Abstract
Background
In the search for anticancer agents, a promising 17-β-estradiol metabolite, 2-methoxyestradiol (2ME2) was found that exerts antiproliferative in vitro and in vivo activity. Since 2ME2 has limited biological accessibility and rapid metabolic degradation, the purpose of this study was to investigate the in vitro influence exerted by an analogue of 2ME2 namely 2-methoxyestradiol-bis-sulphamate (2MEBM) in a breast adenocarcinoma cell line (MCF-7).
Methods
This was conducted by investigating 2MEBM's in vitro influence on cell cycle progression, mitochondrial membrane potential and possible production of reactive oxygen species (ROS) generation. In vitro effects of 2MEBM on cell cycle progression was demonstrated by means of flow cytometry using propidium iodide. Hydrogen peroxide and superoxide production was investigated using 2,7-dichlorofluorescein diacetate and hydroethidine, respectively. The probable reduction in the mitochondrial membrane potential was demonstrated using a MitoCapture™ kit.
Results
Cell cycle progression revealed the presence of a sub-G1 apoptotic peak. Reduction of mitochondrial membrane potential after exposure to 2MEBM was demonstrated and an increase in ROS production was also observed.
Conclusion
This study verified that 2MEBM exposure resulted in apoptosis induction, increased ROS production and reduced mitochondrial membrane potential in a tumorigenic breast epithelial cell line. Data obtained from this project contributes to the unravelling of the in vitro signal transduction of 2MEBM in tumorigenic cell lines.
Keywords: 2-methoxyestradiol-bis-sulphamate, cell cycle progression, reactive oxygen species, mitochondrial integrity, apoptosis, autophagy
Introduction
2-Methoxyestradiol (2ME2), the major derivative of 2-hydroxyestrogens is an endogenous inhibitor of breast cancer development [1]. 2ME2 exerts its antiproliferative, antiangiogenic and anticancer in vitro and in vivo activity in an estrogen receptor independent mode [1-3]. Antiproliferative effects were observed in the estrogen receptor positive human breast adenocarcinoma cell line (MCF-7), highly tumorigenic melanoma cell line (MDA-MB-435) and the human cervical adenocarcinoma cell line (HeLa). In addition, 2ME2 also inhibited in vivo growth of xenografts generated from MDA-MB-435 melanoma cells, murine sarcoma cells (Meth A sarcomas), murine melanoma cells (B16 sarcomas) and the human multiple myeloma KAS-6/1 cell line [3]. 2ME2 (patented as Panzem®) is currently undergoing phase II clinical trials as an anticancer drug for multiple myeloma [2], ovarian cancer [4], glioblastoma multiforme [5], breast- and prostate cancer [6]. However, high doses are needed (up to 6 g/day), since 2ME2 has limited biological accessibility due to the rapid inactivation of the hydroxyl groups at positions C3/C17 by conjugation and oxidation [7].
Low oral bioavailability and fast metabolic degradation of 2ME2 resulted in the chemical production of promising 2ME2 analogues. One of these analogues is 2-methoxyestradiol-bis-sulphamate (2MEBM), a bis-sulphamoylated derivative of 2ME2 that possesses antiproliferative and antitumor activity with enhanced bioavailability [8-11]. In vitro human umbilical vein endothelial cells (HUVEC) growth-inhibition assay studies indicated that 2MEBM is 60-fold more potent than 2ME2 as antiangioangenic agent [3] and tenfold more potent than 2ME2 as an antiproliferative agent [3,12]. Increased potency is related to the additional sulphamate group added to the precursor 2ME2 molecule [3,12].
Antiproliferative effects of 2MEBM have been reported in tumour growth in nude mice bearing xenografts derived from MDA-MB-435 cells, estrogen receptor positive human breast adenocarcinoma wild type cell line (MCF-7wt), mitoxantrone resistant breast adenocarcinoma cell line (MCF-7 MR), drug resistant human adenocarcinoma cell line (MCF-7 DOX40), prostate cancer cell line (LNACaP) and human umbilical vein endothelial cells (HUVEC) [3,13-15]. 2MEBM induces a G2/M arrest with reversible morphology change in dermal fibroblasts from the elongated fibroblast morphology to a cobblestone, morphology with rounding of cells, while the same treatment in human umbilical vein endothelial cells did not induce morphological changes or a G2/M arrest, indicating that the effects of 2MEBM on growth is cell line dependent [16]. A G2/M arrest followed by the induction of apoptosis was however observed in cell lines including tumorigenic estrogen receptor positive breast carcinoma cell line (MCF-7), drug resistant human adenocarcinoma cell line (MCF-7 DOX40), mitoxantrone resistant breast adenocarcinoma cell line (MCF-7 MR) and highly tumorigenic estrogen receptor negative breast carcinoma cell line MDA-MB-231 [8,17].
