SUMMARY
What is known and Objective
Chemotherapeutic drugs for cancer treatment are, of necessity, cytotoxic. Unintended damage to normal central nervous system neuronal structure or function might lead to deleterious adverse effects on cognitive function, a mild form of which is reported by some cancer survivors. Understanding the physiologic connection between cancer chemotherapy and the reported cognitive dysfunction, could help inform choice of drugs, treatment regimens and new drug development. Our objective is to comment on a proposed mechanism for ‘chemo-fog’.
Comment
An increasing number of patients are surviving cancer and are generating a new and rapidly growing category within the healthcare system. Some of these cancer survivors are reporting that they are experiencing residual and lingering effects from the cancer, or from its treatment, and that they now need care as survivors. This has given rise to the new field of ‘survivor care’. Control of chemo-fog is an important aspect and understanding its mechanism, the basis for more rationale therapy. Such insight would also help direct drug-discovery efforts.
What is new and Conclusion
New evidence suggests that ‘chemo-fog’ may be due to excessive cytokine release by the cytotoxic agents. Control of the elevated levels of the blood–brain-barrier-permeable pro-inflammatory cytokines, may help minimize this adverse effect.
WHAT IS KNOWN AND OBJECTIVE
Cancer chemotherapeutic drugs have contributed to the significant improvement in clinical outcome and survival of cancer patients. Life extension can be traced to advances in the selection of drugs, dose regimens and specific combinations used to treat either the primary tumor or its spread. Everyone is grateful that these drugs have helped usher in a new era – an era of increased survival from cancer. But cancer chemotherapeutic drugs, as all drugs, have adverse effects. Some adverse effects are immediate; others can be delayed. Likewise, some are transient; others can linger.
Adverse effects might be reasonably and particularly expected for cancer chemotherapeutic drugs, which must be cytotoxic to be effective. But intended cytotoxicity on cancer cells raises the possibility of unintended toxicity on normal cells. If such ‘collateral’ toxicity occurs to a sufficient extent to central nervous system (CNS) neuronal and support cells, adverse effects that outlast the period of chemotherapeutic drug exposure could result and give rise to chronic dysfunction in sensitive areas of mental performance.
A postulated adverse effect associated with adjuvant cancer chemotherapy is a mild cognitive impairment (variously called ‘chemo-fog’, ‘chemo-brain’ or similar term) (1, 2). The cognitive domains that are most often detected as being impaired include verbal and visual memory, attention, concentration, language, motor skills, multitasking and ability to organize information. Although a causal relationship between cognitive impairment and the chemotherapy per se has not been unequivocally proven, recent imaging studies buttress claims of such a connection (3) and animal models show that these drugs can impair learning or memory – sometimes synergistically – when given in combinations that are used clinically (4). Our objective is to comment on a possible mechanism for ‘chemo-fog’.
COMMENT
The earliest suggestion of a possible link between cancer chemotherapeutic treatment and cognitive impairment as an adverse consequence was likely that of Silberfarb et al. (5), who stated that ‘Chemotherapy was the major variable associated with cognitive impairment in these patients’. In 1998, the seminal paper by van Dam et al. (6) reported cognitive impairment in patients treated for breast cancer. As the patients had neither brain cancer nor brain irradiation, the inference was made stronger. It is now suggested that a subgroup of patients (widely estimated at 4–75%) experience some impairment even years after therapy.
Not much is currently understood about chemo-fog. Basically three very general things are known: (i) some patients who receive cancer chemotherapeutic drugs administered individually or in combinations report mild cognitive impairment during or subsequent to treatment; (ii) standardized tests detect deficits in the cognitive ability of some cancer survivors and (iii) some recent imaging studies report differences in such patients. Little is known beyond this. Nevertheless, the condition has been reported by a sufficiently large number of patients that it merits study so that patients, families, healthcare providers, insurers, employers and others have the best information possible to inform their individual and collective decisions.
MECHANISM?
Understanding the mechanism by which cancer chemotherapeutic drugs might cause cognitive impairment would have multiple benefits: it would inform clinical treatment, improve ‘survivor-care’, and help direct novel drug-discovery efforts. For this reason, the recent proposal by Aluise et al. (7) is particularly exciting. Doxorubicin (DOX), one of the more widely used adjuvant agents for the treatment of breast cancer, impairs learning /memory in rodents (8, 9) and DOX-treated patients test lower in cognitive scores and visuospatial skills (10, 11). Although DOX is cytotoxic (it produces its peripheral cytotoxic effect by interrupting cell replication and tumour growth either by intercalation into DNA or interference with topoisomerase II or by generation of reactive oxygen species), neither it nor its metabolites readily pass the blood–brain barrier (BBB). How then can it cause cognitive impairment? Aluise et al. have proposed a link. DOX stimulates proinflammatory cytokine release in response to cell injury and circulating TNF-α levels rise. Unlike DOX, cytokines pass the BBB and there is strong evidence for excess CNS cytokine-induced cognitive dysfunction (12).
