Abstract
African Americans experience a higher mortality rate and an excess burden of ESRD compared with Caucasians in the general population, but among those treated with dialysis, African Americans typically survive longer than Caucasians. We examined whether differences in inflammation may explain this paradox. We prospectively followed a national cohort of incident dialysis patients in 81 clinics for a median of 3 years (range 4 months to 9.5 years). Among 554 Caucasians and 262 African Americans, we did not detect a significant difference in median CRP between African Americans and Caucasians (3.4 versus 3.9 mg/L). Mortality was significantly lower for African Americans versus Caucasians (34% versus 56% at 5 years); the relative hazard was 0.7 (95% CI, 0.5 to 0.9) after adjusting for age, gender, dialysis modality, smoking, body mass index, diabetes, BP, cholesterol, cardiovascular disease, congestive heart failure, comorbid disease, hemoglobin, albumin, CRP, and IL-6. However, the risk varied by CRP tertile: the relative hazards for African Americans compared with Caucasians were 1.0 (95% CI, 0.7 to 1.4), 0.7 (95% CI, 0.4 to 1.3), and 0.5 (95% CI, 0.3 to 0.8) in the lowest, middle, and highest tertiles, respectively. We obtained similar results when we accounted for transplantation as a competing event, and we examined mortality across tertiles of IL-6. In summary, racial differences in survival among dialysis patients are not present at low levels of inflammation but are large at higher levels. Differences in inflammation may explain, in part, the racial paradox of ESRD survival.
African American individuals are disproportionately represented among ESRD patients in the United States, comprising 32% of the ESRD population1 but only 12% of the general population.2 In general, survival of ESRD patients is poor.1 African Americans have higher prevalence of many intermediate markers of poor dialysis outcomes,3–5 yet African Americans on dialysis have better survival than Caucasians.1,6,7 In 2008, the annual mortality rate was 16% for African Americans and 24% for Caucasians treated with dialysis in the United States.1 This survival advantage is in contrast to the decreased life expectancy that African Americans experience when compared with Caucasians in the general population8 and has been observed in at least one predialysis (estimated GFR, 15 to 29 ml/min per 1.73 m2) population.9 The cause of this paradox is not entirely clear.10
It has been postulated that the African American-Caucasian dialysis survival paradox may be due to racial differences in those who survive to develop ESRD and subsequently undergo dialysis treatment. For example, younger African Americans (less than 65 years) with early-stage chronic kidney disease (CKD) have been shown to experience greater mortality than their Caucasian counterparts11 and African American U.S. veterans with pre-ESRD CKD have greater mortality than Caucasians.12 Therefore, African American patients who survive to develop ESRD may be of overall better health than Caucasian patients. Additionally, access to ESRD care and patient preferences may contribute to the survival paradox. Very ill African American patients may be less likely to be offered or elect to initiate dialysis than similarly ill Caucasian patients. Racial differences in receipt of renal transplantation may contribute to the survival advantage among African Americans as well. It is possible that because African Americans are less likely to receive transplants,1,13,14 those African Americans who initiate and remain on dialysis are of better health than the Caucasians who do not undergo transplantation and remain on dialysis.
A less-studied possible explanation for the survival paradox seen among dialysis patients is that of racial differences in inflammation. C-reactive protein (CRP), a commonly examined inflammatory marker, may be elevated in the setting of conditions such as infections, inflammatory illnesses, periodontal disease, chronic lung disease, and obesity.15 CRP has been studied extensively and found to be an independent predictor of cardiovascular disease (CVD).16 Elevated CRP has been associated with mortality in dialysis patients.17,18 In the general population, African Americans have higher CRP than Caucasians,19 perhaps owing to their greater prevalence of risk factors for inflammation. However, the relationship between race and CRP in the dialysis population and the role this might play in explaining the survival paradox have not been well examined. Therefore, we explored this relationship in a national, prospective cohort study of incident dialysis patients. We hypothesized that levels of inflammation would differ between African American and Caucasian dialysis patients and would provide insights into their observed survival paradox.
RESULTS
Participant Characteristics
There were 554 Caucasian and 262 African American participants in our cohort (Table 1). African Americans were younger, more likely to be female, and more likely to be treated with hemodialysis than were Caucasians. African Americans were more likely to be current smokers, had greater body mass index (BMI) and greater systolic BP than Caucasians. CVD was more prevalent among Caucasian participants. African Americans had greater HDL levels but lower hemoglobin levels than Caucasians. There was a trend toward CRP being greater among Caucasians, although this did not reach statistical significance. IL 6 levels were greater among Caucasians than African Americans (P < 0.01).
Table 1.