Since several questions remain regarding the in vitro influence of 2MEBM, this study aimed to investigate whether 2MEBM alters cell cycle progression and mitochondrial membrane integrity. The influence on reactive oxygen species production in the tumorigenic breast epithelial MCF-7 cell line was also studied.
Materials and methods
Materials
Cell line
MCF-7 is a tumorigenic adherent breast epithelial cell line derived from metastatic sites in adenocarcinoma. MCF-7 cells are able to process estradiol via cytoplasmic estrogen receptors and are capable of forming domes. MCF-7 cells were supplied by Highveld Biological (Pty) Ltd. (Sandringham, South Africa).
Reagents
All required reagents of cell culture analytical grade were purchased from Sigma (St. Louis, United States of America) unless otherwise specified. Heat-inactivated fetal calf serum (FCS), sterile cell culture flasks and plates were purchased from Sterilab Services (Kempton Park, Johannesburg, South Africa). Penicillin, streptomycin and fungizone were obtained from Highveld Biological (Pty) Ltd. (Sandringham, South Africa). 2,7-Dichlorofluorescein diacetate and hydroethidine was acquired from Sigma (St. Louis, United States of America). The MitoCapture ™ Mitochondrial apoptosis detection kit was purchased from BIOCOM biotech (Pty) Ltd. (Clubview, South Africa). Since 2MEBM is not commercially available, it was synthesized by Professor Vleggaar from the Department of Chemistry (University of Pretoria, Pretoria, South Africa). The fluorescence activated cell sorting (FACS) FC500 System flow cytometer equipped with an air-cooled argon laser excited at 488 nm was supplied by Beckman Coulter South Africa (Pty) Ltd. (Pretoria, South Africa).
Cell culture
Cells were grown and maintained in 25 cm2 tissue culture flasks in a humidified atmosphere at 37°C and 5% CO2. MCF-7 cells were cultured in Dulbecco's minimum essential medium eagle (DMEM) and supplemented with 10% heat-inactivated FCS (56°C, 30 min), 100 U/ml penicillin G, 100 μg/ml streptomycin and fungizone (250 μg/l).
General cell culture procedures for experiments
A stock solution of 2 × 10-3M 2MEBM dissolved in dimethyl sulphoxide (DMSO) was prepared and diluted with medium to the desired concentrations prior to exposure of the cells. Medium of control cells was supplemented with an equal volume of DMSO (vehicle control cells). The DMSO content of the final dilutions never exceeded 0.05% (v/v). Experiments were conducted in 25 cm2 cell culture flasks with exponentially growing MCF-7 cells seeded at 1 × 106 cells per 25 cm2 flask to a final volume of 5 ml of maintenance medium. After a 24 h incubation period at 37°C to allow for cell adherence, medium was discarded and cells were exposed to 2MEBM. Previous data obtained in our laboratory by means of crystal violet staining revealed that the 50% growth inhibitory concentration (IC50) of 2MEBM on the tumorigenic MCF-7 cell line was found to be after 48 h of exposure and a concentration a 0.4 μM [18,19]. All subsequent experiments were thus performed at 48 h exposure period with a 0.4 μM 2MEBM on MCF-7 cells. Cells exposed to actinomycin D with a concentration of 0.1 μg/ml in growth medium for 48 h served as a positive control for apoptosis.
Methods
Cell cycle progression
Cell cycle distribution, G2/M block and the detection of a sub-G1 apoptotic peak were analysed by flow cytometry using propidium iodide DNA staining. After 48 h of exposure to 0.4 μM 2MEBM, cells were trypsinized and resuspended in 1 ml growth medium. 1 × 106 cells were centrifuged for 5 min at 300 xg. The pellet resuspended twice in ice-cold phosphate buffer solution (PBS). The supernatant was discarded and the cells were resuspended in 200 μl of ice-cold PBS containing 0.1% FCS. Ice-cold 70% ethanol (4 ml) was added in a drop wise manner and cells were stored at 4°C for 24 h. After 24 h, cells were pelleted by centrifugation for 5 min. The supernatant was removed and cells were resuspended in 1 ml of PBS containing propidium iodide (40 μg/ml) and incubated at 37°C, 5% CO2 for 45 min. Subsequently, cells were analysed by means of FACS FC500 System flow cytometer (Beckman Coulter South Africa (Pty) Ltd) equipped with an air-cooled argon laser excited at 488 nm. Cell cycle distributions were calculated from at least 10 000 cells and were analyzed means of cyflogic version 1.2.1 software (Pertu Therho, Turko, Finland) by assigning relative DNA content per cell to sub-G1, G1, S and G2/M fractions.
