Abstract
The pharmacokinetics and urinary excretion following single intramuscular administration of levofloxacin at a dose of 4 mg/kg was investigated in seven male cross bred calves. Appreciable plasma concentration of levofloxacin (0.38 ± 0.06 µg/ml) was detected at 1 min after injection and the peak plasma level of 3.07 ± 0.08 µg/ml was observed at 1 h. The drug level above MIC90 in plasma was detected up to 12 h after administration. Rapid absorption of the drug was also evident by the high value of the absorption rate constant (2.14 ± 0.24 /h). The overall systemic bioavailability of levofloxacin, after intramuscular administration, was 56.6 ± 12.4%. The high value of AUC (7.66 ± 0.72 mg . h/ml) reflected the vast area of body covered by drug concentration. Extensive distribution of the drug into various body fluids and tissues was noted by the high value of Vdarea (1.02 ± 0.05 l/kg). The high ratio of AUC/MIC (76.6 ± 7.25) obtained in this study indicated excellent clinical and bacteriological efficacy of levofloxacin in calves. The elimination half-life and MRT were 3.67 ± 0.4 h and 5.57 ± 0.51 h, respectively. The total body clearance (ClB) was 204.9 ± 22.6 ml/kg/h. On the basis of the pharmacokinetic parameters, a suitable intramuscular dosage regimen for levofloxacin in calves would be 1.5 mg/kg repeated at 12 h intervals.
Keywords: calves, dosage, levofloxacin, pharmacokinetics, urinary excretion
Introduction
Fluoroquinolone resistance relates directly to human and veterinary usage; emerging bacterial resistance poses the single greatest threat to the future survival of the fluoroquinolone drugs as an effective antibiotic class [6]. Levofloxacin [(-)-9-Fluoro-3-methyl-10-(4-methyl-1-piprazinyl)-7-oxo-2,3-dihydro-7H-pyrido [1,2,3-de] [1,4]-benzoxazine-6-carboxylic acid] a recently introduced second generation fluoroquinolone, possesses excellent activity against gram-positive, gram-negative and anaerobic bacteria [8,17]. As compared to other fluoroquinolones, ofloxacin and ciprofloxacin, it also has more pronounced bactericidal activity against organisms like Pseudomonas, Enterobacteriaceae and Klebsiella [15]. The drug is well distributed to target body tissues and fluids in the respiratory tract, skin, urine and prostrate; in addition, its uptake by cells makes it suitable for use against intracellular pathogens [16]. Levofloxacin is metabolized in the liver to demethyl-levofloxacin and levofloxacin-N-oxide and excreted in the urine [16]. The pharmacokinetics of levofloxacin has been investigated in man [7] and guinea pigs [9]. However, information on the pharmacokinetics of levofloxacin in cattle is completely lacking. In view of the marked species variation in the kinetic data of antimicrobial drugs, the present study was undertaken to determine the pharmacokinetics, urinary excretion and an appropriate dosage regimen, following single intramuscular administration and in vitro plasma protein binding of levofloxacin in cross bred calves.
Materials and Methods
Animals and drug administration
Seven healthy male cross bred calves (Holstein Friesian × Sahiwal), ranging between 1-1.5 years of age with an average body weight of 87.3 ± 10.6 kg were used for this study. The animals were maintained on seasonal green fodder, wheat straw and water ad libitum. The average day temperature in the shed was about 25℃ during the experimental period. The experimental protocol followed the ethical guidelines on the proper care and use of animals. Levofloxacin (Hoechst Marion Roussel, India) was administered intramuscularly at the dose rate of 4 mg/kg body weight into the neck region.
Collection of samples
To conduct the pharmacokinetic study, the animals were kept in metabolic stalls of standard size, designed in such a way that the entire amount of urine excreted naturally by the animals within a period of time was automatically collected without contamination or spillage in the containers placed beneath the stalls. Blood samples (5 ml) were withdrawn from the jugular vein into heparinized glass centrifuge tubes before and at 1, 2.5, 5, 7.5, 10, 15, 20, 30, 45 min and 1, 1.5, 2, 2.5, 3, 4, 6, 8, 10, 12, 16 and 24 h after administration of the drug. Plasma was separated by centrifugation at 2,000 × g for 15 min at room temperature and kept at -20℃ until analysis, which was done usually on the day of specimen collection. Urine samples were also collected simultaneously from the same animals at various predetermined time intervals of 2, 4, 6, 8, 10, 12, 16 and 24 h after administration of the drug. At the end of the given time interval, the volume of total urine voided and colleted in the container, was measured for each animal and after filtration, 10 ml samples were taken for analysis.
