Abstract
Nuclear medicine has become a key part of molecular imaging. In the present review article, we focus on the transbilayer phospholipids as exquisite targets for radiolabelled probes in molecular imaging. Asymmetry of phospholipid distribution is a characteristic of mammalian cell membranes. Phosphatidylcholine and sphyngomyelin cholinophospholipids are primarily located within the external leaflet of the cell membrane. Phosphatidylserine and phosphatidylethanolamine aminophospholipids, and also phosphatidylinositol are primarily located within the internal leaflet of the cell membrane. New radiolabelled tracers have been designed in preclinical and clinical research for PET-CT and SPECT-CT molecular imaging of transbilayer phospholipids.
Keywords: phospholipids, molecular imaging, PET-CT, SPECT-CT
Introduction
In this beginning of the twenty-first century, personalized molecular medicine is the objective of molecular diagnosis, molecular imaging, and molecular therapy [1,2]. Molecular imaging (MI) includes a number of morphological imaging techniques (i.e. ultrasound, computed tomography and magnetic resonance imaging), and optical imaging techniques (i.e. bioluminescence and fluorescence imaging) [3,4]. In addition, nuclear medicine with radiolabelled probes has become a key part of MI, especially with 18F-fluorodeoxyglucose (18F-FDG) metabolic imaging [5]. New radiolabelled tracers have been designed for positron emission tomography-computed tomography (PET-CT) and single-photon emission computed tomography-computed tomography (SPECT-CT) molecular imaging [6]. In this review article, we focus on the transbilayer phospholipids as exquisite targets for radiolabelled probes in molecular imaging.
Molecular imaging
There is no universally accepted definition of molecular imaging [5,7]. In 2000, the Society of Molecular Imaging http://www.molecularimaging.org/ defined molecular imaging as: 'the characterization and measurement of biological processes in living animals at the cellular and molecular level'. In 2005, the European Society for Molecular Imaging http://www.e-smi.eu formulated a definition of molecular imaging as: 'the characterisation of the dynamics of the molecular processes in the living organisms in vivo. In vivo molecular imaging is a science combining molecular biology, cellular biology and physiology with imaging in living subjects'. In 2006, the Federation of Asian Societies for Molecular Imaging (FASMI: http://fasmi.org/) defined molecular imaging as: ' the characterization and measurement of biological processes in living animals at the cellular and molecular level by means of non-invasive (or minimally invasive) imaging'. In 2007, the Society of Nuclear Medicine Molecular Imaging Center of Excellence http://interactive.snm.org/ definitions task force approved this definition of molecular imaging as: 'the visualization, characterization, and measurement of biological processes at the molecular and cellular levels in humans and other living systems' [8]. A MI probe is a molecule used in molecular imaging to deliver a tracer to a specific organ or tissue. A probe typically consists of a ligand containing or linked to a signalling label. The label provides the signal (i.e. electromagnetic wave, light and radiation) that can be picked up by a detector, and the ligand carries the tracer to the site of interest [9]. A MI target used in molecular imaging is a molecule or structure in the body to which binds a probe delivered to a specific organ or tissue. The target may be a peptide, or a glucide, or a lipid; in many cases, the target is a protein [10,11]. Molecular imaging may be a single disease/gene or a general disease/biologic function control point for targeting [12].
Molecular imaging with transbilayer phospholipid targets may be performed with radiolabelled probes such as radiolabelled annexin V or C2A synaptotagmin domain I or beta 2 glycoprotein I, radiolabelled duramycin, radiolabelled hypericin, radiolabelled lactadherin, radiolabelled choline or fluorocholine, radiolabelled diacylglycerols, radiolabelled sphyngomyelin for visualization, characterization and measurement of key biological functions (i.e. apoptosis, necrosis, thrombosis, vasculature endothelium, choline metabolism, myocardial and neuronal phosphoinositide turnover) or for assessing specific diseases (i.e. cancers, immune diseases, inflammatory diseases, infectious diseases, cardiac diseases and neurological diseases).
Membrane bilayer
The membrane bilayer is composed of 40% lipids and glycolipids, and 60% integral proteins and glycoproteins [13]. The lipids in the membrane bilayer are composed of phospholipids (75% to 88%), glycosphyngolipids (2% to 5%) and cholesterol (10% to 20%) [13]. The phospholipids include phosphatidylcholine (45% to 55%), phosphatidylethanolamine (15% to 25%), phosphatidylinositol (10% to 15%), phosphatidylserine (2% to 10%), phosphatidic acid (1% to 2%), sphyngomyelin (5% to 10%) and cardiolipin (2% to 5%). Liposomes are artificial lipid vesicles encapsulating drugs (e.g. chemotherapy drugs, antibiotics, fungicides), enzymes, biological material (e.g. antigens, antibodies) and tracers (e.g. radiolabelled products, contrast agents) [14]. Liposomes are nanoparticles with a diameter < 100 nm characterised by the composition of lipids, the number of membrane bilayers, and the surface charges [7]. The material encapsulated is either dissolved in an aqueous phase or in a lipid phase. Radioactive phospholipid liposomes have been designed for molecular imaging [15].
Phospholipid bilayer
In 1972, Singer and Nicolson developed the fluid mosaic model to explain the composition of the cell membrane bilayer with randomly oriented globular proteins and lipids [16]. According to this thermodynamic model, the phospholipid membrane bilayer is composed of hydrophilic heads and hydrophobic tails. The polar hydrophilic heads are in contact with water (see Figure 1). Recent refinements of the Singer-Nicolson mosaic model suggest a structured dynamic organisation of the membrane bilayer with genetically predefined non-randomly oriented globular proteins into the lipid matrix [17,18]. In mammalians, the asymmetry of phospholipid distribution is a characteristic of cell membranes (see Figure 2). Phosphatidylcholine (PC) and sphyngomyelin (SM) cholinophospholipids are primarily located in the extracellular membrane. Phosphatidylethanolamine (PE) and phosphatidylserine (PS) aminophospholipids as well as phosphatidylinositol (PI) phospholipid are primarily located in the intracellular membrane [19]. Two ATP-dependent translocase enzymes (i.e. cholinophospholipid-specific translocase or floppase, and aminophospholipid-specific translocase or flippase) and a Mg2+-ATPase (i.e. aminophospholipid translocase) maintain the aminophospholipids in the inner leaflet of the membrane bilayer. PE and PS are externalised to the outer leaflet of the cell membrane after inactivation of aminophospholipid translocase or flippase and activation of scramblase, another Ca2+-dependent ATP-independent enzyme [19,20].
Figure 1.
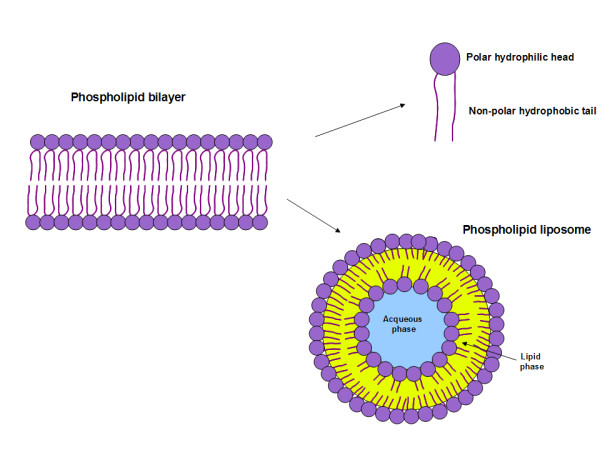
Phospholipid bilayer composed of hydrophobic non-polar tails and hydrophilic polar heads. Liposomes are artificial phospholipid vesicles encapsulating materials either in an aqueous phase or in a lipid phase.
Figure 2.
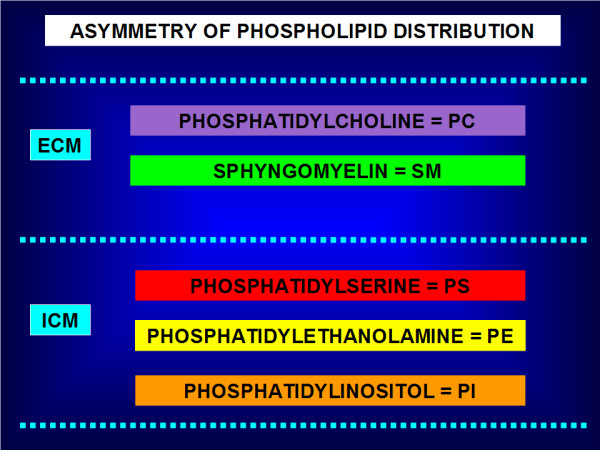
Asymmetry of phospholipid distribution. Phosphatidylcholine (PC) and sphyngomyelin (SM) cholinophospholipids are primarily located in the external leaflet of the cell membrane (ECM). Phosphatidylserine (PS) and phosphatidylethanolamine (PE) aminophospholipids as well as phosphatidylinositol (PI) phospholipid are primarily located in the internal leaflet of the cell membrane (ICM). In mammalians, the asymmetry of phospholipid distribution is maintained by the flip/flop movement of phospholipids. PS and PE are maintained to the inner leaflet of the membrane bilayer by the Mg2+-ATPase (i.e. aminophospholipid translocase), and the ATP-dependent aminophospholipid-specific flippase and cholinophospholipid-specific floppase (i.e. multidrug resistance proteins). In pathophysiological conditions, PS and PE are shuttled to the outer leaflet after inactivation of the aminophospholipid translocase or flippase, and activation of the Ca2+-dependent ATP-independent scramblase.
Phospholipid targets
Phospholipids are composed of a phosphatidyl tail including a free fatty acid, glycerol and phosphate [21]. In the phospholipid polar head, choline, serine, ethanolamine and inositol bind to the phosphatidyl non-polar tail. Sphingomyelin is the only one cell membrane phospholipid not derived from a glycerol but from an aminoalcohol sphingosine; SM is composed of a core ceramide (i.e. sphingosine and free fatty acid tail) and contains a polar head group composed of phosphocholine or phosphoethanolamine [22].
In preclinical and clinical research, phospholipids are used as targets for molecular imaging with radiolabelled probes as illustrated in Figure 3[23,24].
Figure 3.