Mitochondrial membrane potential
Mitochondrial integrity was investigated by means of a unique cationic dye, 5,5',6,6'-tetrachloro-1,1',3,3'-tetraethylbenzimidazolylcarbocyanine iodide. The MitoCapture ™ mitochondrial kit provides quantitive apoptosis information. Reduction of the mitochondrial membrane potential is an early feature during apoptosis due to the loss of the electrochemical gradient across the mitochondrial membrane [20]. Actinomycin D will be utilised as a positive control for reductions in the mitochondrial membrane [21]. After 48 h of exposure to 0.4 μM 2MEBM, cells were trypsinized and centrifuged at 13 000 × g. Cells were resuspended in 1 ml of diluted MitoCapture solution (1 μl MitoCapture: 1 ml pre-warmed incubation buffer), incubated at a humidified atmosphere (37°C, 5% CO2) for 20 min and subsequently centrifuged at 500 × g. After the supernatant was discarded, cells were resuspended in 1 ml of prewarmed incubation buffer (37°C). Cells were analyzed immediately after following the above-mentioned step using fluorescence activated cell sorting (FACS) FC500 System flow cytometer (Beckman Coulter South Africa (Pty) Ltd). Apoptotic cells were detected in the FITC channel (usually FL1) showing diffused green fluorescence. Data from at least 10 000 cells were analyzed by means of cyflogic version 1.2.1 software (Pertu Therho, Turko, Finland).
Hydrogen peroxide measurement
Hydrogen peroxide (H2O2) generation was assessed using 2,7-dichlorofluorescein diacetate (DCFDA), a non-fluorescent probe, which, upon oxidation by ROS and peroxides is converted to the highly fluorescent derivative 2,7-dichlorofluorescein (DCF). After 48 h of treatment, cells were trypsinized and 1 × 106 cells were resuspended in 1 ml PBS. Cells were incubated with 20 μM DCFDA for 25 min at 37°C. Hydrogen peroxide (20 μM) was added 5 min prior to measurement as a positive control for DCF formation. DCF (FL1) fluorescence was measured with a FACS FC500 System flow cytometer (Beckman Coulter South Africa (Pty) Ltd.) equipped with an air-cooled argon laser excited at 488 nm. Data analysis from at least 10 000 cells were conducted using cyflogic version 1.2.1 software (Pertu Therho, Turko, Finland).
Superoxide measurement
Superoxide generation was assessed using hydroethidine (HE). HE is oxidized by superoxide and not by hydroxyl radicals, singlet O2, H2O2 or nitrogen radicals, to a red fluorescent compound. After 48 h of treatment, cells were trypsinized and 1 × 106 cells were resuspended in 1 ml PBS. Cells were incubated with 10 μM HE for 15 min at 37°C. The HE fluorescent product fluorescence (FL2) was measured using a FACS FC500 System flow cytometer (Beckman Coulter South Africa (Pty) Ltd.) equipped with an air-cooled argon laser excited at 488 nm. The information generated from at least 10 000 cells were analyzed by means of cyflogic version 1.2.1 software (Pertu Therho, Turko, Finland).
Statistics
Measurement of FITC-, HE- and DCF-derived fluorescence was expressed as a ratio of the value measured for the 2MEBM-treated cells compared to vehicle-treated exposed cells (mean relative fluorescence). Flow cytometry analysis involved data from at least 10 000 events that was repeated thrice where after a representative figure was chosen for each experiment.
Results
Cell cycle progression
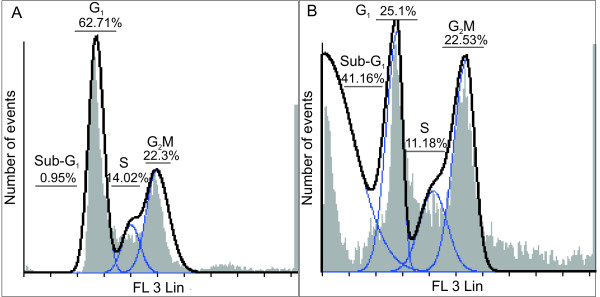
DNA content analyses by means of flow cytometry showed no increase of cells occupying the G2/M phase in 2MEBM-treated cells compared to vehicle-treated cells. However, a statistically significant increase was observed in the sub-G1 population when compared to the vehicle-treated cells. Furthermore, a reduction of 2MEBM-treated cells in the G1-phase was found when compared to the vehicle-treated control (Figure 1A and 1B).