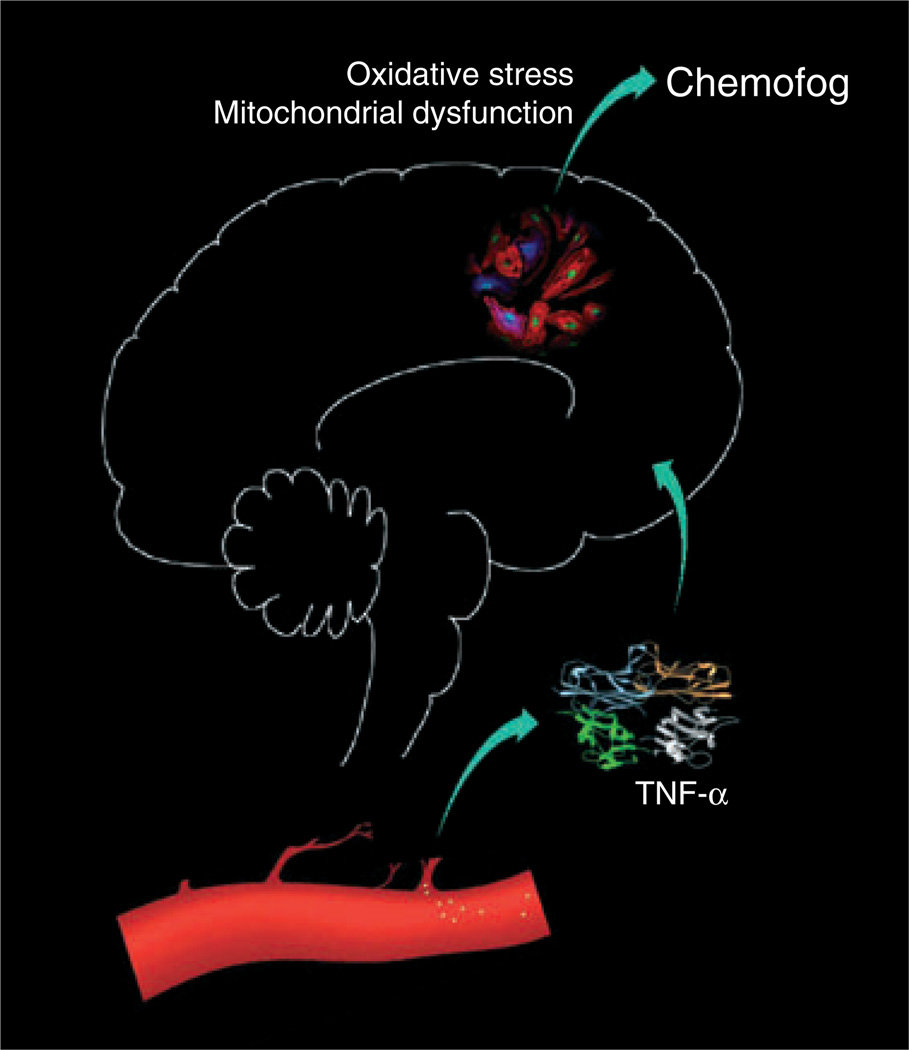
Aluise et al. implicate in particular tumor necrosis factor alpha (TNF-α). Administration of DOX to mice increases the levels of peripheral TNF-α, which is subsequently detected in the brain (e.g., hippocampus and cerebral cortex). Once in the brain, TNF-α activates the following deleterious sequence: glial cell-induced local TNF-α production → induction of NO synthase → the over-production of reactive nitrogen species (e.g., peroxy-nitrite, ONOO−). This sequence is inhibited by anti-TNF-α antibody. DOX also causes anti-TNF-α antibody-sensitive deleterious effects on brain cell mitochondria. Thus, DOX produces biochemical changes that result in the increased production of TNF-α, increased levels of circulating TNF-α, increased passage of TNF-α into the brain (hippocampus and cortex), stimulation of local generation of reactive oxygen and nitrogen species, oxidative stress, a mitochondrial dysfunction and consequent impairment of working memory (Fig 1).
Fig. 1.
Illustration of a postulated link between a cancer chemotherapeutic drug (doxorubicin) and chemofog [highly simplified version of that presented in (7)].