Baseline characteristics of study participants by race
| Characteristicsa | Race |
Pb | |
|---|---|---|---|
| Caucasians (n = 554) | African Americans (n = 262) | ||
| Age, years | 58.7 (14.6) | 54.2 (14.8) | <0.01 |
| Female,% | 43.7 | 53.4 | <0.01 |
| Hemodialysis % | 76.4 | 89.3 | <0.01 |
| Current smoking, % | 15.1 | 18.1 | <0.01 |
| Body mass index, kg/m2 | 26.6 (6.4) | 27.8 (7.5) | 0.03 |
| Systolic BP, mmHg | 149 (19) | 154 (17) | <0.01 |
| Diabetes, % | 53.7 | 55.7 | 0.59 |
| Prevalent CVD, % | 49.0 | 33.0 | <0.01 |
| Congestive heart failure, % | 47.9 | 45.8 | 0.57 |
| Comorbidity score, 0 to 3 | 1.98 (0.81) | 1.91 (0.78) | 0.23 |
| Severe comorbidity (score = 3), % | 31.1 | 26.3 | 0.17 |
| Total cholesterol, mg/dl | 190 (49) | 186 (50) | 0.32 |
| High-density lipoprotein, mg/dl | 42 (15) | 47 (16) | <0.01 |
| CRP, mg/L | 3.9 (1.8, 9.5) | 3.4 (1.5, 9.6) | 0.10 |
| IL-6, pg/ml | 4.2 (2.7, 7.5) | 3.6 (2.4, 6.0) | <0.01 |
| Serum albumin, g/dl | 3.6 (0.4) | 3.6 (0.3) | 0.62 |
| Hemoglobin, g/dl | 10.8 (1.3) | 10.4 (1.3) | <0.01 |
CVD, cardiovascular disease; CRP, C-reactive protein.
aMedian (interquartile range) for CRP, and IL-6, arithmetic mean (SD) for other continuous variables.
bBy t test or chi-squared test.
Overall, the median CRP was 3.8 mg/L (interquartile range, 1.6 to 9.6). A number of participant characteristics varied by CRP tertile (Table 2). Among those in the lowest CRP tertile, African Americans were more likely to be treated with hemodialysis and have greater systolic BP but had less CVD, severe comorbid disease, and hemoglobin levels than did Caucasians. Among those in the middle CRP tertile, African Americans were younger, were more likely to be current smokers, and had greater systolic BP and lower hemoglobin levels than did Caucasians. The highest CRP tertile revealed several important baseline differences between African Americans and Caucasians. African Americans in this tertile were younger, more likely to be treated with hemodialysis, and more likely to be smokers; had greater BMI and prevalence of diabetes; but were less likely to have CVD and had lower hemoglobin levels than their Caucasian counterparts.
Table 2.
Baseline characteristics of study participants by CRP tertile and race
| Characteristicsa | Tertile 1 |
Tertile 2 |
Tertile 3 |
||||||
|---|---|---|---|---|---|---|---|---|---|
| Caucasians (n = 170) | African Americans (n = 102) | Pb | Caucasians (n = 197) | African Americans (n = 77) | Pb | Caucasians (n = 187) | African Americans (n = 83) | Pb | |
| Age, years | 54.9 (15.4) | 52.4 (15.9) | 0.20 | 59.3 (14.2) | 54.4 (14.5) | 0.01 | 61.4 (13.4) | 56.1 (13.4) | <0.01 |
| Female, % | 44.1 | 49.0 | 0.43 | 44.2 | 57.1 | 0.05 | 42.8 | 55.4 | 0.06 |
| Hemodialysis % | 66.5 | 90.2 | <0.01 | 81.7 | 84.4 | 0.60 | 79.7 | 92.8 | <0.01 |
| Smoking, % | 14.6 | 18.6 | 0.68 | 16.8 | 15.8 | 0.01 | 13.9 | 19.8 | 0.02 |
| BMI, kg/m2 | 25.6 (4.7) | 26.1 (5.7) | 0.45 | 27.0 (6.3) | 27.0 (5.7) | 0.99 | 27.3 (7.6) | 30.6 (9.8) | <0.01 |
| Systolic BP, mmHg | 148 (20) | 153 (17) | 0.03 | 150 (19) | 157 (17) | 0.01 | 147 (18) | 152 (16) | 0.07 |
| Diabetes, % | 54.1 | 47.1 | 0.26 | 55.6 | 55.8 | 0.97 | 51.3 | 66.3 | 0.02 |
| Prevalent CVD, % | 41.8 | 25.7 | <0.01 | 45.9 | 33.8 | 0.07 | 58.8 | 41.0 | <0.01 |
| Congestive heart failure, % | 36.5 | 36.3 | 0.97 | 50.0 | 54.6 | 0.50 | 56.2 | 49.4 | 0.30 |
| Comorbidity score (0 to 3, where 3 is most severe) | 1.91 (0.86) | 1.77 (0.74) | 0.18 | 1.95 (0.80) | 1.82 (0.79) | 0.20 | 2.07 (0.77) | 2.16 (0.78) | 0.39 |
| Severe comorbidity, score = 3 (severe) | 32.4 | 18.6 | 0.01 | 27.9 | 23.4 | 0.44 | 33.2 | 38.6 | 0.39 |
| Cholesterol, mg/dl | 191 (53) | 180 (43) | 0.09 | 193 (46) | 190 (59) | 0.66 | 186 (47) | 190 (49) | 0.50 |
| CRP, mg/L | 1.2 (0.9, 1.7) | 1.3 (0.9, 1.6) | 0.98 | 3.8 (3.0, 4.6) | 3.8 (3.1, 4.5) | 0.91 | 15.3 (9.2, 24.0) | 14.6 (10.3, 21.4) | 0.66 |
| IL-6, pg/ml | 2.6 (1.8, 3.9) | 2.5 (1.9, 3.6) | 0.68 | 3.9 (2.9, 5.6) | 3.7 (2.4, 5.5) | 0.78 | 8.1 (5.3, 15.7) | 5.8 (3.8, 11.4) | 0.67 |
| Albumin, g/dl | 3.7 (0.4) | 3.6 (0.4) | 0.53 | 3.6 (0.4) | 3.7 (0.3) | 0.32 | 3.6 (0.4) | 3.6 (0.3) | 0.64 |
| Hemoglobin, g/dl | 11.1 (1.1) | 10.7(1.3) | <0.01 | 10.8 (1.4) | 10.3 (1.3) | <0.01 | 10.6 (1.3) | 10.1 (1.3) | <0.01 |
BMI, body mass index; CVD, cardiovascular disease; CRP, C-reactive protein.
