Abstract
Dietary ω3 fatty acids can modulate substrate availability for cyclooxygenases and lipoxygenases, thus modulating downstream eicosanoid formation. This could be an alternative approach to using NSAIDs and other COX inhibitors for limiting PGE2 synthesis in colon cancer prevention. The aims of this study were to evaluate to what extent cyclooxygenase- and lipoxygenase-derived products could be modulated by dietary fish oil in normal colonic mucosa, and to evaluate the role of COX-1 and COX-2 in formation of these products. Mice (wild-type, COX-1 null, or COX-2 null) were fed a diet supplying a broad mixture of fatty acids present in European/American diets, supplemented with either olive oil (oleate control diet) or menhaden (fish) oil ad libitum for 9–11 wk. Colonic eicosanoid levels were measured by LC-MS/MS, and proliferation was assessed by Ki67 immunohistochemistry. Dietary alteration of colonic arachidonic acid: eicosapentaenoic ratios resulted in large shifts in formation of cyclooxygenase and lipoxygenase metabolites. COX-1 knockout virtually abolished PGE2 formation but interestingly 12-HETE and 15-HETE formation was increased. The large changes in eicosanoid profiles were accompanied by relatively small changes in colonic crypt proliferation, but such changes in eicosanoid formation might have greater biological impact upon carcinogen challenge. These results indicate that in normal colon, inhibition of COX-2 would have little effect on reducing PGE2 levels.
Keywords: colon cancer, fish oil, cyclooxygenase, prostaglandin E2, hydroxyeicosatetraenoic acids, EPA
INTRODUCTION
Prostaglandin E2 (PGE2) has been identified as a key pro-inflammatory signaling molecule involved in the promotion and development of sporadic colorectal cancer [1, 2]. Upregulation of cyclooxygenase-2 (COX-2) and increased PGE2 synthesis are hallmarks of colon cancer progression [1, 3–5]. Relatively less is known about the role of other arachidonic acid (AA) metabolites in colon cancer, but they do play a role in the inflammatory response and in carcinogenesis [6–8]. Despite strong preclinical and clinical data suggesting their potential efficacy as preventive agents against colon cancer, pharmacological agents that reduce colorectal PGE2 concentrations through COX inhibition (NSAIDs) have unacceptable cardiovascular and GI toxicity profiles for daily prescription to otherwise healthy populations [9].
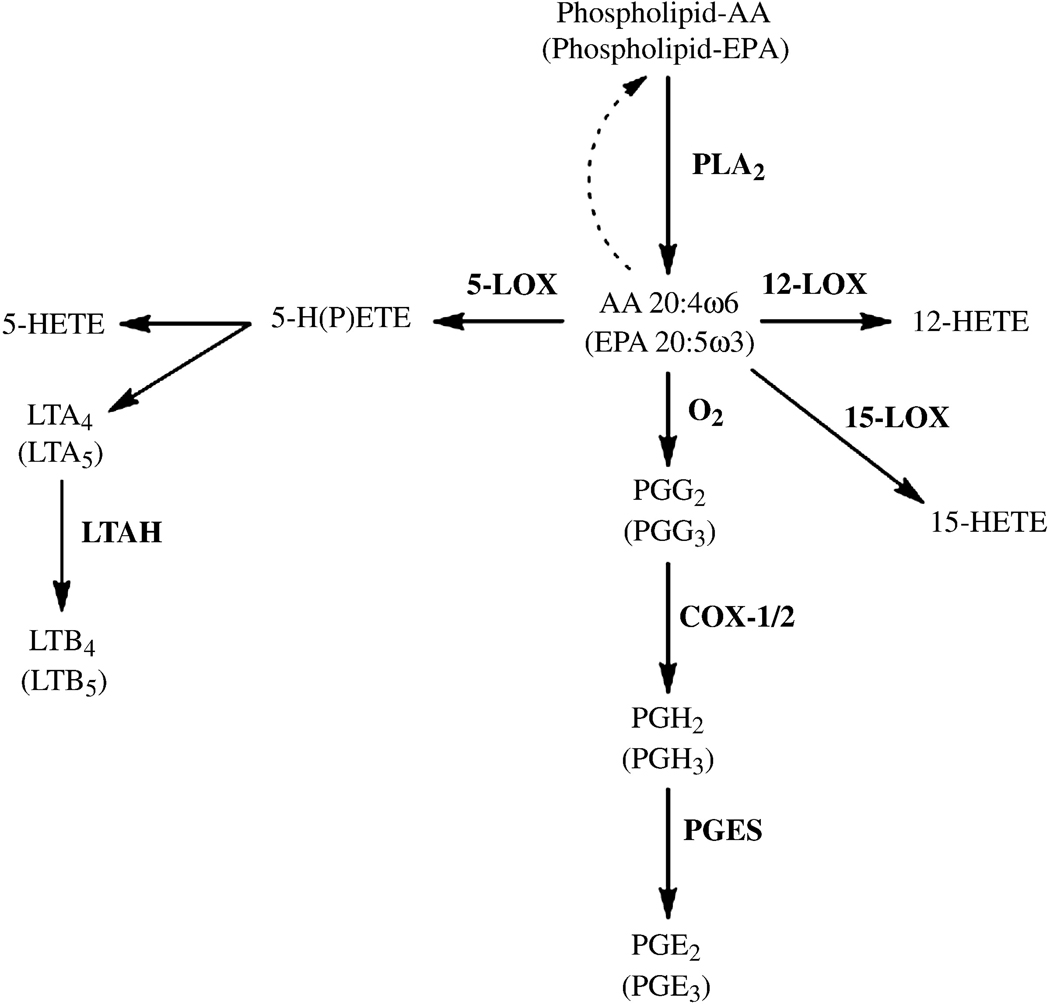
Modification of substrate availability for eicosanoid synthesis is an alternative approach for colon cancer prevention. Dietary ω3 fatty acids can modulate substrate pools available to cyclooxygenases and lipoxygenases, thereby controlling downstream eicosanoid formation and subsequent receptor activation [10] (Fig. 1). The ω3 fatty acid eicosapentaenoic acid (EPA) is a poor substrate for cyclooxygenases and a reasonably good inhibitor of arachidonic acid (ω6) oxygenation [11]. EPA can also serve as a substrate for eicosanoid formation, generating 3-series prostaglandins (PGs), including PGE3, and 5-series leukotrienes (LT) such as LTB5 [12, 13]. The 3-series PG products are generally less pro-inflammatory than the 2-series products [13–15].
Figure 1.
Pathways of eicosanoid biosynthesis from arachidonic and eicosapentaenoic acid precursors. For each arachidonic acid metabolite, the compound in parentheses is the corresponding eicosapentaenoic acid metabolite. Abbreviations: AA, arachidonic acid; COX, cyclooxygenase; PLA2, pholsholipase A2; EPA, eicosapentaenoic acid; HETE, hydroxyeicosatetraenoic acid; H(P)EPE, hydroperoxyeicosapentaenoic acid; H(P)ETE, hydroperoxyeicosatetraenoic acid; LOX, lipoxygenase; LTA, leukotriene A; LTAH, LTA hydrolase; LTB, leukotriene B; PGE, prostaglandin E; PGES, prostaglandin E synthase; PGHS, prostaglandin H synthase.
Typical North American and Western European diets contain high levels of ω6 fatty acids from plant oils. However, diets rich in ω3 fatty acids are associated with reduced risks for colorectal adenocarcinoma as well as other epithelial malignancies [16, 17]. In animal models, data from multiple laboratories demonstrate that there are colon tumor-promoting effects of dietary ω6 fatty acids, saturated fatty acids and Western-style lipid profiles, whereas ω3 fatty acids and fish oil lack colon tumor promoting effects [18, 19]. Also, increasing the ω3:ω6 ratio can reduce local PGE2 concentrations, particularly in the context of a low fat diet when ω6 intakes are low [20]. Supplementation of human diets with fish oil enriched with EPA and docosahexaenoic acid (DHA) reduces PGE2 synthesis and crypt proliferation in human colorectal mucosal [21–24].
Despite experimental evidence of the benefits of shifting the ω3/ω6 balance toward the ω3 fatty acids, little data exists on the resulting alterations to the balance of ω3- versus ω6-derived eicosanoid balance in the colon. The majority of studies have measured PGE2 only. Replacement of corn oil with fish oil in the diets of rats has been shown to reduce colonic PGE2 by 4.5-fold, producing a PGE2/PGE3 ratio of ~2.2:1 [25]. There is relatively less known about the potential impact of fish oil on LT production in the colon, and to what extent ω6 products might be decreased when ω3 products are increased. In addition to the effects of diet, decreased activities of cyclooxygenase-1 and/or -2 might result in shunting of substrate to lipoxygenases.
This study therefore aimed to profile eicosanoid products of arachidonic acid and EPA in histologically normal colon of mice that did or did not carry a genetic deletion of COX-1 or COX-2. The effects of a control diet containing western-blend fat supplemented with oleate were compared with that of a fish oil diet, because modifying substrate pools presented to cyclooxygenase and lipoxygenases should represent a less toxic, physiologic approach for modulating colon cancer risk in normal tissues. The use of mice with genetic knockout of COX-1 or COX-2 also allowed for investigation of possible shunting of the substrate fatty acids to lipoxygenases. We therefore profiled eicosanoid products from arachidonic acid and EPA in histologically normal colon of mice that did or did not carry a genetic deletion of COX-1 or COX-2.
