mechanisms regulated by ERK phosphorylation. Future studies investigating the interaction between MEK/ERK phosphorylation and the mechanics of EVT invasion and movement could provide even more insight into the mechanisms directly controlling trophoblast invasion in both normal and inflammatory states of pregnancy. Additionally, these studies could help to pinpoint exact areas within these pathways that could become possible targets of future treatments for those at risk of developing adverse pregnancy outcomes.
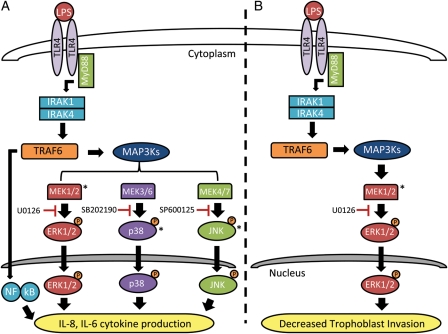
The results from this study suggest that the presence of a bacterial pathogen or bacterial by-product, such as LPS, can activate the TLR4-mediated signaling (Poltorak et al., 1998; Hoshino et al., 1999; Elovitz et al., 2003) cascade resulting in (i) increased cytokine production which initiates an activated innate immune response and recruits neutrophils and macrophages into the placenta and (ii) decreased trophoblast invasion. While the LPS-mediated increase in cytokine production and decrease in trophoblast invasion are both likely initiated by a TLR4 dependent signaling pathway, ultimately alterations in these specific trophoblast functions occurs through different MAPK signaling mechanisms. Increased IL-8 and IL-6 cytokine production seen in the presence of an inflammatory stimulus is mediated by phosphorylation of ERK1/2, p38 MAPK and JNK which translocate to the nucleus and activate transcription factors responsible for cytokine up-regulation (Fig. 7A). On the other hand, the LPS-mediated decrease in EVT invasion is regulated by the phosphorylation of ERK1/2 only (Fig. 7B). While further studies examining the effect of LPS on trophoblast invasion (and specifically the MEK/ERK signaling pathways regulating this interaction) are warranted, the LPS-mediated decrease in invasion of EVT cells, whose primary function is to invade the maternal uterine spiral arteries, would suggest that an altered inflammatory state could lead to placental dysfunction and thus contribute to the development of adverse obstetrical outcomes including preterm birth, pre-eclampsia and IUGR.
Figure 7.
Proposed MAPK pathways and their inhibitors involved in EVT dysfunction during an inflammatory state. (A) LPS binding to the TLR4 receptor activates a downstream signaling cascade resulting in the phosphorylation of specific MAPKs. Phosphorylation and subsequent activation of ERK1/2, p38 MAPK and JNK results in the translocation of these MAPKs into the nucleus where they activate transcription factors and up-regulate the genes responsible for IL-8 and IL-6 cytokine production. Along with the activated MAPKs, LPS binding to TLR4 results in the activation of NF-κB which is shown to represent the classical pathway of LPS/TLR4 activation of pro-inflammatory cytokine production. (B) In the presence of LPS, the activation of TLR4 results in the downstream phosphorylation and activation of ERK1/2 where it crosses into the nucleus, activates transcription factors and alters the genes responsible for decreased EVT invasion. The LPS-mediated increase in cytokine production and decrease in EVT invasion are both initiated by a TLR4-dependent signaling pathway but ultimately alterations in specific trophoblast functions occurs through different MAPK signaling mechanisms. Specific inhibitors of ERK1/2 (U0126), p38 MAPK (SB 202190) and JNK (SP 600125) show points of inhibition within each pathway. Asterisks denote MAPK proteins measured in our study by ELISA.
Authors’ roles
L.A. contributed to the development of the study design, performed all elements of the laboratory based studies, analyzed the data and was primarily responsible for drafting this manuscript. A.G.B. participated in the study design, assisted with laboratory techniques and interpretation of the experimental results. S.P. participated in critical discussion of the experiments and contributed to manuscript editing and critique. M.A.E. is the primary investigator in the laboratory and was responsible for help with designing the study, overseeing all experiments and research progress and contributed significantly to this manuscript.
Funding
This project was supported by an NIH T32 Training Grant 5T32HD007305 25.
