Scientific Abstract
The authors examined pregnancy and obstetric complications in association with autism spectrum disorders (ASD) in children of participants from the Nurses' Health Study II, a prospective national cohort with information collected through biennial mailed questionnaires since 1989. Logistic regression was used to obtain crude and adjusted odds ratios for ASD, and by diagnostic subgroup. 793 cases were reported among 66,445 pregnancies. Pregnancy complications and obstetric suboptimality factors were assessed by maternal report of occurrence in first birth and, in secondary analyses, in any birth. Complications and a suboptimality score were significantly associated with having a child with ASD (OR 1.49, 95% CI 1.26, 1.77, p <.0001 for pregnancy complications in first birth and 2.76, 95% CI 2.04, 3.74, p <.0001 comparing individuals with 4 or more obstetric suboptimality factors in first birth to those with none; results similar when assessed in any birth). In particular, gestational diabetes was associated with a significantly increased risk of ASD in results of primary and sensitivity analyses (OR in primary analysis = 1.76, 95% CI 1.34, 2.32, p <.0001); suboptimal parity and suboptimal age at first birth were also individual factors associated with ASD. Associations were similar by diagnostic subgroup, suggesting autism, Asperger syndrome, and other Pervasive Developmental Disorders are all associated with pregnancy complications. Consistent with previous research, the general class of pregnancy complications was associated with autism spectrum disorders as a whole. Additional work will be required to more fully assess the role of gestational diabetes.
Keywords: Autism Spectrum Disorders, Gestational Diabetes, Obstetric Labor Complications, Pregnancy Complications
The results of numerous case-control (Burd et al., 1999; Glasson et al., 2004; Hultman et al., 2002; Juul-Dam et al., 2001; Maimburg et al., 2006) and cohort studies (Croen et al., 2002; Eaton et al., 2001; Lauritsen et al., 2005) suggest that obstetric complications may be associated with autism spectrum disorders (ASD). A recent meta analysis, which examined 40 studies of prenatal factors in association with ASD, found evidence for association with parental age and gestational diabetes, as well as a few other prenatal factors, but noted the inconsistency in results across studies (Gardener et al., 2009). Further, the authors highlighted the fact that few factors have been examined in multiple rigorous studies, demonstrating the need for replication. Questions remain, including the mechanisms underlying these associations, whether associations differ by diagnostic subgroup, and whether certain obstetric complications confer a higher risk than others (Bolton et al., 1997; Gardener et al., 2009; Gillberg & Cederlund, 2005). Further, the majority of the larger studies examining these factors have been conducted in other countries(Glasson et al., 2004; Hultman et al., 2002; Larsson et al., 2005; Lauritsen et al., 2005), suggesting a need for large cohort studies in the USA.
We sought to replicate previous results and provide additional data on these issues in a large United States cohort, the Nurses' Health Study II. We hypothesized that we would find similar associations across diagnostic subgroups, and that our results would be consistent with previous research demonstrating a significant association between ASD and the general category of pregnancy and obstetric complications, rather than specific factors.
Materials and Methods
Study population
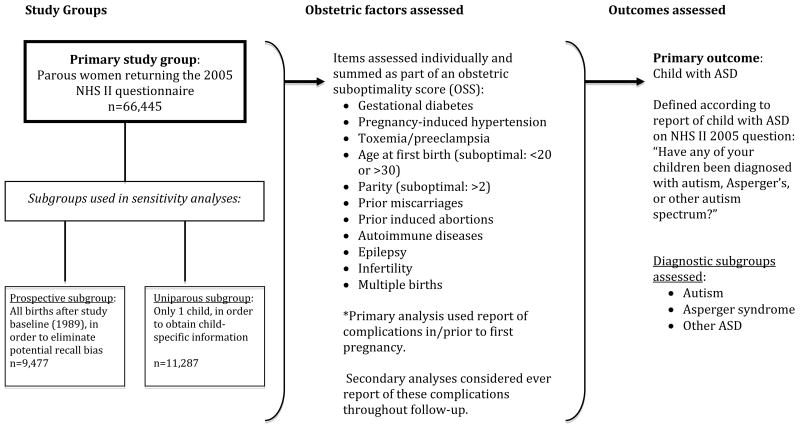
The Nurses' Health Study II (NHS II) is a prospective cohort of 116,608 female nurses aged 25-42 when recruited in 1989, who have been followed by biennial mailed questionnaires to assess the incidence of cancer and other chronic diseases. The Partners Health Care Institutional Review Board has reviewed and approved the methods of this study, and completion and return of questionnaires sent by U.S. mail constitutes implied consent. The 2005 questionnaire included an item asking women if they had a child diagnosed with autism, Asperger syndrome, or “other autism spectrum disorder”. Only parous women with at least one birth before the end of 2003 (to allow time for report of diagnosis by 2005) who also returned the 2005 questionnaire when the outcome was assessed were included; this provided a primary study population of 66,445 women. Among those women, 9,477 had their first birth in 1989 or later and were included in our prospective subgroup sensitivity analysis, while 11,287 women had only one pregnancy and were included in our uniparous sensitivity analysis. Figure 1 summarizes the groups, exposures, and outcomes used in this study.
Figure 1.
Summary of study groups, exposures, and outcome definitions used in primary and sensitivity analyses. Abbreviations: NHS II = Nurses Health Study II. Due to the way in which the NHS II question was asked, we did not know year of birth for the ASD children, except for those women with 1 child (uniparous group); however, using information on age at first and last birth when available (for most women) identified a known period of time in which the child was born, which for most women was within 5 years, and was similar in cases and controls.
Outcome information
Cases were defined as those women meeting inclusion criteria and reporting having had a child with autism, Asperger syndrome, or other autism spectrum, as reported on the 2005 questionnaire. While it is expected that the ‘other autism spectrum’ category includes pervasive-developmental disorder not-otherwise specified (PDD-NOS), this disorder was not specified on the questionnaire. If the ASD question was left blank but the rest of the questionnaire was completed, it was assumed the mother had no ASD affected children. No further information on the affected child was available. Two Center for Disease Control and Prevention (CDC) studies have utilized parental report of ASD, and a high concordance in estimated prevalence of ASD between the two studies was seen, as was a strong correlation between report of diagnosis and developmental and behavioral problems as measured on screening items (Center for Disease Control and Prevention, 2006).
Exposure information
A history of past pregnancies, miscarriages, gestational diabetes, toxemia, and preeclampsia was assessed at baseline and updated every two years. Occurrence of induced abortions was assessed beginning in 1993 (with age ranges) and every questionnaire year thereafter. Pregnancy-related high blood pressure was assessed on the 1993, 1995, and 1997 questionnaires. All NHS questionnaires are available online at http://www.channing.harvard.edu/nhs/questionnaires/index.shtml. In order to ensure exposure occurrence prior to a woman's first delivery, and thus before the birth of the affected child (since for women with multiple children we do not have information on which of the nurses' children may be affected), exposures and other covariates were considered when reported prior to first birth or during first pregnancy, as appropriate (prior to first birth for miscarriages, abortions, and infertility; during first pregnancy for toxemia, gestational diabetes, pregnancy related high blood pressure and twin birth at first birth). In secondary analyses we considered classification of these variables as ‘ever reported’ in any birth/pregnancy through 2003. Exposures of interest in this analysis may therefore have occurred before or after the birth of the child diagnosed with autism, but may be considered useful in determining overall differences between women who have a child with ASD and those who do not, and in comparison to the primary analyses.