METHODS AND MATERIALS
Animals and Diets
All animal protocols for this experiment were approved by the University Committee on Use and Care of Animals at the University of Michigan. Six-week old female C57BL/6 mice (wild-type, COX-1 null, or COX-2 null, n=20 per group) were a generous gift from Dr. Robert Langenbach (University of North Carolina, Chapel Hill, NC). Mice were fed modified AIN-93G diets (Dyets, Bethlehem, PA) comprised of casein (20% by wt.), cornstarch (33.7%), dyetrose (13.2%), sucrose (10%), cellulose (5%), Western fat mixture (7%), tBHQ (0.0026%), salt mix (3.5%), vitamin mix (1%), L-cysteine (0.3%), choline bitartrate (0.25%). This diet also contained either 6% by wt. olive oil [oleate diet, as olive oil contains large amounts of oleic acid (18:1ω9)] or 6% by wt. menhaden oil [fish oil diet, rich in EPA (20:5ω3) and DHA (22:6ω3)]. The Western fat mixture (7% of the diet by wt.) was comprised of a broad mixture of fatty acids present in European and American diets, composed of coconut oil (45 wt. % supplying saturated fats), olive oil (30 wt. % supplying oleate), corn oil (15 wt. % supplying linoleic acid) and soybean oil (10 wt. % supplying α-linolenic acid). Mice were fed one of these two diets (n=10/genotype/diet) ad libitum for 9–11 wk. See Table 1 for the diet compositions. Mice were maintained on a 12 h light/dark cycle. At the end of the study, animals were sacrificed by isoflurane inhalation and decapitation. Blood was collected from the neck into heparin-containing tubes and the colon was immediately removed and immediately rinsed with ice cold PBS containing indomethacin (5.6 µg/mL). A small portion of the lower colon was removed for histological analysis and the remaining tissue was sliced vertically, and the mucosa scraped off. The mucosa was immediately snap frozen in liquid nitrogen and stored at −80°C prior to processing.
Table 1.
Fatty acid composition of the diets. Mice were fed a modified AIN–93 diet containing 6% Western fat blend and either 6% olive oil (oleate diet, representing a diet high in MUFA) or 6% menhaden oil (high PUFA).
| diet |
|||
|---|---|---|---|
| fatty acida | oleate | fish oil | |
| Composition | stearic acid (18:0) | 2.7 | 3.3 |
| (g/kgdiet) | oleic acid (18:1ω9) | 59.3 | 30.2 |
| linoleic acid (18:2ω6) | 15.9 | 11.3 | |
| linolenic acid (18:3ω3) | 1.4 | 1.5 | |
| AA (20:4ω6) | 0 | 0.7 | |
| EPA (20:5ω3) | 0 | 7.9 | |
| DHA (22:6ω3) | 0 | 5.2 | |
| SFA | 36.2 | 46.2 | |
| MUFA | 62.3 | 34.6 | |
| PUFA | 17.1 | 31.3 | |
| ω3 | 1.1 | 17.5 | |
| ω6 | 16.0 | 12.1 | |
| ω9 | 61.9 | 34.4 | |
| Ratios | ω3/ω6 | 0.1 | 1.4 |
| ω9/ω6 | 3.9 | 2.9 | |
| arachidonic acid/EPA | – | 11.3 | |
| EPA/DHA | – | 1.5 | |
SFA: saturated fatty acids, MUFA: mono-unsaturated fatty acids, PUFA: poly-unsaturated fatty acids
Sections of whole colon tissue were fixed for 24 h in neutral buffered formalin, before brief storage in ethanol. Sections were embedded in paraffin blocks for preparation of slides. Frozen mucosal samples (~140 mg) were pulverized using a Multisample Bio-Pulverizer (Research Products International Corp., Mt. Prospect, IL) that was cooled with dry ice and liquid nitrogen, and homogenates were prepared in 500 µL of cold PBS containing 5.6 µg/mL indomethacin. The suspension was subjected to ultrasonication in ice water for 3 min (20 s sonication, 20 s cooling cycle), further diluted with 500 µL cold PBS/indomethacin, snap frozen, and stored at −80°C prior to analysis of eicosanoids. A small portion (10 µL) of the homogenate was analyzed for protein content using Advanced Protein Assay (Cytoskeleton, Denver, CO).
Western Blotting
Western blotting was performed to confirm COX expression in our genetic models. Mucosa homogenates were centrifuged (13,000 × g), and supernatants were analyzed. Supernatant proteins (10 µg) were separated by SDS–PAGE on 10% Tris–HCl gels (Bio-Rad Laboratories, Hercules, CA), transferred onto PVDF membrane (Millipore Corporation, Billerica, MA) and analyzed by immunoblotting using antibodies against rabbit anti-mouse COX-1 antibody (Cayman Chemical, Ann Arbor, MI) and COX-2 antibody (Novus Biologicals, LLC, Littleton, CO). Antibodies against β-actin (Abcam, San Francisco, CA) were used to control for loading. Antigen–antibody complexes were detected using Western Blotting Lumiglo Reagent (Santa Cruz Biotechnology, Santa Cruz, CA). For COX-2 blotting, a lysate of NIH3T3 mouse fibroblasts expressing COX-2 was included as a positive control. Representative COX-1 and COX-2 Western blots of colonic mucosa from COX-1 null, COX-2 null, and wild-type mice are presented in Fig. 2. COX-1 was absent in the mucosa of COX-1 null mice, but was detected in both wild-type and COX-2 null mice. COX-2 was not detected in the mucosa of any of the genotypes, consistent with the low expression of COX-2 in non-transformed mucosa [1, 26, 27].
Figure 2.
Representative COX-1 and COX-2 Western blots of colonic mucosa from wild-type (WT), COX-1 null, and COX-2 null mice fed the oleate diet. For the COX-2 experiment, a lysate of NIH3T3 mouse fibroblast cells known to express COX-2 were included as a positive control.
Total Fatty Acid Analysis by GC-MS
For extraction of total fatty acids, 10 µL of internal standard (17:0, 1 mg/mL in hexane) was added to 150 µL of mucosal homogenate. The sample was then added to 1.5 mL Folch reagent (chloroform: methanol 2:1), vortexed for 1 minute, and centrifuged (200 × g for 5 min). The organic layer was removed to a 12 × 75 mm glass tube and dried in a SpeedVac. The sample was solubilized in 150 µL of hexane: chloroform (1:1), and vortexed. Fatty acid methyl esters (FAMEs) were prepared by adding 10 µL of METH-PREP II derivatization reagent (0.2N methanolic (m-trifluoromethylphenyl)trimethylammonium hydroxide, Alltech, Deerfield, IL), the sample was vortexed (0.5 min) and held at room temperature for 30 min. The organic layer was then removed for analysis. For quantification, standard curves were prepared using mixtures of fatty acids in varying concentrations derivatized in the same manner as tissue extracts.
GC analysis was performed on an Hewlett Packard 5890 GC with a 5971 MSD (Santa Clara CA) and a SP-2330 capillary column 30 m × 0.32 mm, 0.2 µm film thickness (Supelco, Bellefonte, PA) as previously described [28]. The carrier gas was He with column head pressure of 10 psi. Splitless injection was at 220°C using a total flow rate of 50 mL/min. Temperature ramping was performed from 70– 220°C as described previosuly. Total run time was 21.3 min and single ion monitoring was used that was optimized for each fatty acid.
Eicosanoid Analysis by LC-MS
For extraction of eicosanoids, 350 µL of the homogenate was added to 12 × 75 mm glass tubes on ice, along with 1N citric acid (20 µL) and deuterated internal standards (50 ng/mL PGE2-d4, 200 ng/mL LTB4-d4, 100 ng/ml 15-S-HETE-d8, 100 ng/mL 5-S-HETE-d8, 200 ng/ml 12-S-HETE-d8, 400 ng/ml 13-S-HODE-d4, 5 µg/ml arachidonic acid- d8). The resulting solution was extracted three times with 2 mL hexane: ethyl acetate (1:1 v/v, containing 0.1% BHT w/v and 1mM EDTA) by vortexing (3 min), and centrifugation (2000 × g, 10 min, 4°C). The combined extracts were evaporated and reconstituted with 100 µL cold methanol: 10 mM ammonium acetate buffer, pH 8.5 (60:40 v/v), sonicated (3 min in cold water), and centrifuged (13,000 × g, 3 min, 4°C). The extracts were then transferred to deactivated glass HPLC vials for LC-MS/MS analysis (25 µL injected).
