Abstract
The authors hypothesized that the absence of cross-sectional associations of body mass index (BMI; weight (kg)/height (m)2) with peripheral arterial disease (PAD) in prior studies may reflect lower weight among persons who smoke or have poor health status. They conducted an observational study among 5,419 noninstitutionalized residents of 4 US communities aged ≥65 years at baseline (1989–1990 or 1992–1993). Ankle brachial index was measured, and participants reported their history of PAD procedures. Participants were followed longitudinally for adjudicated incident PAD events. At baseline, mean BMI was 26.6 (standard deviation, 4.6), and 776 participants (14%) had prevalent PAD. During 13.2 (median) years of follow-up through June 30, 2007, 276 incident PAD events occurred. In cross-sectional analysis, each 5-unit increase in BMI was inversely associated with PAD (prevalence ratio (PR) = 0.92, 95% confidence interval (CI): 0.85, 1.00). However, among persons in good health who had never smoked, the direction of association was opposite (PR = 1.20, 95% CI: 0.94, 1.52). Similar results were observed between BMI calculated using weight at age 50 years and PAD prevalence (PR = 1.30, 95% CI: 1.11, 1.51) and between BMI at baseline and incident PAD events occurring during follow-up (hazard ratio = 1.32, 95% CI: 1.00, 1.76) among never smokers in good health. Greater BMI is associated with PAD in older persons who remain healthy and have never smoked. Normal weight maintenance may decrease PAD incidence and associated comorbidity in older age.
Keywords: ankle brachial index, body mass index, cardiovascular diseases, peripheral arterial disease
Peripheral arterial disease (PAD) is estimated to affect 15% of the US population, and prevalence increases sharply with age (1). Beyond its associations with cardiovascular disease (CVD) events and all-cause mortality (2), PAD is also strongly associated with functional limitations (3) and poor quality of life (4)—factors that will be of greater importance to public health as the population ages.
Because PAD represents a peripheral manifestation of atherosclerosis, most traditional and novel CVD risk factors are strongly associated with this condition (5–8). One important exception, however, is body mass index (BMI). Most large-scale epidemiologic studies, but not all (9, 10), have observed either no association (6, 8, 11–16) or an inverse association (17–21) between BMI and PAD prevalence. Most of these studies evaluated older persons. Mechanisms underlying the surprising lack of, or even inverse, association are uncertain.
We hypothesized that smoking and poor health status might simultaneously be associated with lower BMI and greater PAD prevalence, thereby obscuring a positive association that might exist if adiposity itself directly or indirectly fosters the development of PAD. Thus, we evaluated the association of BMI with PAD prevalence cross-sectionally among community-living adults aged ≥65 years at baseline who were participating in the Cardiovascular Heath Study. We also evaluated the association of BMI with subsequent incident clinical PAD events occurring during follow-up. Lastly, to preliminarily evaluate whether results may be similar for BMI in middle age, we used self-reported recalled weight at age 50 years to estimate midlife BMI and evaluate its association with PAD prevalence at the baseline examination. We repeated the analyses in a prespecified subset of participants who were never smokers and reported good health status at the baseline examination, to eliminate potential confounding by these factors.
MATERIALS AND METHODS
Participants
The Cardiovascular Health Study is a community-based study of older adults, designed to evaluate risk factors for development and progression of CVD. The study design and protocols have been described previously (22, 23). In brief, eligibility required age ≥65 years, an expectation to remain in the area for 3 years after recruitment, no active cancer treatment, and the ability to provide consent. Between 1989 and 1990, 5,201 participants were recruited from 4 US communities (Sacramento, California; Forsyth County, North Carolina; Washington County, Maryland; and Allegheny County, Pennsylvania). An additional group of 687 African-American participants was recruited in 1992–1993. Participants were sampled from Medicare eligibility lists in each area.
For this analysis, the first visit for each participant (1989–1990 or 1992–1993) was considered the baseline visit. We excluded persons with missing data on baseline ankle brachial index (ABI) (n = 140; 2.4%), BMI (n = 17; 0.3%), weight at age 50 years (n = 228; 3.9%), or covariates (n = 15; 0.3%). In addition, we excluded persons with ABI measurements greater than 1.40 (n = 69; 1.2%), as such persons frequently have medial arterial calcification, which produces artificially high ABI measurements. After these exclusions, 5,419 persons (92%) were included in this analysis. All participants provided written informed consent, and the study was approved by the investigational review boards at the 4 clinical sites, the Data Coordinating Center at the University of Washington, and the University of California, San Diego.
Measurements
Anthropometric data and BMI.
Anthropometric measurements were performed by trained personnel using standardized protocols. During measurement, participants wore examination suits and no shoes. Standing height was measured using a stadiometer calibrated in centimeters. Weight was measured using a balance-beam scale calibrated in kilograms. BMI was calculated as weight in kilograms divided by height in meters squared. BMI at age 50 years was estimated using self-reported weight at age 50 and measured height at the baseline study visit.
ABI and PAD.
The ABI protocol has been described previously (24). Briefly, after at least 5 minutes of rest and with the subject in the supine position, a standard mercury sphygmomanometer and a Doppler stethoscope (8 MHz; Huntleigh Technology, Inc., Luton, United Kingdom) were used to determine systolic blood pressures in the right brachial artery and the right and left leg posterior tibial arteries. Duplicate measurements were obtained and averaged. When blood pressure could not be taken on the right arm, the left arm was used. The ratio of systolic blood pressure in the leg to systolic blood pressure in the arm was used to define the leg-specific ABI. The lower of the leg-specific ABIs was used as the patient-specific ABI for this analysis.