Statistical analyses
Univariate relationships and basic characteristics of exposures and covariates were assessed by descriptive statistics, χ2 tests, and t-tests. Relationships between ASD and reproductive and obstetric complications (as both individual factors and summarized in a suboptimality score) under study were assessed by multivariate logistic regression, with and without adjustment for potential confounders. Potential confounders considered, which have been associated with autism and could affect risk of complications, included nurses' age at baseline (1989) in years, age at first birth in years, race (as binary white/other), marital status (as married, past married, or never married as of first birth), income (in 5 levels of household income), spouse education (in 4 categories: high school or less, 2 or 4 year college, and graduate), and parity (as a continuous variable). When assessing risk of individual complications, the other reproductive factors under study were also considered in multivariate models (for example, when assessing risk of gestational diabetes, adjustment for history of miscarriages, prior abortions, and pregnancy related high blood pressure, etc, was considered). Gestational diabetes, toxemia/preeclampsia, and pregnancy-related hypertension were considered individually, and, to address potential metabolic syndrome, were also assessed as a single ‘pregnancy complications’ variable (1 if reported any and 0, otherwise).
We also created an obstetric suboptimality score (OSS) similar to those used in previous studies for the purposes of comparison to prior work, and to assess the risk associated with increasing number of suboptimality factors (Bolton et al., 1997; Gillberg & Gillberg, 1983; Lord et al., 1991; Piven et al., 1993). Due to limited availability of information, this score was a modified, reduced version of the Gillberg score, although other scores were considered in selecting OSS factors (Gardener, et al, 2009; Glasson et al, 2004; Stein et al., 2006). Our OSS is a sum of the following items (defined to be consistent with the Gillberg and other scores, which were designed to assess optimal birth and pregnancy conditions): suboptimal age at first birth (defined as <20 or >30, (Gillberg and Gillberg, 1983)), suboptimal parity (defined as >2, (Gillberg & Gillberg, 1983)), history of induced abortion, prior miscarriage, history of infertility, pregnancy-related high blood pressure, gestational diabetes, toxemia, twin births, epilepsy, and autoimmune diseases. This score gives an equal weight to each factor, with a possible total score of 11. A second OSS was created which did not include suboptimal age at first birth, for purposes of comparison to subgroups used in sensitivity analyses (due to older age at first birth in these subgroups, which are described below). Many studies have found that a higher number of suboptimal factors is associated with increased risk of ASDs (Brimacombe et al., 2007; Gillberg & Gillberg, 1983), suggesting that the OSS is a useful complement to analyses of individual items. Individuals with 1, 2, 3, and 4 or more factors were compared to those with none, using indicator variables in regression models. The ordinal score test was used to assess evidence for a trend of the OSS. We also examined the potential for a non-linear relationship between the OSS and ASD non-parametrically using cubic splines (Durrleman & Simon, 1989; Govindarajulu et al., 2007).
For all analyses, crude, age-adjusted, age and age-at-first-birth (AFB) adjusted, and multivariate models were compared. Analyses were also conducted by diagnostic subgroup. Missingness was low (<5%) for race and <10% for marital status and paternal education, although income was missing in approximately 20%; the missing indicator method was used to handle the missing covariate data (Miettinen, 1985).
Sensitivity analyses
A number of sensitivity analyses were utilized to test the robustness of results. As we currently have no information on which child is the affected child (the 2005 question asked only whether women had any affected children), analyses were repeated among women who had only one pregnancy (the ‘uniparous group’). For these women, we know the year of birth of the reported child with ASD, and whether any reported complication refers to the child with ASD. As our primary study population included women with children born before 1989, for any exposures reported at baseline which occurred in the past, reporting was retrospective even though we only scored it if it was prior to first birth /during first pregnancy. In order to assess the possibility of recall bias, we created a prospective subgroup and excluded the 56,968 women with any children born before 1989 to ensure that exposures not only occurred prior to first birth/during first pregnancy but were also measured before the first birth (and therefore the outcome).
Results
A total of 793 mothers reported having had a child with an ASD among 66,445 pregnancies through the end of the study period (representing 1.2% of the study group). ASD mothers were, on average, significantly younger at baseline than non-ASD mothers, but had a later age at first birth (Table 1). Crude comparisons demonstrated differences between cases (mothers who reported having a child with ASD) and non-cases (mothers who did not report having a child with ASD) with regard to a number of reproductive factors (Table 1). ASD mothers were more likely to have reported abortions, miscarriages, toxemia, gestational diabetes, pregnancy-related high blood pressure, and to have higher OSS scores. These differences were observed whether exposures were classified as occurring prior to the first birth or at any time. Frequencies of obstetric complications among the non-case pregnancies were consistent with those in the general U.S. population (Hunt & Shuller, 2007; Weinberg & Wilcox, 1998).
Table 1. Reproductive factors and characteristics of the study population.