Figure 1.
Cell cycle histograms (FL3 Log representing propidium iodide detected by flow cytometry) revealed a statistical significant sub-G1 apoptotic increase in 0.4 μM 2ME2BM-treated cells (B) when compared to vehicle-treated cells (A). However, no G2/M block was observed in the 0.4 μM 2ME2BM-treated cells. A decrease in cells present in G1 was found in the 0.4 μM 2ME2BM-treated cells when compared to the vehicle-treated cells.
Mitochondrial integrity
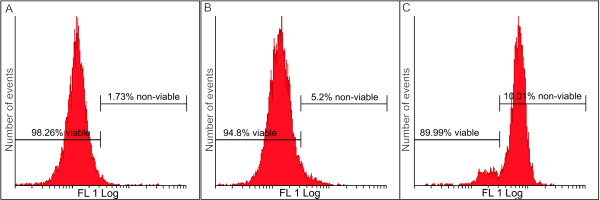
Flow cytometry analyses revealed a statistically insignificant reduction in the mitochondrial membrane potential in the 2MEBM-treated cells when compared to vehicle-treated cells and actinomycin D-treated cells (Figure 2A, B and 2C). The latter is an indicator of apoptosis due to the loss of the electrochemical gradient across the mitochondrial membrane.
Figure 2.
Mitochondrial membrane potential was investigated by means of flow cytometry using the,5',6,6'-tetrachloro-1,1',3,3'- tetraethylbenzimidazolylcarbocyanine iodide dye (FL1 Log represented the dye detected by the flow cytometry). A reduction in mitochondrial membrane potential was observed in the 2ME2BM-treated cells (B) when compared to the vehicle-treated cells (A). Figure C depicts MCF-7 cells exposed to actinomycin D which was used as the positive control for the reduction of the mitochondrial membrane potential.
Hydrogen peroxide
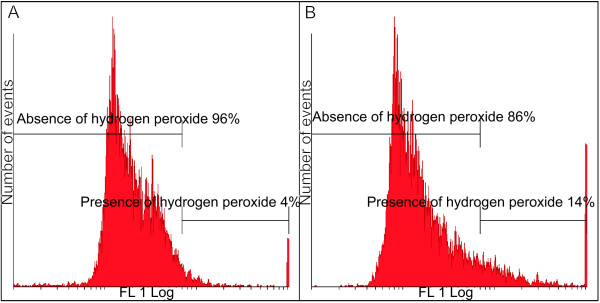
In order to investigate the hydrogen peroxide production in 2MEBM-treated cells compared to vehicle-treated cells, flow cytometry using DCFDA was employed. A statistically insignificant increase in hydrogen peroxide was detected in the 2MEBM-treated cells when compared to the vehicle-treated cells (Figure 3A and 3B).
Figure 3.
Hydrogen peroxide measurement (FL1 Log representing the hydrogen peroxide detected by flow cytometry) in 2ME2BM-treated cells demonstrated a statistically insignificant increase to 14% when compared to the vehicle-treated cells (4%).
Superoxide measurement
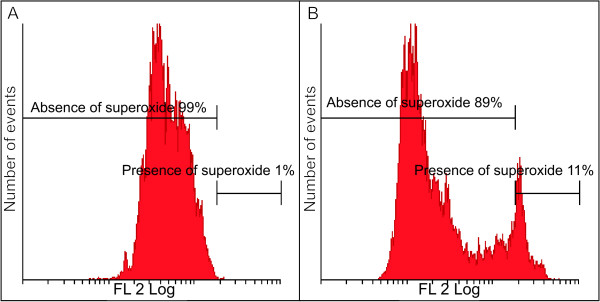
Flow cytometry utilizing HE demonstrated the influence of 2MEBM on superoxide production in MCF-7 cells. The latter was increased in a statistically insignificant manner in 2MEBM-treated cells when compared to the vehicle-treated cells (Figure 4A and 4B).
Figure 4.
Superoxide production detection (FL2 Log representing the superoxide detected by flow cytometry) revealed an increase of 10% in the 2ME2BM-treated cells when compared to the vehicle-treated cells.
Discussion
As previously stated, 2MEBM holds therapeutic potential as an anticancer agent, since it exerts antiproliferative effects in vitro and inhibits tumour growth in vivo [14]. In this article the effects of 2MEBM on the MCF-7 cell line was investigated by demonstrating the influence of 2MEBM on cell cycle progression, membrane integrity and possible production of reactive oxygen species in order to suggests the types of cell death induced by 2MEBM.
