Abstract
Transplantation therapy for human diabetes is limited by the toxicity of immunosuppressive drugs. If toxicity can be minimized, there will still be a shortage of human donor organs. Xenotransplantation of porcine islets is a strategy to overcome supply problems. Xenotransplantation in mesentery of pig pancreatic primordia obtained very early during organogenesis [embryonic day 28 (E28)] is a way to obviate the need for immunosuppression in rats or rhesus macaques and to enable engraftment of a cell component originating from porcine islets implanted beneath the renal capsule of rats. Here, we show engraftment in the kidney of insulin and porcine proinsulin mRNA-expressing cells following implantation of porcine islets beneath the renal capsule of diabetic rhesus macaques transplanted previously with E28 pig pancreatic primordia in mesentery. Donor cell engraftment is confirmed using fluorescent in situ hybridization (FISH) for the porcine X chromosome and is supported by glucose-stimulated insulin release in vitro. Cells from islets do not engraft in the kidney without prior transplantation of E28 pig pancreatic primordia in mesentery. This is the first report of engraftment following transplantation of porcine islets in non-immunosuppressed, immune-competent non-human primates. The data are consistent with tolerance to a cell component of porcine islets induced by previous transplantation of E28 pig pancreatic primordia.
Key words: β cell, diabetes mellitus, non-human primates, transplantation, xenotransplantation
Introduction
In that pigs are plentiful, and because porcine insulin works well in humans, the pig has been suggested to be a pancreas organ donor for humans with diabetes. The severity of humoral rejection due to preexisting natural antibodies effectively precludes their use as whole pancreas donors in non-human primates or humans.1–4 However, isolated islets of Langerhans (islets) can survive following transplantation into immunosuppressed nonhuman primates2–4 or humans with diabetes1 without initiating humoral rejection. Unfortunately, recent experience with pig to primate islet2–4 or neonatal islet3 transplantation shows that sustained insulin independence can be achieved but only through the use of immunosuppressive agents that are not approved for human use or would result in an unacceptable level of morbidity in humans.2–4
Glucose tolerance can be normalized in streptozotocin (STZ)-diabetic (type 1) LEW rats or ZDF (type 2) diabetic rats following transplantation in mesentery of pig pancreatic primordia obtained very early during embryogenesis [on embryonic day 28 (E28), just after the organ differentiates and prior to the time dorsal and ventral anlagen fuse] without host immunosuppression.5–8 Cells expressing insulin and porcine proinsulin mRNA with β-cell morphology engraft in host mesentery, mesenteric lymph nodes, liver and pancreas post-transplantation.5–8 Cells originating from E28 pig pancreatic primordia engraft similarly in non immunosuppressed STZ-diabetic rhesus macaques,9 and glucose tolerance is improved post-transplantation.10
To ascertain whether engraftment of cells originating from E28 pig pancreatic primordia in rats is permissive for engraftment of the same or a similar cell component present in porcine islets from adult swine, we implanted the latter beneath the renal capsule of rats that previously had been transplanted with E28 pig pancreatic primordia in mesentery. Whole islets do not engraft. However, a population of cells originating from donor islets with β-cell morphology that express insulin and porcine proinsulin mRNA does engraft in kidneys of rats transplanted previously with E28 pig pancreatic primordia.8 Here we show similar engraftment of cells originating from porcine islets using diabetic rhesus macaques as hosts.
Results
Three rhesus macaques rendered diabetic (Diab) with STZ were transplanted with pig pancreatic primordia in mesentery. Four weeks later, macaques were transplanted with 15,000–20,000 porcine islet equivalents (IEQ) per kg in the renal subcapsular space of one kidney (Diab-E28-Islets group). Eight weeks later, the macaques in the Diab-E28-Islets group were euthanized and tissues obtained for analysis. Three other STZ-diabetic rhesus macaques underwent porcine islet implantation in the subcapsular space of one kidney without prior transplantation of E28 pig pancreatic primordia (Diab-Islets group). Eight weeks later, macaques in the Diab-Islets group were euthanized and tissues obtained for analysis.
Figure 1 shows mesentery from a macaque in the Diab-E28-Islets group (Fig. 1A) and in the Diab-Islets group (Fig. 1B) immediately after euthanasia. We have shown previously that mesentery of macaques transplanted with E28 pig pancreatic primordia becomes opacified relative to mesentery of non-transplanted macaques and contains enlarged mesenteric lymph nodes.9 Similarly, mesentery of macaques in the Diab-E28-Islets group (Fig. 1A) is opacified relative to mesentery of macaques in the Diab-Islets group (Fig. 1B). An arrow delineates a mesenteric vessel leading to a lymph node (LN) in Figure 1A.
Figure 1.