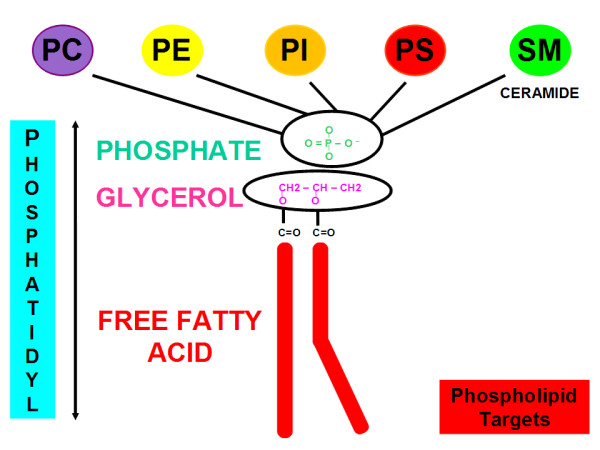
Phospholipid targets are composed of a phosphatidyl tail including free fatty acid, glycerol, and phosphate. In the phospholipid polar head, choline, serine, ethanolamine, and inositol bind to the phosphatidyl non-polar tail. Sphingomyelin is the only one cell membrane phospholipid not derived from a glycerol but from an aminoalcohol sphingosine; SM is composed of a core ceramide (i.e. sphingosine and free fatty acid tail) and contains a polar head group composed of phosphocholine or phosphoethanolamine.
PS target
PS is an aminophospholipid located in the inner leaflet of the cell membrane. In pathophysiological conditions including apoptosis, thrombosis and tumour vasculature, PS is externalised from the inner leaflet to the outer leaflet of the cell membrane [25]. In necrosis, PS is exposed in the inner leaflet of damaged cells. For SPECT-CT and PET-CT imaging, radiolabelled probes have been designed to target exposed PS with or without the breakdown of cell membrane asymmetry.
Annexin V, a 36-kDa serum protein first isolated from human placenta, has been radiolabelled with 99mTc, 111In, 67Ga, 123I, 124I, 11C, 64Cu, 68Ga, 94mTc and 18F for imaging of apoptosis or necrosis, and thrombosis [26-29]. Annexin V binds specifically to PS in presence of Ca2+ ions. Spontaneous and induced apoptotic changes may be assessed qualitatively and quantitatively with radiolabelled annexin V-targeted to PS [30,31]. In preclinical research, 99mTc-HYNIC-annexin V imaging has been used to evaluate cardiovascular models (e.g. heart transplant rejection, myocarditis and mural thrombus), oncology models (e.g. cyclophosphamide induced intramedullary and splenic apoptosis in lymphoma), neurology models (e.g. neonatal brain ischemia), inflammatory/infectious models (e.g. fulminant hepatitis, subacute and acute infection) and immune models (e.g. rheumatoid arthritis treated by corticosteroids). In clinical research, 99mTc-HYNIC-annexin V imaging using a recombinant human form of annexin V has been used to assess myocardial infarction, heart transplant rejection, atherosclerosis, ischemic pre-conditioning and chemotherapy/radiation therapy-induced apoptosis in head and neck cancers, lymphomas, lung cancers, breast cancers, leukaemia and soft tissue sarcomas. 99mTc-HYNIC-annexin V imaging has also been used in patients with dementia and stroke, and in patients with rheumatoid arthritis and Crohn's disease for evaluation of early apoptotic response to Infliximab® therapy (i.e. anti-TNFα monoclonal antibody) [24,26]. Figures 4 and 5 illustrate PS targeting 99mTc-HYNIC-annexin V molecular imaging probe in a patient with a non-small cell lung cancer (NSCLC). PS targeting 99mTc-annexin V-128 with an N-terminal site specific for endogenous binding of technetium-99m has been designed in order to improve the sensitivity of detection of apoptosis or necrosis (i.e. twofold higher than that of 99mTc-HYNIC-annexin V) [32].
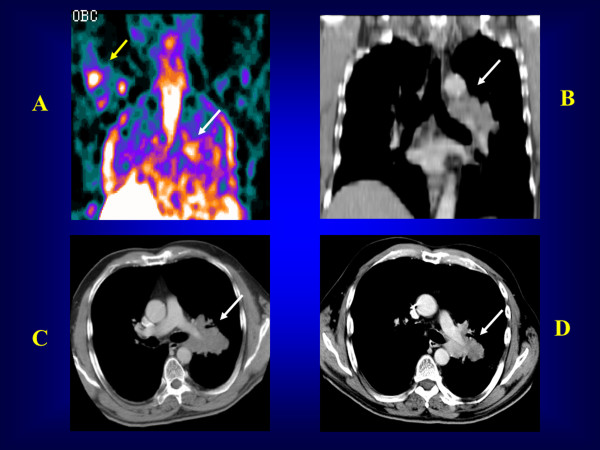
Figure 4.
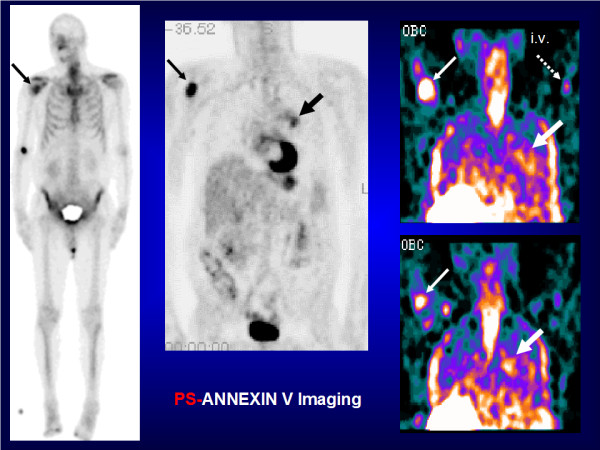
PS-annexin V imaging (North American Scientific 2020 - Phase II-III - Clinical Trial - Antwerp 2003). A patient with a left para-hilar NSCLC (large black arrow) who presented with a bone metastasis in the right upper humerus (fine black arrow) on bone scan and 18FDG PET. 99mTc-HYNIC-annexin V imaging showed tracer uptake in the primary tumour (large white arrow) and the bone metastasis (fine white arrow) as soon as 24 h after the first course of chemotherapy. i.v., intravenous injection (dashed arrow).
Figure 5.
PS-annexin V imaging (North American Scientific 2020 - Phase II-III Clinical Trial - Antwerp 2003). A patient with a left para-hilar NSCLC on pre-chemotherapy CT (upper right: simple white arrow) who presented with 99mTc-HYNIC-annexin V uptake post-chemotherapy in the primary lung tumour (upper left: simple white arrow) and the right upper humerus bone metastasis (upper left: yellow arrow). After the first course of chemotherapy, diagnostic CTs showed a decrease in the primary lung tumour greatest diameter, which was consistent with an apoptotic tumour response to chemotherapy (lower left and lower right: simple white arrows). Upper left (A): post-chemotherapy 99mTc-HYNIC-annexin V scan; upper right (B) and lower left (C): pre-chemotherapy diagnostic CTs; lower right (D): post-chemotherapy diagnostic CT.
C2A domain of synaptotagmin I (C2A) is a 12-kDa protein predominantly located within the synaptic vesicles binding to PS. It has been fused with glutathione-s-transferase (GST) and radiolabelled with 99mTc for imaging of apoptosis or necrosis [33]. In a rat and pig model of acute myocardial infarction, increased 99mTc-C2A-GST uptake was seen in the myocardial infarct area at risk, and was associated with wall dysfunction [34-36]. In a mouse model of NSCLC, increased 99mTc-C2A-GST tumour uptake was noted after paclitaxel chemotherapy-induced apoptosis [37]. Also, C2A-GST has been easily labelled with 18F for early imaging of apoptosis after chemotherapy [38]. In a rabbit model of lung cancer, increased 18F-C2A-GST tumour uptake was detected 72 h after paclitaxel chemotherapy with increased apoptotic index and caspase-3 activity. Recently, the C2A domain of synaptotagmin I has been labelled with a [99mTc(CO)3+] core by using an efficient C-terminal site-specific radiolabelling method for the imaging of cell death [39].
PS is not exposed in normal endothelium, but increased exposure of PS is seen on the tumour endothelium vasculature [40,41]. Bavituximab, a chimeric monoclonal antibody (MW = 145.3 kDa) binding to the beta-2 glycoprotein I domain of PS, has been radiolabelled with the β + emitter arsenicum-74 (74As, T1/2 = 17.8 days) for tumour vasculature PET imaging, and with the β-,γ emitter 77As (T1/2 = 38.8 h) for SPECT imaging and potential tumour vasculature endothelium therapy [42-44]. Increased 74As-bavituximab uptake was seen in a rat model of prostate cancer with the highest tumour-to-background activity ratio at 72 h post-i.v injection (at 72 h p.i., tumour-to-liver ratio = 22, and tumour-to-muscle ratio = 470). Using autoradiography and immunohistochemical studies, 74As-bavituximab was found to specifically bind to the tumour endothelium vasculature. Bavituximab radiolabelled with 77As or 76As (β-, T1/2 = 26.3 h) may be used for dosimetry and immunotherapy.
Hypericin, a non-porphyrin necrosis agent (MW = 504 Da) extracted from St John's wort, has been labelled with 64Cu (β+, T1/2 = 12.7 h) for PET imaging of necrosis. In a female mouse model of BT474 breast xenograft tumour, 64Cu-bis-DOTA-hypericin demonstrated increased uptake at 24 h post-i.v. injection in necrotic injured tissues treated with near infrared photothermal ablation therapy. PET distribution also showed higher uptake in the liver and the kidneys. Bis-DOTA-hypericin had a selective binding affinity for PS and PE phospholipids [45]. In necrosis, PS and PE are exposed to 64Cu-labelled hypericin probe in the inner leaflet of damaged cell membrane. Hypericin derivatives are efficient and yield reproducible results when radiolabelled with 123I (i.e. mono-123I-iodohypericin and mono-123I-iodohypericin monocarboxylic acid). They have been also used in preclinical models of liver necrosis and myocardial infarction as well as in clinical correlates of these pathophysiologic states [46,47]. In a preclinical rat model of liver rhabdomyosarcoma, 131I-labelled hypericin was successfully used in theragnostics with a vascular disrupting agent (i.e. combretastatin A4 phosphate or CA4P); high radiolabelling efficiency was noted with minimum deiodination. 131I-hypericin uptake colocalised tumour necrosis within 24 h post-i.v. injection on co-registered planar γ scintigraphy with CT, MRI, histology and autoradiography [48]. Hypericin has also been labelled with 99mTc (i.e. 99mTc-hypericin or 99mTc-mercaptoacetyldiglycyl-1,5-diaminopentylene hypericin-carboxamide) for visualisation of necrotic tissues in rats with reperfused liver infarct [49]. 99mTc-hypericin, however, was found to be not suitable for imaging of necrosis compared to 123I-hypericin derivatives [50]. Although the mechanism of target uptake is unknown, it may be hypothesised that the PS and PE phospholipid targets are involved for 123I-, 131I- and 99mTc-labelled hypericin derivatives, but this is still to be definitely proven.