HPLC separations for eicosanoids were performed on a Waters 2695 separations module (Milford, MA), employing a Luna Phenyl-Hexyl analytical column (2 × 150 mm, 3 µm particle size) (Phenomenex, Torrance, CA). The column was maintained at 40°C. Gradient elution was performed with a binary solvent system: phase A: 10 mM ammonium acetate (pH 8.5); phase B: MeOH. The system flow rate was 0.2 mL/min. The linear gradient program was as follows: 40% B (0–0.5min 40% B), 70% B 6–11 min), 80% B (14 min), 100% B (16–18 min), and 40% B (20–27 min). Samples were maintained at 10°C prior to injection. The effluent was introduced into a Finnigan TSQ Quantum Ultra triple quadrupole mass spectrometer (Thermo Sci., Waltham, MA) by (−)-ESI. The ESI voltage −2.3 kV, the capillary temperature was 350°C, with N2 as the nebulizing and sheath gases. Following ionization, deprotonated pseudomolecular ions ([M−H]−) were fragmented by collision-induced dissociation using argon gas, and detection was performed by selected reaction monitoring (SRM) of fragment ions (see Table 2). Quantification was performed by the stable isotope dilution method relative to deuterated internal standards. Eicosanoid levels in colon mucosa were normalized to protein levels. Levels for animals fed the diets for 9–11 weeks were not significantly different, and were combined.
Table 2.
SRM transitions for LC-MS/MS analysis of eicosanoids
| Compounda | [M–H]– (m/z) |
SRM Transition | Collision Energy (eV) |
Tube Lens (V) |
Retention Time (min) |
|---|---|---|---|---|---|
| PGE3 | 349 | 349.20 → 269.20 | 18 | −57 | 8.97 |
| PGE2 | 351 | 351.20 → 271.23 | 20 | −55 | 10.80 |
| PGE2-d4 | 355 | 355.20 → 275.20 | 16 | −45 | 10.81 |
| LTB5 | 333 | 333.14 → 195.10 | 23 | −55 | 13.86 |
| LTB4 | 335 | 335.20 → 195.00 | 22 | −70 | 14.82 |
| LTB4-d4 | 339 | 339.20 → 197.00 | 20 | −66 | 14.82 |
| 13-HODE | 295 | 295.24 → 277.18 | 34 | −70 | 16.47 |
| 13-S-HODE-d4 | 299 | 299.24 → 281.18 | 30 | −68 | 16.47 |
| 5-HETEs | 319 | 319.50 → 115.27 | 27 | −42 | 19.09 |
| 5- S-HETE-d8 | 327 | 327.00 → 115.80 | 30 | −50 | 18.94 |
| 12--HETEs | 319 | 319.24 → 179.09 | 26 | −55 | 17.84 |
| 12-S-HETE-d8 | 327 | 327.24 → 184.09 | 22 | −55 | 17.74 |
| 15--HETEs | 319 | 319.31 → 219.30 | 20 | −52 | 17.37 |
| 15- S-HETE-d8 | 327 | 327.20 → 26.05 | 20 | −52 | 17.37 |
| arachidonic acid | 303 | 303.10 → 177.00 | 31 | −75 | 22.55 |
| arachidonic acid-d8 | 311 | 311.10 → 267.48 | 25 | −76 | 22.54 |
Analysis of Epithelial Proliferation in Colonic Crypts
Immunohistochemical (IHC) staining was performed on colon tissue to visualize the Ki67 antigen expressed in proliferating cells. Tissue slices were fixed in formalin and embedded in paraffin. Slices were then deparrafinized, and antigen unmasking was performed by steaming in citrate target retrieval buffer. Ki67 immunostaining using the mib1 Ki67 antibody and the Labelled Streptavidin-Biotin2 System-Horseradish Peroxidase detection protocol (Dako, Carpinteria, CA). Diaminobenzidine (DAB) staining was performed on a Dako stainer, and counterstaining was with Gill’s hematoxylin/eosin (H/E) (Fisher Sci., Pittsburg, PA). Quantification of Ki67 labeling was performed using MCID software (Cambridge, UK). Well-oriented, full-length crypts were scored by setting color thresholds with the software. The extension of positive (Ki67-labeled) cells up the crypt was recorded as well as the number of positive cells in the entire crypt and in each longitudinal fifth of the crypt. Ki67 staining was expressed as labeling index (LI, fraction of Ki67 positive cells in the whole crypt) labeling index in each crypt compartment (fifths) as well as the extent of the proliferative zone (EPZ, number of cells from the crypt base to the highest Ki67 positive cell). Ki67 labeling was measured by 2 analysts whose mean scoring did not differ appreciably (< 10% when LI and EPZ were compared for the same images).
Data Analysis
Statistical analyses were performed by 2-way MANOVA with diet, genotype, and their interaction as the factors (PASW Statistics software v.18, SPSS, Chicago, IL). We ran this analysis for each of the chemical (fatty acids and eicosanoids) as well as biological (IHC) measures separately. Significance was defined as P < 0.05. As the interaction was of primary importance, we present the comparisons of one factor within the levels of the other factor. For colonic fatty acid levels, Box-Cox transformations of raw data were performed prior to MANOVA analysis in order to achieve normality: square root (12:0, 14:0, 16:1, 18:0, 18:1, 20:1, 20:4, and ω3 HUFA score), negative square root (16:0), and log (18:2, 18:3, 20:3, 20:5, 22:6).
RESULTS
Fatty Acid Profiles
Total (membrane bound + free) fatty acid levels in mouse colonic mucosa are shown in Table 3. Fatty acid profiles differed between diets, as expected, as well as between genotypes. The level of AA was roughly 2- to 3-fold greater in mice fed the oleate diet compared to the fish oil diet for the COX-1 and COX-2 null mice, whereas these levels were similar between the two diets for the wild-type mice. As expected, the level of EPA was much higher (7- to 16-fold) in mice fed fish oil compared to oleate diet. However, there were also differences in EPA levels between genotypes in mice fed fish oil, with COX-1 null mice having roughly half the EPA as COX-2 null and wild-type mice. The AA:EPA ratio observed in the mice fed oleate diet (21:1–28:1) was dramatically decreased (to about 0.8:1–1:1) by administration of fish oil, indicating a significant shift in the relative availability of AA and EPA for COX metabolism. This shift in the balance of substrate available for COX metabolism from ω6 to ω3 fatty acids by the fish oil diet is reflected in the % of highly unsaturated fatty acids (HUFA, fatty acids with ≥20 carbons and ≥3 double bonds) that are also ω3 fatty acids (the % ω3 HUFA score) [29]. The ω3 HUFA score was roughly 25–26% for all mice fed the oleate diet, and 68–76% for mice fed the fish oil diet (Table 3).
Table 3.
Levels of fatty acids in colonic mucosa as a % of total fatty acids
| % of total fatty acids†‡§ |
||||||
|---|---|---|---|---|---|---|
| COX-1 null |
COX-2 null |
wild-type |
||||
| fatty acid | oleate | fish oil | oleate | fish oil | oleate | fish oil |
| lauric (12:0) | 2.08 ± 0.287 | 1.71 ± 0.208 | 2.04 ± 0.431 | 1.22 ± 0.241 | 2.03 ± 0.274 | 1.43 ± 0.202 |
| myristic (14:0) | 2.55 ± 0.278 | 3.07 ± 0.244a | 1.91 ± 0.266 | 2.00 ± 0.297c | 2.30 ± 0.250 | 2.32 ± 0.267 |
| palmitic (16:0) | 19.2 ± 1.69 | 20.9 ± 1.96 | 16.3 ± 1.68 | 18.3 ± 2.07 | 17.9 ± 1.98 | 18.8 ± 1.83 |
| palmitoleic (16:1ω7) | 5.16 ± 1.08 | 4.30 ± 0.661 | 2.91 ± 0.261 | 3.61 ± 0.679 | 4.86 ± 0.666 | 4.96 ± 0.686 |
| stearic (18:0) | 5.96 ± 0.870 | 8.08 ± 0.790 | 8.35 ± 1.29 | 10.9 ± 1.28 | 6.50 ± 0.818 | 9.94 ± 0.768b |
| oleic(18:1ω9) | 39.9 ± 2.59 | 35.2 ± 3.62a | 37.9 ± 3.98 | 25.7 ± 3.85bc | 41.8 ± 3.01 | 26.5 ± 3.09b |
| linoleic (18:2ω6) | 13.5 ± 0.766 | 14.9 ± 1.27 | 15.2 ± 0.782 | 17.4 ± 1.16 | 13.9 ± 0.681 | 15.7 ± 1.12 |
| linolenic (18:3ω3) | 0.434 ± 0.0925 | 0.425 ± 0.431 | 0.666 ± 0.174 | 0.638 ± 0.129 | 0.450 ± 0.0666 | 1.15 ± 0.332 |
| eicosenoic (20:1ω9) | 0.743 ± 0.169 | 1.45 ± 0.246ab | 0.761 ± 0.121 | 0.766 ± 0.303c | 0.917 ± 0.132 | 0.794 ± 0.0615 |
| eicosatrienoic (20:3) | 0.670 ± 0.106 | 0.419 ± 0.129a | 0.856 ± 0.170 | 0.622 ± 0.111 | 0.534 ± 0.0932 | 0.738 ± 0.0998 |
| AA (20:4ω6) | 7.28 ± 1.03 | 2.15 ± 0.808ab | 9.53 ± 1.62 | 5.27 ± 0.826bc | 6.53 ± 1.29 | 4.77 ± 0.675 |
| EPA (20:5ω3) | 0.422 ± 0.104 | 2.80 ± 0.997b | 0.487 ± 0.0855ab | 4.74 ± 0.764b | 0.319 ± 0.0917 | 5.21 ± 0.829b |
| DHA (22:6ω3) | 2.19 ± 0.282 | 4.58 ± 1.51b | 3.13 ± 0.635ab | 8.75 ± 1.92b | 1.90 ± 0.380 | 7.81 ± 1.19b |
| % ω3 HUFA□ | 24.8 ± 1.79 | 76.4 ± 2.38b | 26.1 ± 1.32b | 68.4 ± 1.53b | 25.1 ± 2.94 | 69.5 ± 1.82b |
| AA/EPA | 24.6 ± 3.80 | 0.802 ± 0.176 | 21.4 ± 2.00 | 1.13 ± 0.0607 | 28.1 ± 4.72 | 1.03 ± 0.129 |
Total fatty acids were determined by saponification of mucosa, derivatization, and analysis by GC-MS
Values represent mean ± SEM
Means that differ significantly within each row (2-way MANOVA with comparisons of one factor within the levels of the other factor, P<0.05), are denoted as follows:
significantly different than wild-type mice fed the same diet,
significantly different than oleate diet for mice with the same genotype and
Significantly different than COX-1 null mice fed the same diet. Raw data are presented here, but statistical analyses were performed on Box-Cox transformed data as described in the Materials and Methods section. MANOVA was not performed for AA/EPA ratios.