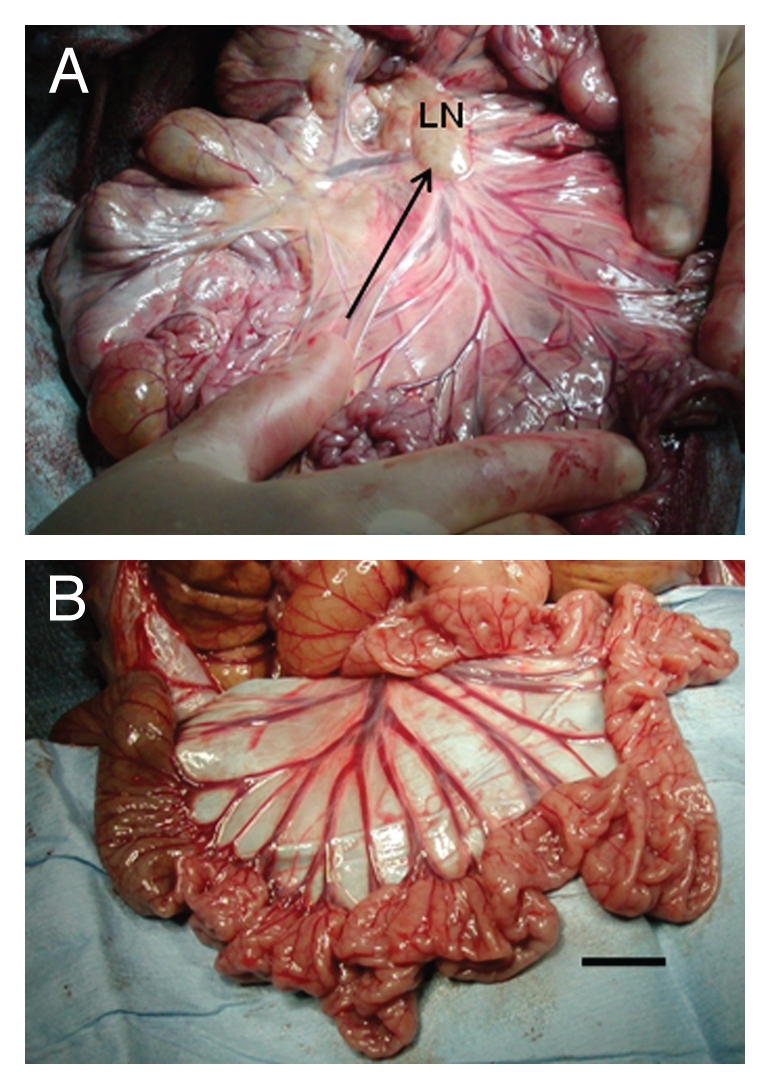
Photographs of mesentery from rhesus macaques. (A) Rhesus macaque transplanted with E28 pig pancreatic primordia in mesentery and implanted with porcine islets in one kidney. Arrow parallels lymphatic vessel (LN, lymph node). (B) Rhesus macaque implanted with porcine islets in one kidney without prior transplantation of E28 pig pancreatic primordia. Scale bar, 2 cm.
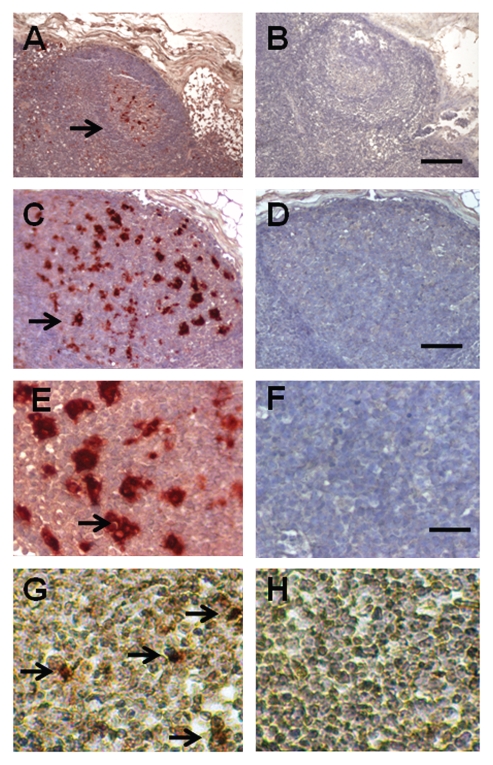
Figure 2 shows sections of a mesenteric lymph node from a macaque in the Diab-E28-Islets group stained using anti-insulin antibodies (Fig. 2A, C and E) or control antiserum (Fig. 2B, D and F) or hybridized to an antisense (Fig. 2G) or sense (Fig. 2H) probe for porcine proinsulin mRNA. As before, in macaques transplanted only with pig pancreatic primordia,9 cells are present with β-cell morphology that stain for insulin (Fig. 2A, C and E, arrows) and to which the antisense porcine proinsulin mRNA probe binds (Fig. 2G, arrows). No staining for insulin is observed in sections incubated with control antiserum (Fig. 2B, D and F). No hybridization is observed if a sense probe is substituted for the antisense probe (Fig. 2H).
Figure 2.
Sections of a mesenteric lymph node from a macaque transplanted with E28 pig pancreatic primordia in mesentery, followed by porcine islets in the renal subcapsular space stained using anti-insulin antibodies (A, C and E) or control antiserum (B, D and F) or hybridized to an antisense (G) or sense (H) probe for porcine proinsulin mRNA. Arrows, positively staining cells (A, C, E and G). Scale bars, 80 µm (A and B); 25 µm (C and D); 15 µm (E–H).
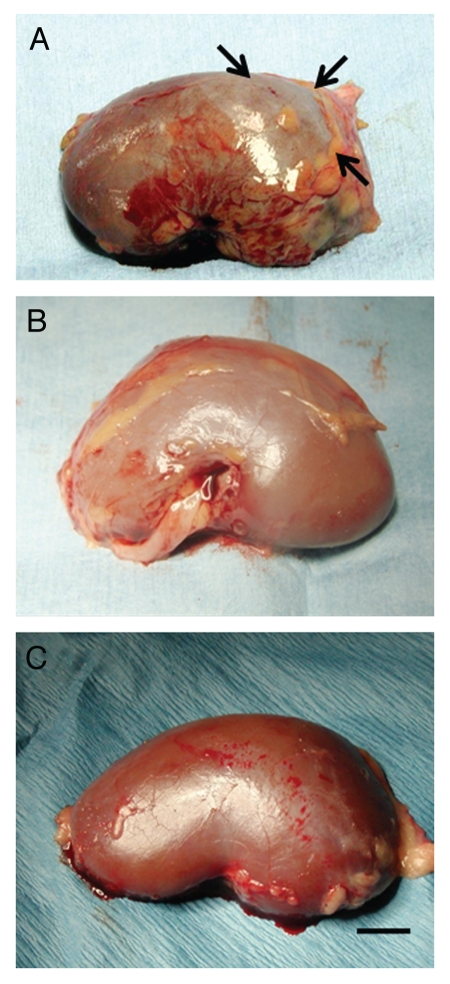
Figure 3 shows a kidney from a rhesus macaque in the DiabE28-Islets group into which islets were implanted (Fig. 3A), a non-implanted (contralateral) kidney from a Diab-E28-Islets group macaque (Fig. 3B) and a kidney implanted with porcine islets without prior transplantation of E28 pig pancreatic primordia (Diab-Islets group) (Fig. 3C). As shown following implantation of porcine islets into kidneys of rats under identical conditions,8 a distinct, whitish, well-demarcated graft can be distinguished from the surrounding renal parenchyma (Fig. 3A, arrows). No graft can be distinguished in the non-implanted (contralateral) kidney (Fig. 3B) or in the kidney from the primate implanted with porcine islets without prior pancreatic primordia transplantation (Fig. 3C).