Lactadherin, a glycoprotein secreted by mammary epithelium, epididymal epithelium, vascular cells and activated macrophages, has been shown to bind in a Ca2+-independent manner and specifically to the C2 domain of PS [51-53]. In vitro, FITC-labelled bovine lactadherin has been used for early detection of apoptosis in leukaemia cell lines treated by etoposide, and also in HeLa cervical cancer lines treated by staurosporine [54,55]. HYNIC-lactadherin has also been successfully labelled with 99mTc (i.e. 99mTc-HYNIC-lactadherin). In vivo, in a mouse model, 99mTc-HYNIC-lactadherin had a lower uptake in the kidneys compared to 99mTc-HYNIC annexin V. However, this new PS-targeting probe had a higher uptake in the liver, which is a disadvantage for imaging of liver and myocardial apoptosis [56].
PSBP-6 or PS-binding peptide-6, a synthetic 14 amino acid sequence (MW = 1.623 kDa; Kd = 100 nM) where Gln is replaced by Ala screened through a peptide library, showed the highest relative binding affinity and stability; a single amino acid chelator (SAAC) was introduced at the N-terminus position of PSBP-6 to form a stable complex with 99mTc (i.e. SAAC-99mTc-PSBP-6), and to improve the labelling efficiency. Unlike annexin V, SAAC-PSBP-6 binding to PS is Ca2+ independent [57]. In DLD1 human colon carcinoma cells pretreated with increased TRAIL doses and also in murine melanoma B16/F10 cells treated with poly(L-glutamic acid) paclitaxel (PG-TXL), SAAC-(99mTc)-PSBP-6 showed selective binding to apoptotic cells in vitro; confirmation was obtained with TUNEL staining and autoradiography. In treated and untreated nude mice, biodistribution data showed increased SAAC-(99mTc)-PSBP-6 uptake in the liver and the kidney in vivo. Preclinical studies are required to assess the sensitivity and specificity of this new PS binding molecular imaging probe. Table 1 summarises radiolabelled PS-based molecular imaging probes.
Table 1.
Phosphatidylserine-based radiolabelled molecular imaging probes
| Molecular target | Molecular probes | AA | MW (kDa) | Affinity (M) | Biological functions | Isotopes | Imaging techniques |
|---|---|---|---|---|---|---|---|
| PS a | annexin V | 319 | 36 | 10-9 | apoptosis, necrosis, thrombosis | 99mTc, 123I, 111In, 67Ga, 18F, 68Ga, 124I, 64Cu, 94mTc | SPECT(CT), PET(CT) |
| PS | C2A domain of synaptotagmin I | 128 | 12 | 10-9 | apoptosis, necrosis | 99mTc | SPECT(CT) |
| 18F | PET(CT) | ||||||
| PS b | bavituximab chimeric antibody | - | 145.3 | 10-9 | tumour endothelium vasculature imaging | 74As c | PET(CT) |
| tumour endothelium vasculature therapy | 77As d | SPECT(CT) | |||||
| PS | hypericin | - | 0.504 | - | necrosis |
99mTc, 123I, 131I 64Cu e |
SPECT(CT) PET(CT) |
| PS | lactadherin | 364 | 47 - 50 | 10-9 | apoptosis | 99mTc f | SPECT(CT) |
| PS | PSBP-6 | 14 | 1.623 | 10-9 | apoptosis, necrosis | 99mTc g | SPECT(CT) |
a Various linkers have been used for the radiolabelling of annexin V [26]. HYNIC is the most used bifunctional chelating agent in preclinical and clinical research. b β2 glycoprotein I domain of phosphatidylserine. c 74As, β + emitter (T1/2 = 17.8 days). d 77As, β- emitter (T1/2 = 1.6 days). e Bis-DOTA has been used as a bifunctional chelating agent for radiolabelling of hypericin. f HYNIC has been used as a bifunctional chelating agent for radiolabelling of lactadherin. g SAAC has been used as a single amino acid chelator for radiolabelling of PSBP-6. AA, amino acids; MW, molecular weight; PET(CT), positron emission tomography (computed tomography); PS, phosphatidylserine; PSBP-6, PS-binding peptide-6; SPECT(CT), single-photon emission computed tomography (computed tomography).
PC target
Choline is a main component of biomembranes targeted by the choline kinase enzyme, and is phosphorylated to intracellular phosphocholine and extracellular phosphatidylcholine [58]. Choline has been labelled with 11C (i.e. 11C-choline) and more recently with 18F (i.e. 18F-fluorocholine) for PET-CT imaging [59,60]. With a longer physical half-life, 18F-fluorocholine (18F-FCH; 18F, T1/2 = 109 min) is more suitable than 11C-choline (11C, T1/2 = 20 min) for PET-CT imaging. In human subjects, 18F-FCH showed a fast blood pool clearance with a peak ≤ 5 min post-i.v. injection, a fast tissue uptake and a predominantly renal excretion. Additionally, 18F-FCH biodistribution changes very slowly later than 10 min after i.v. injection [61].
18F-fluorocholine (18F-FCH) and 11C-choline are incorporated into the membrane phospholipids, and are predominantly used for prostate cancer imaging and brain tumour imaging [62,63]. In prostate cancer patients, 18F-FCH is a promising tracer for detection of primary prostate cancer, staging of lymph node and bone metastases, and detection of recurrence after definitive therapy [62-64]. In brain tumours, this agent may be useful to detect glioblastoma multiforme, to distinguish high-grade gliomas with a characteristic peri-tumoural uptake from metastases and benign lesions, to guide a stereotactic biopsy, and also to differentiate post-radiation necrosis from recurrence [65-68]. Also, 11C-choline and 18F-FCH have been used for hepatocellular carcinoma (HCC) imaging [69-71]. In a woodchuck model, 11C-choline sensitivity for detection of well-differentiated HCCs was higher than that of 18F-FDG, and in 12 patients with moderately differentiated HCCs, the 11C-choline detection rate was better than that of 18F-FDG. 18F-FCH imaging has also successfully been performed for detection of well-differentiated HCC in patients with liver nodules or cirrhosis or chronic liver disease, and for detection of recurrences from HCC [72,73].
Under normal glucose conditions, acetate free fatty acid is metabolised into acetyl-CoA by acetyl-CoA synthetase in both the cytosol and mitochondria for synthesis of cholesterol and fatty acids [74]. In tumours, acetate is metabolised into fatty acids by fatty acid synthetase, which is overexpressed in cancer cells [75]. Acetate is mainly incorporated in the PC microdomains that play a major role for growth and metastasis [76]. Acetoacetate ketone body is metabolised to acetoacetyl-CoA and to acetyl-CoA, and then incorporated into the metabolic pathway for lipid synthesis [77]. Like acetate, acetoacetate could be incorporated into the PC membrane. Acetate and acetoacetate have been labelled with 11C (i.e. 11C-acetate and 11C-acetoacetate) [74,77]. Acetate has also been labelled with 18F (i.e. 18F-fluoroacetate) [78]. 11C-acetate has a rapid blood clearance with a high accumulation in liver and myocardium. In normal physiology, 18F-fluoroacetate is not a functional analogue of 11C-acetate; 18F-fluoroacetate has a longer blood half-life, a rapid liver clearance and low myocardial uptake, extensive excretion into bile and urine, and suffers from skeletal defluorination resulting in skeletal uptake of the formed 18F-fluoride [79]. In preclinical and clinical research, 11C-acetate PET imaging has been successfully used for tumour imaging in cancers of the prostate, kidney, pancreas, as well as gliomas, meningiomas and HCC [80,81]. In preclinical research, 18F-fluoroacetate has been used for imaging of gliosis in glioblastoma, stroke, and ischemia-hypoxia associated with neuroinflammation [82]. In CWR22 tumour-bearing male nu/nu mice, 18F-fluoroacetate has been successfully used for prostate cancer imaging with a higher tumour-to-prostate activity ratio compared to 11C-acetate at 30 min and 2 h post-i.v. injection. In a patient with prostate cancer, 18F-fluoroacetate was also able to detect several but not all bone metastases [83,84]. In other preclinical research models of breast and prostate cancers, 11C-acetoacetate uptake tended to be higher than that of 11C-acetate [85]. Assessment of brain ketone metabolism in rats indicated a 7 to 8 fold increase in 11C-acetoacetate under ketone dietary treatment [86,87]. Table 2 summarises the radiolabelled PC-based molecular imaging probes.
Table 2.
Phosphatidylcholine-based radiolabelled molecular imaging probes
| Molecular target | Molecular probes | Biological functions | Isotopes | Imaging techniques |
|---|---|---|---|---|
| PC | choline | choline metabolism | 11C | PET(CT) |
| PC | fluorocholine | choline metabolism | 18F | PET(CT) |
| PC | acetate | acetate metabolism | 11C | PET(CT) |
| PC | fluoroacetate | acetate metabolism | 18F | PET(CT) |
| PC | acetoacetate | acetoacetate metabolism | 11C | PET(CT) |
PC, phosphatidylcholine; PET(CT), positron emission tomography (computed tomography).
PE target
PE is primarily located in the inner leaflet of the membrane bilayer [88]. Like PS, this major aminophospholipid is externalised from the inner leaflet to the outer leaflet of the cell membrane during apoptosis and tumour vascular endothelium [89,90]. In necrosis, PE is exposed in the inner leaflet of disintegrated cell membrane.