| ASD mothers n=793 | Non-ASD mothers n=65,652 | |
|---|---|---|
| Variable | n(%) or mean (std) | n(%) or mean (std) |
| Age at baseline | ||
| 24-29 | 240 (30.3%) | 11061 (16.9%) |
| 30-35 | 366 (46.2%) | 25552 (38.9%) |
| 36-42 | 187 (23.6%) | 29039 (44.2%) |
|
| ||
| Mean age at baseline | 32.3 (4.4) | 34.6 (4.7) |
|
| ||
| Age at first birth | ||
| <20 | 19 (2.4%) | 3762 (5.7%) |
| 20-<25 | 142 (17.9%) | 19127 (29.1%) |
| 25-<30 | 301 (38.0%) | 26942 (41.0%) |
| 30-<35 | 215 (27.1%) | 11432 (17.4%) |
| 35+ | 116 (14.6%) | 4389 (6.7%) |
|
| ||
| Mean age at first birth | 28.9 (5.1) | 26.6 (4.8) |
|
| ||
| Average parity | 2.48 (1.0) | 2.30 (0.95) |
|
| ||
| Lost pregnancy/miscarriage prior to first birth* | 241 (30.4%) | 15125 (23.0%) |
|
| ||
| Ever had a miscarriage | 395 (49.8%) | 27550 (42.0%) |
|
| ||
| Abortion prior to first pregnancy* | 144 (18.2%) | 7971 (12.1%) |
|
| ||
| Ever had an abortion | 168 (21.2%) | 10266 (15.6%) |
|
| ||
| Any report of ectopic pregnancy | 19 (2.4%) | 1421 (2.2%) |
|
| ||
| Pregnancy complications in first pregnancy1 | 175 (22.1%) | 9944 (15.2%) |
|
| ||
| Any report of pregnancy complications | 224 (28.3%) | 12573 (19.2%) |
|
| ||
| Gestational diabetes at first birth* | 58 (7.3%) | 2111 (3.2%) |
|
| ||
| Any report of gestational diabetes | 83 (10.5%) | 3591 (5.5%) |
|
| ||
| Toxemia at first birth* | 86 (10.8%) | 5882 (9.0%) |
|
| ||
| Any report of toxemia | 134 (16.9%) | 8144 (12.4%) |
|
| ||
| Pregnancy-related HBP at first birth* | 85 (10.7%) | 5799 (8.8%) |
|
| ||
| Pregnancy-related high blood pressure (HBP) | 111 (14.0%) | 6700 (10.2%) |
|
| ||
| Endometriosis | 88 (11.1%) | 6867 (10.5%) |
|
| ||
| Twin birth in 1st pregnancy* | 25 (3.2%) | 1143 (1.7%) |
|
| ||
| Ever report of twin birth | 97 (12.2%) | 4054 (6.2%) |
|
| ||
| Supoptimal parity: >2* 2 | 332 (41.9%) | 22646 (34.5%) |
|
| ||
| Suboptimal maternal age: <20 or >30* | 290 (36.6%) | 16095 (24.5%) |
|
| ||
| Maternal epilepsy* | 21 (2.7%) | 1218 (1.9%) |
|
| ||
| Maternal autoimmune disease*3 | 231 (29.1%) | 16117 (24.6%) |
|
| ||
| Obstetric Suboptimality Score (OSS) total- reporting of complications at or prior to first birth4 | ||
| 0 | 75 (9.5%) | 13279 (20.2%) |
| 1 | 227 (28.6%) | 22594 (34.4%) |
| 2 | 216 (27.2%) | 15833 (24.1%) |
| 3 | 157 (19.8%) | 8892 (13.5%) |
| 4+ | 118 (14.9%) | 5054 (7.7%) |
|
| ||
| Mean OSS | 2.1 (1.3) | 1.6 (1.3) |
All variables are self-reported. Due to lack of information on affected child year of birth, complications in first birth as well as ever reported are compared.
Pregnancy complications include gestational diabetes, pregnancy-related high blood pressure, and toxemia.
Suboptimal parity and age at first birth as defined by Gillberg & Gillberg (C. Gillberg, Gillberg C., 1983).
Autoimmune diseases include: diabetes, thyroid disease, Graves disease, asthma, multiple sclerosis, ulcerative colitis, lupus, and rheumatoid arthritis.
Variables included in the OSS are starred (*) in Table; history of infertility prior to first birth was also included in the score.
In the primary analysis, pregnancy complications and obstetric suboptimality scores were significantly associated with ASD (Table 2), as were the following individual factors: suboptimal age-at-first birth, suboptimal parity, abortions, and autoimmune disease in the mother. When exposures were assessed in secondary analyses as ever reported in any birth, results were similar in both magnitude and direction (supplementary data).
Table 2. Pregnancy complications and obstetric suboptimality factors in or prior to first pregnancy in relation to ASD risk in the study group (N=66,445).
| Variable | Age-adjusted OR | Age & AFB adjusted OR | Fully-adjusteda | |
|---|---|---|---|---|
| OR (95% CI) | p-value | |||
| Miscarriage | 1.35 | 1.11 | 1.00 (0.83, 1.21) | 0.99 |
|
| ||||
| Abortion | 1.47 | 1.25 | 1.26 (1.01, 1.57) | 0.04 |
|
| ||||
| Pregnancy Complications | 1.50 | 1.45 | 1.49 (1.26, 1.77) | <.0001 |
|
| ||||
| Toxemia | 1.22 | 1.30 | 1.36 (1.04, 1.79) | 0.03 |
|
| ||||
| Gestational diabetes | 2.07 | 1.80 | 1.76 (1.34, 2.32) | <.0001 |
|
| ||||
| Pregnancy-related HBP | 1.18 | 1.13 | 0.96 (0.73, 1.26) | 0.76 |
|
| ||||
| OSSb | ||||
| 0 | 1.0 | 1.0 | 1.0 | |
| 1 | 1.71 | 1.62 | 1.61 (1.24, 2.10) | 0.0003 |
| 2 | 2.29 | 2.03 | 2.04 (1.56, 2.66) | <.0001 |
| 3 | 2.88 | 2.32 | 2.33 (1.75, 3.09) | <.0001 |
| 4+ | 3.79 | 2.76 | 2.76 (2.04, 3.74) | <.0001 |
|
| ||||
| OSS-2c | ||||
| 0 | 1.0 | 1.0 | 1.0 | |
| 1 | 1.38 | 1.42 | 1.41 (1.14, 1.74) | 0.002 |
| 2 | 1.93 | 1.82 | 1.81 (1.45, 2.25) | <.0001 |
| 3 | 2.00 | 1.81 | 1.80 (1.39, 2.32) | <.0001 |
| 4+ | 3.13 | 2.75 | 2.74 (2.05, 3.65) | <.0001 |
|
| ||||
| Suboptimal AFB | 1.74 | -- | 1.75 (1.50, 2.05) | <.0001 |
|
| ||||
| Suboptimal parity | 1.30 | 1.73 | 1.52 (1.31, 1.76) | <.0001 |
|
| ||||
| Epilepsy | 1.44 | 1.47 | 1.39 (0.89, 2.16) | 0.14 |
|
| ||||
| Autoimmune disease | 1.40 | 1.43 | 1.37 (1.17, 1.60) | <.0001 |
|
| ||||
| Twin birth -1st pregnancy | 1.53 | 1.20 | 1.30 (0.86, 1.96) | 0.22 |
Table abbreviations: AFB= age at first birth, HBP= high blood pressure, OSS= obstetric suboptimality score. OSS and variables above are reported at or prior to first pregnancy. Values shown are odds ratios, with 95% confidence intervals in parentheses for fully adjusted estimates. Pregnancy complications and obstetric suboptimality factors assessed here were considered if reported during first pregnancy or prior to first birth as applicable.
For individual complications, fully adjusted models include the following demographic variables: race, marital status, income, and spouse education, as well as age, AFB, and parity, and, when not being assessed as the complication of interest, twin births, pregnancy complications, induced abortions, and miscarriages. When omitting adjustment for twin births, results were slightly stronger but did not change overall significance. The fully adjusted OSS models included adjustment for demographic factors as well as age and age at first birth; additional adjustment for parity as a continuous variable slightly attenuated, but did not alter significance of, results.
OSS included the following items, defined in the text and in Table 1: suboptimal age at first birth and parity, maternal epilepsy, maternal autoimmune disease, twin birth, gestational diabetes, toxemia, pregnancy-related HBP, history of infertility, abortion prior to first birth, and miscarriage prior to first birth.
OSS-2 does not include the item suboptimal age at first birth; shown for comparison to Table 3 and supplementary data. Case n for this OSS-2 as follows, from 0 to 4+ factors: 129, 253, 221, 113, 77.