WHAT IS NEW AND CONCLUSION
The increase in cancer survival has led to an increased awareness of the possible chronic effects of cancer chemotherapeutic drugs. Given the well-known susceptibility of neurons and supportive cells to the toxic effects of drugs that are used to treat cancer, it would not be surprising if some of these highly beneficial drugs also impaired certain aspects of cognitive function. The mechanistic link proposed by Aluise et al. (7) adds credence to patient reports of cognitive impairment. Control of the elevated levels of the blood–brain-barrier-permeable pro-inflammatory cytokines, may help minimize this adverse effect. This mechanism suggests targets for novel treatment, prophylaxis, and drug-discovery efforts.
ACKNOWLEDGEMENTS
The author is president of the Forget-Me-Not Foundation, a non-profit devoted to increasing the awareness, study, and treatment of the medical needs of cancer survivors (‘survivor care’) and is co-investigator on NIH R01 award CA–129092 (PI: Ellen A. Walker, PhD, Temple University School of Pharmacy) which tests for cognitive deficits induced by chemotherapeutic agents administered alone or in combination to mice.
REFERENCES
- 1.Raffa RB, Duong PV, Finney J, et al. Is ‘chemo-fog’/’chemo-brain’ caused by cancer chemotherapy? Journal of Clinical Pharmacy and Therapeutics. 2006;31:129–138. doi: 10.1111/j.1365-2710.2006.00726.x. [DOI] [PubMed] [Google Scholar]
- 2.Raffa RB, Tallarida RJ, editors. Chemofog: cancer chemotherapy-related cognitive impairment. Austin: Springer/Landes; 2010. [PubMed] [Google Scholar]
- 3.Raffa RB. Is a picture worth a thousand (forgotten) words?: neuroimaging evidence for the cognitive deficits in ‘chemo-fog’/‘chemo-brain’. Journal of Clinical Pharmacy and Therapeutics. 2010;35:1–9. doi: 10.1111/j.1365-2710.2009.01044.x. [DOI] [PubMed] [Google Scholar]
- 4.Foley JJ, Raffa RB, Walker EA. Effects of chemotherapeutic agents 5-fluorouracil and methotrexate alone and combined in a mouse model of learning and memory. Psychopharmacology. 2008;199:527–538. doi: 10.1007/s00213-008-1175-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Silberfarb PM, Philibert D, Levine PM. Psychosocial aspects of neoplastic disease: II. Affective and cognitive effects of chemotherapy in cancer patients. American Journal of Psychiatry. 1980;137:597–601. doi: 10.1176/ajp.137.5.597. [DOI] [PubMed] [Google Scholar]
- 6.van Dam FS, Schagen SB, Muller MJ, et al. Impairment of cognitive function in women receiving adjuvant treatment for high-risk breast cancer: high-dose versus standard-dose chemotherapy. Journal of the National Cancer Institute. 1998;90:210–218. doi: 10.1093/jnci/90.3.210. [DOI] [PubMed] [Google Scholar]
- 7.Aluise CD, Sultana R, Tangpong J, et al. Chemobrain (chemofog) as a potential side effect of doxorubicin administration: role of cytokine-induced, oxidative/nitrosative stress in cognitive dysfunction. In: Raffa RB, Tallarida RJ, editors. Chemofog: cancer chemotherapy-related cognitive impairment. Austin: Springer/Landes; 2010. pp. 147–153. [Google Scholar]
- 8.Macleod JE, DeLeo JA, Hickey WF, Ahles TA, Saykin AJ, Bucci DJ. Cancer chemotherapy impairs contextual but not cue specific fear memory. Behavioral Brain Research. 2007;181:168–172. doi: 10.1016/j.bbr.2007.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Konat GW, Kraszpulski M, James I, Zhang HT, Abraham J. Cognitive dysfunction induced by chronic administration of common cancer chemotherapeutics in rats. Metabolic Brain Disease. 2008;23:325–333. doi: 10.1007/s11011-008-9100-y. [DOI] [PubMed] [Google Scholar]
- 10.Jansen CE, Dodd MJ, Miaskowski CA, Dowling GA, Kramer J. Preliminary results of a longitudinal study of changes in cognitive function in breast cancer patients undergoing chemotherapy with doxorubicin and cyclophosphamide. Psychooncology. 2008;17:1189–1195. doi: 10.1002/pon.1342. [DOI] [PubMed] [Google Scholar]
- 11.Raffa RB, Tallarida RJ. Effects on the visual system might contribute to some of the cognitive deficits of cancer chemotherapy-induced ‘chemo-fog’. Journal of Clinical Pharmacy and Therapeutics. 2010;35:249–255. doi: 10.1111/j.1365-2710.2009.01086.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Trask PC, Esper P, Riba M, Redman B. Psychiatric side effects of interferon therapy: prevalence, proposed mechanisms, and future directions. Journal of Clinical Oncology. 2000;18:2316–2326. doi: 10.1200/JCO.2000.18.11.2316. [DOI] [PubMed] [Google Scholar]