Duramycin, a 2-kDa small protein produced by Streptovercillium cinnamoneus, has been designed in order to target PE for imaging of apoptosis or necrosis [91]. 99mTc-HYNIC-duramycin has been successfully used in a rat model of ischemia-reperfusion for imaging of myocardial infarction cell death [92]. 99mTc-duramycin is highly stable in vivo, and has favourable pharmacokinetics for early imaging of acute cardiac cell death at 10 min post-i.v. injection with a fast blood clearance, a low liver uptake and a high uptake in apoptotic and necrotic myocardium. Figure 6 illustrates the PE targeting 99mTc-duramycin molecular imaging probe in a rat model of myocardial infarction with correlation by autoradiography.
Figure 6.
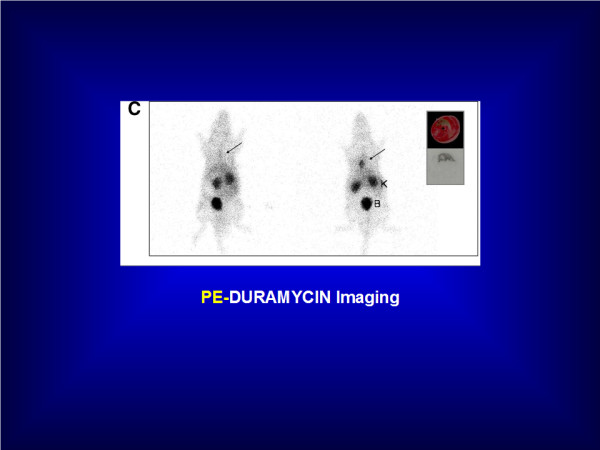
PE-duramycin imaging. Increased 99mTc-Duramycin uptake is seen in acute myocardial infarction of the rat at 120 min post-i.v. injection with excellent infarct-to-non infarct activity ratio (right). Autoradiography confirmed radioactivity uptake within the myocardial infarction. No tracer uptake is seen in the healthy rat (left). B, bladder; K, kidney. Courtesy of Pr. Zhao M (Department of Biophysics, Medical College of Wisconsin, Milwaukee, WI, USA). Previously published in the J Nucl Med 2008, 49(8):1345-52.
In addition to this, radiolabelled ethanolamine (i.e. ethanolamine labelled with 11C or 18F) is a potential new probe in oncology PET-CT imaging for assessment of tumour proliferation [93]. 14C-ethanolamine has been used in a variety of tumour cell types (i.e. melanoma, prostate cancer, glioblastoma multiforme, diffuse large B-cell lymphoma, colorectal adenocarcinoma). 14C-ethanolamine is incorporated into PE and has a two- to sevenfold significantly better uptake into tumour cell types than 14C-choline. In an in vitro model of cultured prostate cancer cells, 14C-ethanolamine and 14C-N, N'-dimethyl-ethanolamine uptake was two- to fourfold better in androgen-dependent and proliferating PC3 cells compared to androgen-independent and growth-arrested LnCap cells.
Hypericin, a PE- and PS-targeting probe, has been radiolabelled with 64Cu (i.e. 64Cu-bis-DOTA-hypericin), 123I (i.e. mono-123I-iodohypericin and mono-123I-iodohypericin monocarboxylic acid), 131I (i.e. 131I-hypericin) and 99mTc (i.e. 99mTc-hypericin) for imaging of necrosis in preclinical and clinical research as described above in the PS target section [45-50]. Table 3 summarises the PE-based radiolabelled molecular probes.
Table 3.
Phosphatidylethanolamine-based radiolabelled molecular imaging probes
| Molecular target | Molecular probes | AA | MW (kDa) | Affinity (M) | Biological functions | Isotopes | Imaging techniques |
|---|---|---|---|---|---|---|---|
| PE | duramycin | 19 | 2 | 10-9 | apoptosis necrosis |
99mTc a | SPECT(CT) |
| PE | ethanolamine N'N-dimethyl ethanolamine |
- | - | - | ethanolamine metabolism (tumour proliferation) | 14C b | PET(CT) |
| PE c | hypericin | - | 0.504 | - | necrosis | 123I, 131I 99mTc | SPECT(CT) |
| 64Cu d | PET(CT) |
a HYNIC has been used as a bifunctional chelating agent for the radiolabelling of duramycin. b ethanolamine or N,N'-dimethylethanolamine labelled with 14C in preclinical in vitro research may be labelled with 11C or 18F for PET(CT) in vivo imaging. c 64Cu-bis-DOTA-hypericin has a selective binding affinity for PS and PE (ref.45); it may be hypothesised that 123I-, 131I- and 99mTc-labelled hypericin derivatives bind to PS and PE. d bis-DOTA has been used as a bifunctional chelating agent for the radiolabelling of hypericin. AA, amino acids; MW, molecular weight; PE, phosphatidylethanolamine; PET(CT), positron emission tomography (computed tomography); SPECT(CT), single-photon emission computed tomography (computed tomography).
PI target
PI phospholipid is located in the inner leaflet of the membrane bilayer [94]. 1,2 Diacylglycerol (DAG) is metabolised to intermediate phosphoinositide metabolites (i.e. PA, PIP, PIP2, PIP3) including phosphatidylinositol (PI) [95]. DAG activates the intracellular protein kinase C (PKC) transduction signalling pathway, which is involved in higher cortical functions (e.g. memory, learning) and cardiac functions (e.g. hypertrophic growth, ventricular remodelling).
Intact 11C-inositol (i.e. non-acetylated inositol) has been suggested as a diagnostic agent in PET imaging for evaluation of PI brain metabolism and its role as second messenger [96]. In a rat and monkey model, 1-[1-11C]-butyryl-2-palmitoyl-rac-glycerol or 11C-DAG has been designed as a PI-targeting probe in PET brain imaging to visualise neuronal PI turnover [97,98]. 11C-DAG levels are low in the brain, and thus it may be appropriately used to visualise PI metabolism in the central nervous system. Dynamic PET imaging showed increased 11C-DAG uptake in the first 15 min with an equilibrium at 16 min in pre-stimulation and post-stimulation conditions, which is suggestive of a membrane trapping mechanism. 11C-DAG uptake increased 20% to 30% after arecoline stimulation (i.e. acetylcholine muscarinic receptor stimulation), which suggests that 11C-DAG is a tracer of the transduction signalling PI-mediated pathway. In resting conditions, 11C-DAG uptake was observed in the visual association area, and increased in the whole brain and the occipital areas after arecoline stimulation. In C6 glioma cells implanted in the rat brain, 11C-DAG was rapidly incorporated in the PI turnover within 5 min post-i.v injection; in a patient with astrocytoma grade III, 11C-DAG was gradually incorporated in the leading PI turnover and the PE-PC secondary pool with an equilibrium period at 32-40 min post-iv. injection [99]. In patients with Alzheimer's disease and ischemic stroke, 11C-DAG evaluated the PI cortical function and neural viability, respectively. In eight patients with Alzheimer's disease, 18F-FDG PET imaging showed bilateral hypometabolism in the parieto-temporal association areas, while 11C-DAG imaging showed spotty uptake in the frontal lobes of the brain suggestive of compensatory plastic process in non-damaged neural circuits to degenerative cognitive impairment [100]. In five patients with cerebral infarction, dynamic PET brain imaging showed decreased 11C-DAG uptake in comparison to normal cortex [101]. 11C-DAG pharmacokinetics demonstrated rapid decrease in the plasma with a peak at 40 s post-i.v. injection, and gradual increase in the brain to reach a plateau at 15 to 20 min post-i.v. injection. Reflecting neural signal transduction activity, the incorporation constant of 11C-DAG (k*DAG) was best correlated with the cerebral metabolic rate of O2 (CMRO2). Maintained PI metabolism suggested preservation of neural viability in the peri-infarct area of the ischemic stroke. Patients with subacute local brain injury, either ischemic stroke or brain tumour, also exhibited 11C-DAG spots located in the associative areas distant from the lesion between 2 weeks and 1 month after injury; one of the possible features of neural recovery in the intact brain related to PI metabolism in PET imaging [102].
In a rat model, 11C-DAG has also been used for assessing the myocardial PI turnover [103]. 11C-DAG incorporation into intermediate PI metabolites was seen both in the infarcted and the non-infarcted myocardium at 7 days after myocardial infarction. Furthermore, 11C-DAG has been used to evaluate the PI-mediated angiotensin II signalling pathway. In a rat model of infarcted myocardium, 11C-DAG assessed the effect of angiotensin-converting enzyme inhibitor (i.e. captropril) on activated PI metabolism in order to reduce left ventricular remodelling in the early phase of myocardial infarction [104]. After 3 weeks of treatment with captropril, a significant decrease of 11C-DAG uptake was seen in the infarcted myocardium compared to the non-infarcted myocardium, while 201Tl imaging showed decreased uptake in the infarcted myocardium with and without captopril treatment. In patients with myocardial infarction, 11C-DAG has been used to evaluate the PI myocardial turnover and the left ventricular remodelling [105]. In 13 patients with myocardial infarction, 11C-DAG was significantly correlated with the myocardium-to-left atrium chamber ratio, the left ventricle ejection fraction, and the brain natriuretic peptide concentration. Significant increase of 11C-DAG uptake was seen in the remote viable region of myocardial infarction patients compared to healthy subjects. DAG has also been labelled with 18F to trace the PI metabolism into the ventricular myocardium [106]. 18F has a longer half-life, and 18F-DAG may be more useful for clinical routine than 11C-DAG. In the rat myocardium, 3, 5, 10, and 30 min after i.v. injection of 1-[4-[18F]fluorobutyryl]-2-palmitoylglycerol (C4, C16), 18F-DAG reflected the myocardium PI metabolism in the early phase (3 to 5 min); in the late phase (≥ 30 min) after i.v. injection,18F-DAG may also reflect the PE metabolism. Quantification of PI metabolism requires further optimization with a 18F-radiolabelled DAG compound closer to the PI metabolism than 18F-DAG (C4, C16) [106]. Table 4 summarises the PI-based radiolabelled molecular imaging probes.
Table 4.