Specifically, pregnancy complications were associated with an approximately 50% increased odds of ASD (both first pregnancy and ever), and this association was similar and remained significant in the prospective and uniparous groups (Table 3). For individual complications, both toxemia and gestational diabetes were significantly associated with increased risk of ASD both during first pregnancy and ever. However, only gestational diabetes remained significantly associated with ASD in the prospective group (OR 1.59, 95% CI 1.05, 2.41, p=0.03). None of the three individual complications assessed was associated with ASD in the uniparous group, although the point estimate for gestational diabetes was similar to that of the prospective group. There were also fewer exposed cases in the prospective subgroup, which negatively affected our power to detect associations by these factors.
Table 3. Pregnancy complications and obstetric suboptimality factors in or prior to first pregnancy in relation to ASD risk in the prospective and uniparous subgroups.
| Prospective (N=9,477)a |
Uniparous (N=11,287)b |
|||||
|---|---|---|---|---|---|---|
| Variable | Case | Fully-adjusted | Case | Fully-adjusted | ||
| n | OR (95% CI) | p-value | n | OR (95% CI) | p-value | |
| Miscarriage | 88 | 0.82 (0.59, 1.14) | 0.23 | 36 | 0.72 (0.43, 1.19) | 0.20 |
|
| ||||||
| Abortion | 22 | 0.90 (0.51, 1.58) | 0.70 | 23 | 1.30 (0.73, 2.29) | 0.37 |
|
| ||||||
| Pregnancy Complications | 61 | 1.50 (1.11, 2.01) | 0.008 | 30 | 1.71 (1.11, 2.65) | 0.02 |
|
| ||||||
| Toxemia | 17 | 1.22 (0.68, 2.18) | 0.50 | 14 | 1.47 (0.72, 2.99) | 0.29 |
|
| ||||||
| Gestational diabetes | 26 | 1.59 (1.05, 2.41) | 0.03 | 8 | 1.51 (0.73, 3.16) | 0.27 |
|
| ||||||
| Pregnancy-related HBP | 23 | 1.62 (0.95, 2.77) | 0.08 | 17 | 1.09 (0.56, 2.10) | 0.80 |
|
| ||||||
| OSS-2c | ||||||
| 0 | 49 | 1.0 | -- | 22 | 1.0 | -- |
| 1 | 62 | 0.95 (0.65, 1.39) | 0.79 | 23 | 0.96 (0.53, 1.73) | 0.87 |
| 2 | 66 | 1.24 (0.85, 1.80) | 0.27 | 27 | 1.28 (0.72, 2.27) | 0.41 |
| 3 | 33 | 1.11 (0.71, 1.74) | 0.66 | 15 | 1.20 (0.61, 2.35) | 0.60 |
| 4+ | 33 | 2.24 (1.42, 3.53) | 0.005 | 15 | 2.43 (1.23, 4.83) | 0.01 |
|
| ||||||
| Suboptimal AFB | 172 | 1.02 (0.72, 1.45) | 0.91 | 77 | 2.56 (1.59, 4.14) | 0.0001 |
|
| ||||||
| Suboptimal parity | 50 | 1.09 (0.78, 1.53) | 0.61 | NA | -- | -- |
|
| ||||||
| Epilepsy | 9 | 1.97 (0.99, 3.92) | 0.05 | 3 | 1.34 (0.41, 4.30) | 0.63 |
|
| ||||||
| Autoimmune disease | 52 | 1.03 (0.75, 1.41) | 0.88 | 31 | 1.21 (0.79, 1.88) | 0.38 |
|
| ||||||
| Twin birth- 1st pregnancy | 19 | 1.37 (0.84, 2.24) | 0.20 | 11 | 1.73 (0.90, 3.34) | 0.10 |
Abbreviations and notes as for Table 2. Fully adjusted as in Table 2 with the exception of no adjustment for parity in the uniparous subgroup.
The prospective subgroup includes 243 cases and 9,234 non-cases with first births by 1989 or later. Due to differing year of first report, prospective analyses for pregnancy-related high blood pressure and abortion includes 111 cases and 3,874 non-cases (first report in 1993).
The uniparous subgroup includes 102 cases and 11,185 non-cases reporting only 1 pregnancy.
Due to age distribution in these groups, OSS-2 does not include age at first birth.
The OSS was significantly associated with ASD in the primary analysis, with a significant dose-response type increase in risk of ASD with increase in OSS, and a nearly tripling of risk with 4 or more obstetric suboptimality factors (Table 2); associations were slightly stronger for ever reported exposures (supplementary data). Although the ordinal trend test was significant (p <0.0001), the relationship between the OSS and ASD was non-linear (p=0.007 for the non-linearity test) (Figure 2). The OSS was significantly associated with an increased risk of ASD in both the prospective and uniparous subgroups for OSS≥4; the association was weaker for OSS of 1-3 in these groups, although numbers and power were reduced (Table 3). Although suboptimal age at first birth and parity, induced abortion, and autoimmune disease were significantly associated with ASD in the primary analysis, only suboptimal age at first birth remained significantly associated with ASD in the uniparous group (OR=2.56, 95% CI 1.59, 4.14); none of these individual items were associated in the prospective group. Ever report of a twin birth was the only additional factor associated with an increased risk of ASD in the secondary analysis of factors occurring in any births (supplementary data).
Figure 2.
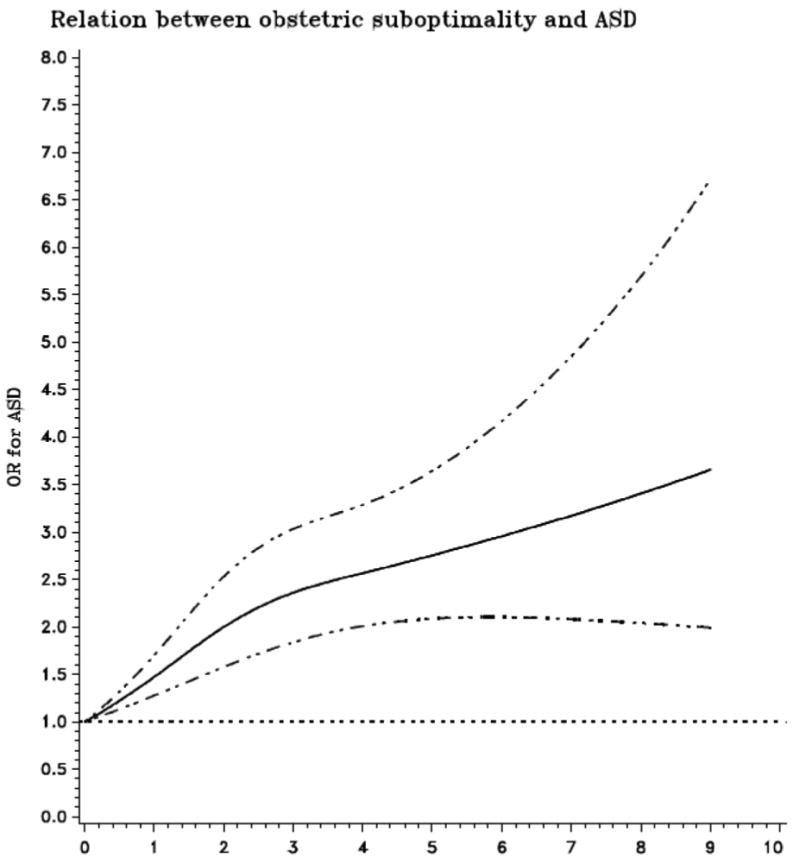
The graph shows the odds of ASD (Y axis) associated with value of obstetric suboptimality score (X axis). ORs on the Y axis represent values from fully adjusted models, as in Tables 3 and 4. The horizontal dotted line represents the null OR value of no association, while dashed lines flanking the solid line indicate 95% confidence intervals.