Previous reports have indicated that 2MEBM induces a G2/M arrest followed by induction of apoptosis in cell lines including MCF-7, MCF-7 DOX40, MCF-7 MR and MDA-MB-231 [8,17]. In addition, 2MEBM caused a significant apoptotic sub-G1 peak after 48 h. Day, et al (2009) observed similar results where 24 h 2MEBM exposure resulted in G2/M arrest; however, 48 h exposure resulted in a sub-G1 peak accompanied with no changes in the cell cycle distribution in the human 2MEBM-resistant cell line (A2780.140) derived from the human ovarian carcinoma cell line (A2780) [13]. B-Cell Lymphoma-2 Family Members (BCL-2) phosphorylation has also been found to be induced by 2MEBM in MCF-7 cells and correlates with a G2/M arrest [22]. Foster, et al (2008) reported similar findings pertaining to the decrease of 2MEBM-treated cells observed in the G1-phase in this study [8,22,23]. Induction of apoptosis by 2MEBM in breast adenocarcinoma CAL51 cell line was demonstrated by Wood, et al (2004) [24]. In this study 2MEBM caused a reduction in the mitochondrial membrane potential, confirming the induction of apoptosis. The mitochondrial membrane potential depolarization is an early event in the intrinsic (mitochondrial) apoptotic pathway [20,25,26]. Our study suggests that 2-methoxyestradiol-bis-sulphamate (2MEBM) influences the mitochondrial membrane potential minimally, thus the involvement of the intrinsic pathway to a lesser extent. Furthermore, exposure to 2MEBM revealed a sub-G1 peak. This is confirmed by studies conducted in our laboratory where flow cytometry utilizing annexin V-FITC demonstrated that 2MEBM induced apoptosis in MCF-7 cells [18]. During apoptosis, the electrochemical mitochondrial membrane gradient collapses [27-30]. The latter precipitates cell death through either the release of molecules involved in apoptosis, or the loss of mitochondrial functions essential for cell survival [31].
This study demonstrates the novel finding that 2MEBM exposure in the MCF-7 cell line resulted in increased ROS production. However, the source and processing of ROS and the role they play in 2MEBM-induced growth inhibition and cell death remains to be elucidated as no reports has been published on this matter. It was previously reported that the precursor molecule, 2ME2, inhibits superoxide dismutase and increased ROS production in human leukaemia cells [32]. 2ME2 also induces ROS generation in cell lines including myeloid leukaemia U937 cells, myeloid leukaemia HL-60 cell line, human acute T cell leukaemia cells (Jurkat cells), HeLa cells and MCF-7 cells [32-35]. Azad, et al (2009) revealed that ROS can induce autophagy by means of Atg4, catalase and the mitochondrial electron transport chain leading to both cell survival and cell death and is a selective towards cancer treatment. ROS generation upregulates Beclin-1 expression by an unknown mechanism increasing the occurrence of autophagy and hydrogen peroxide is essential for starvation-induced autophagy and directly targets Atg4 for oxidation and inactivates Atg4 inducing autophagy [36]. In addition, upregulated mitochondrial matrix calcium leads to increased reactive oxygen species production results in a reduction of the mitochondrial membrane potential (permeability transition pore is triggered). The latter leads to cytochrome c release from the mitochondria and subsequent activation of caspases leading to apoptosis [37,38]. Ling, et al (2003) reported that upregulated reactive oxygen species was accompanied by a reduction in the mitochondrial membrane potential and subsequent apoptosis [39]. Thus, the increased ROS production accompanied with reduced mitochondrial potential found in this study suggests that both of the above-mentioned contribute to the apoptosis induced by 2MEBM. The novel finding regarding the in vitro influence of 2MEBM on either of these reactive oxygen species has not been reported as previously.
Conclusion
In conclusion, the aims of this project namely, to investigate the effects of 2MEBM exposure in the MCF-7 cell line on cell cycle progression, reactive oxygen species production and the possible induction of cell death were achieved. 2MEBM exposure resulted in mitochondrial membrane potential reduction and increased ROS production and apoptosis. The data generated from this project contributes to the known scientific knowledge regarding the in vitro effects of 2MEBM, will possibly lead to new possible in vitro targets for cancer therapies. Future studies concerning the action mechanisms of 2MEBM is necessary to produce a better understanding regarding the treatment of cancer and may possibly contribute to the development and/or improvement of novel chemotherapeutic agents.
Competing interests
The authors declare that they have no competing interests.
Authors' contributions
MHV was involved in the project design, conducted the experiments and data analysis in this project and drafted the article. AMJ was involved in the planning of this project, project supervision, funding acquisition from various received grants, interpretation of data and revision of the manuscript. All authors have contributed and approved the final manuscript.