The % of highly unsaturated fatty acids (≥20 carbons and ≥3 double bonds) that are ω3 (the ω3 HUFA score)
The activities of various fatty acid metabolizing enzymes (desaturases, elongases, etc.) were also estimated by calculating the ratios of products/substrates and comparing between treatments (data not shown). While several differences were observed, the most significant findings were that the activity of the fatty acid Δ5-desaturase (as measured by the 20:4/20:3 ratio) was elevated in mice fed oleate diet vs. mice fed fish oil (11.1–12:1 vs. 5:1–10:1, respectively). A similar pattern was observed for the activity of stearoyl-CoA desaturase (SCD, as measured by the 18:1/18:0 ratio), with ratios of roughly 8:1–9:1 for mice fed oleate diet vs. roughly 3:1–5:1 in mice fed fish oil. Also, within the fish oil diet, COX-1 null mice had increased SCD activity compared to COX-2 null or wild-type mice (5:1 vs. 3:1), which could explain the observation that this group had oleic acid levels similar to the oleate diet animals (see Table 3).
Free arachidonic acid levels in colonic mucosa
Animals fed the oleate diet generally had higher levels of free AA (intracellular AA freed from the membrane by phospholipase A2, and measured by LC-MS without saponification) in the colonic mucosa compared to animals fed fish oil, but the difference between diets was only statistically significant for COX-2 null mice (Fig. 3). The differences in free AA between diet groups reflected reduced total levels of AA produced by the fish oil diet (shown in Table 3). For the oleate diet, COX-2 null mice had elevated free AA, which was statistically significant compared to the wild-type mice. Genotype did not significantly affect free AA levels in mice fed fish oil.
Figure 3.
Levels of free arachidonic acid and measured prostaglandins and leukotrienes in mouse colon mucosa. Eicosanoids were measured LC-MS/MS by the stable isotope dilution method. Error bars represent SEM. Brackets indicate significant differences between means (2-way MANOVA with comparisons of one factor within the levels of the other factor, P<0.05). As no appropriate deuterated internal standard was available for absolute quantification of PGE3 and LTB5, these values are expressed as a % relative to wild-type (WT) mice fed the oleate diet, which was set at 100%.
PGE2 and PGE3
Mice fed oleate diet had 3- to 4-fold higher mucosal PGE2 concentrations compared to mice fed fish oil in wild-type and COX-2 null mice (Fig. 3). The absence of COX-1 resulted in almost total elimination of PGE2 in mice fed either diet, unlike the absence of COX-2, which had virtually no effect compared to wild type. Levels of PGE3 were typically several-fold lower than PGE2 levels in all groups fed the oleate diet and PGE3 was increased in wild-type and COX-2 null mice fed fish oil. However, we did not have an appropriate internal standard for quantification, so the absolute levels of PGE3 were not determined. Similar to PGE2, the absence of COX-1 resulted in almost total lack of PGE3 in the mucosa with either diet.
LTB4 and LTB5
Levels of LTB4 were generally 3- to 5-fold higher in mice fed the oleate fat diet than in mice fed fish oil, whereas genotype appeared to have little effect (Fig. 3). Again, we did not have an appropriate internal standard for quantification of LTB5, but LTB5 levels increased 2–10 fold in wild-type and COX-1 null mice fed the fish oil diet vs. the oleate diet, whereas diet had little impact on LTB5 levels in COX-2 null mice.
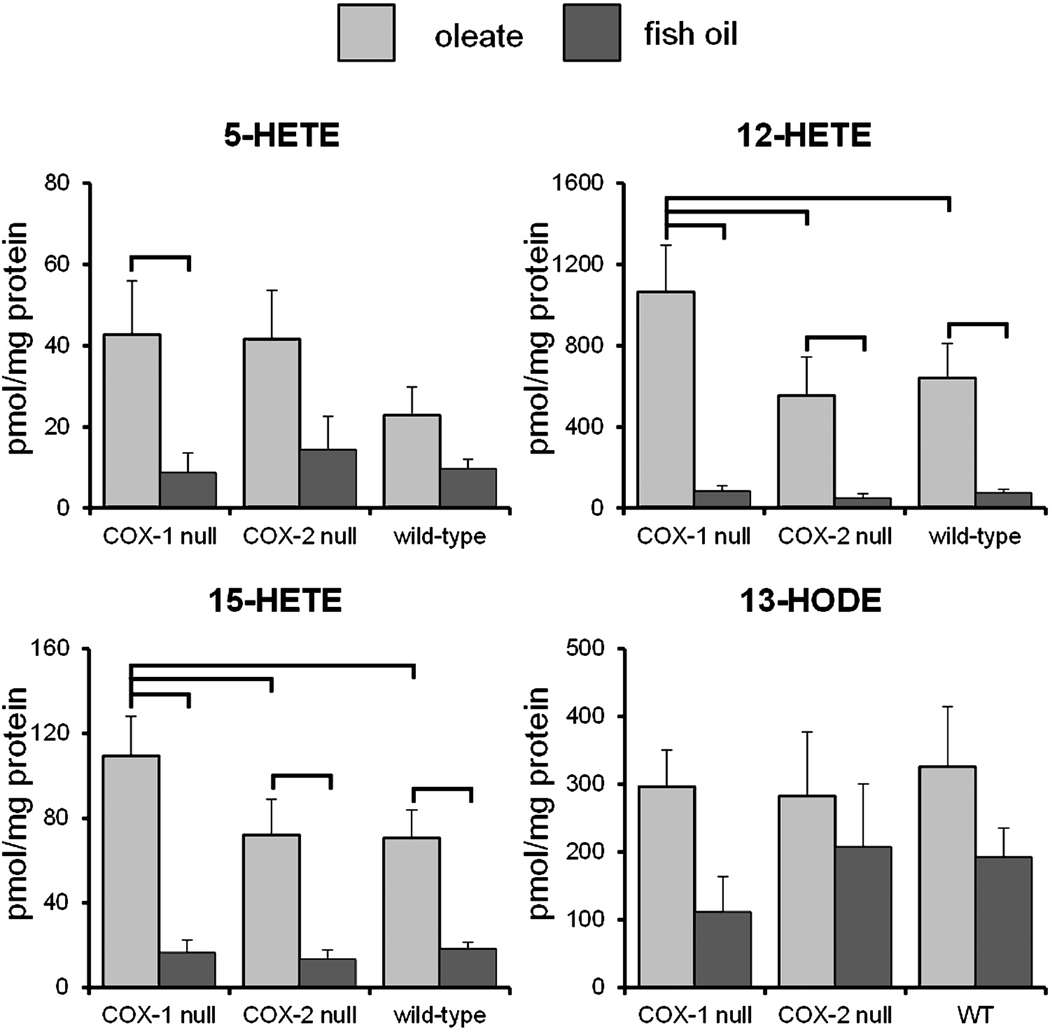
HETEs and 13-HODE
LOX products derived from AA (5-, 12- and 15-HETEs) and linoleic acid (13-HODE) were quantified due to their utility as biomarkers of transformation and inflammation in colonic mucosa [6, 8, 30–35]. The levels of HETEs and 13-HODE in mouse colon mucosa are shown in Fig. 4. 12-HETE was by far the most abundant of all the eicosanoids detected, with 15-HETE and 13-HODE also being present at higher levels than PGE2 or LTB4. Levels of 12-HETE were roughly 7- to 20-fold greater in mice fed the oleate diet versus mice fed fish oil, with statistically significant differences between diets for every genotype. For mice fed the oleate diet, COX-1 null animals had significantly elevated 12-HETE compared to wild-type and COX-2 null animals. Similar results were observed for 15-HETE. Levels of 15-HETE were roughly 4- to 8-fold greater in mice fed the oleate diet than in mice fed fish oil, with statistically significant differences between diets for every genotype. For mice fed the oleate diet, COX-1 null animals had significantly higher 15-HETE levels compared to wild-type and COX-2 null animals.