Results by diagnostic subgroup
Results by diagnostic subgroup in the full study population are shown in Table 4. Overall, autism, Asperger syndrome, and ‘other ASD’ cases had a similar profile of obstetric complications, with OSS of 2 or more factors, suboptimal age at first birth, and parity being the factors most consistently significantly associated with increased risk. In the prospective sensitivity analyses of diagnostic subgroups (supplementary data), OSS of ≥4 was significantly associated for all diagnostic subgroups, with a similar magnitude and strength as the primary analyses, while general pregnancy complications was significantly associated with only ‘other ASD.’ Gestational diabetes was the only individual pregnancy complication that was significantly associated with ASD in this analysis, and only for autism cases. Case numbers by exposure category were small in this prospective sensitivity analysis.
Table 4. Pregnancy complications and obstetric suboptimality factors in or prior to first pregnancy in relation to risk of diagnostic subgroups: autism, asperger syndrome, and other ASDa.
| Variable | Case | Age-adjusted OR | Age & AFB adjusted OR | Fully-adjusted | |
|---|---|---|---|---|---|
| n | OR (95% CI) | p-value | |||
| Autism (228 cases) | |||||
|
| |||||
| Miscarriage | 71 | 1.39 | 1.15 | 1.06 (0.75, 1.49) | 0.74 |
|
| |||||
| Abortion | 41 | 1.44 | 1.23 | 1.19 (0.79, 1.78) | 0.42 |
|
| |||||
| Pregnancy complications | 48 | 1.40 | 1.36 | 1.38 (1.00, 1.90) | 0.05 |
|
| |||||
| Toxemia | 24 | 1.18 | 1.26 | 1.29 (0.78, 2.15) | 0.32 |
|
| |||||
| Gestational diabetes | 18 | 2.21 | 1.93 | 1.88 (1.15, 3.07) | 0.01 |
|
| |||||
| Pregnancy-related HBP | 24 | 1.15 | 1.10 | 0.96 (0.58, 1.59) | 0.87 |
|
| |||||
| OSS | |||||
| 0 | 24 | 1.0 | 1.0 | 1.0 | |
| 1 | 59 | 1.38 | 1.31 | 1.32 (0.82, 2.12) | 0.26 |
| 2 | 63 | 2.07 | 1.85 | 1.85 (1.15, 2.98) | 0.01 |
| 3 | 42 | 2.38 | 1.94 | 1.96 (1.17, 3.28) | 0.01 |
| 4+ | 40 | 3.95 | 2.94 | 2.96 (1.74, 5.03) | <.0001 |
|
| |||||
| Suboptimal AFB | 86 | 1.82 | - | 1.74 (1.30, 2.33) | 0.0002 |
|
| |||||
| Suboptimal parity | 97 | 1.33 | 1.77 | 1.64 (1.24, 2.16) | 0.0004 |
|
| |||||
| Autoimmune disease | 65 | 1.35 | 1.38 | 1.30 (0.97, 1.75) | 0.08 |
|
| |||||
| Asperger syndrome (378 cases) | |||||
|
| |||||
| Miscarriage | 112 | 1.31 | 1.10 | 1.03 (0.79, 1.35) | 0.82 |
|
| |||||
| Abortion | 65 | 1.38 | 1.19 | 1.17 (0.85, 1.62) | 0.33 |
|
| |||||
| Pregnancy complications | 86 | 1.56 | 1.52 | 1.56 (1.22, 1.99) | 0.0004 |
|
| |||||
| Toxemia | 47 | 1.42 | 1.51 | 1.69 (1.17, 2.45) | 0.006 |
|
| |||||
| Gestational diabetes | 30 | 2.27 | 2.01 | 1.95 (1.33, 2.85) | 0.0006 |
|
| |||||
| Pregnancy-related HBP | 40 | 1.16 | 1.12 | 0.83 (0.56, 1.24) | 0.37 |
|
| |||||
| OSS | |||||
| 0 | 35 | 1.0 | 1.0 | 1.0 | |
| 1 | 112 | 1.81 | 1.74 | 1.74 (1.19, 2.54) | 0.005 |
| 2 | 104 | 2.36 | 2.15 | 2.17 (1.47, 3.20) | <.0001 |
| 3 | 70 | 2.75 | 2.32 | 2.32 (1.53, 3.51) | <.0001 |
| 4+ | 57 | 3.90 | 3.03 | 3.01 (1.94, 4.66) | <.0001 |
|
| |||||
| Suboptimal AFB | 129 | 1.56 | - | 1.73 (1.38, 2.17) | <.0001 |
|
| |||||
| Suboptimal parity | 164 | 1.39 | 1.78 | 1.53 (1.24, 1.89) | <.0001 |
|
| |||||
| Autoimmune disease | 116 | 1.49 | 1.52 | 1.46 (1.17, 1.82) | 0.0009 |
|
| |||||
| ‘Other ASD’ (247 cases) | |||||
|
| |||||
| Miscarriage | 76 | 1.35 | 1.08 | 0.89 (0.63, 1.25) | 0.49 |
|
| |||||
| Abortion | 50 | 1.65 | 1.38 | 1.50 (1.02, 2.21) | 0.04 |
|
| |||||
| Pregnancy complications | 61 | 1.71 | 1.65 | 1.71 (1.28, 2.29) | 0.0003 |
|
| |||||
| Toxemia | 26 | 1.18 | 1.28 | 1.17 (0.72, 1.91) | 0.53 |
|
| |||||
| Gestational diabetes | 17 | 1.85 | 1.58 | 1.56 (0.95, 2.58) | 0.08 |
|
| |||||
| Pregnancy-related HBP | 31 | 1.39 | 1.32 | 1.24 (0.79, 1.95) | 0.36 |
|
| |||||
| OSS | |||||
| 0 | 26 | 1.0 | 1.0 | 1.0 | |
| 1 | 65 | 1.40 | 1.29 | 1.27 (0.81, 2.01) | 0.30 |
| 2 | 70 | 2.11 | 1.78 | 1.76 (1.12, 2.79) | 0.02 |
| 3 | 53 | 2.74 | 2.06 | 2.06 (1.27, 3.34) | 0.004 |
| 4+ | 33 | 2.97 | 1.97 | 1.97 (1.15, 3.38) | 0.01 |
|
| |||||
| Suboptimal AFB | 95 | 1.88 | - | 1.68 (1.26, 2.22) | 0.0003 |
|
| |||||
| Suboptimal parity | 96 | 1.13 | 1.56 | 1.35 (1.03, 1.76) | 0.03 |
|
| |||||
| Autoimmune disease | 67 | 1.29 | 1.32 | 1.25 (0.94, 1.67) | 0.12 |
Abbreviations and notes as for Table 2.