aMedian (interquartile range) for CRP, and IL-6, arithmetic mean (SD) for other continuous variables.
bBy t test or chi-squared test.
Mortality
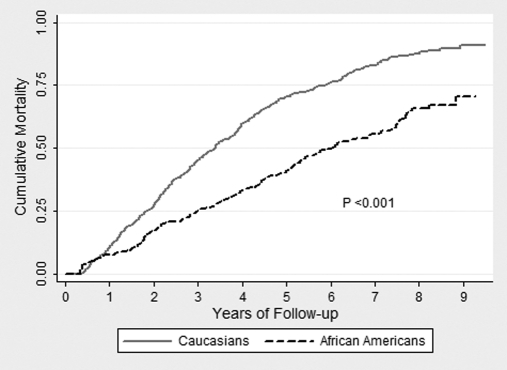
A total of 504 (61.8%) deaths occurred during follow-up. Among Caucasians, there were 370 deaths (66.8%), and among African Americans there were 134 deaths (51.2%). Crude mortality rates at 1, 3, and 5 years of follow-up are shown in Table 3, and Figure 1 illustrates cumulative mortality by race. Mortality rates between African Americans and Caucasians were significantly different (P < 0.001 by log-rank test).
Table 3.
Crude mortality by race and years of follow-up
| Caucasians (n = 554) |
African Americans (n = 262) |
|||||
|---|---|---|---|---|---|---|
| Cumulative Deaths, n | Person Years | Crude Mortality Rate | Cumulative Deaths, n | Person Years | Crude Mortality Rate | |
| 1-year mortality | 48 | 291 | 16.5% | 12 | 144 | 8.3% |
| 3-year mortality | 210 | 968 | 21.7% | 55 | 562 | 9.8% |
| 5-year mortality | 311 | 1297 | 24.0% | 89 | 854 | 10.4% |
Figure 1.
There was a statistically significant difference in unadjusted survival between African Americans and Caucasians.
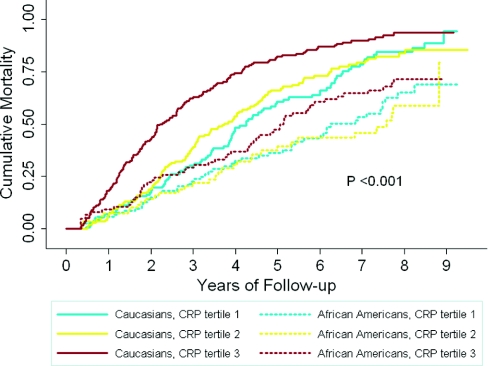
Cox proportional hazards models revealed that, overall, African Americans had a lesser relative hazard (RH) of mortality than did Caucasians (0.68, 95% confidence interval [CI], 0.53 to 0.86 after extensive adjustment for potential confounders) (Table 4). However, when stratified by CRP tertile, the survival advantage observed for African Americans was only seen among those in tertile 3 (Figure 2). After adjustment for confounders, the RH for death was 0.51 (CI, 0.33 to 0.79) comparing African Americans with Caucasians in CRP tertile 3 (Table 4). There was no statistically significant racial survival difference observed in CRP tertiles 1 and 2. Effect modification by CRP tertile and race were particularly evident when tertiles 1 and 3 were compared (P interaction <0.01 in models adjusted for variables beyond age and gender). Additionally, when we examined the continuous relationship of CRP with mortality, there was a significant association in models adjusted for age, gender, and race (RH per log unit high sensitivity CRP was 1.23; CI, 1.15 to 1.32), and there was a significant interaction between CRP and race (P interaction 0.04 in fully adjusted models).
Table 4.
Relative hazard (95% confidence interval) of all-cause mortality comparing African Americans with Caucasians, by CRP tertile
| Overall African Americans Compared with Caucasians (Referent) (n = 816) | By CRP Tertile |
*P for Linear Trend across CRP Tertiles | |||
|---|---|---|---|---|---|
| Tertile 1 (n = 272) | Tertile 2 (n = 274) | Tertile 3 (n = 270) | |||
| Crude | 0.46 (0.39 to 0.54) | 0.53 (0.39 to 0.72) | 0.45 (0.31 to 0.67) | 0.39 (0.29 to 0.54) | 0.05 |
| Model 1a | 0.51 (0.43 to 0.62) | 0.63 (0.46 to 0.85) | 0.47 (0.31 to 0.71) | 0.44 (0.30 to 0.64) | 0.05 |
| Model 2b | 0.61 (0.49 to 0.75) | 0.90 (0.63 to 1.28) | 0.56 (0.34 to 0.92) | 0.48 (0.31 to 0.75) | <0.01 |
| Model 3c | 0.67 (0.53 to 0.85) | 0.89 (0.61 to 1.29) | 0.58 (0.35 to 0.98) | 0.51 (0.33 to 0.80) | <0.01 |
| Model 4d | 0.68 (0.53 to 0.86) | 1.01 (0.72 to 1.40) | 0.73 (0.42 to 1.28) | 0.51 (0.33 to 0.79) | <0.01 |
*P for linear trend test included an interaction term for race and CRP tertile and describes how the effect of CRP varies by race.