Figure 3.
Photographs of kidneys from rhesus macaques. (A) From a macaque transplanted with E28 pig pancreatic primordia in mesentery and implanted with porcine islets in one kidney; A whitish well-demarcated triangle-shaped graft (arrows) is delineated. (B) A non-implanted kidney from a macaque transplanted with E28 pig pancreatic primordia in mesentery and implanted with porcine islets in the contralateral kidney; and (C) From a macaque implanted with porcine islets in the kidney with no prior transplantation of E28 pig pancreatic primordia. Scale bar, 0.8 cm.
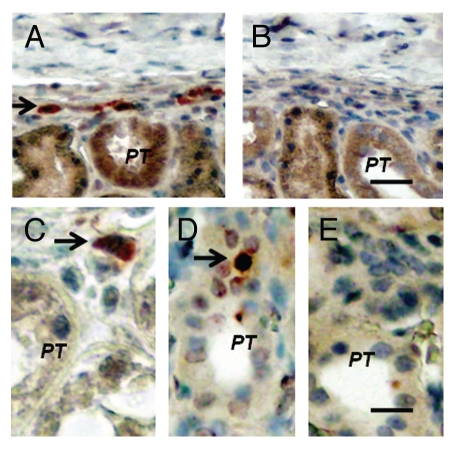
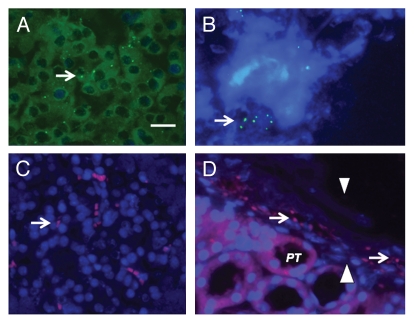
Figure 4 shows sections from islet-implanted kidneys of macaques in the Diab-E28-Islets group. Sections are stained using anti-insulin antibodies (Fig. 4A and C) or control serum (Fig. 4B) or hybridized to an antisense (Fig. 4D) or sense (Fig. 4E) probe for porcine proinsulin mRNA. Cells that stain for insulin are present in the subcapsular space (Fig. 4A, arrow). A high power view of a single insulin-staining cell is shown in Figure 4C (arrow). It is polygonal with a round nucleus and abundant cytoplasm, a β-cell morphology.8,9 A cell in the sub-capsular space to which the antisense porcine proinsulin mRNA probe binds is shown in Figure 4D (arrow). No staining for insulin is observed in sections incubated with control antiserum (Fig. 4B). No hybridization is observed if a sense probe is substituted for the antisense probe (Fig. 4E).
Figure 4.
Sections of the islet-implanted kidney from a macaque transplanted with E28 pig pancreatic primordia in mesentery followed by porcine islets in the renal subcapsular space stained using anti-insulin antibodies (A and C) or control antiserum (B) or hybridized to an anti-sense (D) or sense (E) probe for porcine proinsulin mRNA (PT, proximal tubule). Arrows, positively staining cells (A, C and D). Scale bars 15 µm (A and B); 7.5 µm (C–E).
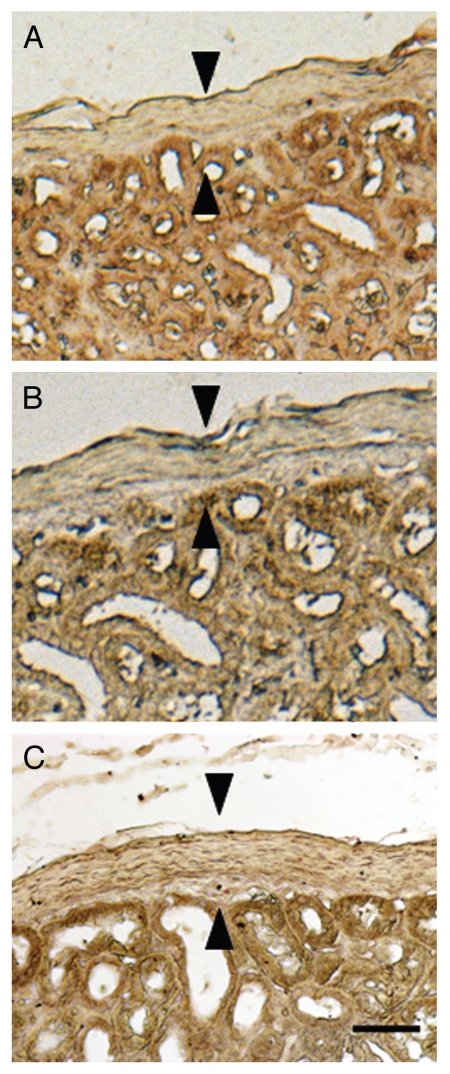
Figure 5 shows sections from the contralateral (non-implanted) kidney from a macaque in the Diab-E28-Islets group stained using anti-insulin antibodies (Fig. 5A) or control serum (Fig. 5B) and from an islet-implanted kidney from a macaque in the Diab-Islets group (Fig. 5C) stained using anti-insulin antibodies. No cells that stain for insulin are present in the sub-capsular space of the non-implanted (contralateral) kidney (compare Fig. 4A with 5A) or the kidney implanted with islets in the macaque not previously transplanted with E28 pig pancreatic primordia in mesentery (compare Fig. 4A with 5C).
Figure 5.
Sections of the non-implanted kidney (A and B) from a macaque transplanted with E28 pig pancreatic primordia in mesentery, followed by porcine islets in the renal subcapsular space of the contralateral kidney, or the implanted kidney from a macaque implanted with porcine islets without prior transplantation of E28 pig pancreatic primordia (C) stained using anti-insulin antibodies (A and C) or control antiserum (B) Arrowheads, subcapsular space (A–C). Scale bar 15 µm.