Figure 4.
Levels of HETEs and 13-HODE in mouse colon mucosa. Eicosanoids were measured LC-MS/MS by the stable isotope dilution method. Error bars represent SEM. Brackets indicate significant differences between means (2-way MANOVA with comparisons of one factor within the levels of the other factor, P<0.05).
Levels of 5-HETE were generally greater in mice fed the oleate diet than in mice fed fish oil, although the only significant difference between diets was observed in COX-1 null animals. No significant differences were observed between genotypes, although COX-1 and COX-2 null mice fed the oleate diet had slightly elevated 5-HETE compared to wild-type mice fed the oleate diet. No statistically significant differences were observed for 13-HODE levels between diets or genotypes, although mice fed the oleate diet had generally greater levels compared to mice fed fish oil. The differences in membrane linoleic acid concentrations (the substrate for 13-HODE synthesis, Table 3) were generally not reflected in 13-HODE levels.
Colonic Proliferation
Proliferation was quantified using Ki67 IHC to assess the biological impact of altering substrate ratios and resultant eicosanoid profiles on normal mucosa. PGE2 is known to stimulate proliferation in colonic epithelial cells [36, 37], while EPA-derived eicosanoids such a PGE3 stimulates proliferation less effectively [13, 15]. Apoptosis was not measured due to the extremely low apoptotic rate in normal mucosa (typically <1 apoptotic cell/crypt) [38–40]. Representative Ki67 imuunohistochemical staining of colonic tissue is shown in Fig. 5. Overall, diet and genotype had minimal effects on colonic proliferation patterns as quantified by Ki67 IHC (Fig. 6). The extent of the proliferative zone (EPZ, the number of cells from the crypt base to the highest Ki67-positive cell, expressed as a fraction of crypt height) was similar (0.60–0.79) for all groups. The EPZ was slightly, but not significantly, lower for mice fed oleate diet than mice fed fish oil for each genotype. Total labeling indices (LI, the fraction of Ki-67 positive cells in the entire crypt) were between 0.35–0.46. Compartment LI values generally decreased towards the top of the crypt, as expected (compartment 1: crypt base, compartment 5: top of crypt). However, the Φ40 values (the LI in the upper 40% of the crypt, i.e. compartments 4 and 5) did vary greatly (0.03–0.21) between groups, with COX-2 null mice fed fish oil having a higher Φ40 than any other group. It should be noted that the fish oil diet had higher levels of saturated fat than the oleate diet (46.2 vs. 36.2 g/kg, respectively). Saturated fat and fish oil/ω-3 PUFAs have been shown to have opposite anti-carcinogenic and anti-inflammatory effects in the colon, and this may be an additional explanation for the lack of observed effects on colonic proliferation between diets [41, 42].
Figure 5.
Representative images of Ki67 immunohistochemistry (IHC) for each treatment. Nuclei are stained blue-purple (hematoxylin), while Ki67+ (proliferating) nuclei are stained brown. Magnification is 40×.
Figure 6.
Proliferation in colon mucosa epithelium as measured by Ki67 labeling of well-oriented, full-length crypts: labeling index (LI, fraction of Ki67positive cells) for whole crypts and crypt compartments (fifths) as well as Φ40 (the LI in the upper 40% of the crypt, i.e. compartments 4 and 5) and the extent of the proliferative zone (EPZ, number of cells from the crypt base to the highest Ki67 positive cell). Error bars represent SEM. Brackets indicate significant differences between means (P<0.05). Note the distinct scales for graphs of EPZ, total LI, and LIs for compartments 1–4 (0–1) vs. graphs of compartment 5 and Φ40 (0–0.4). Brackets indicate significant differences between means (2-way MANOVA with comparisons of one factor within the levels of the other factor, P<0.05). Note that scoring could not be performed for all animals due to poor slicing and arrangement of tissue architecture.
The largest and perhaps most important differences were observed in the absolute size of the crypts, shown in Fig. 7. For mice fed the oleate diet, the mean crypt height was significantly higher in COX-2 null mice than in COX-1 null or wild-type mice (34 ± 1.9 cells vs. 19 ± 2.7 and 22 ± 1.2 cells, respectively). This reflects the trend of PGE2 and arachidonic acid (membrane and free) levels that were highest in COX-2 null mice (Fig. 3). Fish oil appeared to dampen the effect of genotype on crypt height.
Figure 7.
Crypt height (number of cells in a full-length crypt) in colonic mucosa as measured by H/E staining of well-oriented, full-length crypts. Brackets indicate significant differences between means (2-way MANOVA with comparisons of one factor within the levels of the other factor, P<0.05). Note that scoring could not be performed for all animals due to poor slicing and arrangement of tissue architecture.
DISCUSSION
Eicosanoids and Fatty Acids
Administration of fish oil reduced the amount of free AA in colonic mucosa. Free AA levels were surprisingly higher in COX-2 null mice than in wild-type mice fed the oleate diet. This may be due in part to the fact that genetic deletion of COX-1 or COX-2 can induce compensatory upregulation of cPLA2 [43–45], resulting in increased release of AA from membrane phospholipids. However, the slight increase in the level of free AA in the mucosa of COX-1 null mice was not statistically significant compared to wild-type mice. Fish oil dramatically altered the AA:EPA ratio relative to the oleate diet. The changes in the AA:EPA ratio are correlated with reduced levels of mucosal PGE2 and increases in PGE3. These results are consistent with a previous study in wild-type mice treated with the carcinogen azoxymethane, in which dietary fish oil reduced the PGE2:PGE3 ratio from 736:1 to 2.3:1in colonic mucosa [25]. In our study, it appears that these changes were largely mediated through COX-1, as deletion of COX-1 dramatically reduced PGE2 and PGE3. Deletion of COX-2 did not affect PGE2 or PGE3 synthesis compared to wild-type mice for either diet, suggesting that COX-2 is a minor pathway leading to PGH2 substrate for E-series PG synthesis in normal mucosa. This is consistent with low expression of COX-2 in normal colonic mucosa of mice (see Fig. 2) [1, 26, 27]. The small increase in PGE2 levels in COX-2 null versus wild-type mice fed oleate fat diets may result from the observation that genetic deletion of COX-1 and COX-2 can induce compensatory upregulation of the other COX isoform, as well as mPGES-1, to maintain PGE2 synthesis [43–45]. The PGE2 data further suggest that COX-1, and not COX-2, would be the major targetable COX isoform for dietary modulation of eicosanoid profiles in normal colonic mucosa.
Similar to the prostanoids, the fish oil diet generally reduced synthesis of AA-derived LTB4 and increased synthesis of EPA-derived LTB5 relative to the oleate diet. The fish oil diet also significantly reduced the levels of 12- and 15-HETEs in colonic mucosa, and the same trend was observed for the minor product 5-HETE. The HETEs data for oleate diet animals are consistent with a previous report that the relative levels of HETEs in both normal and inflamed colonic mucosa of rodents are 12-HETE > 15-HETE > 5-HETE [46]. In human colonic mucosa, reported 15-HETE levels are between 2- and 100-fold higher than 12-HETE in both normal and inflamed mucosa, with 5-HETE being present at much lower concentrations [8, 46–49]. These differences between rodents and humans are likely due in part to the fact that 12- and 15-S-HETEs are produced by a 12/15-LOX in rodents (which produces predominantly 12-HETE), and by distinct enzymes in humans with 15-HETE predominating [50, 51]. Therefore, in humans, the relative levels of 12- and 15-HETEs are likely to be reversed from those seen in mice. The relatively low levels of 5-HETE relative to the other HETEs may be due to the low expression of 5-LOX in normal mucosa [33].
The HETEs data also suggest possible shunting of arachidonic acid substrate to LOX metabolism when COX-1 is absent, since COX-1 null mice fed the oleate diet had virtually no PGE2 synthesis and significantly elevated synthesis of 12-HETE and 15-HETE relative to wild-type and COX-2 null animals in the context of similar AA levels. A similar but non-significant trend in shunting from COX to LOX in was also observed for 5-HETE, but the effect of shunting on eicosanoids derived from 5-LOX was likely minimal as COX-1 null and wild-type animals fed oleate diet had similar LTB4 levels.
It is interesting to note that the mean net loss of PGE2 synthesis in COX-1 null mice was roughly 35 pmol/mg (96% of wild-type PGE2 levels), while the mean increase in total HETEs synthesis was ~482 pmol/mg [1217 pmol/mg total HETEs, a 66% increase from the total HETEs levels in wild-type animals (735 pmol/mg)]. The mean net increases in 5-, 12-, and 15-HETEs were ~20 pmol/mg (an 87% increase from wild-type), 423 pmol/mg (66%), and 39 pmol/mg (55%), respectively. Therefore, substrate availability, binding affinity, enzymatic efficiency, and compartmentalization of enzymes could play a role in shunting of substrate from COX to LOX pathways in normal mucosa, as the increase in HETEs synthesis was 12-fold greater than the decrease in PGE2 synthesis.