Contributor Information
Michelle H Visagie, Email: michellevisagie@yahoo.com.
Anna M Joubert, Email: annie.joubert@up.ac.za.
Acknowledgements
The flow cytometer (Department of Pharmacology, University of Pretoria, Pretoria, South Africa) was employed during the research project. This study was supported by grants from the Medical Research Council of South Africa, the Cancer Association of South Africa, Research committee of University of Pretoria (RESCOM), National Research Foundation and the Struwig-Germeshuysen Cancer Research Trust of South Africa.
References
- Newman SP, Leese MP, Purohit A, James DRC, Rennie CE, Potter BV, Reed MJ. Inhibition of in vitro angiogenesis by 2-Methoxy- and 2-ethyl-estrogen sulphamates. Int J Cancer. 2004;109:533–540. doi: 10.1002/ijc.20045. [DOI] [PubMed] [Google Scholar]
- Funakoshi T, Birsner AE, D'Amato RJ. Antiangiogenic effect of oral 2-methoxyestradiol on choroidal neovascularization in mice. Exp Eye Res. 2006;83(5):1102–1107. doi: 10.1016/j.exer.2006.05.016. [DOI] [PubMed] [Google Scholar]
- Ireson CR, Chander SK, Purohit A, Perera S, Newman SP, Parish D, Leese MP, Smith AC, Potter BV, Reed MJ. Pharmacokinetics and efficacy of 2-methoxyoestradiol and 2-methoxyoestradiol-bis-sulphamate in vivo in rodents. Br J Cancer. 2004;90:932–937. doi: 10.1038/sj.bjc.6601591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matei D, Schilder J, Sutton G, Perkins S, Breen T, Quon C, Sidor C. Activity of 2 methoxyestradiol (Panzem® NCD) in advanced, platinum-resistant ovarian cancer and primary peritoneal carcinomatosis: A hoosier oncology group trial. Gynaecol Oncol. 2009;115(1):90–96. doi: 10.1016/j.ygyno.2009.05.042. [DOI] [PubMed] [Google Scholar]
- Lakka SS, Rao JS. Angiogenic therapy in brain tumors. Expert Rev Neurother. 2008;8(10):1457–1473. doi: 10.1586/14737175.8.10.1457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sutherland TE, Anderson RL, Hughes RA, Altmann E, Schuliga M, Ziogas J, Stewart AG. 2-Methoxyestradiol- a unique blend of activities generating a new class of anti-tumour/anti-inflammatory agents. Drug Discov Today. 2007;12(13/14):577–584. doi: 10.1016/j.drudis.2007.05.005. [DOI] [PubMed] [Google Scholar]
- Chander SK, Foster PA, Leese MP, Newman SP, Potter BVL, Purohit A, Reed MJ. In vivo inhibition of angiogenesis by sulphamoylated derivatives of 2-methoxyestradiol. Br J Cancer. 2007;96:1368–1376. doi: 10.1038/sj.bjc.6603727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Foster PA, Newman SP, Leese MP, Bernetiere S, Diolez C, Camara J, Hacher B, Baronnet MM, Ali T, Potter BV, Reed MJ, Purohit A. A new micronized formulation of 2-methoxyestradiol-bis-sulphamate (STX140) is therapeutically potent against breast cancer. Anticancer Res. 2008;28:577–581. [PubMed] [Google Scholar]
- Kirches E, Wraich-Kirches M. 2-Methoxyestradiol as a potential cytostatic drug in gliomas? Anticancer Agents Med Chem. 2009;9(1):55–65. doi: 10.2174/187152009787047725. [DOI] [PubMed] [Google Scholar]
- Lakhani NJ, Lepper ER, Sparreboom A, Dahut WL, Venitz J, Figg WD. Determination of 2-methxyestradiol in human plasma, using liquid chromatography/tandem mass spectrometry. Rapid Commun Mass Spectrom. 2005;19:1176–1182. doi: 10.1002/rcm.1902. [DOI] [PubMed] [Google Scholar]
- Reed MJ, Purohit A, Woo LWL, Newman SP, Potter BVL. Steroid Sulfatase: Molecular Biology, regulation, and inhibition. Endocr Rev. 2005;26(2):171–202. doi: 10.1210/er.2004-0003. [DOI] [PubMed] [Google Scholar]
- Utsumi T, Leese MP, Chander SK, Gaukroger K, Purohit A, Newman SP, Potter BV, Reed MJ. The effects of 2-methoxyoestrogen sulphamates on the in vitro and in vivo proliferation of breast cancer cells. J Steroid Biochem Mol Biol. 2005;94:219–227. doi: 10.1016/j.jsbmb.2005.01.022. [DOI] [PubMed] [Google Scholar]