aModel 1, adjusted for age and sex.
bModel 2, adjusted for model 1, dialysis modality, and traditional cardiovascular disease (CVD) risk factors (smoking status, body mass index, diabetes, blood pressure, cholesterol), prevalent CVD, and congestive heart failure.
cModel 3, adjusted for model 2 plus comorbidity score, hemoglobin, and serum albumin.
dModel 4, adjusted for model 3 and CRP, and IL-6 levels.
Figure 2.
Unadjusted analysis of cumulative mortality by race and C-reactive protein (CRP) tertile revealed a survival advantage for African Americans compared to Caucasians only among CRP tertile 3.
Subgroup Analyses
Stratified and interaction analysis showed that the survival advantage for African Americans as compared with Caucasians was most evident among those older than 60 years of age, men, those with no history of CVD, and those with diabetes (data not shown).
Sensitivity Analyses
When accounting for transplantation as a potential informative event in competing risk models, the RH for death continued to only be significantly less for African Americans compared with Caucasians when CRP tertile 3 was examined in a fully adjusted model (Table 5). When participants were stratified by IL-6 tertile (as opposed to CRP tertile), a survival advantage for African Americans was only seen among those in tertile 3. After adjustment for confounders, the RH for death was 0.56, 95% CI 0.36 to 0.89 comparing African Americans to Caucasians. There was no statistically significant racial survival difference observed in IL-6 tertiles 1 and 2. Among the subset of participants for whom baseline vascular access status was known and adjusted (n = 583), a similar trend was present (data not shown).
Table 5.
Competing risk analysis of the relative hazard of all-cause mortality comparing African Americans with Caucasians, by CRP tertile (transplantation is the competing event)
| Overall (n = 816) | CRP Tertile African Americans Compared with Caucasians (Referent) |
|||
|---|---|---|---|---|
| Tertile 1 (n = 272) | Tertile 2 (n = 274) | Tertile 3 (n = 270) | ||
| Crude | 0.61 (0.50 to 0.74) | 0.81 (0.58 to 1.14) | 0.53 (0.36 to 0.78) | 0.51 (0.38 to 0.68) |
| Adjusteda | 0.77 (0.60 to 0.98) | 1.30 (0.90 to 1.87) | 0.74 (0.41 to 1.33) | 0.60 (0.39 to 0.91) |
aAdjusted for age, sex, dialysis modality, traditional cardiovascular disease (CVD) risk factors (smoking status, body mass index, diabetes, blood pressure, and cholesterol), prevalent CVD, congestive heart failure, comorbidity score, hemoglobin, serum albumin, CRP, and IL-6 levels.
DISCUSSION
In a national prospective cohort study of incident dialysis patients and in contrast to what is seen in the general population,19 we observed that Caucasians had greater levels of inflammatory markers than African Americans in the first few months after dialysis initiation. We also found that the survival advantage frequently observed among African Americans as compared with Caucasians was only present in the setting of higher levels of inflammation as measured by CRP and IL-6. When African Americans and Caucasians with lesser inflammation were compared, greater survival for African Americans was less profound and was similar among patients with the lowest levels inflammation. Our findings persisted with adjustment for factors known both to impact survival and to have unequal distribution between African Americans and Caucasians treated with dialysis. Additionally, our results were not explained by racial differences in access to transplantation.
Importantly, we observed a survival advantage for African Americans despite them having lesser hemoglobin and equivalent albumin values to Caucasians in the overall cohort. This is in contrast to some studies that have found African Americans treated with dialysis to have higher albumin and other markers of better nutritional status20,21 that may relate to their greater survival. Furthermore, African Americans with the highest markers of inflammation, where we found the greatest survival advantage, were more likely to have diabetes than were Caucasians. We noted that the African American-Caucasian survival advantage in the setting of significant inflammation was particularly strong among individuals older than 60 years of age, men, and those with diabetes. Consistent with our findings is a recent study of BP among hemodialysis patients by Myers et al.22 that also found the survival advantage for African Americans to be only among their older age group (greater than 40 years) regardless of systolic BP. Furthermore, African American patients with diabetic nephropathy (pre-ESRD) treated in the Department of Veterans Affairs were found to have decreased risk of CVD and mortality when compared with Caucasians.23
The possible differential relationship between inflammation and mortality among African Americans as compared with Caucasians treated with dialysis has been discussed in two recent reports in the literature. Noori et al.24 examined a prospective cohort of prevalent hemodialysis patients and found that the relationship between the highest (versus lowest) quartile of IL-6 and mortality among African Americans was nearly half that of the same among Caucasians. The authors concluded that African Americans may be more resilient against the death effect of inflammation. Furthermore, Streja et al.21 evaluated data from prevalent hemodialysis patients treated by a United States-based large dialysis organization and reported that the survival advantage among African Americans was reversed (with greater survival among Caucasians) when adjustment was made for surrogates of the malnutrition-inflammation complex syndrome. Here, the authors report being limited by a lack of measurement of a marker of inflammation (e.g. CRP or IL-6) but conclude that better nutritional and inflammatory status may be the main cause of the survival advantage observed among African Americans.