To provide additional evidence that cells in mesenteric lymph nodes and the islet-implanted kidneys of macaques in the Diab-E28-Islets group originate from the pig, we performed fluorescent in situ hybridization using probes specific for the pig X chromosome (Fig. 6). Shown in Figures 6A and C are pig X chromosomes in nuclei of cells from a normal porcine pancreas (arrow positive controls) stained using two different probes (green A; pink C). Figures 6B and D show pig X chromosomes (arrows) in the nuclei of cells in the mesenteric lymph node (Fig. 6B) and renal subcapsular space (Fig. 6D, arrowheads). As before, in rat renal tubules,8 autofluorescence of macaque renal tubular cytoplasm is observed. However, pig X chromosomes are not present in macaque renal tubules.
Figure 6.
Fluorescence microscopy of tissue sections originating from (A and C) a normal porcine pancreas or (B) a mesenteric lymph node from a rhesus macaque that had been transplanted with embryonic pig pancreas in mesentery and subsequently with porcine islets in kidney or (D) a subcapsular section from a kidney of a rhesus macaque transplanted with embryonic pig pancreas in mesentery and subsequently with porcine islets in that kidney (PT, proximal tubule). Arrows delineate pig X chromosomes. Arrowheads renal capsule (D). Scale bar 10 µm.
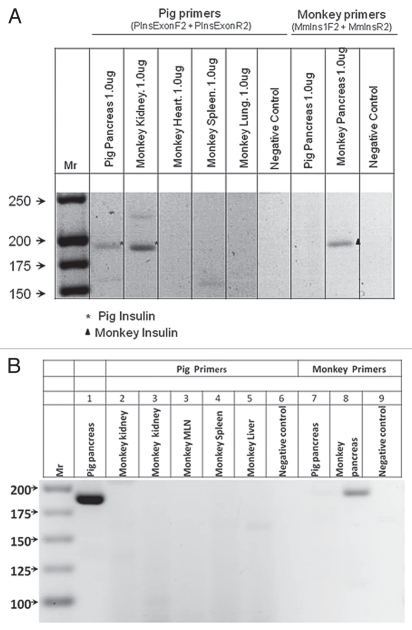
Multiple organs were excised from a macaque transplanted with E28 pig pancreatic primordia in mesentery and, subsequently, with porcine islets in the renal subcapsular space of one kidney (Diab-E28-Islets group). Tissues were homogenized individually and total RNA purified. RT-PCR was performed using primers for pig or monkey proinsulin mRNA (intron-spanning to eliminate the possibility of false amplification from genomic DNA). Results are shown in Figure 7A. Products were sequenced to confirm identities. The pig primers amplify a band of 193 bps in RNA originating from pig pancreas, corresponding to pig pro-insulin insulin mRNA. The rhesus macaque (monkey) primers amplify a band of 199 bps corresponding to monkey proinsulin insulin mRNA in monkey pancreas. Pig proinsulin mRNA is also detected in the islet-implanted monkey kidney. Multiple organs were excised from a macaque transplanted with porcine islets in the renal subcapsular space of one kidney with no prior transplantation of E28 pig pancreatic primordia in mesentery (Diab-Islets group, and RT-PCR was performed as above. As shown in Figure 7B, no pig proinsulin mRNA was detected in any monkey organ including the transplanted kidney.
Figure 7.
RT-PCR: (A) Shown left to right are DNA molecular weights (Mr); amplification of bands using primers specific for porcine proinsulin from 1 µg RNA extracted from pig pancreas; or from a rhesus macaque (monkey) transplanted with E28 pig pancreatic primordia in mesentery, followed by implantation of porcine islets in the renal subcapsular space (Diab-E28-Islets), kidney, heart, spleen, lung, a negative control for porcine-specific primers (no RNA); and amplification of bands using primers specific for monkey proinsulin from 1 µg of pig pancreas RNA, monkey pancreas and a second negative control for macaque-specific primers. (B) Shown left to right are DNA molecular weights (Mr); amplification of bands using primers specific for porcine proinsulin from 2 µg RNA extracted from pig pancreas; or from a rhesus macaque (monkey) implanted with porcine islets in the renal subcapsular space with no previous transplantation of E28 pig pancreatic primordia (Diab-Islets), kidneys, mesenteric lymph node (MLN) spleen, liver, a negative control for porcine-specific primers (no RNA); and amplification of bands using primers specific for monkey proinsulin from 2 µg of pig pancreas RNA, monkey pancreas and a second negative control for macaque-specific primers. Pig primers amplify a 193 bps band. Monkey primers amplify a 199 bps band.
Figure 8 is an electron micrograph of the renal subcapsular space from a rhesus macaque kidney into which porcine islets were implanted a month following transplantation of E28 pig pancreatic primordia in mesentery. Shown is a cell with encapsulated granules (arrows) characteristic of endocrine secretory granules.6
Figure 8.
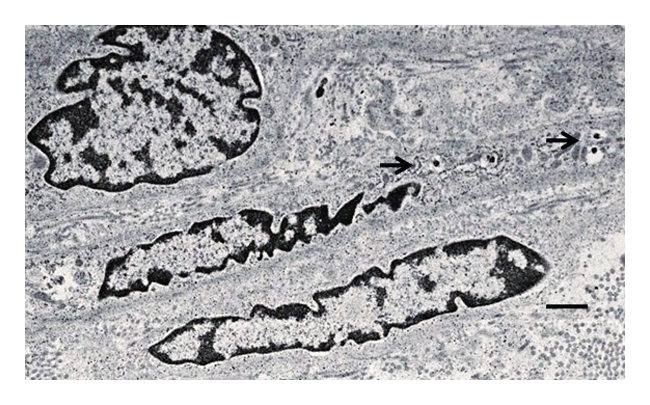
Electron micrograph of rhesus macaque kidney following sequential transplantation of E28 pig pancreatic primordia in mesentery and implantation of porcine islets in the kidney. Arrows, endocrine granules. Scale bar a 2 µm.