References
- Abrahams VM, Bole-Aldo P, Kim YM, Straszewski-Chavez SL, Chaiworapongsa T, Romero R, Mor G. Divergent trophoblast responses to bacterial products mediated by TLRs. J Immunol. 2004;173:4286–4296. doi: 10.4049/jimmunol.173.7.4286. [DOI] [PubMed] [Google Scholar]
- Abrahams VM, Visintin I, Aldo PB, Guller S, Romero R, Mor G. A role for tlrs in the regulation of immune cell migration by first trimester trophoblast cells. J Immunol. 2005;175:8096–8104. doi: 10.4049/jimmunol.175.12.8096. [DOI] [PubMed] [Google Scholar]
- Adams Waldorf KM, Persing D, Novy MJ, Sadowsky DW, Gravett MG. Pretreatment with toll-like receptor 4 antagonist inhibits lipopolysaccharide-induced preterm uterine contractility, cytokines, and prostaglandins in rhesus monkeys. Reprod Sci. 2008;15:121–127. doi: 10.1177/1933719107310992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arechavaleta-Velasco F, Koi H, Strauss JF, Parry S. Viral infection of the trophoblast: time to take a serious look at its role in abnormal implantation and placentation? J Reprod Immunol. 2002;55:113–121. doi: 10.1016/s0165-0378(01)00143-7. [DOI] [PubMed] [Google Scholar]
- Auphan N, DiDonato JA, Rosette C, Helmberg A, Karin M. Immunosuppression by glucocorticoids: inhibition of NF-kB activity through induction of IKB synthesis. Science. 1995;270:286–290. doi: 10.1126/science.270.5234.286. [DOI] [PubMed] [Google Scholar]
- Brosens I, Robertson W, Dixon H. The physiological response to the vessels of the placental bed to normal pregnancy. Clin Obstet Gynecol. 1967;77:573–593. doi: 10.1002/path.1700930218. [DOI] [PubMed] [Google Scholar]
- Chakraborty C, Barbin YP, Chakrabarti S, Chidiac P, Dixon SJ, Lala PK. Endothelin-1 promotes migration and induces elevation of [ca2+]i and phosphorylation of map kinase of a human extravillous trophoblast cell line. Mol Cell Endocrinol. 2003;201:63–73. doi: 10.1016/s0303-7207(02)00431-8. [DOI] [PubMed] [Google Scholar]
- Dadelszen Pv, Magee LA. Could an infectious trigger explain the differential maternal response to the shared placental pathology of preeclampsia and normotensive intrauterine growth restriction? Acta Obstet Gynecol Scand. 2002;81:642–648. [PubMed] [Google Scholar]
- Dadelszen P, Magee LA, Krajden M, Alasaly K, Popovska V, Devarakonda RM, Money DM, Patrick DM, Brunham RC. Levels of antibodies against cytomegalovirus and chlamydophila pneumoniae are increased in early onset pre-eclampsia. Int J Obstet Gynaecol. 2003;110:725–730. [PubMed] [Google Scholar]
- Elovitz MA, Wang Z, Chien EK, Rychlik DF, Phillippe M. A new model for inflammation-induced preterm birth: the role of platelet-activating factor and toll-like receptor-4. Am J Pathol. 2003;163:2103–2111. doi: 10.1016/S0002-9440(10)63567-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Genbacev O, Jensen K, Powlin S, Miller R. In vitro differentiation and ultrastructure of human extravillous trophoblast (EVT) cells. Placenta. 1993;14:463–475. doi: 10.1016/s0143-4004(05)80466-7. [DOI] [PubMed] [Google Scholar]
- Getsios S, Chen GT, Huang DT, MacCalman CD. Regulated expression of cadherin-11 in human extravillous cytotrophoblasts undergoing aggregation and fusion in response to transforming growth factor beta 1. J Reprod Fertil. 1998;114:357–363. doi: 10.1530/jrf.0.1140357. [DOI] [PubMed] [Google Scholar]