What is it about the inflammatory milieu of ESRD that could lead to these observations? It is likely related to the decreased risk of cardiovascular disease that has been noted for African Americans as compared with Caucasians treated with dialysis.25,26 It has been postulated that because African Americans tend to have more organ-limited kidney diseases as a cause of ESRD (e.g. focal segmental glomerulosclerosis), their burden of extrarenal atherosclerosis may be less than that of Caucasians.27 In our study, for example, we found that 49% of Caucasians versus 33% of African Americans had CVD at the start of dialysis. A lesser prevalence of CVD may explain the recent report that a cohort of African Americans with moderate CKD attributed to hypertension were more likely to progress to ESRD than to die,28 which is in contrast to the general CKD population.29–32 Therefore, it is possible that inflammation present in African Americans with ESRD is most often due to causes other than CVD, thus potentially leading to less detrimental effects on survival. It is also possible that the origin of inflammation does not differ between African Americans and Caucasians, but rather the difference lies in the consequences or handling of inflammation. Perhaps yet-to-be-described genetic polymorphisms may explain the apparent racial differences in response to inflammation.
There are some limitations of our study that deserve mention. First, potential causal relationships should be interpreted with caution given the observational design. Second, we analyzed a single measurement of CRP and IL-6 during the first few months of dialysis initiation. Examination of these laboratory measures throughout the dialysis experience could alter our results. Third, we had limited data on treatment-related factors and lacked power to assess cause-specific mortality. Fourth, we lacked subclinical measures of CVD, thus residual confounding could have limited our adjustment for CVD. There are several strengths of our study, however, including that it is one of few examining the contribution of inflammation to the racial survival paradox seen in ESRD. The prospective collection of data and the national multi-center design with characteristics comparable with the United States dialysis population33 also allowed for a well characterized, generalizable cohort of dialysis patients. Additionally, we were able to assess the contribution of transplantation and the effects on multiple important subgroups.
In our study, the survival advantage among African American dialysis patients was found to only exist in the setting of high levels of inflammation. Racial differences in the presence and response to inflammation may underlie the long-observed survival paradox seen among dialysis patients and is worthy of further investigation.
CONCISE METHODS
Study Design and Population
We examined the relationship between race, inflammation, and mortality in a national prospective cohort study of incident dialysis patients. The Choices for Healthy Outcomes in Caring for ESRD (CHOICE) study was initiated in 1995 to examine treatment choices and outcomes of dialysis care.34 Eligibility criteria for enrollment included establishment of long-term outpatient dialysis in the preceding 3 months, ability to provide informed consent for participation, age older than 17 years, and ability to speak English or Spanish. Median time from dialysis initiation to enrollment was 45 days. Informed consent was obtained from each participant, and the institutional review boards of The Johns Hopkins University and the participating dialysis facilities approved the study protocol.
From October 1995 to June 1998, 1041 participants from 19 states were enrolled at 81 dialysis clinics associated with Dialysis Clinic Inc. (DCI, Nashville, TN; n = 923), New Haven CAPD (New Haven, CT; n = 86) or Saint Raphael's Hospital (New Haven, CT; n = 32). The total number of enrolled participants represents about two-thirds of eligible patients at these clinics, and those not enrolled were of similar age and gender to those who were enrolled.35 For the purposes of this study, we limited our sample to participants who were of self-reported African American or Caucasian race (51 who were of “other” race were excluded), underwent CRP measurement, and survived at least 4 months from the date of dialysis initiation. We required at least 4 months of survival because of the well established high mortality rate known to occur within the first few months of dialysis initiation,36 which likely primarily reflects the burden of predialysis comorbidities.
Measurements
During enrollment, study participants completed a baseline questionnaire and provided demographics and medical/social history. Height and weight, used to calculate BMI, were obtained from the Center for Medicare Services form 2728 (which is completed at the initiation of outpatient dialysis therapy). Vascular access data were obtained via review of discharge summaries, dialysis flow, and clinic notes.37 Baseline nonfasting venous blood specimens were routinely collected at the DCI facilities just before a dialysis session. Laboratory values for albumin, hemoglobin, total cholesterol, and HDL were obtained from monthly dialysis laboratory tests or sent to Quest Diagnostics (Baltimore, MD). Those obtained from monthly tests were averaged for the first 3 months on dialysis therapy. A specimen bank was established to store blood samples from the DCI enrollees, with specimens obtained for 97% of the total cohort. For participants with samples sent to the specimen bank, specimens were also sent overnight to the central laboratory, where they were stored at −80°C. More than 95% of samples were frozen within 48 hours of venipuncture. For assessment of inflammation, high-sensitivity CRP was measured using a colorimetric competitive enzyme-linked immunosorbent assay (coefficient of variation, 8.9%), and IL-6 was measured using an ultrasensitive enzyme-linked immunosorbent assay (coefficient of variation, 7%). Both CRP and IL-6 were measured at the Laboratory for Clinical Biochemistry Research at the University of Vermont (Colchester, VT).