Phosphatidylinositol-based radiolabelled molecular imaging probes
| Molecular target | Molecular probes | Biological functions | Isotopes | Imaging techniques |
|---|---|---|---|---|
| PI | inositol a | PI brain metabolism | 11C | PET(CT) |
| PI | DAG | PI myocardial turnover | 11C, 18F | PET(CT) |
| PI | DAG | PI neuronal turnover | 11C | PET(CT) |
a intact inositol: no acetylated analogues. DAG, diacylglycerol; PET(CT), positron emission tomography (computed tomography); PI, phosphatidylinositol.
SM target
SM is a cholinophospholipid located in the outer leaflet of the membrane bilayer [19]. Liposomes composed of SM have been experimentally used for tumour imaging. In a mouse tumour model, serum stable SM liposomes-encapsulating 67Ga prepared in a lipid phase with cholesterol (SM/cholesterol molar ratio, 2:1) enhanced blood circulation and increased 67Ga delivery to the tumour [107]. Tumour-to-blood activity ratio (T/B) and tumour index (TI = T/B × percentage dose per gram) were higher at 24, 48 and 72 h post-injection.
Neutral SM liposomes with encapsulated 67Ga in an aqueous phase offer better radiolabelling properties and 67Ga accumulation for tumour imaging [108]. Table 5 summarises the SM-based radiolabelled molecular imaging probes.
Table 5.
Sphyngomyelin-based radiolabelled molecular imaging probes
| Molecular target | Molecular probes | Biological functions | Isotopes | Imaging techniques |
|---|---|---|---|---|
| SM | liposomes a | tumour imaging | 67Ga | SPECT(CT) |
a nanocarrier SM liposomes encapsulating 67Ga in lipid phase or aqueous phase. SM, sphyngomyelin; SPECT(CT), single-photon emission computed tomography (computed tomography).
Conclusion
In nuclear medicine, the transbilayer phospholipid targets are used in preclinical and clinical research for SPECT-CT and PET-CT molecular imaging with radiolabelled probes to assess key biological functions:
1. the PS target allows the visualisation, characterisation, and measurement of apoptosis or necrosis, and thrombosis with radiolabelled annexin V and the 99mTc or 18F-labelled C2A domain of synaptotagmin I and the 99mTc-labelled HYNIC-lactadherin; 99mTc-labelled synthetic PSBP-6-SAAC probe has been designed for molecular imaging of cell death. PS targeting probes may allow imaging of tumour endothelium vasculature with 74As-bavituximab. 77/76As-bavituximab may also serve for β- radioimmunotherapy of tumours with PS exposed endothelium tumour vasculature.
2. the PE target allows imaging of apoptosis or necrosis with 99mTc-HYNIC-duramycin. It may also allow tumour imaging with radiolabelled ethanolamine and N,N' dimethylethanolamine.
3. the PS and PE targets allow imaging of necrosis with the radiolabelled 64Cu-bis-DOTA hypericin.
4. the PC target allows assessment of choline metabolism with 11C-choline or 18F-fluorocholine, and also the evaluation of the acetate and acetoacetate metabolism with 11C-acetate or 18F-fluoroacetate and 11C-acetoacetate.
5. the PI target allows evaluation of the neuronal and myocardial PI turnover with 11C or 18F-labelled DAG, and also with 11C-inositol.
6. the SM target may be used with liposomes encapsulating 67Ga for imaging purposes.
7. Translation of preclinical research to clinical research will be necessary to optimally assess the pharmacokinetics of radiolabelled probes for molecular imaging of transbilayer phospholipids.
Abbreviations
AA: amino acids; ATP: adenosine triphosphate; CT: computed tomography; C2A-GST: C2A Synaptotagmin domain I glutathione-S-transferase; CA4P: combretastatin A4 phosphate; DAG: diacylglycerol; DOTA: 1,4,7,10-tetraazacyclododecane-N, N', N″, N″'-tetraacetic acid; ECM: external leaflet of the cell membrane; FITC: fluorescein isothiocyanate; 18F-FCH: 18F-fluorocholine; 18F-FDG: 18F-fluorodeoxyglucose; kDa: kilodalton; HYNIC: hydrazinonicotinamide; i.e.: id est (in example with complete enumeration); e.g.: exampli gratia (for example with incomplete enumeration); ICM: internal leaflet of the cell membrane; M: molar; 10-9 M: nanomolar; MI: molecular imaging; MRI: magnetic resonance imaging; MW: molecular weight; NSCLC: non-small cell lung cancer; PA: phosphatidic acid; PC: phosphatidylcholine; PE: phosphatidylethanolamine; PET-CT: positron emission tomography-computed tomography; p.i: post-intra-venous injection; PI: phosphatidylinositol; PIP: phosphatidylinositol 4-phosphate; PIP 2: phosphatidylinositol 4,5-diphosphate; PIP 3: phosphatidylinositol 3,4,5-triphosphate; PS: phosphatidylserine; SAAC: single amino acid chelator; SM: sphyngomyelin; SPECT-CT: single-photon emission computed tomography-computed tomography; TRAIL: tumour necrosis factor-related apoptosis-inducing ligand; TUNEL: terminal deoxynucleotidyl transferase-mediated dUTP nick end labelling.
Competing interests
The authors declare that they have no competing interests.
Authors' contributions
TB has made substantial contribution in drafting the manuscript. FP has made substantial contribution in reviewing the manuscript. All authors read and approved the final manuscript.
Authors' information
TB is nuclear medicine physician (MD, PhD) and Adjunct Professor at The University of Western Ontario in the department of Medical Imaging (London, ON, Canada).
FP is imaging program leader (PhD), Assistant Scientific Director at The Lawson Health Research Institute (LHRI), Professor at The University of Western Ontario in the departments of Medical Imaging, Medical BioPhysics and Physics, and Chief Medical Physicist at St. Joseph's Health Care and London Health Sciences Centre (London, ON, Canada).
Contributor Information
Tarik Z Belhocine, Email: tbelhocine@yahoo.ca.
Frank S Prato, Email: prato@lawsonimaging.ca.
Acknowledgements
The authors would like to acknowledge the contribution of Dr. Kimberley J. Blackwood (PhD), post-doctoral associate at The University of Western Ontario and The Lawson Health Research Institute, for her valuable assistance in the manuscript editing.
References
- Jain KK. Personalized medicine. Curr Opin Mol Ther. 2002;4:548–558. Review. [PubMed] [Google Scholar]
- Basu S. Personalized versus evidence-based medicine with PET-based imaging. Nat Rev Clin Oncol. 2010;7:665–668. doi: 10.1038/nrclinonc.2010.121. [DOI] [PubMed] [Google Scholar]
- Hoffman JM, Gambhir SS. Molecular imaging: the vision and opportunity for radiology in the future. Radiology. 2007;244:39–47. doi: 10.1148/radiol.2441060773. Review. [DOI] [PubMed] [Google Scholar]
- Anderson CJ. A new vision for the MICoE. J Nucl Med. 2010;51 19N, 24N. [PubMed] [Google Scholar]
- Hnatowich DJ. Observations on the role of nuclear medicine in molecular imaging. J Cell Biochem Suppl. 2002;39:18–24. doi: 10.1002/jcb.10400. Review. [DOI] [PubMed] [Google Scholar]
- Pappas V. Renewed energy for nuclear medicine and molecular imaging. J Nucl Med. 2011;52:20N. [PubMed] [Google Scholar]
- Pomper MG, Gelovani JG. Molecular Imaging in Oncology. Informa Healthcare USA, Inc; 2008. [Google Scholar]
- Mankoff David A. A definition of molecular imaging. J Nucl Med. 2007;48:18N, 21N. [PubMed] [Google Scholar]
- Reubi JC, Maecke HR. Peptide-based probes for cancer imaging. J Nucl Med. 2008;49:1735–1738. doi: 10.2967/jnumed.108.053041. Review. [DOI] [PubMed] [Google Scholar]
- Tait JF. Imaging of Apoptosis. J Nucl Med. 2008;49:1573–1576. doi: 10.2967/jnumed.108.052803. [DOI] [PubMed] [Google Scholar]
- Anderson CJ, Bulte JW, Chen K, Chen X, Khaw BA, Shokeen M, Wooley KL, VanBrocklin HF. Design of targeted cardiovascular molecular imaging probes. J Nucl Med. 2010;51(Suppl 1):3S–17S. doi: 10.2967/jnumed.109.068130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eckelman WC, Reba RC, Kelloff GJ. Targeted imaging: an important biomarker for understanding disease progression in the era of personalized medicine. Drug Discov Today. 2008;13:748–759. doi: 10.1016/j.drudis.2008.05.009. [DOI] [PubMed] [Google Scholar]
- Vance JE, Steenbergen R. Metabolism and functions of phosphatidylserine. Prog Lipid Res. 2005;44:207–234. doi: 10.1016/j.plipres.2005.05.001. Review. [DOI] [PubMed] [Google Scholar]
- Ulrich AS. Biophysical aspects of using liposomes as delivery vehicles. Biosci Rep. 2002;22:129–150. doi: 10.1023/A:1020178304031. Review. [DOI] [PubMed] [Google Scholar]