- Gleeson LM, Chakraborty C, McKinnon T, Lala PK. Insulin-like growth factor-binding protein 1 stimulates human trophoblast migration by signaling through alpha 5 beta 1 integrin via mitogen-activated protein kinase pathway. J Clin Endocrinol Metab. 2001;86:2484–2493. doi: 10.1210/jcem.86.6.7532. [DOI] [PubMed] [Google Scholar]
- Goldenberg R, Hauth J, Andrews W. Intrauterine infection and preterm delivery. N Engl J Med. 2000;342:1500–1507. doi: 10.1056/NEJM200005183422007. [DOI] [PubMed] [Google Scholar]
- Gonçalves LF, Chaiworapongsa T, Romero R. Intrauterine infection and prematurity. Ment Retard Dev Disabil Res Rev. 2002;8:3–13. doi: 10.1002/mrdd.10008. [DOI] [PubMed] [Google Scholar]
- Graham CH, Lala PK. Mechanism of control of trophoblast invasion in situ. J Cell Physiol. 1991;148:228–234. doi: 10.1002/jcp.1041480207. [DOI] [PubMed] [Google Scholar]
- Graham C, Lala P. Mechanisms of placental invasion of the uterus and their control. Biochem Cell Biol. 1992;70:867–874. doi: 10.1139/o92-135. [DOI] [PubMed] [Google Scholar]
- Graham CH, Lysiak JJ, McCrae KR, Lala PK. Localization of transforming growth factor-beta at the human fetal-maternal interface: role in trophoblast growth and differentiation. Biol Reprod. 1992;46:561–572. doi: 10.1095/biolreprod46.4.561. [DOI] [PubMed] [Google Scholar]
- Hoshino K, Takeuchi O, Kawai T, Sanjo H, Ogawa T, Takeda Y, Takeda K, Akira S. Cutting edge: toll-like receptor 4 (TLR 4)-deficient mice are hyporesponsive to lipopolysaccharide: evidence for tlr4 as the LPS gene product. J Immunol. 1999;162:3749–3752. [PubMed] [Google Scholar]
- Janeway CA, Medzhitov R. Innate immune recognition. Ann Rev Immunol. 2002;20:197–216. doi: 10.1146/annurev.immunol.20.083001.084359. [DOI] [PubMed] [Google Scholar]
- Jiang K, Chen Y, Jarvis JN. Soluble factors from LPS- and PHA-activated PBMC induce MAPK, Stat1 and Stat3 phosphorylation in primary cultures of human term placental trophoblasts: implications for infection and prematurity. Placenta. 2007;28:538–542. doi: 10.1016/j.placenta.2006.06.013. [DOI] [PubMed] [Google Scholar]
- Jovanovic M, Vicovac L. Interleukin-6 stimulates cell migration, invasion and integrin expression in HTR-8/svneo cell line. Placenta. 2009;30:320–328. doi: 10.1016/j.placenta.2009.01.013. [DOI] [PubMed] [Google Scholar]
- Jovanovic M, Stefanoska I, Radojcic L, Vicovac L. Interleukin-8 (CXCL8) stimulates trophoblast cell migration and invasion by increasing levels of matrix metalloproteinase (MMP)2 and MMP9 and integrins a5 and b1. Reproduction. 2010;139:789–798. doi: 10.1530/REP-09-0341. [DOI] [PubMed] [Google Scholar]
- Kaga N, Katsuki Y, Obata M, Shibutani Y. Repeated administration of low-dose lipopolysaccharide induces preterm delivery in mice: a model for human preterm parturition and for assessment of the therapeutic ability of drugs against preterm delivery. Am J Obstet Gynecol. 1996;174:754–759. doi: 10.1016/s0002-9378(96)70460-x. [DOI] [PubMed] [Google Scholar]
- Kang S-S, Woo SS, Im J, Yang JS, Yun C-H, Ju HR, Son CG, Moon E-Y, Han SH. Human placenta promotes IL-8 expression through activation of JNK/SAPK and transcription factors NF-kappab and AP-1 in PMA-differentiated THP-1 cells. Int Immunopharmacol. 2007;7:1488–1495. doi: 10.1016/j.intimp.2007.07.011. [DOI] [PubMed] [Google Scholar]
- Kaufmann P, Castellucci M. Extravillous trophoblasts in the human placenta. Troph Res. 1997;10:21–65. [Google Scholar]