Estimation of drug
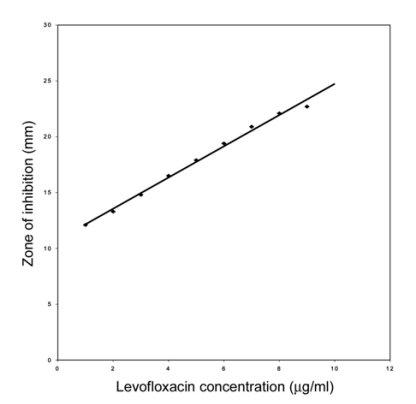
The concentration of levofloxacin in plasma and urine samples was estimated by a standard microbiological assay technique [3] using Escherichia coli (ATCC 10536) as the test organism. This method estimated the level of drug having antibacterial activity, without differentiating between the parent drug and its active metabolites. The assay could detect a minimum of 0.1 µg/ml of levofloxacin. For each sample, nine replicates were analyzed and corrected for the zone of inhibition by standard reference solution. The standard curve of levofloxacin is given in Fig. 1. The concentrations of levofloxacin in the samples were calculated as µg/ml of plasma or urine.
Fig. 1.
Standard curve of levofloxacin in plasma of cross bred calves. Each point represents the mean of the results from 14 assays.
In vitro plasma protein binding
In vitro binding of levofloxacin to the plasma protein of calves was determined by employing the equilibrium dialysis technique [15] at plasma concentrations of 2, 5, 10, 20, 50 and 100 µg/ml and the constants for protein binding were obtained.
Pharmacokinetic variables and dosage regimen
The plasma concentration-time profile of levofloxacin after intramuscular administration in each animal was used to establish various pharmacokinetic determinants and mean kinetic variables were obtained by averaging the variables calculated for individual animals. Overall systemic bioavailability was calculated by using the values of the area under the plasma concentration-time curve (AUC) and β obtained after single intravenous administration of levofloxacin in the same animals used for intramuscular study of levofloxacin at an interval of 30 days. Pharmacokinetic parameters were calculated manually by the computed least-squares linear regression technique [10]. The maintenance (D') dose of levofloxacin was calculated according to the equation:

The priming dose was obtained by omitting -1 from the above equation. The Cp (min)∝ is the minimum therapeutic concentration of levofloxacin, the β is the elimination rate constant and τ is the dosing interval [5].
Results
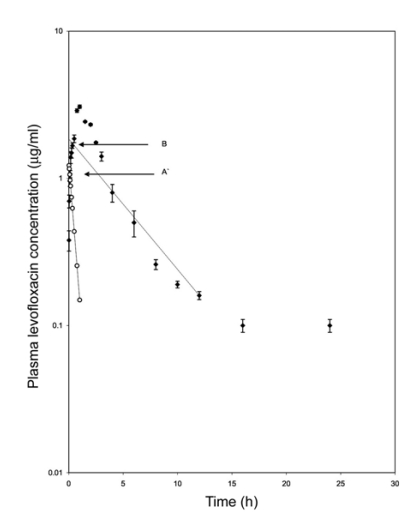
The mean plasma levels of levofloxacin at different time intervals following a single intramuscular injection with a dose of 4 mg/kg body weight are presented on a semilogarithmic scale in Fig. 2. Intramuscular injection resulted into appreciable plasma concentration of the drug (0.38 ± 0.06 µg/ml) at 1 min and a peak plasma level of 3.07 ± 0.08 µg/ml was attained at 1 h post administration. The plasma levels then declined gradually to 0.16 µg/ml at 12 h. Evaluation of the results revealed that the disposition pattern of levofloxacin followed a one-compartment open model that was adequately described by the equation: Cp = Be-βt - A'e-Kat where, Cp is the plasma level of levofloxacin at time t and e represents the base of natural logarithm, A' and B are the extrapolated zero-time intercepts of the absorption and elimination phases, respectively, Ka and β are the absorption and elimination rate constants, respectively. Table 1 shows the pharmacokinetic parameters that describe the absorption and elimination pattern of levofloxacin in the calves. A graphical representation of the mean urine concentration of levofloxacin along with cumulative per cent of drug excreted in urine at different time intervals is shown in Fig. 3.
Fig. 2.