Comorbidity was assessed using the Index of Coexistent Disease,35,38 a medical record-derived index demonstrated to predict death in dialysis populations.39 Index of Coexistent Disease scores range from 0 to 3, with 3 as the highest severity level. A trained research nurse abstracted medical records to determine these scores on the basis of 19 medical conditions. Individual comorbidities were abstracted from dialysis clinic records, hospital discharge summaries, medication lists, consultation notes, diagnostic imaging, and cardiac imaging reports, which were collected at each dialysis unit and sent to New England Medical Center for abstraction and scoring. Reliability of data abstraction and severity scoring was assessed using masked recoding of 45 medical records.39 Interrater reliability for scoring was high (κ = 0.93).
Outcomes Ascertainment
The observation period for each participant began at enrollment and continued until December 31, 2004. Median follow-up was 3.0 years (range approximately 4 months to 9.5 years), for our study population. The primary outcome of interest was mortality, which was ascertained from clinic reports, medical records, Center for Medicare Services, and the National Death Index. Follow-up for mortality continued until death, transplantation, or the last follow-up date. Transplantation was a secondary outcome of interest and was ascertained from clinic reports and the United States Renal Data System.
Statistical Analyses
Baseline characteristics of the sample stratified by race and CRP tertile were compared using χ2 tests for categorical variables and t tests for continuous variables. Cox proportional hazards regression was used to examine the relationship between race, CRP tertile (and continuous CRP), and mortality, including interaction terms to test for effect modification. Survival time was calculated from 4 months after the date of dialysis initiation until death. In the primary analyses, participants were censored at last follow-up or kidney transplant.
Potential confounders were identified as those factors noted in the literature to be associated with race, inflammation, or mortality in dialysis patients. On the basis of this criterion, confounders included in the Cox models were age, gender, dialysis modality (hemodialysis versus peritoneal dialysis), traditional CVD risk factors (smoking status, body mass index, diabetes, BP, and cholesterol), prevalent CVD (including cerebrovascular and peripheral vascular disease), congestive heart failure, comorbidity score, hemoglobin, serum albumin, CRP, and IL-6 levels. Subgroup analyses of age, gender, CVD, diabetes, and dialysis modality groups were performed because extensive renal epidemiologic literature suggests that mortality often varies by these factors.
Sensitivity analyses were performed to test the robustness of our findings. First, analyses of mortality were performed while accounting for the competing event of transplantation using the method of Fine and Gray,40 because transplantation may be an informative censoring event. Second, IL-6 was examined as the primary inflammatory marker, and the relationship between race, IL-6 tertile, and mortality was analyzed in Cox proportional hazards models. Finally, analyses including adjustment for baseline vascular access status was conducted among the subset of patients for whom these data were known.
In all analyses, the possibility of confounding by dialysis clinic was controlled with fixed-effects modeling, clustered on clinic, which accounted for within-clinic correlation and between-clinic differences in outcomes.41 A two-sided P < 0.05 was used as the level of significance for all tests. Statistical analyses were performed using Stata IC, version 11 (StataCorp, College Station, TX).
DISCLOSURES
None.
Acknowledgments
We thank the patients, staff, laboratory, and medical directors of the participating clinics at Dialysis Clinic, Inc., and Saint Raphael's Hospital who contributed to the study.
This work was supported by Grants RO1 DK 59616 and RO1 DK 080123 from the National Institute of Diabetes and Digestive and Kidney Diseases, Grant RO1 HL 62985 from the National Heart, Lung and Blood Institute (Bethesda, MD), and Grant RO1 HS 08365 from the Agency for Health Care Research and Quality (Rockville, MD).
D.C.C. is supported by the Harold Amos Medical Faculty Development Program of the Robert Wood Johnson Foundation (Princeton, NJ). N.R.P. is supported, in part, by Grant K24 DK 02643 from the National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health (Bethesda, MD).
Footnotes
Published online ahead of print. Publication date available at www.jasn.org.
See related editorial, “Is the Malnutrition-Inflammation Complex the Secret behind Greater Survival of African-American Dialysis Patients?,” on pages 2150–2152.