Proliferation
The fish oil diet, genotype status, and resulting changes in eicosanoid profiles showed little correlation with the proliferation indices in colonic crypts. It is important to note that these animals were not exposed to any carcinogens or inflammatory agents and therefore represent normal colonic mucosa that is not transformed or inflamed. The only statistically significant changes were on absolute crypt size and Φ40. Deletion of COX-2 resulted in increased crypt height in mice fed the oleate diet, with increases in crypt height reflecting the elevated free arachidonic acid and PGE2 levels in these animals. The fish oil diet attenuated this effect. In humans, increased crypt height has been associated with reduced colon cancer risk [52, 53], while results in animals have been mixed [54–59]. In one study of fish oil feeding, decreased mucosal AA:EPA ratio was associated with shorter crypts, unlike in our study, but increased mucosal PGE2 levels were associated with larger crypts [60]. Although PGE2 in colonic pathology is generally associated with inflammation, it should be noted that in other conditions PGE2 serves to limit inflammation. This makes it difficult to interpret the effects of PGE2 in normal colonic mucosa.
The increased LI in compartment 5 (top 1/5th of the crypt) and Φ40 in COX-2 null mice fed fish oil was unexpected, given that PGE2 values were not elevated in this group. It is important to note that these animals were sickly and had a lower survival rate than the other groups (5/10 animals survived for this group, whereas all animals survived in all other groups except COX-2 null mice fed oleate diet, with 9/10 surviving). The increased proliferation may therefore be due to an underlying pathology confounding the experimental treatment in these mice. In addition, COX-2 has been shown to have a protective role in resolution of inflammation and survival of colitis [61–63]. In comparing the effects of genotype on proliferation in animals fed a oleate blend fat diet, COX-2 may have subtle physiological role that is critical for maintaining proliferation at low levels and crypt height at normal levels (EPZ and crypt height were higher in COX-2 nulls). In models of allergic inflammation and colitis, PGE2 is well known to have a protective role in limiting inflammation [61–65]. This is unlike initiated colon tissue where highly up-regulated COX-2 is known to have tumor-promoting effects [2, 3, 66]. This calls into question whether prevention in normal risk individuals who do not have any defined pathological changes in the colon would benefit from COX-1 or COX-2 inhibition.
Due to the small sample size of this study, no adjustment was made for multiple comparisons in the statistical analysis. If adjustments for multiple comparisons had been made using the Bonferroni method, the single comparison between the two diets within the same genotype would be the same as the present analysis. The three comparisons between genotypes within each diet would result in the loss of statistical significance for some outcomes. However, the main findings of this study would still be statistically significant.
For fatty acids, the increased % ω3 HUFA, the lower levels of EPA and the lower AA:EPA ratios for the fish oil diet versus the oleate diet would still be significant within each genotype. For the prostaglandins, the near complete elimination of PGE2 and PGE3 synthesis in COX-1 null mice, and the reduced PGE2 and PGE3 synthesis in mice fed fish oil versus oleate diet in the COX-2 null and wild-type mice would remain significant. The reduction in HETEs synthesis between diets would remain significant for 12- and 15-HETE within each genotype except for 12-HETE in COX-2 null mice. However, the shunting effect from COX to LOX metabolism in COX-1 null mice would no longer be statistically significant. For proliferation, the increased Φ40 in COX-2 null mice versus wild-type mice would remain significant, as well as the increased crypt height in COX-2 null mice versus COX-1 null and wild-type mice fed the oleate diet.
In conclusion, the changes in eicosanoid profiles induced by diet and genotype were profound, while corresponding changes in proliferation were slight. The changes induced by fish oil could protect the colon upon carcinogen challenge. Inhibition of COX-2, however, might not have preventive effects in low-risk settings.
Acknowledgements
We thank Dr. Robert Langenbach for providing the transgenic animals and Dr. William Lands for advice on formulation of the rodent diets. Patrick Brown assisted with statistical data analysis, and Dr. Kathleen Noon assisted with chiral chromatographic LC-MS-MS methods (Biomedical Mass Spectrometry Facility).
Financial Support: Supported by the Michigan Institute for Clinical & Health Research (grant GM 48864), the University of Michigan Comprehensive Cancer Center (grant P30-CA46592), NCI grant RO1 CA120381, NIH grant R01 GM68848, and the Kutsche Memorial Endowment in Internal Medicine at the University of Michigan Medical School. A. Neilson was supported by the NCI T32 Cancer Biology Training Program at the University of Michigan (grant 5T32CA009676-18).
Abbreviations
- AA
arachidonic acid
- AIN
American Institute of Nutrition
- COX
cyclooxygenase
- DAB
3,3'-Diaminobenzidine
- DHA
docosahexaenoic acid
- EPA
eicosapentaenoic acid
- EPZ
extent of the proliferative zone
- H/E
hematoxylin/eosin
- HETE
hydroxyeicosatetraenoic acid
- HODE
hydroxyoctadecadienoic acid
- HUFA
highly unsaturated fatty acid
- IHC
immunohistochemistry
- LI
labeling index
- LT
leukotriene
- NSAIDs
non-steroidal anti-inflammatory drugs
- PBS
phosphate-buffered saline
- PG
prostaglandin
- PUFA
polyunsaturated fatty acid
- SCD
stearoyl-CoA desaturase
REFERENCES
- 1.Ishikawa T-O, Herschman HR. Tumor formation in a mouse model of colitis-associated colon cancer does not require COX-1 or COX-2 expression. Carcinogenesis. 2010;31:729–736. doi: 10.1093/carcin/bgq002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Nakanishi M, Montrose DC, Clark P, Nambiar PR, Belinsky GS, Claffey KP, et al. Genetic Deletion of mPGES-1 Suppresses Intestinal Tumorigenesis. Cancer Res. 2008;68:3251–3259. doi: 10.1158/0008-5472.CAN-07-6100. [DOI] [PubMed] [Google Scholar]
- 3.McLean MH, Murray GI, Fyfe N, Hold GL, Mowat NAG, El-Omar EM. COX-2 expression in sporadic colorectal adenomatous polyps is linked to adenoma characteristics. Histopathology. 2008;52:806–815. doi: 10.1111/j.1365-2559.2008.03038.x. [DOI] [PubMed] [Google Scholar]
- 4.Pugh S, Thomas GA. Patients with adenomatous polyps and carcinomas have increased colonic mucosal prostaglandin E2. Gut. 1994;35:675–678. doi: 10.1136/gut.35.5.675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Giardiello FM, Spannhake EW, Dubois RN, Hylind LM, Robinson CR, Hubbard WC, et al. Prostaglandin levels in human colorectal mucosa - Effects of sulindac in patients with familial adenomatous polyposis. Digest Dis Sci. 1998;43:311–316. doi: 10.1023/a:1018898120673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Shureiqi I, Wojno KJ, Poore JA, Reddy RG, Moussalli MJ, Spindler SA, et al. Decreased 13-S-hydroxyoctadecadienoic acid levels and 15-lipoxygenase-1 expression in human colon cancers. Carcinogenesis. 1999;20:1985–1995. doi: 10.1093/carcin/20.10.1985. [DOI] [PubMed] [Google Scholar]
- 7.Magnusson C, Ehrnstrom R, Olsen J, Sjolander A. An increased expression of cysteinyl leukotriene 2 receptor in colorectal adenocarcinomas correlates with high differentiation. Cancer Res. 2007;67:9190–9198. doi: 10.1158/0008-5472.CAN-07-0771. [DOI] [PubMed] [Google Scholar]