All comparison mothers (n=65,652) were used for these analyses; case group n varied by diagnostic subgroup. Case numbers for diagnostic subgroups add up to greater than the total 793 cases due to 53 women reporting in multiple categories (7 women reported in all 3 diagnostic groups). Results were overall similar when mutually-exclusive case groups were utilized. Epilepsy and twin births were the two individual factors not assessed by diagnostic subgroup due to small n.
Discussion
In this large cohort of nurses, we found that women with a history of pregnancy and obstetric complications were more likely to have a child with an autism spectrum disorder than women with a normal obstetric history, consistent with previous findings (Bolton et al., 1997; Brimacombe et al., 2007; Gardener et al., 2009; Juul-Dam et al., 2001). This association was independent of maternal age. The individual pregnancy complication that was most consistently significantly associated across the full and subgroups was a history of gestational diabetes, indicating as much as a doubling of the risk of having a child with ASD.
This study has a number of strengths, including a large sample size, a national cohort of women with a high level of health education and good access to health care, detailed and prospectively collected obstetric history, and information on potential confounding variables. However, a number of limitations should be noted. We relied on maternal report of ASD status. Results from a pilot study among these women including Autism Diagnostic Interview-Revised (ADI-R) validation of maternal report of ASD, however, suggests very high accuracy of reported diagnoses; 10 of 11 individuals deemed affected by maternal report met full criteria according to ADI-R. Further, maternally-reported ASD and developmental disorders have been demonstrated to be reliable in previous reports in other populations (Center for Disease Control and Prevention, 2006; Faraone et al., 1995). To the extent that diagnostic misclassification may have led to the inclusion of some borderline ASD cases, this would tend to weaken rather than strengthen the observed associations. Another related limitation is that we did not have data on the child's date of birth, which could improve information on timing of exposures, and, for multiparous women we did not have the ability to determine which child carried the ASD diagnosis. However, the main results were confirmed in sensitivity analyses restricted to women with only one pregnancy, which are not affected by this limitation. It should also be noted that the magnitude of associations that we found are consistent with those of investigations with more complete data on the affected children (Eaton et al., 2001; Glasson et al., 2004).
All data in this study were self-reported via mailed questionnaires; miscategorization of pregnancy complications, obstetrical history, and other model covariates is therefore possible. However, validation studies conducted within this population of nurses have repeatedly demonstrated reliability of self-reported health information (Colditz et al., 1997), including pregnancy-specific information (Tomeo et al., 1999). As results of the prospective sensitivity analyses that excluded women with births prior to exposure reporting were similar to those of the primary analysis, recall bias is not likely to have occurred, with the possible exception of history of induced abortion in the full study population, as this factor did not remain significantly associated with risk for ASD in the prospective subgroup analysis.
We did not have information on paternal age, which has been reported as a risk factor for ASD in a number of studies (Kolevzon et al., 2007; Lauritsen et al., 2005; Reichenberg et al., 2006; Sasanfar et al, 2010). However, paternal age does not appear to be associated with obstetric complications (Tarín et al., 1998) and it is therefore unlikely that paternal age would affect results after adjustment for maternal age. We did not have information on birth order, though we did account for parity; in addition, our uniparous group cannot be confounded by birth order. The role of genetic factors cannot be ruled out and would need to be addressed in a separate study. While we adjusted for race and income, our study population is composed primarily of well-educated, mid- to high-SES Caucasian women. Thus, it is uncertain to what extent the results of this study can be generalized to different social and ethnic groups.
General pregnancy and obstetric complications have been implicated in a number of other studies but results have been inconsistent for specific factors (Gillberg & Gillberg, 1983; Glasson et al., 2004; Kolevzon et al., 2007; Stein et al., 2006; Wallace et al., 2008). In our study, gestational diabetes was consistently associated with risk of ASD, and, in particular, for autism even in the smaller prospective group. A recent meta-analysis pooling available data from 6 studies with data on gestational diabetes found this to be one of the few significantly associated prenatal factors, with a summary estimate indicating a doubling in risk of autism (Gardener et al., 2009). Our results are consistent with these findings. It is possible that nutritional factors or elevated hormone levels associated with gestational diabetes could interact with a genetic predisposition for ASD. Future studies should assess gestational diabetes individually and with adequate control for potential confounders. Given the robustness of our gestational diabetes finding across the multiple analyses, one possible preventative measure might be to target women at risk for gestational diabetes in order to intervene early. Our results confirm previous findings of no relationship between ASD and pregnancy-related high blood pressure and toxemia (Eaton et al., 2001; Gardener et al., 2009; Glasson et al., 2004; Hultman et al., 2002; Stein et al., 2006; Wallace et al., 2008).
Our OSS was significantly associated with ASD in all analyses for 4 or more factors and, in the primary analyses, for 2 or more factors. Our results demonstrated a significant dose-response relationship between the OSS and ASD, suggesting that a greater number of complications is associated with greater risk for ASD (though the increase in risk appeared to attenuate with very large OSS), a trend which has been found in other studies (Gardener et al., 2009; Gillberg & Gillberg, 1983; Lord et al., 1991). While our score did not include information on birth weight, APGAR scores, and other factors often assessed in optimality or suboptimality scores, many of the previous studies either did not separately assess the effect of the individual factors that make up the score (Bolton et al., 1997; Piven et al., 1993; Zwaigenbaum et al., 2002), provide estimates adjusted for a host of potential confounders (Piven et al., 1993; Stein et al., 2006), or consider a potential non-linear relationship between ASD and OSS (Zwaigenbaum et al., 2002), which could lead to model misspecification and invalid estimates. In assessing individual suboptimality factors, a fairly consistent association between increased maternal age and ASD has been noted; our finding in the primary analysis of an approximate doubling of odds with suboptimal age at first birth (which was primarily age at first birth >30), is consistent with those reports (Bhasin & Schendel, 2006; Croen et al., 2002; Glasson et al., 2004; Maimburg & Vaeth, 2006). In our score, we defined suboptimal age at first birth as <20 or >30 in order to be consistent with the previously utilized suboptimality scores and reports of increased risk associated with both younger (Larsson et al., 2005) and older mothers (Croen et al., 2002; Eaton et al., 2001; Glasson et al., 2004); however, defining this item in the score as only age at first birth >30, which has been more consistently associated with increased risk of autism, did not materially alter results. Further, when excluding ‘suboptimal’ parity from the OSS (as both higher and lower parity and birth order have been inconsistently associated with ASD (Gardener et al., 2009)), results were slightly attenuated but remained significant. Although history of infertility was included in our OSS, these results are not presented here as they have been reported in a separate paper (Lyall et al, submitted manuscript). Maternal autoimmune disease has also been suggested to be associated with autism in prior work, though findings are not consistent (Atladottir et al., 2009; Croen et al., 2005). Our results suggested a moderate but significant association in primary analyses, but no association was seen in the sensitivity analyses of prospective or uniparous subgroups.