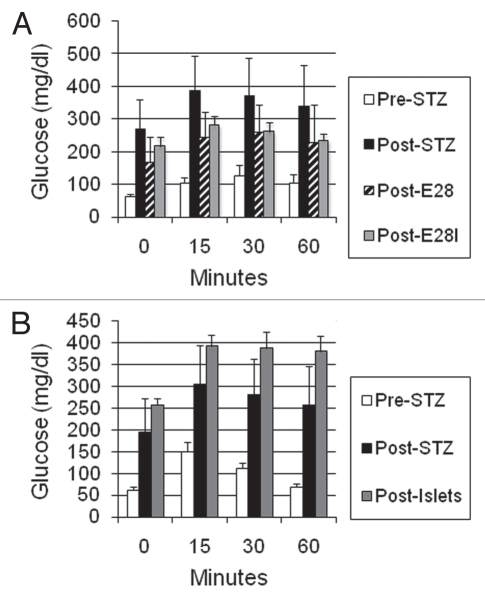
Figures 9A and B illustrate GTTs performed on macaques in the Diab-E28-Islets and Diab-Islets groups, respectively, prior to administration of STZ (pre-STZ) and following administration of STZ (post-STZ) immediately before transplantation. In the Diab-E28-Islets group macaques, GTTs were also performed 4 weeks following transplantation of E28 pig pancreatic primordia prior to implantation of islets (post-E28) and again 7 weeks following subsequent implantation of porcine islets (post-E28-I). In the Diab-Islets group macaques, GTTs were performed 7 weeks following implantation of porcine islets (post-islets). Multiple comparisons were performed (Bonferroni) between mean glucose levels (times 0, 15, 30 and 60) among the Diab-E28-Islets group macaques (pre-STZ, post-STZ, post-E28; and Post-E28-I) (Fig. 9A). Levels were elevated significantly post-STZ relative to pre-STZ (p < 0.001). Thus, administration of STZ rendered macaques diabetic. Levels were reduced significantly post-E28 (p < 0.001) or post-E28-I (p < 0.01) relative to post-STZ but remained elevated relative to pre-STZ (p < 0.001 for each comparison). Thus transplantation of E28 pig pancreatic primordia with or without subsequent implantation of porcine islets improved glucose tolerance but did not render it normal. Levels post-E28 were not different from levels post-E28-I. Thus, implantation of islets subsequent to transplantation of E28 pig pancreatic primordia did not improve glucose tolerance. Multiple comparisons were performed between mean glucose levels (times 0, 15, 30 and 60) among the Diab-Islets group macaques (pre-STZ, post-STZ and post-islets) (Fig. 9B). Levels were elevated significantly (p < 0.001) post-STZ relative to pre-STZ and were further elevated post-islets relative to post-STZ (p < 0.01). Thus, implantation of porcine islets alone did not improve glucose tolerance.
Figure 9.
Intravenous glucose tolerance in rhesus macaques. Glucose in peripheral venous blood was measured in (A) three macaques in the Diab-E28-Islets group and (B) three macaques in the Diab-Islets group before any transplantations prior to administration STZ (pre-STZ) and 5 days following administration of STZ (post-STZ). Glucose tolerance was measured in Diab-E28 Islets macaques 4 weeks post-transplantation of E28 pig pancreatic primordia (post-E28) and again 7 weeks after subsequent implantation of porcine islets (post-E28-I) and in Diab-Islets group macaques 7 weeks following implantation of porcine islets (post-islets). Data are shown as mean ± SE (three macaques).
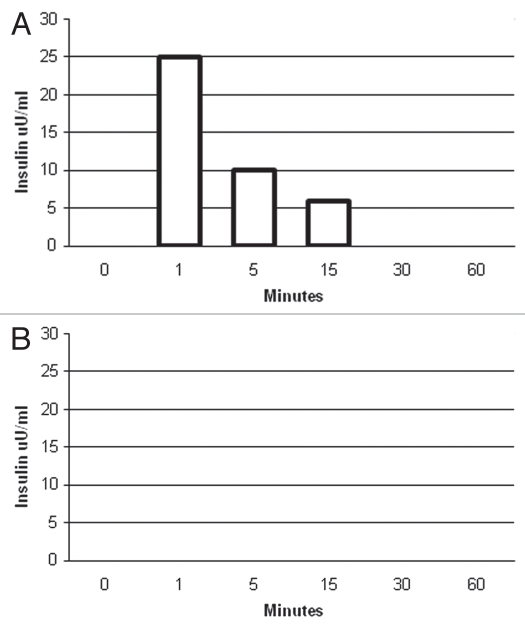
To show that pig cells within transplanted diabetic macaque kidneys are capable of releasing insulin in response to glucose, we incubated slices from (1) the kidney implanted with porcine islets or (2) the contralateral kidney from one rhesus macaque into which E28 pig pancreatic primordia had been transplanted previously, for 1 h at 37°C in DMEM containing 3 mM glucose. We took aliquots immediately prior to (time 0) and following addition of glucose to incubations so as to render the glucose concentration 20 mM. As illustrated in a representative experiment using weight-matched tissue (Fig. 10A), no insulin could be detected at time 0 in supernatants from the implanted kidney. However, insulin was detectable by 1 min after increasing the DMEM glucose level, consistent with a first-phase insulin release characteristic of β cells. No insulin was detected at any time in any supernatants from the non-implanted kidney (Fig. 10B) or in any supernatants from a kidney of a macaque in which porcine islets were implanted without prior transplantation of E28 pig pancreatic primordia in mesentery (not shown).
Figure 10.
Levels of insulin (µU/ml) measured over 60 min in DMEM-containing weight-matched tissue originating from (A) a kidney implanted with porcine islets (159 mg) and (B) the contralateral non-implanted kidney (158 mg) of the same macaque in the Diab-E28-Islets group. Immediately after measurements were made at time 0, glucose was added to DMEM so as to render the glucose concentration 20 mM. Data are representative of three determinations.