Semilogarithmic plot of plasma concentration-time profile of levofloxacin following its single intramuscular injection of 4 mg/kg b.w. in cross bred calves. Values are presented as mean ± SE (n = 7). The data were analyzed according to the onecompartment open model. Absorption and elimination phases are represented by least square regression lines. The calculated points (o) of distribution phases were obtained by residual technique. Constants A' and B are zero-time intercepts of absorption and elimination phases, respectively.
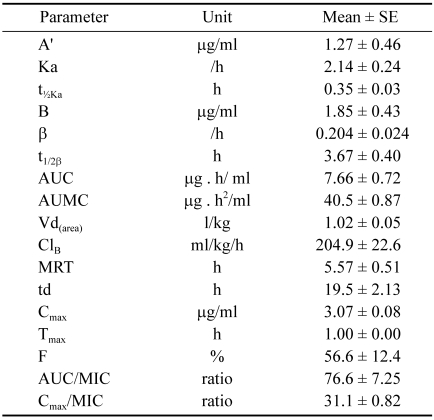
Table 1.
Pharmacokinetic parameters of levofloxacin in cross bred calves following single intramuscular administration (n = 7)
A' and B: zero-time plasma drug concentration intercepts of the regression lines of absorption and elimination phases, respectively; Ka and β: absorption and elimination rate constants, respectively; t½Ka: absorption half-life; t½β: elimination half-life; AUC; area under the plasma concentration-time curve; AUMC: area under the first moment curve; Vdarea: apparent volume of distribution; ClB: total body clearance; MRT: mean residence time; td: duration of therapeutic effect; Cmax and tmax: peak plasma drug concentration and time required to attain the peak concentration, respectively; MIC: minimum inhibitory concentration of drug in plasma; F: overall systemic bioavailability.
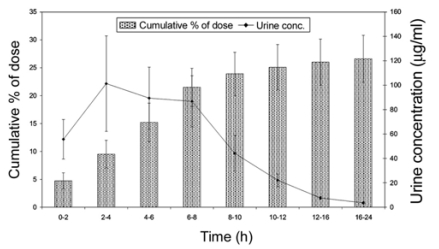
Fig. 3.
Concentration and cumulative per cent of total dose of levofloxacin excreted in urine of cross bred calves following its single intravenous dose of 4 mg/kg b.w. Each point and bar represents the mean ± SE (n = 7).
Discussion
The rapid appearance of levofloxacin in plasma following its intramuscular administration suggested that the drug rapidly entered into the systemic circulation. The high value for the absorption rate constant further confirmed rapid absorption of levofloxacin. Rapid absorption after intramuscular injection has also been reported for other fluoroquinolones, marbofloxacin [18,19] and ciprofloxacin [21] in cattle. An average plasma concentration of 0.032-0.5 µg/ml has been reported to be the minimum therapeutic concentration (MIC90) of levofloxacin against most gram positive, gram negative and atypical bacteria [7]. Keeping in mind the synergistic effects of the immune system and other in vivo factors, and to cover most of the susceptible organisms, in this discussion, the MIC90 of 0.1 µg/ml of levofloxacin has been taken into consideration. The peak plasma level of levofloxacin attained in the present study was approximately 30 fold higher than the MIC of levofloxacin and the drug was detected above the minimum therapeutic plasma level up to 12 h of administration. Similar to our findings, a peak plasma concentration of 3.4 µg/ml was attained after single intraperitoneal injection of levofloxacin in pneumonic guinea pigs [9]. The high value of AUC (7.66 ± 0.72 µg · h/ml) and apparent volume of distribution (1.02 ± 0.05 l/kg) obtained in the present study reflected the vast body area covered by the drug concentration and extensive penetration of levofloxacin into various body fluids and tissues after intramuscular injection.