- Kim YM, Romero R, Oh SY, Kim CJ, Kilburn BA, Armant DR, Nien JK, Gomez R, Mazor M, Saito S, et al. Toll-like receptor 4: a potential link between ‘Danger signals,’ the innate immune system, and preeclampsia? Am J Obstet Gynecol. 2005;193 doi: 10.1016/j.ajog.2005.07.076. 921.e921–921.e928. [DOI] [PubMed] [Google Scholar]
- Kline JN, Cowden JD, Hunninghake GW, Schutte BC, Watt JL, Wohlford-Lenane CL, Powers LS, Jones MP, Schwartz DA. Variable airway responsiveness to inhaled lipopolysaccharide. Am J Respir Crit Care Med. 1999;160:297–303. doi: 10.1164/ajrccm.160.1.9808144. [DOI] [PubMed] [Google Scholar]
- Koi H, Zhang J, Makrigiannakis A, Getsios S, MacCalman CD, Strauss JF, III, Parry S. Syncytiotrophoblast is a barrier to maternal-fetal transmission of herpes simplex virus. Biol Reprod. 2002;67:1572–1579. doi: 10.1095/biolreprod.102.004325. [DOI] [PubMed] [Google Scholar]
- Kurman R. The morphology, biology, and pathology of intermediate trophoblast: a look back to the present. Hum Pathol. 1992;22:847–855. doi: 10.1016/0046-8177(91)90173-m. [DOI] [PubMed] [Google Scholar]
- LaMarca HL, Dash PR, Vishnuthevan K, Harvey E, Sullivan DE, Morris CA, Whitley GSJ. Epidermal growth factor-stimulated extravillous cytotrophoblast motility is mediated by the activation of PI3-k, AKT and both p38 and p42/44 mitogen-activated protein kinases. Hum Reprod. 2008;23:1733–1741. doi: 10.1093/humrep/den178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamont RF. Infection in the prediction and antibiotics in the prevention of spontaneous preterm labour and preterm birth. Br J Obstet Gynaecol. 2003;110:71–75. doi: 10.1016/s1470-0328(03)00034-x. [DOI] [PubMed] [Google Scholar]
- Lappas M, Permezel M, Rice GE. Mitogen-activated protein kinase proteins regulate LPS-stimulated release of pro-inflammatory cytokines and prostaglandins from human gestational tissues. Placenta. 2007;28:936–945. doi: 10.1016/j.placenta.2007.02.009. [DOI] [PubMed] [Google Scholar]
- Lien E, Means T, Heine H, Yoshimura A, Kusumoto S, Fukase K, Fenton M, Oikawa M, Qureshi N, Monks B, et al. Toll-like receptor 4 imparts ligand-specific recognition of bacterial lipopolysaccharide. J Clin Investig. 2000;105:497–504. doi: 10.1172/JCI8541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lucchi NW, Moore JM. LPS induces secretion of chemokines by human syncytiotrophoblast cells in a MAPK-dependent manner. J Reprod Immunol. 2007;73:20–27. doi: 10.1016/j.jri.2006.05.005. [DOI] [PubMed] [Google Scholar]
- Ma Y, Mor G, Abrahams VM, Buhimschi IA, Buhimschi CS, Guller S. Alterations in syncytiotrophoblast cytokine expression following treatment with lipopolysaccharide. Am J Reprod Immunol. 2006;55:12–18. doi: 10.1111/j.1600-0897.2005.00347.x. [DOI] [PubMed] [Google Scholar]
- McKinnon T, Chakraborty C, Gleeson LM, Chidiac P, Lala PK. Stimulation of human extravillous trophoblast migration by IGF-ii is mediated by IGF type 2 receptor involving inhibitory g protein(s) and phosphorylation of MAPK. J Clin Endocrinol Metab. 2001;86:3665–3674. doi: 10.1210/jcem.86.8.7711. [DOI] [PubMed] [Google Scholar]
- Medzhitov R, Janeway CA., Jr Decoding the patterns of self and nonself by the innate immune system. Science. 2002;296:298–300. doi: 10.1126/science.1068883. [DOI] [PubMed] [Google Scholar]