Discussion
Transplantation of embryonic pancreas (primordia) to replace the function of diseased organs offers theoretical advantages relative to transplantation of either pluripotent ES cells or of fully differentiated (adult) pancreas or islets:10 (1) Primordia differentiate along defined organ-committed lines with no requirement to steer differentiation and no risk of teratoma formation. The glucose-sensing and insulin-releasing functions of β cells that differentiate from primordia are functionally linked; (2) The growth potential of cells within embryonic pancreas is enhanced relative to those in terminally differentiated pancreas or islets; (3) The cellular immune response to transplanted primordia obtained early during embryogenesis is attenuated relative to that directed against adult islets; (4) The ability of avascular primordia to attract a host vasculature renders them less susceptible to humoral rejection than is adult pancreas with donor blood vessels transplanted across a discordant xenogeneic barrier, and (5) Primordia differentiate selectively. In the case of E28 embryonic pig pancreas, exocrine tissue does not differentiate following transplantation, obviating complications such as the enzymatic autodigestion of host tissues.5–9,11–14
While glucose tolerance can be normalized in non-immunosuppressed diabetic rats by transplantation of E28 pig pancreatic primordia5–8 and improved in rhesus macaques following transplantation of E28 pig pancreatic primordia10 (Fig. 9A), it has proven to be more difficult to render macaques nonmoglycemic without an exogenous insulin requirement.9 The most likely explanation is the size difference between the species, with macaques weighing 20–30 times as much as rats.8 A STZ-diabetic rat can be rendered normoglycemic by transplantation of 5–8 pig pancreatic primordia.8 Extrapolating, it would take 100 primordia to normalize glucose tolerance in a macaque. In lieu of increasing the number of transplanted primordia, we determined whether porcine islets, a more easily obtainable and possibly more robust source of insulin-producing cells, could be substituted in animals transplanted previously with E28 pig pancreatic primordia. Our first step was to show, using rats, that prior transplantation of primordia permits engraftment of a β-cell component from porcine islets.8 Here, we document using immunohistochemistry for insulin and in situ hybridization for porcine proinsulin mRNA (Fig. 4); fluorescent in situ hybridization (Fig. 6) and RT-PCR for porcine proinsulin mRNA (Fig. 7) that pig cells engraft in macaque kidney following transplantation of E28 pig pancreatic primordia in mesentery and subsequent implantation of porcine islets in kidney. Pig cells do not engraft in kidney following implantation of islets without prior transplantation of E28 pig pancreatic primordia or in the contralateral (non-implanted) kidney (Fig. 5). Thus, cells illustrated in Figure 4 are almost certainly derived from porcine islets and not from embryonic pancreas transplants. Furthermore, the cells engrafted in macaque kidney release insulin in a glucose-dependent manner in vitro (Fig. 10A). The ELISA we used for these studies is sensitive to porcine insulin but also detects primate insulin. While it is possible that the implanted kidney contains native cells that secrete primate insulin rather than implanted cells that secrete porcine insulin, this is rendered unlikely by the absence of any insulin in the non-implanted kidney (Fig. 10B).
In rodent models, the renal subcapsular space is the most common site for islet implantation. However, in large animals and humans, islet transplantation beneath the kidney capsule is limited due in part to the success of intraportal infusion.15 Islets in the portal vein release insulin into the portal vein, from which it accesses the liver directly, an advantage relative to release into the systemic circulation as occurs following renal subcapsular implantation.15 We utilized the renal subcapsular site in our previous study8 and in the present studies, because β cells originating from E28 pig pancreatic primordia transplanted in mesentery do not engraft in kidney,8,9 permitting the distinction between cells originating from E28 pig pancreatic primordia and porcine islets at the renal subcapsular site. Glucose tolerance in diabetic rats not fully normalized by prior transplantation of E28 pig pancreatic primordia in mesentery, was corrected following subsequent implantation of porcine islets in kidney.8 In contrast, such a correction was not observed following E28 pig pancreatic primordia transplantation and porcine islets implantation in macaques (Fig. 9A), consistent with previous observations that glucose tolerance is more difficult to correct in macaques than in rats.9 Our present experiments were designed to demonstrate engraftment of a cell component originating from porcine islets in kidneys of diabetic macaques transplanted previously with E28 pig pancreatic primordia, not necessarily to normalize glucose tolerance. It is possible that the cell component, although of sufficient mass to normalize glucose tolerance in diabetic rats following implantation of islets [5,000 IEQ in 150 g rats (approximately 33,000 IEQ per kg)],8 is insufficient following implantation of 15–20,000 IEQ/kg in rhesus macaques. Implantation of more islets (isolated from more than one adult pig pancreas) in kidney or, alternatively, infusion of porcine islets into the portal vein of macaques15 following transplantation of E28 pig pancreatic primordia in mesentery may be a better way to normalize glucose tolerance.
Though not observed under all conditions,11–14 engraftment of pancreatic progenitors transplanted across a xenogeneic barrier to non-immunosuppressed immune-sufficient hosts has been reported twice previously. Eloy et al. described normalization of glucose post-transplantation of E15 but not E18 embryonic chick pancreas into liver of non-immunosuppressed STZ-diabetic rats.16 Abraham et al. described successful xenoengraftment in multiple organs of human pancreatic islet-derived progenitor cells infused in non-immunosuppressed mice. Xenotransplantation of fetal pancreas may be particularly suited to induction of tolerance. However, neither Eloy et al. nor Abraham et al. nor we define an immunological mechanism. We have proposed10 that transplantation of E28 pig pancreatic primordia in the mesentery and migration of cells to mesenteric lymph nodes and liver recapitulates events that occur during induction of oral tolerance, the induction of which is dependent on antigen transport via afferent lymphatics into the draining mesenteric lymph nodes. Harada et al. have proposed a similar co-opting of oral tolerance to explain the muted immune response in vivo and by cells from mesenteric lymph nodes in vitro to a colon carcinoma of BALB/c origin or a human CD80-transfected DBA/2 mastocytoma injected into the subserosal space of cecum in BALB/c mice relative to tumors injected subcutaneously.18
In previous studies that did not employ transplantation of E28 pig pancreatic primordia prior to implantation of porcine islets in non-human primates,19–21 complete destruction of grafts was evident within two weeks. We found no evidence that porcine islets survive post-implantation in macaque kidneys without prior transplantation of E28 pig pancreatic primordia transplantation in mesentery (Figs. 5 and 7). However, a β cell-like component (containing insulin and porcine proinsulin mRNA) originating from porcine islets engrafts if macaques are transplanted previously with E28 pig pancreatic primordia in mesentery. To our knowledge, ours is the first report describing sustained survival of such cells following transplantation of porcine islets beneath the renal capsule of non-immunosuppressed immune sufficient non-human primates. The findings establish a rationale for further investigations to ascertain whether porcine islets can normalize glucose tolerance in diabetic primates transplanted previously with E28 pig pancreatic primordia.
Methods
Animal care and induction of diabetes mellitus.