Prevalent clinical PAD was defined as either an ABI less than 0.90 at the baseline examination or both exertional leg pain relieved by rest and a physician's diagnosis of PAD. In addition, any of the following were considered sufficient to validate a PAD diagnosis, with evidence that the test was prompted by the participant's complaint of leg pain: Doppler ultrasound showing an obstruction of ≥75% of the cross-sectional area of the artery or showing an ulcerated plaque; angiography showing ≥50% obstruction of the diameter or ≥75% of the artery cross-sectional area or an ulcerated plaque; absence of a Doppler pulse in any major vessel; a positive exercise test for claudication; or bypass surgery, angioplasty, amputation, or thrombolysis for the indication of PAD.
During follow-up through June 30, 2007, potential incident clinical PAD events were identified by participant report at the annual clinic visits and in 6-month telephone calls, during medical record review of other events, or as part of the regular review of Centers for Medicare and Medicaid Services records (identified on the basis of International Classification of Diseases, Ninth Revision, diagnostic codes 440.2 (atherosclerosis of the native arteries of the extremities) and 443.9 (peripheral vascular disease, unspecified)). Medical records pertaining to each potential PAD event were obtained and reviewed by physicians using the prespecified criteria described above and were adjudicated by the Morbidity Subgroups of the Cardiovascular Health Study Clinical Events Subcommittee.
Other measurements.
Age, sex, and race were determined by self-report. Participants were asked to rate their health status on a 5-point scale (excellent, very good, good, fair, or poor). This variable has been demonstrated to be strongly associated with subsequent mortality in the Cardiovascular Health Study (25). Smoking history was determined by questionnaire and was categorized as current smoker, former smoker, or never smoker. Among past or former smokers, the number of cigarettes smoked per day was ascertained by questionnaire, and pack-years of smoking were calculated. Participants were asked, “In the last year, have you gained or lost more than 10 pounds?”, “Was diet for the purposes of gaining or losing weight a major factor in your weight change?”, and “Was exercise a major factor in your weight change?” Participants who responded that they had lost more than 10 pounds (>4.5 kg) in the last year that was not the result of diet, exercise, or both were classified as having unintentional weight loss (26).
Statistical analysis
We began by summarizing the distribution means and percent prevalences of major demographic variables and CVD risk factors among participants with and without prevalent PAD and with and without obesity (defined as BMI ≥30) (27) at the baseline examination. In exploratory analysis, we evaluated associations of BMI with PAD prevalence and incidence separately in men and women and also evaluated multiplicative interaction terms. In all cases, results showed similar functional form, and no evidence of effect modification was observed (all P’s for interaction ≥ 0.19). Thus, we evaluated both sexes together to maximize statistical power. To allow for nonlinear relations, we categorized BMI in sex-specific quartiles with the lowest quartile serving as the reference category. When quartile analyses demonstrated a fairly linear functional form, we repeated the analyses modeling BMI on a continuous scale to maximize statistical power.
For evaluation of prevalent PAD, we used general linear models with a log link, assuming a binomial distribution. Robust standard error estimates were used. For evaluation of incident clinical PAD events, we used Cox proportional hazards models. Log-minus-log plots and plots of Schoenfeld residuals versus survival time were used to evaluate proportional hazards assumptions. When violations were observed, we stratified by follow-up time and present the results separately by time period.
Sequential models were developed. An initial model adjusted for age, sex, race, and field center. A subsequent model additionally adjusted for smoking status (current, former, or never smoker), pack-years of smoking, and self-reported health status (excellent, very good, good, fair, or poor). In companion analyses, we also evaluated a prespecified subgroup of persons who were never smokers, reported good, very good, or excellent health, and did not report unintentional weight loss in the year preceding the baseline visit (hereafter referred to as persons with “good health status”). We elected not to statistically test for an interaction in this subgroup compared with the remainder of participants. We stratified the data to decrease residual confounding by reported health status and smoking history, not because we believe adiposity is unimportant in the development of PAD among persons with poor health or tobacco use. We also elected not to adjust for other CVD risk factors—such as blood pressure, diabetes, inflammatory cytokines, and lipid levels—because at least some of these factors, and potentially all of them, may lie within the causal pathway between BMI and PAD, and thus adjustment may obscure relations of BMI with PAD. Lastly, to provide insight into the differences between results from the prevalent PAD analyses and the incident PAD analyses, we evaluated the association of unintentional weight loss with PAD prevalence at the baseline examination and the change in BMI from age 50 years to baseline, adjusting for age, sex, race, and field center. Stata, version 10.1 (StataCorp LP, College Station, Texas), was used for all analyses.
RESULTS
Among the 5,419 study participants, the mean age at the baseline examination was 72.7 years (standard deviation (SD), 5.5). A total of 830 participants (15%) were African-American. The mean BMI was 26.6 (SD, 4.6), and 1,039 participants (19%) were obese (BMI ≥30). We classified 776 participants (14%) as having prevalent PAD at baseline—643 on the basis of ABI measurements less than 0.90, 46 on the basis of confirmed clinical PAD, and an additional 87 participants who had both confirmed clinical PAD and an ABI less than 0.90.
Table 1 shows the distribution of demographic variables and traditional CVD risk factors by obesity and PAD status at the baseline visit. Compared with nonobese participants, those who were obese were younger, were more frequently female and African-American, were less likely to smoke, and had poorer self-reported health status. With the exception of smoking, obese participants had higher prevalences of all traditional CVD risk factors. Compared with subjects without prevalent PAD, those with PAD were older, were more frequently male and African-American, and had higher prevalences of all traditional CVD risk factors except BMI and diastolic blood pressure.