Few studies have assessed pregnancy and obstetric complications by diagnostic subgroup; those which have tend to have small sample sizes or focus on one diagnostic subgroup rather than comparing the full spectrum of ASD within one study (Ghaziuddin et al., 1995; Gillberg, 1989). Because our questionnaire asked for autism, Asperger syndrome, and ‘other autism spectrum’ it is possible that the latter category included not only PDD-NOS but also other non-ASD conditions. However, such miscategorization is expected to be relatively low in this study of medically trained nurses, who are less likely to confuse other disorders with autism spectrum disorders. A few previous reports have suggested a relationship between severity of ASD and number of obstetric complications (Glasson et al., 2004; Wallace et al., 2008). While results for our OSS were somewhat weaker in our ‘other ASD’ group, we did not note such a clear relationship between ASD severity and complications, assuming reported diagnosis of autism versus Asperger's or other autism spectrum correlates with severity. In general, our results seem to support the suggestion of Eaton and colleagues of overall similarity rather than differences by diagnostic groups (Eaton et al., 2001).
In our study, the mothers of children with ASD were more likely to have reported pregnancy complications in first pregnancy, or in any pregnancy as assessed over multiple questionnaire years, than were comparison mothers. These results are consistent with a previous observation that both ASD cases and their unaffected siblings had more obstetric complications than unaffected controls (Zwaigenbaum et al., 2002). However, confounding by genetic factors that may predispose to both ASD and obstetric suboptimality, cannot be ruled out. Additional work is needed to tease apart the underlying reasons for associations between pregnancy complications and obstetric suboptimality factors and ASD. Future studies could benefit from taking into account obstetric complications in order to learn more about potential subgroups within the autism spectrum.
In summary, in this large cohort of U.S. nurses, we found that women with a history of pregnancy complications were significantly more likely to have a child diagnosed with ASD than women with uncomplicated pregnancies. Our results by diagnostic subgroup suggest this association holds regardless of the type of autism spectrum diagnosis. Future work assessing gestational diabetes in particular is warranted given its strength and consistency as an individual risk factor in these analyses.
Supplementary Material
Acknowledgments
The work reported in this manuscript was funded in part by CA50385, the main Nurses' Health Study II grant, grants #1788 and #2210 from the Autism Speaks Foundation, and A-14917 from the U.S. Army Medical Research and Material Command (USAMRMC). Role of the funding organizations: funding organizations have reviewed and approved the design and conduct of the overall NHS II, but were not involved in the collection, management, analysis, or interpretation of the data; nor were they involved in the preparation, review, or approval of this manuscript.
References
- Atladóttir HO, Pedersen MG, Thorsen P, Mortensen PB, Deleuran B, Eaton WW, Parner ET. Association of family history of autoimmune diseases and autism spectrum disorders. Pediatrics. 2009;124(2):687–694. doi: 10.1542/peds.2008-2445. [DOI] [PubMed] [Google Scholar]
- Bhasin T, Schendel D. Sociodemographic risk factors for autism in a US metropolitan area. J Autism Dev Disord. 2006;37(4):667–77. doi: 10.1007/s10803-006-0194-y. [DOI] [PubMed] [Google Scholar]
- Bolton PF, Murphy M, MacDonald H, Whitlock B, Pickles A, Rutter M. Obstetric complications in autism: Consequences or causes of the condition? J Am Acad Child Adolesc Psychiatry. 1997;36(2):272–281. doi: 10.1097/00004583-199702000-00018. [DOI] [PubMed] [Google Scholar]
- Brimacombe M, Ming X, Lamendola M. Prenatal and birth complications in autism. Matern Child Health J. 2007;11:73–79. doi: 10.1007/s10995-006-0142-7. [DOI] [PubMed] [Google Scholar]
- Burd L, Severud R, Kerbeshian J, Klug MG. Prenatal and perinatal risk factors for autism. J Perinat Med. 1999;27:441–450. doi: 10.1515/JPM.1999.059. [DOI] [PubMed] [Google Scholar]
- Center for Disease Control and Prevention. Mental health in the United States: parental report of diagnosed autism in children aged 4-17 years--United States, 2003-2004. MMWR Morb Mortal Wkly Rep. 2006;55(17):481–486. [PubMed] [Google Scholar]
- Colditz GA, Manson JE, Hankinson SE. The Nurses' Health Study: 20-year contribution to the understanding of health among women. J Womens Health. 1997;6(1):49–62. doi: 10.1089/jwh.1997.6.49. [DOI] [PubMed] [Google Scholar]
- Croen L, Grether JK, Selvin S. Descriptive epidemiology of autism in a California population: Who is at risk? J Autism Dev Disord. 2002;32(3):217–224. doi: 10.1023/a:1015405914950. [DOI] [PubMed] [Google Scholar]
- Croen LA, Grether JK, Yoshida CK, Odouli R, Van de Water J. Maternal autoimmune diseases, asthma and allergies, and childhood autism spectrum disorders: a case-control study. Arch Pediatr Adolesc Med. 2005;159(2):151–157. doi: 10.1001/archpedi.159.2.151. [DOI] [PubMed] [Google Scholar]
- Durrleman S, Simon R. Flexible regression models with cubic splines. Statistics in Medicine. 1989;8:551–561. doi: 10.1002/sim.4780080504. [DOI] [PubMed] [Google Scholar]
- Eaton WW, Mortensen PB, Thomsen PH, Frydenberg M. Obstetric complications and risk for severe psychopathology in childhood. J Autism Dev Disord. 2001;31(3):279–285. doi: 10.1023/a:1010743203048. [DOI] [PubMed] [Google Scholar]
- Faraone SV, Biederman J, Milberger S. How reliable are maternal reports of their children's psychopathology? One-year recall of psychiatric diagnoses of ADHD children. J Am Acad Child Adolesc Psychiatry. 1995;34(8):1001–1008. doi: 10.1097/00004583-199508000-00009. [DOI] [PubMed] [Google Scholar]