- Phillips WT, Goins BA, Bao A. Radioactive liposomes. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2009;1:69–83. doi: 10.1002/wnan.3. Review. [DOI] [PubMed] [Google Scholar]
- Singer SJ, Nicolson GL. The fluid mosaic model of the structure of cell membranes. Science. 1972;175:720–731. doi: 10.1126/science.175.4023.720. [DOI] [PubMed] [Google Scholar]
- Bagatolli LA, Ipsen JH, Simonsen AC, Mouritsen OG. An outlook on organization of lipids in membranes: searching for a realistic connection with the organization of biological membranes. Prog Lipid Res. 2010;49:378–389. doi: 10.1016/j.plipres.2010.05.001. [DOI] [PubMed] [Google Scholar]
- Vereb G, Szöllosi J, Matkó J, Nagy P, Farkas T, Vigh L, Mátyus L, Waldmann TA, Damjanovich S. Dynamic, yet structured: the cell membrane three decades after the Singer-Nicolson model. Proc Natl Acad Sci USA. 2003;100:8053–8058. doi: 10.1073/pnas.1332550100. Review. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamaji-Hasegawa A, Tsujimoto M. Asymmetric distribution of phospholipids in biomembranes. Biol Pharm Bull. 2006;29:1547–1553. doi: 10.1248/bpb.29.1547. Review. [DOI] [PubMed] [Google Scholar]
- Balasubramanian K, Schroit AJ. Aminophospholipid asymmetry: a matter of life and death. Annu Rev Physiol. 2003;65:701–734. doi: 10.1146/annurev.physiol.65.092101.142459. Review. [DOI] [PubMed] [Google Scholar]
- Garcia-Manyes S, Redondo-Morata L, Oncins G, Sanz F. Nanomechanics of lipid bilayers: heads or tails? J Am Chem Soc. 2010;132:12874–12886. doi: 10.1021/ja1002185. [DOI] [PubMed] [Google Scholar]
- van Meer G, Voelker DR, Feigenson GW. Membrane lipids: where they are and how they behave. Nat Rev Mol Cell Biol. 2008;9:112–124. doi: 10.1038/nrm2330. Review. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Groves AM, Win T, Haim SB, Ell PJ. Non-[18F]FDG PET in clinical oncology. Lancet Oncol. 2007;8:822–830. doi: 10.1016/S1470-2045(07)70274-7. Review. [DOI] [PubMed] [Google Scholar]
- Belhocine TZ, Blankenberg FG. The imaging of apoptosis with the radiolabelled annexin A5: a new tool in translational research. Curr Clin Pharmacol. 2006;1:129–137. doi: 10.2174/157488406776872541. Review. [DOI] [PubMed] [Google Scholar]
- Zwaal RF, Schroit AJ. Pathophysiologic implications of membrane phospholipid asymmetry in blood cells. Blood. 1997;89:1121–1132. Review. [PubMed] [Google Scholar]
- Lahorte CM, Vanderheyden JL, Steinmetz N, Van de Wiele C, Dierckx RA, Slegers G. Apoptosis-detecting radioligands: current state of the art and future perspectives. Eur J Nucl Med Mol Imaging. 2004;31:887–919. doi: 10.1007/s00259-004-1555-4. Review. [DOI] [PubMed] [Google Scholar]
- Belhocine T, Steinmetz N, Hustinx R, Bartsch P, Jerusalem G, Seidel L, Rigo P, Green A. Increased uptake of the apoptosis-imaging agent (99m)Tc recombinant human Annexin V in human tumors after one course of chemotherapy as a predictor of tumor response and patient prognosis. Clin Cancer Res. 2002;8:2766–2774. [PubMed] [Google Scholar]
- Boersma HH, Kietselaer BL, Stolk LM, Bennaghmouch A, Hofstra L, Narula J, Heidendal GA, Reutelingsperger CP. Past, present, and future of annexin A5: from discovery to clinical applications. J Nucl Med. 2005;46:2035–2050. Review. [PubMed] [Google Scholar]
- Sarda-Mantel L, Coutard M, Rouzet F, Raguin O, Vrigneaud JM, Hervatin F, Martet G, Touat Z, Merlet P, Le Guludec D, Michel JB. 99mTc.annexin-V functional imaging of luminal thrombus activity in abdominal aortic aneurysms. Arterioscler Thromb Vasc Biol. 2006;26:2153–2159. doi: 10.1161/01.ATV.0000237605.25666.13. [DOI] [PubMed] [Google Scholar]
- Kartachova MS, Valdés Olmos RA, Haas RL, Hoebers FJ, van Herk M, Verheij M. 99mTc-HYNIC-rh-annexin-V scintigraphy: visual and quantitative evaluation of early treatment-induced apoptosis to predict treatment outcome. Nucl Med Commun. 2008;29:39–44. doi: 10.1097/MNM.0b013e3282f1bc22. [DOI] [PubMed] [Google Scholar]
- Vermeersch H, Ham H, Rottey S, Lahorte C, Corsetti F, Dierckx R, Steinmetz N, Van de Wiele C. Intraobserver, interobserver, and day-to-day reproducibility of quantitative 99mTc-HYNIC annexin V imaging in head and neck carcinoma. Cancer Biother Radiopharm. 2004;19:205–210. doi: 10.1089/108497804323071986. [DOI] [PubMed] [Google Scholar]
- Tait JF, Smith C, Levashova Z, Patel B, Blankenberg FG, Vanderheyden JL. Improved detection of cell death in vivo with annexin V radiolabeled by site-specific methods. J Nucl Med. 2006;47:1546–1553. [PubMed] [Google Scholar]
- Zhao M, Zhu X, Ji S, Zhou J, Ozker KS, Fang W, Molthen RC, Hellman RS. 99mTc-labelled C2A domain of synaptotagmin I as a target-specific molecular probe for noninvasive imaging of acute myocardial infarction. J Nucl Med. 2006;47:1367–1374. [PubMed] [Google Scholar]
- Zhu X, Li Z, Zhao M. Imaging acute cardiac cell death: temporal and spatial distribution of 99mTc-labelled C2A in the area at risk after myocardial ischemia and reperfusion. J Nucl Med. 2007;48:1031–1036. doi: 10.2967/jnumed.106.037754. [DOI] [PubMed] [Google Scholar]
- Fang W, Wang F, Ji S, Meier HT, Hellman RS, Brindle KM, Davletov B, Zhao M. SPECT imaging of myocardial infarction using 99mTc-labelled C2A domain of synaptotagmin I a porcine ischemia-reperfusion model. Nucl Med Biol. 2007;34:917–923. doi: 10.1016/j.nucmedbio.2007.06.014. [DOI] [PubMed] [Google Scholar]
- Zhu X, Migrino RQ, Hellman RS, Brahmbhatt T, Zhao M. Early uptake of 99mTc-C2A: in the acute phase of myocardial infarction as a prognostic indicator for follow-up cardiac dysfunction. Nucl Med Commun. 2008;29:764–769. doi: 10.1097/MNM.0b013e3283025085. [DOI] [PubMed] [Google Scholar]
- Wang F, Fang W, Zhao M, Wang Z, Ji S, Li Y, Zheng Y. Imaging paclitaxel (chemotherapy)-induced tumor apoptosis with 99mTc C2A, a domain of synaptotagmin I: a preliminary study. Nucl Med Biol. 2008;35:359–364. doi: 10.1016/j.nucmedbio.2007.12.007. [DOI] [PubMed] [Google Scholar]
- Wang F, Fang W, Zhang MR, Zhao M, Liu B, Wang Z, Hua Z, Yang M, Kumata K, Hatori A, Yamasaki T, Yanamoto K, Suzuki K. Evaluation of chemotherapy response in VX2 rabbit lung cancer with 18F-labelled C2A domain of synaptotagmin I. J Nucl Med. 2011;52:592–599. doi: 10.2967/jnumed.110.081588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tavaré R, Torres Martin De Rosales R, Blower PJ, Mullen GE. Efficient site-specific radiolabeling of a modified C2A domain synaptotagmin I with [99mTc(CO)3]+: a new radiopharmaceutical for imaging cell death. Bioconjug Chem. 2009;20:2071–2081. doi: 10.1021/bc900160j. [DOI] [PubMed] [Google Scholar]
- Ran S, Downes A, Thorpe PE. Increased exposure of anionic phospholipids on the surface of tumor blood vessels. Cancer Res. 2002;62:6132–6140. [PubMed] [Google Scholar]
- Ran S, Thorpe PE. Phosphatidylserine is a marker of tumor vasculature and a potential target for cancer imaging and therapy. Int J Radiat Oncol Biol Phys. 2002;54:1479–1484. doi: 10.1016/S0360-3016(02)03928-7. [DOI] [PubMed] [Google Scholar]
- Ran S, He J, Huang X, Soares M, Scothorn D, Thorpe PE. Antitumor effects of a monoclonal antibody that binds anionic phospholipids on the surface of tumor blood vessels in mice. Clin Cancer Res. 2005;11:1551–1562. doi: 10.1158/1078-0432.CCR-04-1645. [DOI] [PubMed] [Google Scholar]
- Jennewein M, Lewis MA, Zhao D, Tsyganov E, Slavine N, He J, Watkins L, Kodibagkar VD, O'Kelly S, Kulkarni P, Antich PP, Hermanne A, Rösch F, Mason RP, Thorpe PE. Vascular imaging of solid tumors in rats with a radioactive arsenic-labelled antibody that binds exposed phosphatidylserine. Clin Cancer Res. 2008;14:1377–1385. doi: 10.1158/1078-0432.CCR-07-1516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chopra A. Molecular Imaging and Contrast Agent Database (MICAD) [Internet] Bethesda (MD): National Center for Biotechnology Information (US); 2004-2010. [74]-Labeled monoclonal antibody against anionic phospholipids. [PubMed] [Google Scholar]
- Song S, Xiong C, Zhou M, Lu W, Huang Q, Ku G, Zhao J, Flores LG Jr, Ni Y, Li C. Small-animal PET of tumor damage induced by photothermal ablation with 64cu-bis-dota-hypericin. J Nucl Med. 2011;52:792–799. doi: 10.2967/jnumed.110.086116. [DOI] [PubMed] [Google Scholar]
- Ni Y, Huyghe D, Verbeke K, de Witte PA, Nuyts J, Mortelmans L, Chen F, Marchal G, Verbruggen AM, Bormans GM. First preclinical evaluation of mono-[123I]iodohypericin as a necrosis-avid tracer agent. Eur J Nucl Med Mol Imaging. 2006;33:595–601. doi: 10.1007/s00259-005-0013-2. [DOI] [PubMed] [Google Scholar]