Table 1.
Distribution of Demographic Factors and Traditional Cardiovascular Disease Risk Factors According to Obesity Status and Peripheral Arterial Disease at Baseline, Cardiovascular Health Study, 1989–2007
| Obesity (Body Mass Indexa ≥30) |
Prevalent PADb |
|||||||||||
| No |
Yes |
No |
Yes |
|||||||||
| No. | % | Mean (SD) | No. | % | Mean (SD) | No. | % | Mean (SD) | No. | % | Mean (SD) | |
| No. and % of participants | 4,380 | 81 | 1,039 | 19 | 4,643 | 86 | 776 | 14 | ||||
| Age, years | 73 (5.6) | 71.6 (4.9) | 72.3 (5.2) | 75.6 (6.3) | ||||||||
| Female sex | 2,423 | 55 | 691 | 67 | 2,714 | 58 | 400 | 52 | ||||
| African-American race | 572 | 13 | 258 | 25 | 647 | 14 | 183 | 24 | ||||
| Smoking status | ||||||||||||
| Never smoker | 1,977 | 45 | 527 | 51 | 2,212 | 48 | 292 | 38 | ||||
| Current smoker | 562 | 13 | 82 | 8 | 494 | 11 | 150 | 19 | ||||
| Former smoker | 1,841 | 42 | 430 | 41 | 1,937 | 42 | 334 | 43 | ||||
| Unintentional weight loss of >10 pounds (>4.5 kg) | 291 | 7 | 41 | 4 | 253 | 6 | 79 | 11 | ||||
| Prevalent cardiovascular disease | 962 | 22 | 241 | 23 | 898 | 19 | 305 | 39 | ||||
| Fair or poor self-reported health status | 1,003 | 23 | 331 | 32 | 1,026 | 22 | 308 | 40 | ||||
| Hypertension | 2,439 | 56 | 734 | 71 | 2,593 | 56 | 580 | 75 | ||||
| Diabetes | 570 | 13 | 286 | 28 | 663 | 14 | 193 | 25 | ||||
| Body mass index | 24.9 (2.9) | 33.8 (3.5) | 26.7 (4.7) | 25.9 (4.4) | ||||||||
| Systolic blood pressure, mm Hg | 135.8 (22.0) | 138.5 (20.5) | 134.9 (21.1) | 145.2 (23.1) | ||||||||
| Diastolic blood pressure, mm Hg | 70.3 (11.5) | 72.6 (10.9) | 70.8 (11.2) | 70.5 (12.6) | ||||||||
| Total cholesterol, mg/dL | 211.1 (39.2) | 213.0 (39.4) | 210.8 (38.5) | 215.7 (43.4) | ||||||||
| High density lipoprotein cholesterol, mg/dL | 55.2 (16.1) | 50.5 (13.3) | 54.7 (15.6) | 51.4 (15.7) | ||||||||
Abbreviations: PAD, peripheral arterial disease; SD, standard deviation.
Weight (kg)/height (m)2.
Includes participants with an ankle brachial index less than 0.90 (n = 643) or a history of PAD at baseline (n = 46) or both (n = 87).
BMI and PAD prevalence at baseline
Table 2 shows the cross-sectional associations of BMI with prevalent PAD at the baseline examination. The crude prevalence of PAD was lower among persons with higher BMI. In models adjusting for demographic variables, smoking, and self-reported health status, each 5-unit increase in BMI was associated with a 9% lower PAD prevalence. However, the direction of this association differed in persons who had never smoked and reported good health status, among whom those with a higher BMI trended towards a higher PAD prevalence, although the latter association was not statistically significant.
Table 2.
Cross-Sectional Association of Body Mass Index With Prevalent Peripheral Arterial Disease at the Baseline Visit, Cardiovascular Health Study, 1989–2007
| BMIa Measure | Sex and BMI Category |
All Participants |
Never Smokers in Good Healthb |
|||||||||
| PAD Prevalence |
Demographic Factor-Adjustedc |
Multivariable-Adjustedd |
PAD Prevalence |
Demographic Factor-Adjustedc |
||||||||
| Women (n = 3,114) | Men (n = 2,305) | % | No. With PAD/Total | PR | 95% CI | PR | 95% CI | % | No. With PAD/Total | PR | 95% CI | |
| Quartile | ||||||||||||
| 1 | <23.1 | <23.9 | 17 | 233/1,356 | 1 | Reference | 1 | Reference | 10 | 45/464 | 1 | Reference |
| 2 | 23.1–25.9 | 23.9–26.0 | 14 | 196/1,354 | 0.97 | 0.81, 1.18 | 0.91 | 0.75, 1.09 | 8 | 37/449 | 1.43 | 0.76, 2.69 |
| 3 | 26.0–29.4 | 26.1–28.3 | 14 | 190/1,355 | 0.94 | 0.78, 1.14 | 1.00 | 0.82, 1.21 | 8 | 41/488 | 1.58 | 0.84, 2.97 |
| 4 | ≥29.5 | ≥28.4 | 12 | 157/1,354 | 0.80 | 0.64, 1.01 | 0.81 | 0.66, 1.01 | 8 | 34/437 | 1.70 | 0.79, 3.67 |
| Continuous (per 5-unit increase) | 0.91 | 0.84, 0.99 | 0.92 | 0.85, 1.00 | 1.20 | 0.94, 1.52 | ||||||
Abbreviations: BMI, body mass index; CI, confidence interval; PAD, peripheral arterial disease; PR, prevalence ratio.