Animal care followed Institutional Animal Care and Use Committee (IACUC) standards. Insulin-dependent diabetes mellitus was induced in prepubescent (2.5–3.5 kg) rhesus (Rh) macaques (Macaca mulatta) obtained from Alphagenesis Yemasee SC by infusion of STZ intravenously in a single 60 mg/kg bolus.9 Macaques had free access to water, were fed Harlan 2050 Teklad Global 20% protein diet (3.5% of their body weight per day divided in two meals, each after administration of insulin) and fresh fruit and were monitored twice daily for non-fasting blood glucose. In all diabetic macaques, blood glucose was maintained at or below 200 mg/dl using two daily Lantus insulin injections plus 1–2 units of Lispro as needed.
Isolation of pig pancreatic primordia and porcine islets and transplantation/implantation into diabetic rhesus macaques.
Pancreatic primordia from 20–30 E28 Yorkshire pig embryos (Oak Hill Genetics) were isolated and transplanted between tissue planes of mesentery without suture, exactly as described previously in reference 9. Islets of Langerhans from adult female Yorkshire pigs were isolated as before8 and implanted beneath the kidney capsule. A midline abdominal incision (10–15 cm) was made and the kidney exposed, with care taken not to tear the capsule surrounding the kidney. A small incision was made in the capsule of one kidney (usually the left kidney) and a 22G catheter placed under the capsule through which all islets isolated from one adult pig pancreas (15,000 to 20,000 IEQ per kg) were infused. The capsule was closed with a single suture, and the abdomen was closed in standard fashion.
Glucose tolerance testing.
Levels of glucose were measured using the Hemocue B-glucose Analyzer (Hemocue) in whole blood. Intravenous glucose tolerance testing (IV GTT) was performed as before.10 Exogenous insulin was held for 24 hours in macaques fasted for 18 hours prior to administration of glucose (0.5 g/kg) intravenously over 30 seconds as 50% dextrose. Peripheral venous blood samples were taken prior to injection (Time 0) and at several times after administration. Multiple comparisons were performed using the Bonferroni multiple comparisons test (GraphPad Instat 3). Differences were considered significant if p < 0.05 by two-tailed analysis.
Histology.
Tissue fixation, sectioning, staining and photography were performed as in previous studies in reference 9. Polyclonal rabbit anti-insulin antiserum that recognizes both porcine and rhesus macaque insulin was obtained from Accurate Chemicals. Normal rabbit serum was substituted for the anti-insulin serum for control stains.5–7 Hematoxylin was used as a counterstain in Figures 2, 4 and 5.
Detection of insulin transcripts using RT-PCR or in situ hybridization.
Tissues were excised, RNA was purified and reactions carried out as before.9 Products were separated by electrophoresis on 3% agarose gels and their identities confirmed by sequencing in the Washington University Core Protein and Nucleic Acid Chemistry Laboratory. The primers are intron-spanning to eliminate the possibility of false amplification from genomic DNA. For amplification of pig proinsulin transcripts (GenBank, AY044828), the primers used are SsInsF2: 5′-AAC CCT CAG GCA GGT GCC-3′; SsInsR2: 5′-GGG GTG CGG GGA GCA GCA-3′, using conditions: 30 minutes at 55°C, 2 minutes at 94°C and followed by 40 cycles of 94°C, 15″; 64°C, 30″; 68°C, 25″. For rhesus macaque (monkey) proinsulin transcripts (Human Genome Sequencing Center Baylor College of Medicine: Contigs 422635 and 421061), the primers used are MmIns1F2: 5′-GAC CCT CAG GTG GGG CAG-3′; MmInsR2: 5′-AGG AGG CGG AGG GTG TGG-3′ using conditions: 30 minutes at 55°C, 2 minutes at 94°C and followed by 40 cycles of 94°C, 15″; 61°C, 35″; 68°C, 25″. The pig primers amplify a band of 193 bps in RNA originating from pig pancreas corresponding to pig proinsulin mRNA. The monkey primers amplify a band of 199 bps corresponding to monkey proinsulin mRNA in monkey pancreas RNA.
In situ hybridization was performed on 5 µm paraffin-embedded sections using digoxin-labeled antisense probes that are specific for porcine proinsulin mRNA (do not hybridize to rhesus macaque proinsulin mRNA).9 A sense probe was used for control stains.
Fluorescent in situ hybridization.
Fluorescence in situ hybridization in paraffin-embedded tissue sections was performed exactly as in our previous studies in reference 8 and visualized using an Olympus BX61 epifluorescence microscope system.
Electron microscopy.
Electron microscopy was performed by one of us (H.L.) exactly as in previous studies in reference 8.
Detection of insulin in supernatants.
Kidney tissue was removed from diabetic rhesus macaques, and tissue samples were placed individually in 1 ml of Dulbecco's modified Eagle's medium (DMEM) containing 3 mM glucose at 37°C as before.7 After 1 h, glucose was added to the incubation to make the total concentration 20 mM, and 100 µl aliquots were removed beginning 1 min later. Levels of insulin in aliquots were measured using a porcine insulin-sensitive ELISA (DAKO, #K6219, sensitivity 0.5 µU/ml).
Institutional review board approval.
Animal studies were approved by the Washington University Animal Studies Committee (Approval 20100212).
Acknowledgments
Supported by George M. O'Brien Center DK079333 and grant 1-2008-37 from the Juvenile Diabetes Research Foundation (JDRF). We are grateful for the technical assistance of Nick Benshoff.
Abbreviation
- E
embryonic day
- IEQ
islet equivalents
- STZ
streptozotocin
Disclosure of Potential Conflicts of Interest
No potential conflicts of interest were disclosed.