These observations are in accordance with the similar values of AUC of marbofloxacin (7.648 mg · h/ml ) in cows [18] and the high Vdarea of danofloxacin in goats [1] reported after intramuscular injection. The high value of AUC/MIC90 (76.6 ± 7.25) and Cmax/MIC90 (31.1 ± 0.82) obtained in the present study, provides support for excellent clinical and bacteriological efficacy of levofloxacin in calves. High values of AUC/MIC have also been reported for other fluoroquinolones. After intramuscular injection the AUC/MIC ratio was 40.7 for marbofloxacin in cows [18] and 55.9 for danofloxacin in sheep [20]. In agreement with the present results, a Cmax/MIC ratio of more than 10 has been reported following subcutaneous administration of both danofloxacin and enrofloxacin in calves [22]. The total body clearance of levofloxacin in the present study was 204.9 ± 22.6 ml/kg/h and the elimination half-life (3.67 ± 0.4 h) was comparable to the t½β of 2.53 h for marbofloxacin [18] and 2.4 h for norfloxacin [12] in cattle and 4.41 h for danofloxacin in goats [1] observed after intramuscular administration. Among various pharmacokinetic parameters, bioavailability plays an important role in the therapeutic efficacy of a drug. On the basis of AUC and β after single intravenous (10.5 ± 0.2 µg · h/ml and 0.288 ± 0.001 /h, respectively) and intramuscular (Table 1) administration in calves, the systemic bioavailability of levofloxacin was calculated to be 56.6 ± 12.4% in the calves. This finding was greater than the systemic bioavailability of ciprofloxacin (21.6%) but lower the value of 73% for norfloxacin and 91% for danofloxacin reported after their intramuscular administration in cattle [2,12,21]. The concentration of levofloxacin-equivalent inhibitory units in the urine was very high, at 36 times the MIC90 (minimum inhibitory concentration) even 24 h after administration. High urinary concentration of danofloxacin (58.58 µg/ml) has also been reported after intravenous doses of 1.25 mg/kg in goats [4]. Approximately 26.6% of the microbiological activity of the administered drug was recovered in the urine of calves within 24 h. These findings suggest that levofloxacin may be an appropriate drug for treating urinary tract infections in cattle. The extent of binding of levofloxacin to the plasma proteins of calves (17.0 ± 1.2%) in the present study was in accordance to the corresponding values of 24-38% for levofloxacin in man [16] and 26% for danofloxacin [11] and 36-45% for enrofloxacin [13] in cattle. On the basis of the present study, the priming and maintenance doses of levofloxacin, at a dosage interval of 12 h, were calculated to be 1.48 and 1.38 mg/kg, respectively, or under field conditions, for most bacteria sensitive to levofloxacin, the most appropriate dosage regimen for levofloxacin, would be 1.5 mg/kg repeated at 12 h intervals for the treatment of respiratory, gastrointestinal, urinary tract and other infections in calves. Lack of local reaction or any other adverse effect, rapid absorption, moderate bioavailability, large volume of distribution and high urinary concentration of levofloxacin observed in the present study demonstrated that levofloxacin may be effectively employed by the intramuscular route for the treatment of bacterial infections in calves.
References
- 1.Aliabadi FS, Lees P. Pharmacokinetics and pharmacodynamics of danofloxacin in serum and tissue fluids of goats following intravenous and intramuscular administration. Am J Vet Res. 2001;62:1979–1989. doi: 10.2460/ajvr.2001.62.1979. [DOI] [PubMed] [Google Scholar]
- 2.Apley MD, Upson DW. Lung tissue concentrations and plasma pharmacokinetics of danofloxacin in calves with acute pneumonia. Am J Vet Res. 1993;54:937–943. [PubMed] [Google Scholar]
- 3.Arret B, Johnson DP, Kirshbaum A. Outline of details for microbiological assays of antibiotics: second revision. J Pharm Sci. 1971;60:1689–1694. doi: 10.1002/jps.2600601122. [DOI] [PubMed] [Google Scholar]
- 4.Atef M, El-Gendi AY, Aziza, Amer MM, El-Aty AM. Some pharmacokinetic data for danofloxacin in healthy goats. Vet Res Commun. 2001;25:367–377. doi: 10.1023/a:1010642726054. [DOI] [PubMed] [Google Scholar]