Weight (kg)/height (m)2.
Limited to persons who had never smoked, reported good, very good, or excellent health, and reported no unintentional weight loss of >10 pounds (>4.5 kg).
Adjusted for age, sex, race, and field center.
Adjusted for age, sex, race, field center, smoking status (current, former, or never smoker), pack-years of smoking, and self-reported health status (excellent, very good, good, fair, or poor).
BMI at age 50 years and PAD prevalence at baseline
The mean estimated BMI at age 50 years was 25.5 (SD, 3.8). In the full study sample, PAD prevalences at the baseline examination were similar across quartiles of BMI assessed at age 50 (Table 3). However each 5-unit increase in BMI at age 50 was associated with approximately 30% greater PAD prevalence among persons who had never smoked and reported good health status at the baseline examination (P < 0.001).
Table 3.
Association of Body Mass Index at Age 50 Years With Prevalence of Peripheral Arterial Disease at the Baseline Examination, Cardiovascular Health Study, 1989–2007
| BMIa Measure | Sex and BMI Category |
All Participants |
Never Smokers in Good Healthb |
|||||||||
| PAD Prevalence |
Demographic Factor-Adjustedc |
Multivariable-Adjustedd |
PAD Prevalence |
Demographic Factor-Adjustedc |
||||||||
| Women (n = 3,114) | Men (n = 2,305) | % | No. With PAD/Total | PR | 95% CI | PR | 95% CI | % | No. With PAD/Total | PR | 95% CI | |
| Quartile | ||||||||||||
| 1 | <22.3 | <23.9 | 14 | 192/1,358 | 1 | Reference | 1 | Reference | 8 | 36/440 | 1 | Reference |
| 2 | 22.3–24.3 | 23.9–25.6 | 13 | 170/1,353 | 1.01 | 0.81, 1.25 | 0.97 | 0.80, 1.19 | 6 | 30/479 | 1.10 | 0.57, 2.13 |
| 3 | 24.4–26.9 | 25.7–27.7 | 15 | 201/1,354 | 1.13 | 0.93, 1.38 | 1.05 | 0.86, 1.27 | 10 | 50/489 | 1.72 | 0.95, 3.12 |
| 4 | ≥27.0 | ≥27.8 | 16 | 213/1,354 | 1.15 | 0.94, 1.40 | 1.04 | 0.86, 1.26 | 10 | 41/430 | 1.74 | 0.97, 3.14 |
| Continuous (per 5-unit increase) | 1.05 | 0.97, 1.14 | 1.00 | 0.92, 1.09 | 1.30 | 1.11, 1.51 | ||||||
Abbreviations: BMI, body mass index; CI, confidence interval; PAD, peripheral arterial disease; PR, prevalence ratio.
Weight (kg)/height (m)2.
Limited to persons who had never smoked, reported good, very good, or excellent health, and reported no unintentional weight loss of >10 pounds (>4.5 kg).
Adjusted for age, sex, race, and field center.
Adjusted for age, sex, race, field center, smoking status (current, former, or never smoker), pack-years of smoking, and self-reported health status (excellent, very good, good, fair, or poor).
BMI and incident clinical PAD events
Among the 4,643 participants without prevalent PAD at baseline, and during a median follow-up period of 13.2 years (interquartile range, 7.7–17.2), there were 276 adjudicated incident clinical PAD events. In the full study sample, we observed no association of BMI with incident clinical PAD events in either demographic factor-adjusted models or fully adjusted models. However, among persons who had never smoked and reported good health, each 5-unit increase in BMI was again associated with an approximately 30% greater risk of incident clinical PAD events (Table 4).
Table 4.
Association of Body Mass Index at the Baseline Examination With Incident Clinical Peripheral Arterial Disease Events, Cardiovascular Health Study, 1989–2007
| BMIa Measure | Sex and BMI Category |
All Participants |
Never Smokers in Good Healthb |
|||||||||
| Demographic Factor-Adjustedc |
Multivariable-Adjustedd |
PAD Prevalence |
Demographic Factor-Adjustedc |
|||||||||
| Women (n = 3,114) | Men (n = 2,305) | No. of Incident PAD Events | Person-Years of Follow-up | HR | 95% CI | HR | 95% CI | No. of Incident PAD Events | Person-Years of Follow-up | HR | 95% CI | |
| Quartile | ||||||||||||
| 1 | <23.1 | <23.9 | 67 | 14,784 | 1 | Reference | 1 | Reference | 9 | 5,699 | 1 | Reference |
| 2 | 23.1–25.9 | 23.9–26.0 | 67 | 15,885 | 0.96 | 0.68, 1.34 | 1.03 | 0.73, 1.46 | 14 | 5,951 | 1.62 | 0.70, 3.76 |
| 3 | 26.0–29.4 | 26.1–28.3 | 69 | 16,298 | 0.96 | 0.68, 1.34 | 1.12 | 0.79, 1.59 | 19 | 6,630 | 1.83 | 0.82, 4.09 |
| 4 | ≥29.5 | ≥28.4 | 73 | 15,990 | 1.02 | 0.73, 1.43 | 1.10 | 0.78, 1.57 | 20 | 5,698 | 2.21 | 0.98, 4.98 |
| Continuous (per 5-unit increase) | 1.02 | 0.89, 1.16 | 1.05 | 0.92, 1.20 | 1.32 | 1.00, 1.76 | ||||||
Abbreviations: BMI, body mass index; CI, confidence interval; HR, hazard ratio; PAD, peripheral arterial disease.