- Gardener H, Munger K, Chitnis T, Michels K, Spiegelman D, Ascherio A. Prenatal and Perinatal Factors and Risk of Multiple Sclerosis. Epidemiology. 2009;20(4):611–618. doi: 10.1097/EDE.0b013e31819ed4b9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gardener H, Spiegelman D, Buka S. Prenatal risk factors for autism: comprehensive meta-analysis. Brit J Psychiatry. 2009;195:7–14. doi: 10.1192/bjp.bp.108.051672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghaziuddin M, Shakal J, Tsai L. Obstetric factors in Asperger syndrome: comparison with high-functioning autism. J Intellect Disabil Res. 1995;39:538–543. doi: 10.1111/j.1365-2788.1995.tb00575.x. [DOI] [PubMed] [Google Scholar]
- Gillberg C. Asperger syndrome in 23 Swedish children. Dev Med Child Neurol. 1989;31:520–531. doi: 10.1111/j.1469-8749.1989.tb04031.x. [DOI] [PubMed] [Google Scholar]
- Gillberg C, Cederlund M. Asperger syndrome: Familial and pre- and perinatal factors. J Autism Dev Disord. 2005;35(2):159–166. doi: 10.1007/s10803-004-1993-7. [DOI] [PubMed] [Google Scholar]
- Gillberg C, Gillberg C. Infantile autism: A total population study of reduced optimality in the Pre-, Peri-, and Neonatal period. J Autism Dev Disord. 1983;13(2):153–166. doi: 10.1007/BF01531816. [DOI] [PubMed] [Google Scholar]
- Glasson EJ, Bower C, Petterson B, de Klerk N, Chaney G, Hallmayer JF. Perinatal factors and the development of autism: a population study. Arch Gen Psychiatry. 2004;61(6):618–627. doi: 10.1001/archpsyc.61.6.618. [DOI] [PubMed] [Google Scholar]
- Govindarajulu U, Spiegelman D, Thurston SW, Ganguli B, Eisen EA. Comparing smoothing techniques in Cox models for exposure-response relationships. Stat Med. 2007;26(20):3735–3752. doi: 10.1002/sim.2848. [DOI] [PubMed] [Google Scholar]
- Hultman CM, Sparen P, Cnattingius S. Perinatal risk factors for infantile autism. Epidemiology. 2002;13(4):417–423. doi: 10.1097/00001648-200207000-00009. [DOI] [PubMed] [Google Scholar]
- Hunt KJ, S K. The increasing prevalence of diabetes in pregnancy. Obstet Gynecol Clin North Am. 2007;34(2):173–199. doi: 10.1016/j.ogc.2007.03.00. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Juul-Dam N, Townsend J, Courchesne E. Prenatal, perinatal, and neonatal factors in autism, pervasive developmental disorder-not otherwise specified, and the general population. Pediatrics. 2001;107(4):E63. doi: 10.1542/peds.107.4.e63. [DOI] [PubMed] [Google Scholar]
- Kolevzon A, Gross R, Reichenberg A. Prenatal and perinatal risk factors for autism: a review and integration of findings. Arch Pediatr Adolesc Med. 2007;161(4):326–333. doi: 10.1001/archpedi.161.4.326. [DOI] [PubMed] [Google Scholar]
- Larsson HJ, Eaton WW, Madsen KM, Vestergaard M, Olesen AV, Agerbo E, Schendel D, Thorsen P, Mortensen PB. Risk factors for autism: perinatal factors, parental psychiatric history, and socioeconomic status. Am J Epidemiol. 2005;161(10):916–925. doi: 10.1093/aje/kwi123. discussion 926-918. [DOI] [PubMed] [Google Scholar]
- Lauritsen MB, Pedersen CB, Mortensen PB. Effects of familial risk factors and place of birth on the risk of autism: a nationwide register-based study. J Child Psychol Psychiatry. 2005;46(9):963–971. doi: 10.1111/j.1469-7610.2004.00391.x. [DOI] [PubMed] [Google Scholar]
- Lord C, Mulloy C, Wendelboe M, Schopler E. Pre- and perinatal factors in high-functioning females and males with autism. J Autism Dev Disord. 1991;21(2):197–209. doi: 10.1007/BF02284760. [DOI] [PubMed] [Google Scholar]
- Maimburg RD, Vaeth M. Perinatal risk factors and infantile autism. Acta Psychiatr Scand. 2006;114:257–264. doi: 10.1111/j.1600-0447.2006.00805.x. [DOI] [PubMed] [Google Scholar]
- Mental health in the United States: parental report of diagnosed autism in children aged 4-17 years-United States, 2003-2004. MMWR Morb Mortal Wkly Rep. 2006 2008. [PubMed] [Google Scholar]
- Miettinen OS. Theoretical epidemiology: principles of cccurrence research in medicine. New York: Wiley & Sons; 1985. [Google Scholar]
- Piven J, Simon J, Chase GA, Wzorek M, Landa R, Gayle J, Folstein S. The etiology of autism: pre-, peri-, and neonatal factors. J Am Acad Child Adolesc Psychiatry. 1993;32(6):1256–1263. doi: 10.1097/00004583-199311000-00021. [DOI] [PubMed] [Google Scholar]
- Reichenberg A, Gross R, Weiser M, Bresnahan M, Silverman J, Harlap S, Rabinowitz J, Shulman C, Malaspina D, Lubin G, Knobler H, Davidson M, Susser E. Advancing paternal age and autism. Arch Gen Psychiatry. 2006;63:1026–1032. doi: 10.1001/archpsyc.63.9.1026. [DOI] [PubMed] [Google Scholar]
- Sasanfar R, Haddad S, Tolouei A, Ghadami M, Santangelo SL. Paternal age increases the risk for autism in an Iranian population sample. Mol Autism. 2010;1(1):2–11. doi: 10.1186/2040-2392-1-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stein D, Weizman A, Ring A, Barak Y. Obstetric complications in individuals diagnosed with autism and in healthy controls. Compr Psychiatry. 2006;47(1):69–75. doi: 10.1016/j.comppsych.2005.01.001. [DOI] [PubMed] [Google Scholar]
- Tarín J, Brines J, Cano A. Long-term effects of delayed parenthood. Hum Reprod. 1998;13(9):2371–2376. doi: 10.1093/humrep/13.9.2371. [DOI] [PubMed] [Google Scholar]
- Tomeo CA, Rich-Edwards JW, Michels KB, Berkey CS, Hunter DJ, Frazier AL, Willett WC, Buka SL. Reproducibility and validity of maternal recall of pregnancy-related events. Epidemiology. 1999;10(6):774–777. [PubMed] [Google Scholar]
- Wallace A, Anderson G, Dubrow R. Obstetric and parental psychiatric variables as potential predictors of autism severity. J Autism Dev Disord. 2008;38(8):1542–1554. doi: 10.1007/s10803-007-0536-4. [DOI] [PubMed] [Google Scholar]
- Weinberg C, Wilcox A. Reproductive Epidemiology. In: Rothman K, Greenland S, editors. Modern Epidemiology. 2nd. Philadelphia: Lippincott Williams & Wilkins; 1998. pp. 585–608. [Google Scholar]
- Zwaigenbaum L, Szatmari P, Jones MB, Bryson SE, MacLean JE, Mahoney WJ, Bartolucci G, Tuff L. Pregnancy and birth complications in autism and liability to the broader autism phenotype. J Am Acad Child Adolesc Psychiatry. 2002;41(5):572–579. doi: 10.1097/00004583-200205000-00015. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.