- 8.Shureiqi I, Chen DN, Day RS, Zuo XS, Hochman FL, Ross WA, et al. Profiling lipoxygenase metabolism in specific steps of colorectal tumorigenesis. Cancer Prev Res. 2010;3:829–838. doi: 10.1158/1940-6207.CAPR-09-0110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Vonkeman HE, van de Laar M. Nonsteroidal Anti-Inflammatory Drugs: Adverse Effects and Their Prevention. Semin Arthritis Rheum. 2010;39:294–312. doi: 10.1016/j.semarthrit.2008.08.001. [DOI] [PubMed] [Google Scholar]
- 10.Moonen H, Dommels Y, van Zwam M, van Herwijnen M, Kleinjans J, Alink G, et al. Effects of polyunsaturated fatty acids on prostaglandin synthesis and cyclooxygenase-mediated DNA adduct formation by heterocyclic aromatic amines in human adenocarcinoma colon cells. Molecular Carcinogenesis. 2004;40:180–188. doi: 10.1002/mc.20032. [DOI] [PubMed] [Google Scholar]
- 11.Nieves D, Moreno JJ. Effect of arachidonic and eicosapentaenoic acid metabolism on RAW 264.7 macrophage proliferation. Journal of Cellular Physiology. 2006;208:428–434. doi: 10.1002/jcp.20678. [DOI] [PubMed] [Google Scholar]
- 12.Strasser T, Fischer S, Weber PC. Leukotriene-B5 is formed in human-neutrophils after dietary supplementation with eicosapentaenoic acid. P Natl Acad Sci USA. 1985;82:1540–1543. doi: 10.1073/pnas.82.5.1540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yang P, Chan D, Felix E, Cartwright C, Menter DG, Madden T, et al. Formation and antiproliferative effect of prostaglandin E3 from eicosapentaenoic acid in human lung cancer cells. Journal of Lipid Research. 2004;45:1030–1039. doi: 10.1194/jlr.M300455-JLR200. [DOI] [PubMed] [Google Scholar]
- 14.Wada M, DeLong CJ, Hong YH, Rieke CJ, Song I, Sidhu RS, et al. Enzymes and receptors of prostaglandin pathways with arachidonic acid-derived versus eicosapentaenoic acid-derived substrates and products. J Biol Chem. 2007;282:22254–22266. doi: 10.1074/jbc.M703169200. [DOI] [PubMed] [Google Scholar]
- 15.Bagga D, Wang L, Farias-Eisner R, Glaspy JA, Reddy ST. Differential effects of prostaglandin derived from omega-6 and omega-3 polyunsaturated fatty acids on COX-2 expression and IL-6 secretion. P Natl Acad Sci USA. 2003;100:1751–1756. doi: 10.1073/pnas.0334211100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Nkondjock A, Shatenstein B, Maisonneuve P, Ghadirian P. Specific fatty acids and human colorectal cancer: an overview. Cancer Detect Prev. 2003;27:55–66. doi: 10.1016/s0361-090x(02)00179-4. [DOI] [PubMed] [Google Scholar]
- 17.Bartsch H, Nair J, Owen RW. Dietary polyunsaturated fatty acids and cancers of the breast and colorectum: emerging evidence for their role as risk modifiers. Carcinogenesis. 1999;20:2209–2218. doi: 10.1093/carcin/20.12.2209. [DOI] [PubMed] [Google Scholar]
- 18.Chapkin RS, McMurray DN, Lupton JR. Colon cancer, fatty acids and anti-inflammatory compounds. Curr Opin Gastroen. 2007;23:48–54. doi: 10.1097/MOG.0b013e32801145d7. [DOI] [PubMed] [Google Scholar]
- 19.Calviello G, Serini S, Piccioni E. n-3 polyunsaturated fatty acids and the prevention of colorectal cancer: Molecular mechanisms involved. Curr Med Chem. 2007;14:3059–3069. doi: 10.2174/092986707782793934. [DOI] [PubMed] [Google Scholar]
- 20.Broughton KS, Wade JW. Total fat and (n-3):(n-6) fat ratios influence eicosanoid production in mice. J Nutr. 2002;132:88–94. doi: 10.1093/jn/132.1.88. [DOI] [PubMed] [Google Scholar]
- 21.Anti M, Armelao F, Marra G, Percesepe A, Bartoli GM, Palozza P, et al. Effects of different doses of fish-oil on rectal cell-proliferation in patients with sporadic colonic adenomas. Gastroenterology. 1994;107:1709–1718. doi: 10.1016/0016-5085(94)90811-7. [DOI] [PubMed] [Google Scholar]
- 22.Anti M, Marra G, Armelao F, Bartoli GM, Ficarelli R, Percesepe A, et al. Effect of omega-3-fatty-acids on rectal mucosal cell-proliferation in subjects at risk for colon cancer. Gastroenterology. 1992;103:883–891. doi: 10.1016/0016-5085(92)90021-p. [DOI] [PubMed] [Google Scholar]
- 23.Huang YC, Jessup JM, Forse RA, Flickner S, Pleskow D, Anastopoulos HT, et al. n-3 Fatty acids decrease colonic epithelial cell proliferation in high-risk bowel mucosa. Lipids. 1996;31:S313–S317. doi: 10.1007/BF02637099. [DOI] [PubMed] [Google Scholar]
- 24.Bartram HP, Gostner A, Scheppach W, Reddy BS, Rao CV, Dusel G, et al. Effects of fish-oil on rectal cell-proliferation, mucosal fatty-acids, and prostaglandin-E2 release in healthy-subjects. Gastroenterology. 1993;105:1317–1322. doi: 10.1016/0016-5085(93)90135-y. [DOI] [PubMed] [Google Scholar]
- 25.Vanamala J, Glagolenko A, Yang P, Carroll RJ, Murphy ME, Newman RA, et al. Dietary fish oil and pectin enhance colonocyte apoptosis in part through suppression of PPAR{delta}/PGE2 and elevation of PGE3. Carcinogenesis. 2008;29:790–796. doi: 10.1093/carcin/bgm256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Al-Salihi MA, Terrece Pearman A, Doan T, Reichert EC, Rosenberg DW, Prescott SM, et al. Transgenic expression of cyclooxygenase-2 in mouse intestine epithelium is insufficient to initiate tumorigenesis but promotes tumor progression. Cancer Lett. 2009;273:225–232. doi: 10.1016/j.canlet.2008.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Oshima M, Dinchuk JE, Kargman SL, Oshima H, Hancock B, Kwong E, et al. Suppression of intestinal polyposis in Apc”716 knockout mice by inhibition of cyclooxygenase 2 (COX-2) Cell. 1996;87:803–809. doi: 10.1016/s0092-8674(00)81988-1. [DOI] [PubMed] [Google Scholar]
- 28.Djuric Z, Ren JW, Blythe J, VanLoon G, Sen A. A Mediterranean dietary intervention in healthy American women changes plasma carotenoids and fatty acids in distinct clusters. Nutrition Research. 2009;29:156–163. doi: 10.1016/j.nutres.2009.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Stark KD. The percentage of n-3 highly unsaturated fatty acids in total HUFA as a biomarker for omega-3 fatty acid status in tissues. Lipids. 2008;43:45–53. doi: 10.1007/s11745-007-3128-3. [DOI] [PubMed] [Google Scholar]
- 30.Wu Y, Fang B, Yang XQ, Wang L, Chen D, Krasnykh V, et al. Therapeutic molecular targeting of 15-lipoxygenase-1 in colon cancer. Mol Ther. 2008;16:886–892. doi: 10.1038/mt.2008.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Shureiqi I, Jiang W, Zuo XS, Wu YQ, Stimmel JB, Leesnitzer LM, et al. The 15-lipoxygenase-1 product 13-S-hydroxyoctadecadienoic acid down-regulates PPAR-delta to induce apoptosis in colorectal cancer cells. P Natl Acad Sci USA. 2003;100:9968–9973. doi: 10.1073/pnas.1631086100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Yuri M, Sasahira T, Nakai K, Ishimaru S, Ohmori H, Kuniyasu H. Reversal of expression of 15-lipoxygenase-1 to cyclooxygenase-2 is associated with development of colonic cancer. Histopathology. 2007;51:520–527. doi: 10.1111/j.1365-2559.2007.02799.x. [DOI] [PubMed] [Google Scholar]
- 33.Melstrom LG, Bentrem DJ, Salabat MR, Kennedy TJ, Ding XZ, Strouch M, et al. Overexpression of 5-Lipoxygenase in colon polyps and cancer and the effect of 5-LOX inhibitors in vitro and in a murine model. Clin Cancer Res. 2008;14:6525–6530. doi: 10.1158/1078-0432.CCR-07-4631. [DOI] [PubMed] [Google Scholar]
- 34.Kennedy TJ, Talamonti M, Ujiki M, Ding XZ, Ternent CA, Bell RH, et al. Lipoxygenase expression in colon polyps and inhibition of colon cancer growth by lipoxygenase blockade. J Am Coll Surgeons. 2004;199 S78-S. [Google Scholar]
- 35.Merchant N, Chung DH, Townsend CM, Heslin MJ. Tumor-associated down-regulation of 15-lipoxygenase-1 is reversed by celecoxib in colorectal cancer - Discussion. Ann Surg. 2005;241:946–947. doi: 10.1097/01.sla.0000164177.95620.c1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Dommels YEM, Haring MMG, Keestra NGM, Alink GM, van Bladeren PJ, van Ommen B. The role of cyclooxygenase in n-6 and n-3 polyunsaturated fatty acid mediated effects on cell proliferation, PGE(2) synthesis and cytotoxicity in human colorectal carcinoma cell lines. Carcinogenesis. 2003;24:385–392. doi: 10.1093/carcin/24.3.385. [DOI] [PubMed] [Google Scholar]