- Day JM, Foster PA, Tutill HJ, Newman SP, Ho YT, Leese MP, Potter BM, Reed MJ, Purohit A. BCRP expression does not result in resistance to STX140 in vivo, despite the increased expression of BCRP in A2780 cells in vitro after long-term STX140 exposure. Br J Cancer. 2009;100:476–486. doi: 10.1038/sj.bjc.6604873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newman SP, Ireson CR, Tutill HJ, Day JM, Parsons MFC, Leese MP, Potter BVL, Reed MJ, Purohit A. The role of 17β-hydroxysteriod dehydrogenase in modulating the activity of 2-methoxyestradiol in breast cancer cells. Cancer Res. 2006;66(1):324–330. doi: 10.1158/0008-5472.CAN-05-2391. [DOI] [PubMed] [Google Scholar]
- Tagg SL, Foster PA, Leese MP, Potter BV, Reed MJ, Purohit A, Newman SP. 2-Methoxyoestradiol-3,27-O,O-bis-sulphamate and 2-deoxy-D-glucose in combination: a potential treatment for breast and prostate cancer. Br J Cancer. 2008;99(11):1842–1848. doi: 10.1038/sj.bjc.6604752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ho YT, Newman SP, Purohit A, Leese MP, Potter BV, Reed MJ. The effects of 2-methoxy oestrogens and their sulphamoylated derivatives in conjunction with TNF-alpha on endothelial and fibroblast cell growth, morphology and apoptosis. J Steroid Biochem Mol Biol. 2003;86:189–196. doi: 10.1016/S0960-0760(03)00269-3. [DOI] [PubMed] [Google Scholar]
- Suzuki RN, Newman SP, Purohit A, Leese MP, Potter BV, Reed MJ. Growth inhibition of multi-drug-resistant breast cancer cells by 2-methoxyoestradiol-bis-sulphamate and 2-ethyloestradiol-bis-sulphamate. J Steroid Biochem Mol Biol. 2003;84:269–278. doi: 10.1016/S0960-0760(03)00035-9. [DOI] [PubMed] [Google Scholar]
- Visagie MH, Joubert AM. 2-Methoxyestradiol-bis-sulfamate induces apoptosis and autophagy in a tumorigenic breast epithelial cell line. Mol Cell Biochem. 2011. in press . [DOI] [PubMed]
- Visagie MH, Joubert AM. The In vitro effects of 2-Methoxyestradiol-bis-sulphamate on cell numbers, membrane integrity and cell morphology, and the possible induction of apoptosis and autophagy in a non-tumorigenic breast epithelial cell line. Cell Mol Biol Lett. 2010;15:564–581. doi: 10.2478/s11658-010-0030-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galluzzi L, Zamzami N, de la Motte Rouge T, Lemaire C, Brenner C, Kroemer G. Methods for assessment of mitochondrial membrane permeabilization in apoptosis. Apoptosis. 2007;12:803–813. doi: 10.1007/s10495-007-0720-1. [DOI] [PubMed] [Google Scholar]
- Bouchier-Hayes L, Muñoz-Pinedo C, Connell S, Green DR. Measuring apoptosis at the single cell level. Methods. 2008;44(3):222–228. doi: 10.1016/j.ymeth.2007.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raobaikady B, Reed MJ, Leese MP, Potter BVL, Purohit A. Inhibition of MDA-MB-321 cell cycle progression and cell proliferation by C-2 -substituted oestradiol mono- and bis-3-O-sulphamates. Int J Cancer. 2005;117:150–159. doi: 10.1002/ijc.21066. [DOI] [PubMed] [Google Scholar]
- Foster PA, Ho YT, Newman SP, Kasprzyk MP, Leese MP, Potter BV, Reed MJ, Purohit A. 2ME2BM and 2-EtE2bisMATE induce cell cycle arrest and apoptosis in breast cancer xenografts as shown by a novel ex vivo technique. Breast Cancer Res Treat. 2008;111:251–260. doi: 10.1007/s10549-007-9791-5. [DOI] [PubMed] [Google Scholar]
- Wood L, Leese MP, Mouzakiti A, Purohit A, Potter BV, Reed MJ, Packham G. 2ME2BM induces caspase-dependent apoptosis in CAL51 breast cancer cells and overcomes resistance to TRAIL via cooperative activation of caspases. Apoptosis. 2004;9:323–332. doi: 10.1023/b:appt.0000025809.80684.bd. [DOI] [PubMed] [Google Scholar]
- Elmore S. Apoptosis: a review of programmed cell death. Toxicol Pathol. 2007;35:495–516. doi: 10.1080/01926230701320337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin Z, El-Deiry WS. Overview of cell death signaling pathways. Cancer Biol Ther. 2005;4:139–163. doi: 10.4161/cbt.4.2.1508. [DOI] [PubMed] [Google Scholar]