Weight (kg)/height (m)2.
Limited to persons who had never smoked, reported good, very good, or excellent health, and reported no unintentional weight loss of >10 pounds (>4.5 kg).
Adjusted for age, sex, race, and field center.
Adjusted for age, sex, race, field center, smoking status (current, former, or never smoker), pack-years of smoking, and self-reported health status (excellent, very good, good, fair, or poor).
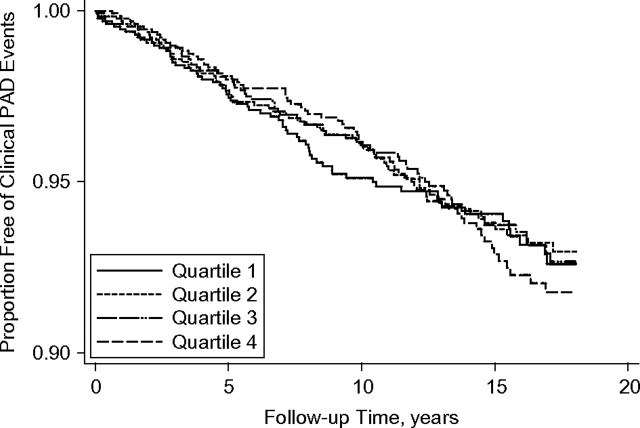
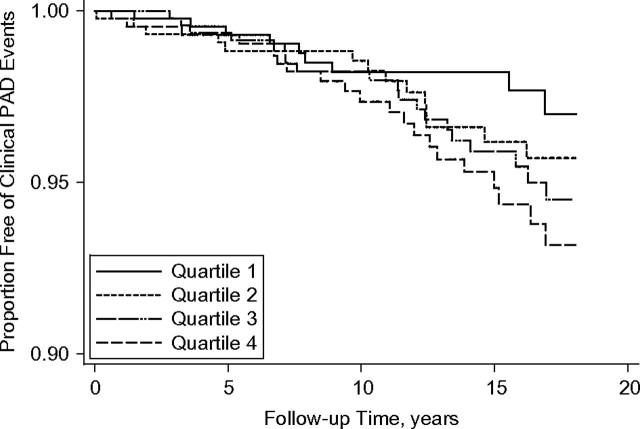
Kaplan-Meier analysis demonstrated that associations differed during the follow-up period. Specifically, in the entire study sample, the association of BMI with clinical PAD events appeared null throughout the available follow-up time (Figure 1). However, in the subset of healthy participants who had never smoked, there was no discernable association of BMI with incident clinical PAD events during approximately the first 8 years of follow-up, whereas thereafter persons in the higher BMI quartiles had a higher risk of PAD events (Figure 2).
Figure 1.
Kaplan-Meier curve for time to first clinical peripheral arterial disease (PAD) event by sex-specific quartile of body mass index (weight (kg)/height (m)2), Cardiovascular Health Study, 1989–2007. For women, body mass index quartiles 1–4 were defined as <23.1, 23.1–25.9, 26.0–29.4, and ≥29.5; for men, they were defined as <23.9, 23.9–26.0, 26.1–28.3, and ≥28.4.
Figure 2.
Kaplan-Meier curve for time to first clinical peripheral arterial disease (PAD) event by sex-specific quartile of body mass index (weight (kg)/height (m)2) in participants who had never smoked and reported good health status, Cardiovascular Health Study, 1989–2007. For women, body mass index quartiles 1–4 were defined as <23.1, 23.1–25.9, 26.0–29.4, and ≥29.5; for men, they were defined as <23.9, 23.9–26.0, 26.1–28.3, and ≥28.4.
Weight change and PAD
We also evaluated weight change in persons with prevalent PAD. On average, subjects reported a gain in BMI (1.1 units (SD, 3.5)) from age 50 years to the baseline examination. However, subjects with prevalent PAD reported a 0.7-unit lower weight gain than those without prevalent PAD in models adjusted for age, sex, race, and field center (P < 0.001). Similarly, in models adjusted for the same covariates, persons reporting unintentional weight loss of >10 pounds in the year preceding the baseline visit were 40% more likely to have prevalent PAD (prevalence ratio = 1.40, 95% confidence interval: 1.15, 1.72).
DISCUSSION
In a large sample of community-living older adults, we observed no associations of BMI with PAD prevalence or incidence when all participants were evaluated together. However, among persons who had good health status and had never smoked, we observed positive associations of BMI with PAD prevalence and incidence. In addition, persons who had prevalent PAD were less likely to have gained weight since age 50 years and more likely to report unintentional weight loss. Collectively, these findings demonstrate that smoking, poor health status, weight loss, and PAD often coexist in older persons and complicate the evaluation of associations of BMI with PAD. When evaluating persons without these confounding factors, we found that greater adiposity was associated with subsequent PAD, even in older age.
Most prior studies evaluating the association of BMI with PAD evaluated older populations and used cross-sectional study designs. These studies have shown inverse, U-shaped, or altogether absent associations of BMI with PAD prevalence (6, 8, 11–20). Few studies have evaluated the association of BMI with PAD incidence (16), and none to our knowledge have evaluated the associations separately by health and smoking status. Our findings were similar to those in other studies when we evaluated all participants together. However, we consistently observed positive associations of BMI with PAD in the prespecified healthier, nonsmoking subgroup.