- 37.Qiao L, Kozoni V, Tsioulias GJ, Koutsos MI, Hanif R, Shiff SJ, et al. Selected eicosanoids increase the proliferation rate of human colon carcinoma cell lines and mouse colonocytes in vivo. BBA-Lipid Lipid Met. 1995;1258:215–223. doi: 10.1016/0005-2760(95)00100-q. [DOI] [PubMed] [Google Scholar]
- 38.Strater J, Koretz K, Gunthert AR, Moller P. In-situ detection of enterocytic apoptosis in normal colonic mucosa and in familial adenomatous polyposis. Gut. 1995;37:819–825. doi: 10.1136/gut.37.6.819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Risio M, Lipkin M, Newmark H, Yang K, Rossini FP, Steele VE, et al. Apoptosis, cell replication, and western-style diet-induced tumorigenesis in mouse colon. Cancer Res. 1996;56:4910–4916. [PubMed] [Google Scholar]
- 40.Courtney E, Matthews S, Finlayson C, Di Pierro D, Belluzzi A, Roda E, et al. Eicosapentaenoic acid (EPA) reduces crypt cell proliferation and increases apoptosis in normal colonic mucosa in subjects with a history of colorectal adenomas. International Journal of Colorectal Disease. 2007;22:765–776. doi: 10.1007/s00384-006-0240-4. [DOI] [PubMed] [Google Scholar]
- 41.Vijay-Kumar M, Vanegas SM, Patel N, Aitken JD, Ziegler TR, Ganji V. Fish oil rich diet in comparison to saturated fat rich diet offered protection against lipopolysaccharide-induced inflammation and insulin resistance in mice.(Research) Nutrition & Metabolism. 2011;8:16. doi: 10.1186/1743-7075-8-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Baltgalvis KA, Berger FG, Peña MMO, Davis JM, Carson JA. The Interaction of a High-Fat Diet and Regular Moderate Intensity Exercise on Intestinal Polyp Development in ApcMin/+ Mice. Cancer Prev Res. 2009;2:641–649. doi: 10.1158/1940-6207.CAPR-09-0017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Sandee D, Sivanuntakorn S, Vichai V, Kramyu J, Kirtikara K. Up-regulation of microsomal prostaglandin E synthase-1 in COX-1 and COX-2 knock-out mouse fibroblast cell lines. Prostag Oth Lipid M. 2009;88:111–116. doi: 10.1016/j.prostaglandins.2008.12.001. [DOI] [PubMed] [Google Scholar]
- 44.Zhang J, Goorha S, Raghow R, Ballou LR. The tissue-specific, compensatory expression of cyclooxygenase-1 and -2 in transgenic mice. Prostag Oth Lipid M. 2002;67:121–135. doi: 10.1016/s0090-6980(01)00177-0. [DOI] [PubMed] [Google Scholar]
- 45.Kirtikara K, Morham SG, Raghow R, Laulederkind SJF, Kanekura T, Goorha S, et al. Compensatory prostaglandin E-2 biosynthesis in cyclooxygenase 1 or 2 null cells. Journal of Experimental Medicine. 1998;187:517–523. doi: 10.1084/jem.187.4.517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Zijlstra FJ, Vandijk APM, Garrelds IM, Ouwendijk RJT, Wilson JHP. Species-differences in the pattern of eicosanoids produced by inflamed and noninflamed tissue. Agents Actions. 1992:C73–C75. [PubMed] [Google Scholar]
- 47.Zijlstra FJ, Wilson JHP. 15-HETE is the main eicosanoid present in mucus of ulcerative proctocolitis. Prostag Leukotr Ess. 1991;43:55–59. doi: 10.1016/0952-3278(91)90133-p. [DOI] [PubMed] [Google Scholar]
- 48.Zijlstra FJ, Vandijk APM, Wilson JHP, Vanriemsdijkoverbeeke IC, Vincent JE, Ouwendijk RJT. 15-HETE is the main eicosanoid formed by human colonic mucosa. Agents Actions. 1992:C53–C59. [PubMed] [Google Scholar]
- 49.Zijlstra FJ, Vandijk APM, Ouwendijk RJT, Vanriemsdijkoverbeeke IC, Wilson JHP. Eicosanoid production by the mucosa in inflammatory bowel-disease after 5-ASA treatment. Agents Actions. 1993;38:C122–C124. doi: 10.1007/BF01991158. [DOI] [PubMed] [Google Scholar]
- 50.Brash AR. Lipoxygenases: Occurrence, functions, catalysis, and acquisition of substrate. J Biol Chem. 1999;274:23679–23682. doi: 10.1074/jbc.274.34.23679. [DOI] [PubMed] [Google Scholar]
- 51.Funk CD, Chen X-S, Johnson EN, Zhao L. Lipoxygenase genes and their targeted disruption. Prostag Oth Lipid M. 2002:68–69. 303–312. doi: 10.1016/s0090-6980(02)00036-9. [DOI] [PubMed] [Google Scholar]
- 52.Lipkin M, Uehara K, Winawer S, Sanchez A, Bauer C, Phillips R, et al. 7th-Day Adventist vegetarians have a quiescent proliferative activity in colonic mucosa. Cancer Lett. 1985;26:139–144. doi: 10.1016/0304-3835(85)90019-9. [DOI] [PubMed] [Google Scholar]
- 53.McTiernan A, Yasui Y, Sorensen B, Irwin ML, Morgan A, Rudolph RE, et al. Effect of a 12-month exercise intervention on patterns of cellular proliferation in colonic crypts: A randomized controlled trial. Cancer Epidem Biomar. 2006;15:1588–1597. doi: 10.1158/1055-9965.EPI-06-0223. [DOI] [PubMed] [Google Scholar]
- 54.Hong MY, Turner ND, Carroll RJ, Chapkin RS, Lupton JR. Differential response to DNA damage may explain different cancer susceptibility between small and large intestine. Exp Biol Med. 2005;230:464–471. doi: 10.1177/153537020523000704. [DOI] [PubMed] [Google Scholar]
- 55.Barnes CJ, Lee M, Hardman WE, Cameron IL. Aspirin suppresses 1,2-dimethylhydrazine-induced alteration of proliferative parameters in rat colonic crypts. Cell Proliferat. 1996;29:467–473. doi: 10.1111/j.1365-2184.1996.tb00989.x. [DOI] [PubMed] [Google Scholar]
- 56.Dirks P, Freeman HJ. Effects of differing purified cellulose, pectin and hemicellulose fiber diets on mucosal morphology in the rat small and large-intestine. Clin Invest Med. 1987;10:32–38. [PubMed] [Google Scholar]
- 57.Heitman DW, Ord VA, Hunter KE, Cameron IL. Effect of dietary cellulose on cell-proliferation and progression of 1,2-dimethylhydrazine-induced colon carcinogenesis in rats. Cancer Res. 1989;49:5581–5585. [PubMed] [Google Scholar]
- 58.Salim EI, Wanibuchi H, Morimura K, Murai T, Makino S, Nomura T, et al. Induction of tumors in the colon and liver of the immunodeficient (SCID) mouse by 2-amino-3-methylimidazo 4,5-f quinoline (IQ) - modulation by long-chain fatty acids. Carcinogenesis. 2002;23:1519–1529. doi: 10.1093/carcin/23.9.1519. [DOI] [PubMed] [Google Scholar]
- 59.Torzsas TL, Kendall CWC, Sugano M, Iwamoto Y, Rao AV. The influence of high and low molecular weight chitosan on colonic cell proliferation and aberrant crypt foci development in CF1 mice. Food Chem Toxicol. 1996;34:73–77. doi: 10.1016/0278-6915(95)00083-6. [DOI] [PubMed] [Google Scholar]
- 60.Lee D-YK, Lupton JR, Aukema HM, Chapkin RS. Dietary Fat and Fiber Alter Rat Colonic Mucosal Lipid Mediators and Cell Proliferation. J Nutr. 1993;123:1808–1817. doi: 10.1093/jn/123.11.1808. [DOI] [PubMed] [Google Scholar]
- 61.Reuter BK, Asfaha S, Buret A, Sharkey KA, Wallace JL. Exacerbation of inflammation-associated colonic injury in rat through inhibition of cyclooxygenase-2. J Clin Invest. 1996;98:2076–2085. doi: 10.1172/JCI119013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Morteau O, Morham SG, Sellon R, Dieleman LA, Langenbach R, Smithies O, et al. Impaired mucosal defense to acute colonic injury in mice lacking cyclooxygenase-1 or cyclooxygenase-2 (vol 105, pg 469, 2000) J Clin Invest. 2000;105:1023. doi: 10.1172/JCI6899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Guslandi M. Exacerbation of inflammatory bowel disease by nonsteroidal anti-inflammatory drugs and cyclooxygenase-2 inhibitors: Fact or fiction? World J Gastroentero. 2006;12:1509–1510. doi: 10.3748/wjg.v12.i10.1509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Aggarwal S, Moodley YP, Thompson PJ, Misso NL. Prostaglandin E2 and cysteinyl leukotriene concentrations in sputum: association with asthma severity and eosinophilic inflammation. Clin Exp Allergy. 2010;40:85–93. doi: 10.1111/j.1365-2222.2009.03386.x. [DOI] [PubMed] [Google Scholar]
- 65.Gauvreau Gail M, Watson Rick M, O'Byrne Paul M. Protective Effects of Inhaled PGE2 on Allergen-induced Airway Responses and Airway Inflammation. Am J Resp Crit Care. 1999;159:31–36. doi: 10.1164/ajrccm.159.1.9804030. [DOI] [PubMed] [Google Scholar]
- 66.Kawamori T, Rao CV, Seibert K, Reddy BS. Chemopreventive Activity of Celecoxib, a Specific Cyclooxygenase-2 Inhibitor, against Colon Carcinogenesis. Cancer Res. 1998;58:409–412. [PubMed] [Google Scholar]