- 5.Baggot JD. Principles of Drug Disposition in Domestic Animals. Philadelphia: Saunders; 1977. pp. 190–218. [Google Scholar]
- 6.Bakken JS. The fluoroquinolones: how long will their utility last? Scand J Infect Dis. 2004;36:85–92. doi: 10.1080/00365540410019039. [DOI] [PubMed] [Google Scholar]
- 7.Chulavatnatol S, Chindavijak B, Vibhagool A, Wananukul W, Sriapha C, Sirisangtragul C. Pharmacokinetics of levofloxacin in healthy Thai male volunteers. J Med Assoc Thai. 1999;82:1127–1135. [PubMed] [Google Scholar]
- 8.Davis R, Bryson HM. Levofloxacin. A review of its antibacterial activity, pharmacokinetics and therapeutic efficacy. Drugs. 1994;47:677–700. doi: 10.2165/00003495-199447040-00008. [DOI] [PubMed] [Google Scholar]
- 9.Edelstein PH, Edelstein MA, Lehr KH, Ren J. In-vitro activity of levofloxacin against clinical isolates of Legionella spp, its pharmacokinetics in guinea pigs, and use in experimental Legionella pneumophila pneumonia. J Antimicrob Chemother. 1996;37:117–126. doi: 10.1093/jac/37.1.117. [DOI] [PubMed] [Google Scholar]
- 10.Gibaldi M, Perrier D. Pharmacokinetics. 2nd ed. New York: Marcel Dekker; 1982. pp. 433–444. [Google Scholar]
- 11.Giles CJ, Magonigle RA, Grimshaw WTR, Tanner AC, Risk JE, Lynch MJ, Rice JR. Clinical pharmacokinetics of parenterally administered danofloxacin in cattle. J Vet Pharmacol Ther. 1991;14:400–410. doi: 10.1111/j.1365-2885.1991.tb00854.x. [DOI] [PubMed] [Google Scholar]
- 12.Gips M, Soback S. Norfloxacin nicotinate pharmacokinetics in unweaned and weaned calves. J Vet Pharmacol Ther. 1996;19:130–134. doi: 10.1111/j.1365-2885.1996.tb00025.x. [DOI] [PubMed] [Google Scholar]
- 13.Kaartinen L, Salonen M, Alli L, Pyorala S. Pharmacokinetics of enrofloxacin after single intravenous, intramuscular and subcutaneous injections in lactating cows. J Vet Pharmacol Ther. 1995;18:357–362. doi: 10.1111/j.1365-2885.1995.tb00604.x. [DOI] [PubMed] [Google Scholar]
- 14.Klesel N, Geweniger KH, Koletzki P, Isert D, Limbert M, Markus A, Riess G, Schramm H, Iyer P. Chemotherapeutic activity of levofloxacin (HR 355, DR-3355) against systemic and localized infections in laboratory animals. J Antimicrob Chemother. 1995;35:805–819. doi: 10.1093/jac/35.6.805. [DOI] [PubMed] [Google Scholar]
- 15.Kunin CM, Dornbush AC, Finland M. Distribution and excretion of four tetracycline analogues in normal young men. J Clin Invest. 1959;38:1950–1963. doi: 10.1172/JCI103974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Langtry HD, Lamb HM. Levofloxacin. Its use in infections of the respiratory tract, skin, soft tissues and urinary tract. Drugs. 1998;56:487–515. doi: 10.2165/00003495-199856030-00013. [DOI] [PubMed] [Google Scholar]
- 17.North DS, Fish DN, Redington JJ. Levofloxacin, a second-generation fluoroquinolone. Pharmacotherapy. 1998;18:915–935. [PubMed] [Google Scholar]
- 18.Schneider M, Valle M, Woehrle F, Boisrame B. Pharmacokinetics of marbofloxacin in lactating cows after repeated intramuscular administrations and pharmacodynamics against mastitis isolated strains. J Dairy Sci. 2004;87:202–211. doi: 10.3168/jds.S0022-0302(04)73159-8. [DOI] [PubMed] [Google Scholar]
- 19.Shem-Tov M, Ziv G, Glickman A, Saran A. Pharmacokinetics and penetration of marbofloxacin from blood into the milk of cows and ewes. Zentralbl Veterinarmed A. 1997;44:511–519. doi: 10.1111/j.1439-0442.1997.tb01137.x. [DOI] [PubMed] [Google Scholar]
- 20.Shojaee Aliabadi F, Landoni MF, Lees P. Pharmacokinetics (PK), pharmacodynamics (PD) and PK-PD integration of danofloxacin in sheep biological fluids. Antimicrob Agents Chemother. 2003;47:626–635. doi: 10.1128/AAC.47.2.626-635.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Singh K, Srivastava AK. Plasma levels, pharmacokinetics, urinary excretion and dosage regimen of ciprofloxacin in cross bred cow calves. J Punjab Acad Sci. 2000;2:105–107. [Google Scholar]
- 22.TerHune TN, Skogerboe TL, Shostorm VK, Weigel DJ. Comparison of pharmacokinetics of danofloxacin and enrofloxacin in calves challenged with Mannheimia haemolytica. Am J Vet Res. 2005;66:342–349. doi: 10.2460/ajvr.2005.66.342. [DOI] [PubMed] [Google Scholar]