References
- 1.Groth CG, Korsgren O, Tibell A, Tollemar J, Moller E, Bolinder J, et al. Transplantation of porcine fetal pancreas to diabetic patients. Lancet. 1994;344:1402–1404. doi: 10.1016/s0140-6736(94)90570-3. [DOI] [PubMed] [Google Scholar]
- 2.Hering B, Wijkstrom M, Graham M, Hardstedt M, Aasheim TC, Jie T, et al. Prolonged diabetes reversal after intraportal xenotransplantation of wild-type porcine islets in immunosuppressed nonhuman primates. Nat Med. 2006;12:301–303. doi: 10.1038/nm1369. [DOI] [PubMed] [Google Scholar]
- 3.Cardona K, Korbutt GS, Milas Z, Lyon J, Cano J, Jiang W, et al. Long-term survival of neonatal porcine islets in non-human primates by targeting costimulation pathways. Nat Med. 2006;12:304–306. doi: 10.1038/nm1375. [DOI] [PubMed] [Google Scholar]
- 4.van der Windt DJ, Bottino R, Casu A, Campanile N, Smetanka C, He J, et al. Long term controlled normoglycemia in diabetic non-human primates after transplantation with hCD46 transgenic porcine islets. Am J Transpl. 2009;9:2716–2726. doi: 10.1111/j.1600-6143.2009.02850.x. [DOI] [PubMed] [Google Scholar]
- 5.Rogers SA, Chen F, Talcott M, Hammerman MR. Islet cell engraftment and control of diabetes in rats following transplantation of pig pancreatic anlagen. Am J Physiol. 2004;286:502–509. doi: 10.1152/ajpendo.00445.2003. [DOI] [PubMed] [Google Scholar]
- 6.Rogers SA, Liapis H, Hammerman MR. Normalization of glucose post-transplantation of pig pancreatic anlagen into non-immunosuppressed diabetic rats depends on obtaining anlagen prior to embryonic day 35. Transpl Immunol. 2005;14:67–75. doi: 10.1016/j.trim.2005.02.004. [DOI] [PubMed] [Google Scholar]
- 7.Rogers SA, Chen F, Talcott M, Liapis H, Hammerman MR. Glucose tolerance normalization following transplantation of pig pancreatic primordia into non-immunosuppressed diabetic ZDF rats. Transpl Immunol. 2006;16:176–184. doi: 10.1016/j.trim.2006.08.007. [DOI] [PubMed] [Google Scholar]
- 8.Rogers SA, Mohanakumar T, Liapis H, Hammerman MR. Engraftment of cells from porcine islets of Langerhans and normalization of glucose tolerance following transplantation of pig pancreatic primordia in non-immunosuppressed diabetic rats. Am J Pathology. 2010;177:854–864. doi: 10.2353/ajpath.2010.091193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rogers SA, Chen F, Talcott MR, Faulkner C, Thomas JM, Thevis M, et al. Long-term engraftment following transplantation of pig pancreatic primordia into non-immunosuppressed diabetic rhesus macaques. Xenotransplantation. 2007;14:591–602. doi: 10.1111/j.1399-3089.2007.00429.x. [DOI] [PubMed] [Google Scholar]
- 10.Hammerman MR. Organogenetic tolerance. Organogenesis. 2010;6:270–275. doi: 10.4161/org.6.4.13283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hecht G, Eventov-Friedman S, Rosen C, Shezen E, Tchorsh D, Aronovich A, et al. Embryonic pig pancreatic tissue for the treatment of diabetes in a nonhuman primate model. Proc Natl Acad Sci USA. 2009;106:8659–8664. doi: 10.1073/pnas.0812253106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Eventov-Friedman S, Katchman H, Shezen E, Arnovich A, Tchorsh D, Dekel B, et al. Embryonic pig liver, pancreas and lung as a source for transplantation: Optimal organogenesis without teratoma depends on distinct time windows. Proc Natl Acad Sci USA. 2005;102:2928–2933. doi: 10.1073/pnas.0500177102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Eventov-Friedman S, Tchorsh D, Katchman H, Shezan E, Arnovich A, Hecht G, et al. Embryonic pig pancreatic tissue transplantation for the treatment of diabetes. PLoS Medicine. 2006;7:1165–1177. doi: 10.1371/journal.pmed.0030215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tchorsh-Yutsis D, Hecht G, Aronovich A, Shezen E, Klionsky Y, Rosen C, et al. Pig embryonic pancreatic tissue as a source for transplantation in diabetes: transient treatment with anti-LFA1, anti-CD48 and FTY720 enables long-term graft maintenance in mice with only mild ongoing immunosuppression. Diabetes. 2009;58:1585–1594. doi: 10.2337/db09-0112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dirk J, van der Windt DJ, Echeverri GJ, Ijzermans JNM, Cooper DCK. The choice of anatomical site for islet transplantation. Cell Transplant. 2008;17:1005–1014. doi: 10.3727/096368908786991515. [DOI] [PubMed] [Google Scholar]
- 16.Eloy R, Haffen K, Kedinger M, Griener JF. Chick embryo pancreatic transplants reverse experimental diabetes of rats. J Clin Invest. 1979;64:361–373. doi: 10.1172/JCI109470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Abraham EJ, Kodama S, Lin JC, Ubeda M, Raustman DL, Habener JF. Human pancreatic islet-derived progenitor cell engraftment in immunocompetent mice. Am J Pathology. 2004;164:817–830. doi: 10.1016/S0002-9440(10)63170-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Harada M, Matsunaga K, Oguchi Y, Iijima O, Ito O, Tamada K, et al. The involvement of transforming growth factor β in the impaired antitumor T-cell response at the gut-associated lymphoid tissue (GALT) Cancer Res. 1995;55:6146–6151. [PubMed] [Google Scholar]
- 19.Dufrane D, Goebbels RM, Saliez A, Guiot Y, Gianello P. Six-month survival of microencapsulated pig islets and alginate biocompatibility in primates: proof of concept. Transplantation. 2006;81:1345–1353. doi: 10.1097/01.tp.0000208610.75997.20. [DOI] [PubMed] [Google Scholar]
- 20.Kirchhof N, Shibata S, Wijkstrom M, Kulick DM, Salerno CT, Clemmings SM, et al. Reversal of diabetes in non-immunosuppressed rhesus macaques by intraportal porcine islet xenografts precedes acute cellular rejection. Xenotransplantation. 2004;11:396–407. doi: 10.1111/j.1399-3089.2004.00157.x. [DOI] [PubMed] [Google Scholar]
- 21.Cantarovich D, Blancho G, Potiron N, Jugeau N, Fiche M, Chagneau C, et al. Rapid failure of pig islet transplantation in non human primates. Xenotransplantation. 2002;9:25–35. doi: 10.1034/j.1399-3089.2002.0o144.x. [DOI] [PubMed] [Google Scholar]