- Basanez G, Nechushtan A, Drozhinin O, Chanturiya A, Choe E, Tutt S, Wood KA, Hsu YT, Zimmerberg J, Youle RJ. Bax, but not Bcl-XL decreases the lifetime of planar phospholipid bilayer membranes at subnanomolar concentrations. Proc Natl Acad Sci USA. 1999;96:5492–5497. doi: 10.1073/pnas.96.10.5492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desagher S, Osen-Sand A, Nichols A, Eskes R, Montessuit S, Lauper S, Maundrell K Antonsson B, Martinou JC. Bid-induced conformational change of Bax is responsible for mitochondrial cytochrome c release during apoptosis. J Cell Biol. 1999;144(5):891–901. doi: 10.1083/jcb.144.5.891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo X, Budihardio I, Zou H, Slaughter C, Wang X. Bid, a Bcl-2 interacting protein, mediates cytochrome c release from mitochondria in response to activation of cell surface death receptors. Cell. 1998;94:481–490. doi: 10.1016/S0092-8674(00)81589-5. [DOI] [PubMed] [Google Scholar]
- Narita M, Shimizu S, Ito T, Chittenden T, Lutz RJ, Matsuda H, Tsujimoto Y. Bax interacts with the permeability transition pore to induce permeability transition and cytochrome c release in isolated mitochondria. Proc Natl Acad Sci USA. 1998;95:14681–14686. doi: 10.1073/pnas.95.25.14681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green DR, Kroemer G. The pathophysiology of mitochondrial cell death. Science. 2004;305:626–9. doi: 10.1126/science.1099320. [DOI] [PubMed] [Google Scholar]
- Gao N, Ranhmani M, Dent P, Grant S. 2-Methoxyestradiol-induced apoptosis in human leukemia cells proceeds through a reactive oxygen species and Akt-dependent process. Oncogene. 2005;24:3797–3809. doi: 10.1038/sj.onc.1208530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- She MR, Li JG, Guo KY, Lin W, Du X, Niu XQ. Requirement of reactive oxygen species generation in apoptosis of leukemia cells induced by 2-methoxyestradiol. Acta Pharmacol Sin. 2007;28(7):1037–1044. doi: 10.1111/j.1745-7254.2007.00604.x. [DOI] [PubMed] [Google Scholar]
- Simizu S, Takada M, Umezawa K, Imoto M. Requirement of caspase-3 (like) protease-mediated hydrogen peroxide production for apoptosis induced by various anticancer drugs. J Biol Chem. 1998;273:26900–26907. doi: 10.1074/jbc.273.41.26900. [DOI] [PubMed] [Google Scholar]
- Stander BA, Marais S, Vorster CJ, Joubert AM. In Vitro effects of 2-methoxyestradiol on morphology, cell cycle progression, cell death and gene expression changes in the tumorigenic MCF-7 breast epithelial cell line. J Steroid Mol Biol. 2010;119:149–160. doi: 10.1016/j.jsbmb.2010.02.019. [DOI] [PubMed] [Google Scholar]
- Azad MB, Chen Y, Gibson B. Regulation of autophagy by reactive oxygen species (ROS): implications for cancer progression and cancer. Antioxid Redox Signal. 2009;11(4):1–14. doi: 10.1089/ars.2008.2270. [DOI] [PubMed] [Google Scholar]
- Chua YS, Chua YL, Hagen T. Structure activity analysis of 2-methoxyestradiol analogues reveals targeting of microtubules as major mechanism of antiproliferative and proapoptotic activity. Mol Cancer Ther. 2010;9:224–235. doi: 10.1158/1535-7163.MCT-09-1003. [DOI] [PubMed] [Google Scholar]
- Brooks PS, Yoon Y, Robotham JL, Anders MW, Sheu SS. Calcium, ATP, and ROS: a mitochondrial love-hate triangle. Am J Physiol Cell Physiol. 2004;287:C817–833. doi: 10.1152/ajpcell.00139.2004. [DOI] [PubMed] [Google Scholar]
- Ling YH, Liebes L, Zou Y, Perez-Soler R. Reactive oxygen species generation and mitochondrial dysfunction in the apoptotic response to botrezomib, a novel proteosome inhibitor, in human H460 non-small cell lung cancer cells. J Biol Chem. 2003;278:33714–33723. doi: 10.1074/jbc.M302559200. [DOI] [PubMed] [Google Scholar]