In the context of these findings, it is noteworthy that the few studies that have observed positive associations of adiposity with PAD have evaluated middle-aged populations, in which the degree of comorbidity and lifetime smoking exposure are expected to be lower. Bowlin et al. (10) reported that each 5-unit increase in BMI was associated with 24% greater odds of intermittent claudication among Israeli men aged 40–65 years, an association that remained statistically significant after adjustment for smoking status and pack-years (10). Among 1,170 Brazilians (mean age approximately 45 years), greater BMI was associated with PAD prevalence, but adjustment for smoking status was not reported (9). Results from middle-aged US populations are more difficult to interpret. In the Framingham Offspring Study (mean age 59 years), each 5-unit increase in BMI was associated with 40% greater odds of PAD prevalence in age- and sex-adjusted models, but the association lost statistical significance when results were simultaneously adjusted for smoking status, pack-years, and other traditional CVD risk factors (11). Similarly, in the Atherosclerosis Risk in Communities Study (mean age 45 years), greater waist:hip ratio was associated with incident PAD in models adjusting for age, sex, race, and field center; however, the association lost statistical significance when results were adjusted simultaneously for smoking and traditional CVD risk factors (28). Thus, in these studies, smoking and other traditional CVD risk factors were entered into the models simultaneously. Unlike smoking, hypertension, hyperlipidemia, insulin resistance/diabetes, and inflammation may be consequences of adiposity and may lie on the causal pathway between adiposity and PAD. Future studies that evaluated never smokers separately would be useful to evaluate the potential relation of adiposity with PAD in midlife.
Persons in our study who had prevalent PAD were less likely to gain weight and more likely to report unintentional weight loss. The association of BMI with incident clinical PAD events was not evident during approximately the first 8 years of follow-up, even in healthy never smokers. It is possible that some persons with a greater burden of disease (even if subclinical) may have lost weight and had a lower BMI at the time of the baseline examination. If so, this may have obscured the association of BMI with short-term PAD incidence, since reverse causality may have been present; PAD and/or associated illness might be leading to weight loss. Greater BMI was more strongly associated with longer-term clinical PAD risk within the healthy never smoker subgroup. Among these participants, BMI may have been measured before they developed illnesses and associated weight loss. Future studies with frequent measurement of BMI and comorbidity are required in order to confirm these hypotheses.
Strengths of this study include the large sample size; the extended follow-up time, which provided sufficient statistical power for subgroup analysis; the availability of adjudicated clinical PAD events; the evaluation of functional form; and the representation of both sexes. The study also had important limitations. Information on weight at age 50 years was gathered by self-report and was combined with height at the baseline study visit to estimate BMI in midlife. The relation of self-reported weight to true weight at age 50 years in the Cardiovascular Health Study is unknown. Prior reports from other studies have questioned the accuracy of weight recall (29). However, the associations of BMI at age 50 years with PAD prevalence were consistent with findings obtained in several other studies of middle-aged cohorts. The ABI protocol used the right arm systolic blood pressure only, and persons with PAD are more likely to have subclavian artery stenosis (30). Any bias due to the use of this method should have lowered the ABI in persons with subclavian stensosis and therefore would likely have biased the results towards the null hypothesis. PAD is strongly associated with mortality (31), and age ≥65 years was an inclusion criterion for this study. Although PAD is less common in younger persons, persons with PAD in midlife may have been less likely to survive to an age allowing study participation or may not have participated because of comorbidity, debility, and/or amputation. We specified subgroup analysis in healthy nonsmokers a priori, yet the number of PAD events in this subgroup, particularly for longitudinal analyses evaluating incident PAD events, was relatively low. Results should be confirmed in future studies. Lastly, all participants were older, community-living, and largely of Caucasian or African-American race. Results may differ in other populations.
In conclusion, in a large sample of community-dwelling older persons who reported good health status and were never smokers, greater BMI in midlife and in older age were associated with PAD prevalence and incidence. Findings of null associations in prior cross-sectional studies may have reflected residual confounding and reverse causality by comorbid medical conditions, poor health status, and smoking and by adjustment for factors on the causal pathway between adiposity and PAD. If our findings are confirmed, preventive strategies focused on maintenance of normal weight may decrease PAD incidence and associated comorbidity in later life.
Acknowledgments
Author affiliations: Nephrology Section, Veterans Affairs San Diego Healthcare System, San Diego, California (Joachim H. Ix); Division of Nephrology, Department of Medicine, School of Medicine, University of California, San Diego, San Diego, California (Joachim H. Ix); Division of Preventive Medicine, Department of Family and Preventive Medicine, School of Medicine, University of California, San Diego, San Diego, California (Joachim H. Ix, Michael H. Criqui); Department of Biostatistics, School of Public Health, University of Washington, Seattle, Washington (Mary L. Biggs); Departments of Medicine and Epidemiology, School of Public Health, University of Washington, Seattle, Washington (Davis S. Siscovick); Departments of Medicine and Public Health, Weill Medical College of Cornell University, New York, New York (Jorge R. Kizer); Division of General Medicine and Primary Care, Beth Israel Deaconess Medical Center and Harvard Medical School, Boston, Massachusetts (Kenneth J. Mukamal); Division of Aging, Brigham and Women's Hospital, Harvard Medical School and Boston VA Healthcare System, Boston, Massachusetts (Luc Djousse); Divisions of Geriatrics and Cardiology, Department of Medicine, School of Medicine, Johns Hopkins University, Baltimore, Maryland (Susan J. Zieman); Division of Nephrology, Kidney Research Institute, Department of Medicine, School of Medicine, University of Washington, Seattle, Washington (Ian H. de Boer); Colorado School of Public Health and Department of Health and Exercise Science, College of Applied Human Sciences, Colorado State University, Fort Collins, Colorado (Tracy L. Nelson); Department of Epidemiology, Graduate School of Public Health, University of Pittsburgh, Pittsburgh, Pennsylvania (Anne B. Newman); and Division of Cardiology, Department of Medicine, School of Medicine, University of California, San Diego, San Diego, California (Michael H. Criqui).