- Ness RB, Sibai BM. Shared and disparate components of the pathophysiologies of fetal growth restriction and preeclampsia. Am J Obstet Gynecol. 2006;195:40–49. doi: 10.1016/j.ajog.2005.07.049. [DOI] [PubMed] [Google Scholar]
- Neudeck H, Oei SL, Stiemer B, Hopp H, Graf R. Binding of antibodies against high and low molecular weight cytokeratin proteins in the human placenta with special reference to infarcts, proliferation and differentiation processes. Histochem J. 1997;29:419–430. doi: 10.1023/a:1026499203743. [DOI] [PubMed] [Google Scholar]
- Nicola C, Chirpac A, Lala PK, Chakraborty C. Roles of rho guanosine 5′-triphosphatase a, rho kinases, and extracellular signal regulated kinase (1/2) in prostaglandin E2-mediated migration of first-trimester human extravillous trophoblast. Endocrinology. 2008;149:1243–1251. doi: 10.1210/en.2007-1136. [DOI] [PubMed] [Google Scholar]
- Pålsson-McDermott EM, O'Neill LAJ. Signal transduction by the lipopolysaccharide receptor, toll-like receptor-4. Immunology. 2004;113:153–162. doi: 10.1111/j.1365-2567.2004.01976.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poltorak A, He X, Smirnova I, Liu M-Y, Huffel CV, Du X, Birdwell D, Alejos E, Silva M, Galanos C, et al. Defective LPS signaling in C3H/hej and C57bl/10sccr mice: mutations in Tlr4 gene. Science. 1998;282:2085–2088. doi: 10.1126/science.282.5396.2085. [DOI] [PubMed] [Google Scholar]
- Prast J, Saleh L, Husslein H, Sonderegger S, Helmer H, Knofler M. Human chorionic gonadotropin stimulates trophoblast invasion through extracellularly regulated kinase and akt signaling. Endocrinology. 2008;149:979–987. doi: 10.1210/en.2007-1282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qiu Q, Yang M, Tsang BK, Gruslin A. Both mitogen-activated protein kinase and phosphatidylinositol 3-kinase signalling are required in epidermal growth factor-induced human trophoblast migration. Mol Hum Reprod. 2004a;10:677–684. doi: 10.1093/molehr/gah088. [DOI] [PubMed] [Google Scholar]
- Qiu Q, Yang M, Tsang BK, Gruslin A. EGF-induced trophoblast secretion of MMP-9 and TIMP-1 involves activation of both pi3k and MAPK signalling pathways. Reproduction. 2004b;128:355–363. doi: 10.1530/rep.1.00234. [DOI] [PubMed] [Google Scholar]
- Ray A, Prefontaine KE. Physical association and functional antagonism between the p65 subunit of transcription factor NF-kb and the glucocorticoid receptor. Proc Natl Acad Sci USA. 1994;91:752–756. doi: 10.1073/pnas.91.2.752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Regan C, Li W, Boucher D, Spatz S, Su M, Kuida K. Erk5 null mice display multiple extraembryonic vascular and embryonic cardiovascular defects. Proc Natl Acad Sci USA. 2002;99:9248–9253. doi: 10.1073/pnas.142293999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Renaud SJ, Postovit L-M, Macdonald-Goodfellow SK, McDonald GT, Caldwell JD, Graham CH. Activated macrophages inhibit human cytotrophoblast invasiveness in vitro. Biol Reprod. 2005;73:237–243. doi: 10.1095/biolreprod.104.038000. [DOI] [PubMed] [Google Scholar]
- Renaud SJ, Macdonald-Goodfellow SK, Graham CH. Coordinated regulation of human trophoblast invasiveness by macrophages and interleukin 10. Biol Reprod. 2007;76:448–454. doi: 10.1095/biolreprod.106.055376. [DOI] [PubMed] [Google Scholar]