- Fonge H, Vunckx K, Wang H, Feng Y, Mortelmans L, Nuyts J, Bormans G, Verbruggen A, Ni Y. Non-invasive detection and quantification of acute myocardial infarction in rabbits using mono-[123I]iodohypericin microSPECT. Eur Heart J. 2008;29:260–269. doi: 10.1093/eurheartj/ehm588. [DOI] [PubMed] [Google Scholar]
- Li J, Sun Z, Zhang J, Shao H, Miranda Cona M, Wang H, Marysael T, Chen F, Prinsen K, Zhou L, Huang D, Nuyts J, Yu J, Meng B, Bormans G, Fang Z, de Witte P, Li Y, Verbruggen A, Wang X, Mortelmans L, Xu K, Marchal G, Ni Y. A dual-targeting anticancer approach: soil and seed principle. Radiology. 2011;260:799–807. doi: 10.1148/radiol.11102120. [DOI] [PubMed] [Google Scholar]
- Chopra A. Molecular Imaging and Contrast Agent Database (MICAD) [Internet] Bethesda (MD): National Center for Biotechnology Information (US); 2004-2010. [99mTc]-Mercaptoacetyldiglycyl-1,5-diaminopentylene hypericincarboxamide. [PubMed] [Google Scholar]
- Fonge H, Jin L, Wang H, Bormans G, Ni Y, Verbruggen A. Synthesis and preliminary biological evaluation of a 99mTc-labelled hypericin derivative as a necrosis avid imaging agent. J Label Comp Radiopharm. 2008;51:33–40. doi: 10.1002/jlcr.1468. [DOI] [Google Scholar]
- Häggqvist B, Näslund J, Sletten K, Westermark GT, Mucchiano G, Tjernberg LO, Nordstedt C, Engström U, Westermark P. Medin: an integral fragment of aortic smooth muscle cell-produced lactadherin forms the most common human amyloid. Proc Natl Acad Sci USA. 1999;96:8669–8674. doi: 10.1073/pnas.96.15.8669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hou J, Fu Y, Zhou J, Li W, Xie R, Cao F, Gilbert GE, Shi J. Lactadherin functions as a probe for phosphatidylserine exposure and as an anticoagulant in the study of stored platelets. Vox Sang. 2011;100:187–195. doi: 10.1111/j.1423-0410.2010.01375.x. [DOI] [PubMed] [Google Scholar]
- Schutters K, Reutelingsperger C. Phosphatidylserine targeting for diagnosis and treatment of human diseases. Apoptosis. 2010;15:1072–1082. doi: 10.1007/s10495-010-0503-y. Review. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi J, Shi Y, Waehrens LN, Rasmussen JT, Heegaard CW, Gilbert GE. Lactadherin detects early phosphatidylserine exposure on immortalized leukemia cells undergoing programmed cell death. Cytometry A. 2006;69:1193–1201. doi: 10.1002/cyto.a.20345. [DOI] [PubMed] [Google Scholar]
- Waehrens LN, Heegaard CW, Gilbert GE, Rasmussen JT. Bovine lactadherin as a calcium-independent imaging agent of phosphatidylserine expressed on the surface of apoptotic HeLa cells. J Histochem Cytochem. 2009;57:907–914. doi: 10.1369/jhc.2009.953729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Falborg L, Waehrens LN, Alsner J, Bluhme H, Frøkiaer J, Heegaard CW, Horsman MR, Rasmussen JT, Rehling M. Biodistribution of 99mTc-HYNIC-lactadherin in mice--a potential tracer for visualizing apoptosis in vivo. Scand J Clin Lab Invest. 2010;70:209–216. doi: 10.3109/00365511003663648. [DOI] [PubMed] [Google Scholar]
- Xiong C, Brewer K, Song S, Zhang R, Lu W, Wen X, Li C. Peptide-based imaging agents targeting phosphatidylserine for the detection of apoptosis. J Med Chem. 2011;54:1825–1835. doi: 10.1021/jm101477d. [DOI] [PubMed] [Google Scholar]
- Price DT, Coleman RE, Liao RP, Robertson CN, Polascik TJ, DeGrado TR. Comparison of [18F]fluorocholine and [18F]fluorodeoxyglucose for positron emission tomography of androgen dependent and androgen independent prostate cancer. J Urol. 2002;168:273–280. doi: 10.1016/S0022-5347(05)64906-3. [DOI] [PubMed] [Google Scholar]
- Leung K. Molecular Imaging and Contrast Agent Database (MICAD) [Internet] Bethesda (MD): National Center for Biotechnology Information (US); 2004-2011. [11C]Choline. [PubMed] [Google Scholar]
- Leung K. Molecular Imaging and Contrast Agent Database (MICAD) [Internet] Bethesda (MD): National Center for Biotechnology Information (US); 2004-2011. [18F]Fluorocholine. [PubMed] [Google Scholar]
- DeGrado TR, Reiman RE, Price DT, Wang S, Coleman RE. Pharmacokinetics and radiation dosimetry of 18F-fluorocholine. J Nucl Med. 2002;43:92–96. [PubMed] [Google Scholar]
- Mertens K, Slaets D, Lambert B, Acou M, De Vos F, Goethals I. PET with (18)F-labelled choline-based tracers for tumour imaging: a review of the literature. Eur J Nucl Med Mol Imaging. 2010;37:2188–2193. doi: 10.1007/s00259-010-1496-z. [DOI] [PubMed] [Google Scholar]
- Chiti A, Picchio M. The rising PET: the increasing use of choline PET/CT in prostate cancer. Eur J Nucl Med Mol Imaging. 2011;38:53–54. doi: 10.1007/s00259-010-1662-3. [DOI] [PubMed] [Google Scholar]
- Picchio M, Briganti A, Fanti S, Heidenreich A, Krause BJ, Messa C, Montorsi F, Reske SN, Thalmann GN. The role of choline positron emission tomography/computed tomography in the management of patients with prostate-specific antigen progression after radical treatment of prostate cancer. Eur Urol. 2011;59:51–60. doi: 10.1016/j.eururo.2010.09.004. [DOI] [PubMed] [Google Scholar]
- Kwee SA, Coel MN, Lim J, Ko JP. Combined use of F-18 fluorocholine positron emission tomography and magnetic resonance spectroscopy for brain tumor evaluation. J Neuroimaging. 2004;14:285–289. doi: 10.1177/1051228404264957. [DOI] [PubMed] [Google Scholar]
- Hara T, Kondo T, Hara T, Kosaka N. Use of 18F-choline and 11C-choline as contrast agents in positron emission tomography imaging-guided stereotactic biopsy sampling of gliomas. J Neurosurg. 2003;99:474–479. doi: 10.3171/jns.2003.99.3.0474. [DOI] [PubMed] [Google Scholar]
- Kwee SA, Ko JP, Jiang CS, Watters MR, Coel MN. Solitary brain lesions enhancing at MR imaging: evaluation with fluorine 18 fluorocholine PET. Radiology. 2007;244:557–565. doi: 10.1148/radiol.2442060898. [DOI] [PubMed] [Google Scholar]
- Lam WW, Ng DC, Wong WY, Ong SC, Yu SW, See SJ. Promising role of [18F] fluorocholine PET/CT vs [18F] fluorodeoxyglucose PET/CT in primary brain tumors-Early experience. Clin Neurol Neurosurg. 2011;113:156–161. doi: 10.1016/j.clineuro.2010.09.012. [DOI] [PubMed] [Google Scholar]
- Kuang Y, Salem N, Tian H, Kolthammer JA, Corn DJ, Wu C, Wang F, Wang Y, Lee Z. Imaging lipid synthesis in hepatocellular carcinoma with [methyl-11c]choline: correlation with on vivo metabolic studies. J Nucl Med. 2011;52:98–106. doi: 10.2967/jnumed.110.080366. [DOI] [PubMed] [Google Scholar]
- Salem N, Kuang Y, Wang F, Maclennan GT, Lee Z. PET imaging of hepatocellular carcinoma with 2-deoxy-2[18F]fluoro-d-deoxyglucose, 6-deoxy-6[18F] fluoro-d-glucose, [1-11C]-acetate and [N-methyl-11C]-choline. Q J Nucl Med Mol Imaging. 2009;53:144–156. [PubMed] [Google Scholar]
- Yamamoto Y, Nishiyama Y, Kameyama R, Okano K, Kashiwagi H, Deguchi A, Kaji M, Ohkawa M. Detection of hepatocellular carcinoma using 11C-choline PET: comparison with 18F-FDG PET. J Nucl Med. 2008;49:1245–1248. doi: 10.2967/jnumed.108.052639. [DOI] [PubMed] [Google Scholar]
- Talbot JN, Gutman F, Fartoux L, Grange JD, Ganne N, Kerrou K, Grahek D, Montravers F, Poupon R, Rosmorduc O. PET/CT in patients with hepatocellular carcinoma using [(18)F]fluorocholine: preliminary comparison with [(18)F]FDG PET/CT. Eur J Nucl Med Mol Imaging. 2006;33:1285–1289. doi: 10.1007/s00259-006-0164-9. [DOI] [PubMed] [Google Scholar]
- Talbot JN, Fartoux L, Balogova S, Nataf V, Kerrou K, Gutman F, Huchet V, Ancel D, Grange JD, Rosmorduc O. Detection of hepatocellular carcinoma with PET/CT: a prospective comparison of 18F-fluorocholine and 18F-FDG in patients with cirrhosis or chronic liver disease. J Nucl Med. 2010;51:1699–1706. doi: 10.2967/jnumed.110.075507. [DOI] [PubMed] [Google Scholar]
- Leung K. Molecular Imaging and Contrast Agent Database (MICAD) [Internet] Bethesda (MD): National Center for Biotechnology Information (US); 2004-2010. [11C]Acetate. [PubMed] [Google Scholar]
- Yoshimoto M, Waki A, Yonekura Y, Sadato N, Murata T, Omata N, Takahashi N, Welch MJ, Fujibayashi Y. Characterization of acetate metabolism in tumor cells in relation to cell proliferation: acetate metabolism in tumor cells. Nucl Med Biol. 2001;28:117–122. doi: 10.1016/S0969-8051(00)00195-5. [DOI] [PubMed] [Google Scholar]