This publication was supported by grant R01 HL094555 from the National Heart, Lung, and Blood Institute (to Drs. Ix, Kizer, Mukamal, Djousse, and Zieman). The Cardiovascular Health Study was supported by contracts N01 HC-85079 through N01HC-85086, N01-HC-35129, N01 HC-15103, N01 HC-55222, N01 HC-75150, N01 HC-54133, and N01-HC-80007 and grant U01 HL080295 from the National Heart, Lung, and Blood Institute, with additional contributions from the National Institute of Neurological Disorders and Stroke. The current study was partly supported by the VA San Diego Healthcare System.
The authors thank Clydene Nee for administrative review and assistance with the manuscript.
Dr. Mary Lou Biggs had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Conflict of interest: none declared.
Glossary
Abbreviations
- ABI
ankle brachial index
- BMI
body mass index
- CVD
cardiovascular disease
- PAD
peripheral arterial disease
- SD
standard deviation
References
- 1.Selvin E, Erlinger TP. Prevalence of and risk factors for peripheral arterial disease in the United States: results from the National Health and Nutrition Examination Survey, 1999–2000. Circulation. 2004;110(6):738–743. doi: 10.1161/01.CIR.0000137913.26087.F0. [DOI] [PubMed] [Google Scholar]
- 2.Fowkes FG, Murray GD, Butcher I, et al. Ankle brachial index combined with Framingham Risk Score to predict cardiovascular events and mortality: a meta-analysis. JAMA. 2008;300(2):197–208. doi: 10.1001/jama.300.2.197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.McDermott MM, Fried L, Simonsick E, et al. Asymptomatic peripheral arterial disease is independently associated with impaired lower extremity functioning: the Women's Health and Aging Study. Circulation. 2000;101(9):1007–1012. doi: 10.1161/01.cir.101.9.1007. [DOI] [PubMed] [Google Scholar]
- 4.Regensteiner JG, Hiatt WR, Coll JR, et al. The impact of peripheral arterial disease on health-related quality of life in the Peripheral Arterial Disease Awareness, Risk, and Treatment: New Resources for Survival (PARTNERS) Program. Vasc Med. 2008;13(1):15–24. doi: 10.1177/1358863X07084911. [DOI] [PubMed] [Google Scholar]
- 5.Criqui MH, Fronek A, Klauber MR, et al. The sensitivity, specificity, and predictive value of traditional clinical evaluation of peripheral arterial disease: results from noninvasive testing in a defined population. Circulation. 1985;71(3):516–522. doi: 10.1161/01.cir.71.3.516. [DOI] [PubMed] [Google Scholar]
- 6.Ix JH, Allison MA, Denenberg JO, et al. Novel cardiovascular risk factors do not completely explain the higher prevalence of peripheral arterial disease among African Americans. The San Diego Population Study. J Am Coll Cardiol. 2008;51(24):2347–2354. doi: 10.1016/j.jacc.2008.03.022. [DOI] [PubMed] [Google Scholar]
- 7.Ix JH, Katz R, De Boer IH, et al. Association of chronic kidney disease with the spectrum of ankle brachial index: the Cardiovascular Health Study. J Am Coll Cardiol. 2009;54(13):1176–1184. doi: 10.1016/j.jacc.2009.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Newman AB, Siscovick DS, Manolio TA, et al. Ankle-arm index as a marker of atherosclerosis in the Cardiovascular Health Study. Cardiovascular Heart Study (CHS) Collaborative Research Group. Circulation. 1993;88(3):837–845. doi: 10.1161/01.cir.88.3.837. [DOI] [PubMed] [Google Scholar]
- 9.Makdisse M, Pereira Ada C, Brasil Dde P, et al. Prevalence and risk factors associated with peripheral arterial disease in the Hearts of Brazil Project. Arq Bras Cardiol. 2008;91(6):370–382. doi: 10.1590/s0066-782x2008001800008. [DOI] [PubMed] [Google Scholar]
- 10.Bowlin SJ, Medalie JH, Flocke SA, et al. Epidemiology of intermittent claudication in middle-aged men. Am J Epidemiol. 1994;140(5):418–430. doi: 10.1093/oxfordjournals.aje.a117264. [DOI] [PubMed] [Google Scholar]
- 11.Murabito JM, Evans JC, Nieto K, et al. Prevalence and clinical correlates of peripheral arterial disease in the Framingham Offspring Study. Am Heart J. 2002;143(6):961–965. doi: 10.1067/mhj.2002.122871. [DOI] [PubMed] [Google Scholar]
- 12.Meijer WT, Grobbee DE, Hunink MG, et al. Determinants of peripheral arterial disease in the elderly: the Rotterdam Study. Arch Intern Med. 2000;160(19):2934–2938. doi: 10.1001/archinte.160.19.2934. [DOI] [PubMed] [Google Scholar]
- 13.Ingolfsson IO, Sigurdsson G, Sigvaldason H, et al. A marked decline in the prevalence and incidence of intermittent claudication in Icelandic men 1968–1986: a strong relationship to smoking and serum cholesterol—the Reykjavik Study. J Clin Epidemiol. 1994;47(11):1237–1243. doi: 10.1016/0895-4356(94)90128-7. [DOI] [PubMed] [Google Scholar]