REFERENCES
- 1. U.S. Renal Data System: USRDS 2010 annual data report: Atlas of end-stage renal disease in the United States, National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Disease; Bethesda, MD, 2010 [Google Scholar]
- 2. U.S. Census Bureau: 2006–2008 American Community Survey. Available at: http://factfinder.census.gov/ Accessed November 1, 2010
- 3. Owen WF, Jr, Chertow GM, Lazarus JM, Lowrie EG. Dose of hemodialysis and survival: Differences by race and sex. JAMA 280: 1764–1768, 1998 [DOI] [PubMed] [Google Scholar]
- 4. Frankenfield DL, Rocco MV, Frederick PR, Pugh J, McClellan WM, Owen WF, Jr: Racial/ethnic analysis of selected intermediate outcomes for hemodialysis patients: Results from the 1997 ESRD Core Indicators Project. Am J Kidney Dis 34: 721–730, 1999 [DOI] [PubMed] [Google Scholar]
- 5. Kinchen KS, Sadler J, Fink N, Brookmeyer R, Klag MJ, Levey AS, Powe NR: The timing of specialist evaluation in chronic kidney disease and mortality. Ann Intern Med 137: 479–486, 2002 [DOI] [PubMed] [Google Scholar]
- 6. Agodoa L, Eggers P: Racial and ethnic disparities in end-stage kidney failure-survival paradoxes in African-Americans. Semin Dial 20: 577–585, 2007 [DOI] [PubMed] [Google Scholar]
- 7. Kalantar-Zadeh K, Kovesdy CP, Derose SF, Horwich TB, Fonarow GC: Racial and survival paradoxes in chronic kidney disease. Nat Clin Pract Nephrol 3: 493–506, 2007 [DOI] [PubMed] [Google Scholar]
- 8. Arias E, Rostron BL, Tejada-Vera B: United States life tables, 2005. Natl Vital Stat Rep 58: 1–132, 2010 [PubMed] [Google Scholar]
- 9. Newsome BB, McClellan WM, Coffey CS, Allison JJ, Kiefe CI, Warnock DG: Survival advantage of black patients with kidney disease after acute myocardial infarction. Clin J Am Soc Nephrol Sep 1: 993–999, 2006 [DOI] [PubMed] [Google Scholar]
- 10. Powe NR: Reverse race and ethnic disparities in survival increase with severity of chronic kidney disease: What does this mean? Clin J Am Soc Nephrol 1: 905–906, 2006 [DOI] [PubMed] [Google Scholar]
- 11. Mehrotra R, Kermah D, Fried L, Adler S, Norris K: Racial differences in mortality among those with CKD. J Am Soc Nephrol 19: 1403–1410, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Choi AI, Rodriguez RA, Bacchetti P, Bertenthal D, Hernandez GT, O'Hare AM: White/black racial differences in risk of end-stage renal disease and death. Am J Med 122: 672–678, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Epstein AM, Ayanian JZ, Keogh JH, Noonan SJ, Armistead N, Cleary PD, Weissman JS, David-Kasdan JA, Carlson D, Fuller J, Marsh D, Conti RM. Racial disparities in access to renal transplantation: Clinically appropriate or due to underuse or overuse? N Engl J Med 343: 1537–1544, 2000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Gaylin DS, Held PJ, Port FK, Hunsicker LG, Wolfe RA, Kahan BD, Jones CA, Agodoa LY: The impact of comorbid and sociodemographic factors on access to renal transplantation. JAMA 269: 603–608, 1993 [PubMed] [Google Scholar]
- 15. Windgassen EB, Funtowicz L, Lunsford TN, Harris LA, Mulvagh SL: C-reactive protein and high-sensitivity C-reactive protein: An update for clinicians. Postgrad Med 123: 114–119, 2011 [DOI] [PubMed] [Google Scholar]
- 16. Kaptoge S, Di Angelantonio E, Lowe G, Pepys MB, Thompson SG, Collins R, Danesh J: C-reactive protein concentration and risk of coronary heart disease, stroke, and mortality: An individual participant meta-analysis. Lancet 375: 132–140, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Parekh RS, Plantinga LC, Kao WH, Meoni LA, Jaar BG, Fink NE, Powe NR, Coresh J, Klag MJ: The association of sudden cardiac death with inflammation and other traditional risk factors. Kidney Int 74: 1335–1342, 2008 [DOI] [PubMed] [Google Scholar]
- 18. Zimmermann J, Herrlinger S, Pruy A, Metzger T, Wanner C: Inflammation enhances cardiovascular risk and mortality in hemodialysis patients. Kidney Int 55: 648–658, 1999 [DOI] [PubMed] [Google Scholar]
- 19. Folsom AR, Aleksic N, Catellier D, Juneja HS, Wu KK: C-reactive protein and incident coronary heart disease in the Atherosclerosis Risk In Communities (ARIC) study. Am Heart J 144: 233–238, 2002 [DOI] [PubMed] [Google Scholar]
- 20. Robinson BM, Joffe MM, Pisoni RL, Port FK, Feldman HI: Revisiting survival differences by race and ethnicity among hemodialysis patients: The Dialysis Outcomes and Practice Patterns Study. J Am Soc Nephrol 17: 2910–2918, 2006 [DOI] [PubMed] [Google Scholar]