- Saba-El-Leil MK, Vella FDJ, Vernay B, Voisin L, Chen L, Labreque N, Ang S, Meloche S. An essential function of the mitogen-activated protein kinase Erk2 in mouse trophoblast develepment. EMBO Rep. 2003;4:964–986. doi: 10.1038/sj.embor.embor939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scheinman RI, Cogswell PC, Lofquist AK, Baldwin AS., Jr Role of transcriptional activation of i kappa b alpha in mediation of immunosuppression by glucocorticoids. Science. 1995;270:283–286. doi: 10.1126/science.270.5234.283. [DOI] [PubMed] [Google Scholar]
- Schwartz DA. The role of TLR4 in endotoxin responsiveness in humans. J Endotoxin Res. 2001;7:389–393. [PubMed] [Google Scholar]
- Shimoya K, Matsuzaki N, Taniguchi T, Kameda T, Koyama M, Neki R, Saji F, Tanizawa O. Human placenta constitutively produces interleukin-8 during pregnancy and enhances its production in intrauterine infection. Biol Reprod. 1992;47:220–226. doi: 10.1095/biolreprod47.2.220. [DOI] [PubMed] [Google Scholar]
- Shimoya K, Moriyama A, Matsuzaki N, Ogata I, Koyama M, Azuma C, Saji F, Murata Y. Human placental cells show enhanced production of interleukin (IL)-8 in response to lipopolysaccharide (LPS), IL-1 and tumour necrosis factor (TNF)-{alpha}, but not to IL-6. Mol Hum Reprod. 1999;5:885. doi: 10.1093/molehr/5.9.885. [DOI] [PubMed] [Google Scholar]
- Shoji T, Yoshida S, Mitsunari M, Miyake N, Tsukihara S, Iwabe T, Harada T, Terakawa N. Involvement of p38 map kinase in lipopolysaccharide-induced production of pro- and anti-inflammatory cytokines and prostaglandin e2 in human choriodecidua. J Reprod Immunol. 2007;75:82–90. doi: 10.1016/j.jri.2007.05.002. [DOI] [PubMed] [Google Scholar]
- Sohn S, Sarvis B, Cado D, Winoto A. Erk5 mapk regulates embryonic angiogenesis and acts as a hypoxia-sensitive repressor of vascular endothelial growth factor expression. J Biol Chem. 2002;277:43344–43351. doi: 10.1074/jbc.M207573200. [DOI] [PubMed] [Google Scholar]
- Takeda K, Kaisho T, Akira S. Toll-like receptors. Ann Rev Immunol. 2003;21:335–376. doi: 10.1146/annurev.immunol.21.120601.141126. [DOI] [PubMed] [Google Scholar]
- Tapping RI, Akashi S, Miyake K, Godowski PJ, Tobias PS. Toll-like receptor 4, but not toll-like receptor 2, is a signaling receptor for escherichia and salmonella lipopolysaccharides. J Immunol. 2000;165:5780–5787. doi: 10.4049/jimmunol.165.10.5780. [DOI] [PubMed] [Google Scholar]
- Tjoa ML, Oudejans CBM, van Vugt JMG, Blankenstein MA, van Wijk IJ. Markers for presymptomatic prediction of preeclampsia and intrauterine growth restriction. Hypertens Pregnancy. 2004;23:171–189. doi: 10.1081/PRG-120028292. [DOI] [PubMed] [Google Scholar]
- Wood GW, Hausmann E, Choudhuri R. Relative role of CSF-1, MCP-1/JE, and RANTES in macrophage recruitment during successful pregnancy. Mol Reprod Dev. 1997;46:62–70. doi: 10.1002/(SICI)1098-2795(199701)46:1<62::AID-MRD10>3.0.CO;2-5. [DOI] [PubMed] [Google Scholar]
- Yan L, Carr J, Ashby P, Murry-Tait V, Thompson C, Arthur JS. Knockout of Erk5 causes multiple defects in placental and embryonic development. BMC Dev Biol. 2003;3:11. doi: 10.1186/1471-213X-3-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoon BH, Romero R, Park JS, Kim M, Oh S-Y, Kim CJ, Jun JK. The relationship among inflammatory lesions of the umbilical cord (funisitis), umbilical cord plasma interleukin 6 concentration, amniotic fluid infection, and neonatal sepsis. Am J Obstet Gynecol. 2000;183:1124–1129. doi: 10.1067/mob.2000.109035. [DOI] [PubMed] [Google Scholar]