- 14.Hooi JD, Kester AD, Stoffers HE, et al. Incidence of and risk factors for asymptomatic peripheral arterial occlusive disease: a longitudinal study. Am J Epidemiol. 2001;153(7):666–672. doi: 10.1093/aje/153.7.666. [DOI] [PubMed] [Google Scholar]
- 15.Ness J, Aronow WS, Ahn C. Risk factors for symptomatic peripheral arterial disease in older persons in an academic hospital-based geriatrics practice. J Am Geriatr Soc. 2000;48(3):312–314. doi: 10.1111/j.1532-5415.2000.tb02652.x. [DOI] [PubMed] [Google Scholar]
- 16.Curb JD, Masaki K, Rodriguez BL, et al. Peripheral artery disease and cardiovascular risk factors in the elderly. The Honolulu Heart Program. Arterioscler Thromb Vasc Biol. 1996;16(12):1495–1500. doi: 10.1161/01.atv.16.12.1495. [DOI] [PubMed] [Google Scholar]
- 17.Kannel WB, McGee DL. Update on some epidemiologic features of intermittent claudication: the Framingham Study. J Am Geriatr Soc. 1985;33(1):13–18. doi: 10.1111/j.1532-5415.1985.tb02853.x. [DOI] [PubMed] [Google Scholar]
- 18.Beks PJ, Mackaay AJ, de Neeling JN, et al. Peripheral arterial disease in relation to glycaemic level in an elderly Caucasian population: the Hoorn Study. Diabetologia. 1995;38(1):86–96. doi: 10.1007/BF02369357. [DOI] [PubMed] [Google Scholar]
- 19.Fowkes FG, Housley E, Riemersma RA, et al. Smoking, lipids, glucose intolerance, and blood pressure as risk factors for peripheral atherosclerosis compared with ischemic heart disease in the Edinburgh Artery Study. Am J Epidemiol. 1992;135(4):331–340. doi: 10.1093/oxfordjournals.aje.a116294. [DOI] [PubMed] [Google Scholar]
- 20.Tseng CH. Prevalence and risk factors of peripheral arterial obstructive disease in Taiwanese type 2 diabetic patients. Angiology. 2003;54(3):331–338. doi: 10.1177/000331970305400309. [DOI] [PubMed] [Google Scholar]
- 21.Criqui MH, Vargas V, Denenberg JO, et al. Ethnicity and peripheral arterial disease: the San Diego Population Study. Circulation. 2005;112(17):2703–2707. doi: 10.1161/CIRCULATIONAHA.105.546507. [DOI] [PubMed] [Google Scholar]
- 22.Fried LP, Borhani NO, Enright P, et al. The Cardiovascular Health Study: design and rationale. Ann Epidemiol. 1991;1(3):263–276. doi: 10.1016/1047-2797(91)90005-w. [DOI] [PubMed] [Google Scholar]
- 23.Tell GS, Fried LP, Hermanson B, et al. Recruitment of adults 65 years and older as participants in the Cardiovascular Health Study. Ann Epidemiol. 1993;3(4):358–366. doi: 10.1016/1047-2797(93)90062-9. [DOI] [PubMed] [Google Scholar]
- 24.Newman AB, Shemanski L, Manolio TA, et al. Ankle-arm index as a predictor of cardiovascular disease and mortality in the Cardiovascular Health Study. The Cardiovascular Health Study Group. Arterioscler Thromb Vasc Biol. 1999;19(3):538–545. doi: 10.1161/01.atv.19.3.538. [DOI] [PubMed] [Google Scholar]
- 25.Fried LP, Kronmal RA, Newman AB, et al. Risk factors for 5-year mortality in older adults: the Cardiovascular Health Study. JAMA. 1998;279(8):585–592. doi: 10.1001/jama.279.8.585. [DOI] [PubMed] [Google Scholar]
- 26.Fried LP, Tangen CM, Walston J, et al. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci. 2001;56(3):M146–M156. doi: 10.1093/gerona/56.3.m146. [DOI] [PubMed] [Google Scholar]
- 27.World Health Organization. Obesity: Preventing and Managing the Global Epidemic. (WHO technical report series, no. 894) Geneva, Switzerland: World Health Organization; 2000. [PubMed] [Google Scholar]
- 28.Wattanakit K, Folsom AR, Selvin E, et al. Risk factors for peripheral arterial disease incidence in persons with diabetes: the Atherosclerosis Risk in Communities (ARIC) Study. Atherosclerosis. 2005;180(2):389–397. doi: 10.1016/j.atherosclerosis.2004.11.024. [DOI] [PubMed] [Google Scholar]
- 29.Bayomi DJ, Tate RB. Ability and accuracy of long-term weight recall by elderly males: the Manitoba Follow-up Study. Ann Epidemiol. 2008;18(1):36–42. doi: 10.1016/j.annepidem.2007.06.009. [DOI] [PubMed] [Google Scholar]
- 30.Shadman R, Criqui MH, Bundens WP, et al. Subclavian artery stenosis: prevalence, risk factors, and association with cardiovascular diseases. J Am Coll Cardiol. 2004;44(3):618–623. doi: 10.1016/j.jacc.2004.04.044. [DOI] [PubMed] [Google Scholar]
- 31.Criqui MH, Langer RD, Fronek A, et al. Mortality over a period of 10 years in patients with peripheral arterial disease. N Engl J Med. 1992;326(6):381–386. doi: 10.1056/NEJM199202063260605. [DOI] [PubMed] [Google Scholar]