- Swinnen JV, Van Veldhoven PP, Timmermans L, De Schrijver E, Brusselmans K, Vanderhoydonc F, Van de Sande T, Heemers H, Heyns W, Verhoeven G. Fatty acid synthase drives the synthesis of phospholipids partitioning into detergent-resistant membrane microdomains. Biochem Biophys Res Commun. 2003;302:898–903. doi: 10.1016/S0006-291X(03)00265-1. [DOI] [PubMed] [Google Scholar]
- Leung K. Molecular Imaging and Contrast Agent Database (MICAD) [Internet] Bethesda (MD): National Center for Biotechnology Information (US); 2004-2010. [11C]Acetoacetate. [PubMed] [Google Scholar]
- Leung K. Molecular Imaging and Contrast Agent Database (MICAD) [Internet] Bethesda (MD): National Center for Biotechnology Information (US); 2004-2010. 2-[18F]Fluoroacetate. 2007 Mar 22 [updated 2008 Jun 24] [PubMed] [Google Scholar]
- Lindhe O, Sun A, Ulin J, Rahman O, Långström B, Sörensen J. [(18)F]Fluoroacetate is not a functional analogue of [(11)C]acetate in normal physiology. Eur J Nucl Med Mol Imaging. 2009;36:1453–1459. doi: 10.1007/s00259-009-1128-7. [DOI] [PubMed] [Google Scholar]
- Jadvar H. Prostate cancer: PET with 18F-FDG, 18F. or 11C-Acetate, and 18F. or 11C-choline. J Nucl Med. 2011;52:81–89. doi: 10.2967/jnumed.110.077941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu RS. Clinical applications of C-11-acetate in oncology [abstract] Clin Positron Imaging. 2000;3:185. doi: 10.1016/S1095-0397(00)00097-2. [DOI] [PubMed] [Google Scholar]
- Marik J, Ogasawara A, Martin-McNulty B, Ross J, Flores JE, Gill HS, Tinianow JN, Vanderbilt AN, Nishimura M, Peale F, Pastuskovas C, Greve JM, van Bruggen N, Williams SP. PET of glial metabolism using 2-18F-fluoroacetate. J Nucl Med. 2009;50:982–989. doi: 10.2967/jnumed.108.057356. [DOI] [PubMed] [Google Scholar]
- Ponde DE, Dence CS, Oyama N, Kim J, Tai YC, Laforest R, Siegel BA, Welch MJ. 18F-fluoroacetate: a potential acetate analog for prostate tumor imaging--in vivo evaluation of 18F-fluoroacetate versus 11C-acetate. J Nucl Med. 2007;48:420–428. [PubMed] [Google Scholar]
- Matthies A, Ezziddin S, Ulrich EM, Palmedo H, Biersack HJ, Bender H, Guhlke S. Imaging of prostate cancer metastases with 18F-fluoroacetate using PET/CT. Eur J Nucl Med Mol Imaging. 2004;31:797. doi: 10.1007/s00259-003-1437-1. [DOI] [PubMed] [Google Scholar]
- Authier S, Tremblay S, Dumulon V, Dubuc C, Ouellet R, Lecomte R, Cunnane SC, Benard F. [(11)C] Acetoacetate utilization by breast and prostate tumors: a PET and biodistribution study in mice. Mol Imaging Biol. 2008;10:217–223. doi: 10.1007/s11307-008-0143-6. [DOI] [PubMed] [Google Scholar]
- Pifferi F, Tremblay S, Plourde M, Tremblay-Mercier J, Bentourkia M, Cunnane SC. Ketones and brain function: possible link to polyunsaturated fatty acids and availability of a new brain PET tracer, 11C-acetoacetate. Epilepsia. 2008;49(Suppl 8):76–79. doi: 10.1111/j.1528-1167.2008.01842.x. Review. [DOI] [PubMed] [Google Scholar]
- Bentourkia M, Tremblay S, Pifferi F, Rousseau J, Lecomte R, Cunnane S. PET study of 11C-acetoacetate kinetics in rat brain during dietary treatments affecting ketosis. Am J Physiol Endocrinol Metab. 2009;296:E796–801. doi: 10.1152/ajpendo.90644.2008. [DOI] [PubMed] [Google Scholar]
- Vance JE. Phosphatidylserine and phosphatidylethanolamine in mammalian cells: two metabolically related aminophospholipids. J Lipid Res. 2008;49:1377–1387. doi: 10.1194/jlr.R700020-JLR200. Review. [DOI] [PubMed] [Google Scholar]
- Marconescu A, Thorpe PE. Coincident exposure of phosphatidylethanolamine and anionic phospholipids on the surface of irradiated cells. Biochim Biophys Acta. 2008;1778:2217–2224. doi: 10.1016/j.bbamem.2008.05.006. [DOI] [PubMed] [Google Scholar]
- Stafford JH, Thorpe PE. Increased exposure of phosphatidylethanolamine on the surface of tumor vascular endothelium. Neoplasia. 2011;13:299–308. doi: 10.1593/neo.101366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leung K. Molecular Imaging and Contrast Agent Database (MICAD) [Internet] Bethesda (MD): National Center for Biotechnology Information (US); 2004-2010. 99mTc-(Hydrazinonicotinic acid-duramycin)(tricine)(TPPTS) [PubMed] [Google Scholar]
- Zhao M, Li Z, Bugenhagen S. 99mTc-labelled duramycin as a novel phosphatidylethanolamine-binding molecular probe. J Nucl Med. 2008;49:1345–1352. doi: 10.2967/jnumed.107.048603. [DOI] [PubMed] [Google Scholar]
- Mintz A, Wang L, Ponde DE. Comparison of radiolabeled choline and ethanolamine as probe for cancer detection. Cancer Biol Ther. 2008;7:742–747. doi: 10.4161/cbt.7.5.5746. [DOI] [PubMed] [Google Scholar]
- Vance JE, Vance DE. Phospholipid biosynthesis in mammalians cells. Biochem Cell Biol. 2004;82:113–128. doi: 10.1139/o03-073. Review. [DOI] [PubMed] [Google Scholar]
- Imahori Y, Fujii R, Ueda S, Matsumoto K, Wakita K, Ido T, Nariai T, Nakahashi H. Membrane trapping of carbone-11-labelled 1,2-diacylglycerols as a basic concept for assessing phosphatidylinositol turnover in neurotransmission process. J Nucl Med. 1992;33:413–422. [PubMed] [Google Scholar]
- Sasaki T, Ogihara-Umeda I, Kojima S, Nishigori H. Sequential enzymatic synthesis and biodistribution of radiolabelled inositol and inositol analogs. Int J Rad Appl Instrum A. 1991;42:97–101. doi: 10.1016/0883-2889(91)90130-S. [DOI] [PubMed] [Google Scholar]
- Fujii R, Ueda S, Kanatsuna T, Imahori Y, Ido T, Wakita K, Horii H, Yagyu T, Higashi N, Mabuchi H, Ohmori Y. [Carbon-11 labelled diacylglycerol for signal transduction imaging by positron CT: evaluation of the quality and safety for clinical use] Kaku Igaku. 1995;32:191–198. Japanese. [PubMed] [Google Scholar]
- Imahori Y, Fujii R, Ueda S, Ohmori Y, Wakita K, Matsumoto K. Phosphoinositide turnover imaging linked to muscarinic cholinergic receptor in the central nervous system by positron emission tomography. J Nucl Med. 1993;34:1543–1551. [PubMed] [Google Scholar]
- Imahori Y, Ohmori Y, Fujii R, Matsumoto K, Ueda S. Rapid incorporation of carbon-11-labeled diacylglycerol as a probe of signal transduction in glioma. Cancer Res. 1995;55:4225–4229. [PubMed] [Google Scholar]
- Kondo M, Imahori Y, Mori S, Ueda Y, Fujii R, Nakajima K. Aberrant plasticity in Alzheimer's disease. Neuroreport. 1999;10:1481–1484. doi: 10.1097/00001756-199905140-00016. [DOI] [PubMed] [Google Scholar]
- Matsumoto K, Imahori Y, Fujii R, Ohmori Y, Sekimoto T, Ueda S, Mineura K. Evaluation of phosphoinositides turnover on ischemic human brain with 1-[1-11]-butyryl-2-palmitoyl-rac-glycerol using PET. J Nucl Med. 1999;40:1590–1594. [PubMed] [Google Scholar]
- Imahori Y, Fujii R, Kondo M, Ohmori Y, Nakajima K. Neural features of recovery from CNS injury revealed by PET in human brain. Neuroreport. 1999;10:117–121. [PubMed] [Google Scholar]
- Chida M, Kagaya Y, Imahori Y, Namiuchi S, Fujii R, Fukuchi M, Takahashi C, Tezuka F, Ido T, Shirato K. Visualization of myocardial phosphoinositide turn over with 1-[1-(11)C]-butyryl-2-palmitoyl-rac-glycerol in rats with myocardial infarction. J Nucl Med. 2000;41:2063–2068. [PubMed] [Google Scholar]
- Kagaya Y, Chida M, Imahori Y, Fujii R, Namiuchi S, Takeda M, Yamane Y, Otani H, Watanabe J, Fukuchi M, Tezuka F, Ido T, Shirato K. Effect of angiotensin converting enzyme inhibition on myocardial phosphoinositides metabolism visualised with 1-[1-11C]-butyryl-2-palmitoyl-rac-glycerol in myocardial infarction in the rat. Eur J Nucl Med Mol Imaging. 2002;29:1516–1522. doi: 10.1007/s00259-002-0933-z. [DOI] [PubMed] [Google Scholar]
- Otani H, Kagaya Y, Imahori Y, Yasuda S, Fujii R, Chida M, Namiuchi S, Takeda M, Sakuma M, Watanabe J, Ido T, Nonogi H, Shirato K. Myocardial 11C-diacylglycerol accumulation and left ventricular remodelling in patients after myocardial infarction. J Nucl Med. 2005;46:553–559. [PubMed] [Google Scholar]
- Chida M, Kagaya Y, Nagata S, Mukoyoshi M, Namiuchi S, Yamane Y, Ishide N, Watanabe J, Takahashi T, Ido T, Shirato K. [18F] labelled diacylglycerol analogue as a potential agent to trace myocardial phosphoinositide metabolism. Nucl Med Biol. 2001;28:815–819. doi: 10.1016/S0969-8051(01)00250-5. [DOI] [PubMed] [Google Scholar]
- Ogihara-Umeda I, Kojima S. Increased delivery of gallium-67 to tumors using serum-stable liposomes. J Nucl Med. 1988;29:516–523. [PubMed] [Google Scholar]
- Kostenikov NA, Fadeev NP, Serzhanina VA, Koresakova LN, Rozenberg OA. [An experimental study of the tumor affinity of gallium-67-labelled sphyngomyelin liposomes] Vopr Onkol. 1992;38:1080–1084. Russian. [PubMed] [Google Scholar]