- 21. Streja E, Kovesdy CP, Molnar MZ, Norris KC, Greenland S, Nissenson AR, Kopple JD, Kalantar-Zadeh K: Role of nutritional status and inflammation in higher survival of African American and hispanic hemodialysis patients. Am J Kidney Dis 57: 883–893, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Myers OB, Adams C, Rohrscheib MR, Servilla KS, Miskulin D, Bedrick EJ, Zager PG: Age, race, diabetes, blood pressure, and mortality among hemodialysis patients. J Am Soc Nephrol 21: 1970–1978, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Young BA, Maynard C, Boyko EJ: Racial differences in diabetic nephropathy, cardiovascular disease, and mortality in a national population of veterans. Diabetes Care 26: 2392–2399, 2003 [DOI] [PubMed] [Google Scholar]
- 24. Noori N, Kovesdy CP, Dukkipati R, Feroze U, Molnar MZ, Bross R, Nissenson AR, Kopple JD, Norris KC, Kalantar-Zadeh K: Racial and ethnic differences in mortality of hemodialysis patients: Role of dietary and nutritional status and inflammation. Am J Nephrol 33: 157–167, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Parekh RS, Zhang L, Fivush BA, Klag MJ: Incidence of atherosclerosis by race in the dialysis morbidity and mortality study: A sample of the US ESRD population. J Am Soc Nephrol 16: 1420–1426, 2005 [DOI] [PubMed] [Google Scholar]
- 26. Young BA, Rudser K, Kestenbaum B, Seliger SL, Andress D, Boyko EJ: Racial and ethnic differences in incident myocardial infarction in end-stage renal disease patients: The USRDS. Kidney Int 69: 1691–1698, 2006 [DOI] [PubMed] [Google Scholar]
- 27. Buckalew VM, Jr., Freedman BI: Reappraisal of the impact of race on survival in patients on dialysis. Am J Kidney Dis 55: 1102–1110, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Alves TP, Wang X, Wright JT, Jr., Appel LJ, Greene T, Norris K, Lewis J. Rate of ESRD exceeds mortality among African Americans with hypertensive nephrosclerosis. J Am Soc Nephrol 21: 1361–1369, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Keith DS, Nichols GA, Gullion CM, Brown JB, Smith DH: Longitudinal follow-up and outcomes among a population with chronic kidney disease in a large managed care organization. Arch Intern Med 164: 659–663, 2004 [DOI] [PubMed] [Google Scholar]
- 30. Foley RN, Murray AM, Li S, Herzog CA, McBean AM, Eggers PW, Collins AJ: Chronic kidney disease and the risk for cardiovascular disease, renal replacement, and death in the United States Medicare population, 1998 to 1999. J Am Soc Nephrol 16: 489–495, 2005 [DOI] [PubMed] [Google Scholar]
- 31. Hemmelgarn BR, Manns BJ, Lloyd A, James MT, Klarenbach S, Quinn RR, Wiebe N, Tonelli M: Relation between kidney function, proteinuria, and adverse outcomes. JAMA 303: 423–429, 2010 [DOI] [PubMed] [Google Scholar]
- 32. Dalrymple LS, Katz R, Kestenbaum B, Shlipak MG, Sarnak MJ, Stehman-Breen C, Seliger S, Siscovick D, Newman AB, Fried L: Chronic kidney disease and the risk of end-stage renal disease versus death. J Gen Intern Med 26: 379–385, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Liu Y, Coresh J, Eustace JA, Longenecker JC, Jaar B, Fink NE, Tracy RP, Powe NR, Klag MJ: Association between cholesterol level and mortality in dialysis patients: Role of inflammation and malnutrition. JAMA 291: 451–459, 2004 [DOI] [PubMed] [Google Scholar]
- 34. Powe NR, Klag MJ, Sadler JH, Anderson GF, Bass EB, Briggs WA, Fink NE, Levey AS, Levin NW, Meyer KB, Rubin HR, Wu AW: Choices for healthy outcomes in caring for end stage renal disease. Semin Dialysis 9: 9–11, 1996 [Google Scholar]
- 35. Miskulin DC, Meyer KB, Athienites NV, Martin AA, Terrin N, Marsh JV, Fink NE, Coresh J, Powe NR, Klag MJ, Levey AS: Comorbidity and other factors associated with modality selection in incident dialysis patients: The CHOICE Study. Choices for Healthy Outcomes in Caring for End-Stage Renal Disease. Am J Kidney Dis 39: 324–336, 2002 [DOI] [PubMed] [Google Scholar]
- 36. Soucie JM, McClellan WM: Early death in dialysis patients: Risk factors and impact on incidence and mortality rates. J Am Soc Nephrol 7: 2169–2175, 1996 [DOI] [PubMed] [Google Scholar]
- 37. Astor BC, Eustace JA, Powe NR, Klag MJ, Fink NE, Coresh J: Type of vascular access and survival among incident hemodialysis patients: The Choices for Healthy Outcomes in Caring for ESRD (CHOICE) Study. J Am Soc Nephrol 16: 1449–1455, 2005 [DOI] [PubMed] [Google Scholar]
- 38. Athienites NV, Miskulin DC, Fernandez G, Bunnapradist S, Simon G, Landa M, Schmid CH, Greenfield S, Levey AS, Meyer KB: Comorbidity assessment in hemodialysis and peritoneal dialysis using the index of coexistent disease. Semin Dial 13: 320–326, 2000 [DOI] [PubMed] [Google Scholar]
- 39. Miskulin DC, Meyer KB, Martin AA, Fink NE, Coresh J, Powe NR, Klag MJ, Levey AS: Comorbidity and its change predict survival in incident dialysis patients. Am J Kidney Dis 41: 149–161, 2003 [DOI] [PubMed] [Google Scholar]
- 40. Fine JP, Gray RJ: A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc 94: 496–509, 1999 [Google Scholar]
- 41. Breslow NE, Day NE: Statistical methods in cancer research. Volume I: The analysis of case-control studies. IARC Sci Publ 32: 5–338, 1980 [PubMed] [Google Scholar]