Abstract
Background
Patients with type 2 diabetes have an increased risk of cardiovascular disease. Few studies have evaluated the cardiovascular disease (CVD) risk simultaneously using the United Kingdom Prospective Diabetes Study (UKPDS) risk engine and non-invasive vascular tests in patients with newly diagnosed type 2 diabetes.
Methods
Participants (n=380; aged 20 to 81 years) with newly diagnosed type 2 diabetes were free of clinical evidence of CVD. The 10-year coronary heart disease (CHD) and stroke risks were calculated for each patient using the UKPDS risk engine. Carotid intima media thickness (CIMT), flow mediated dilation (FMD), pulse wave velocity (PWV) and augmentation index (AI) were measured. The correlations between the UKPDS risk engine and the non-invasive vascular tests were assessed using partial correlation analysis, after adjusting for age, and multiple regression analysis.
Results
The mean 10-year CHD and 10-year stroke risks were 14.92±11.53% and 4.03±3.95%, respectively. The 10-year CHD risk correlated with CIMT (P<0.001), FMD (P=0.017), and PWV (P=0.35) after adjusting for age. The 10-year stroke risk correlated only with the mean CIMT (P<0.001) after adjusting for age. FMD correlated with age (P<0.01) and systolic blood pressure (P=0.09). CIMT correlated with age (P<0.01), HbA1c (P=0.05), and gender (P<0.01).
Conclusion
The CVD risk is increased at the onset of type 2 diabetes. CIMT, FMD, and PWV along with the UKPDS risk engine should be considered to evaluate cardiovascular disease risk in patients with newly diagnosed type 2 diabetes.
Keywords: Atherosclerosis; Cardiovascular risk; Diabetes mellitus, type 2; United Kingdom Prospective Diabetes Study risk engine; Vascular function
INTRODUCTION
The prevalence of cardiovascular disease is 2 to 5 times higher in diabetics compared to non-diabetics [1-3]. Diabetes with no history of myocardial infarction are equally at risk for myocardial infarction as are non-diabetic subjects with a history of myocardial infarction, and they are both classified in the same coronary artery disease risk group [4].
Therefore, in order to prevent cardiovascular disease in type 2 diabetes patients, identifying and correcting risk factors are considered important. In the past the studies have been mainly focused to find cardiovascular disease in type 2 diabetes. Currently, identifying patients who are at high risk for cardiovascular disease prior to development of apparent cardiovascular disease and preventing the disease have taken a higher priority. However, identifying patients who are at high risk for cardiovascular disease and require active prevention is not easy. A few cardiovascular risk models and vascular tests have been suggested to evaluate the risk of cardiovascular disease.
Among cardiovascular disease risk models, through the United Kingdom Prospective Diabetes Study (UKPDS), the UKPDS risk engine was developed for estimating risk of coronary heart disease and stroke for 10 years in type 2 diabetes [5,6]. UKPDS enrolled 5,100 patients who were newly diagnosed with type 2 diabetes and was originally aimed to know whether glycemic control could decrease morbidity and mortality that were associated with diabetic complications [7]. Through the UKPDS, the relationships between cardiovascular diseases and identified general risk factors in type 2 diabetes was investigated and the UKPDS risk engine was developed.
In vascular examinations to predict cardiovascular disease, flow-mediated dilation (FMD) assesses early endothelial cell dysfunction, and pulse wave velocity (PWV) and augmentation index (AI) assess arterial stiffness. Carotid intima-media thickness (CIMT) is used for early detection of atherosclerosis [8]. Vascular examinations evaluate the risk of cardiovascular disease because vascular dysfunction and changes in the vascular structure occur before the onset of cardiovascular disease. These changes are not isolated but represent changes that occur in blood vessels throughout the entire body [9].
There are no studies on the correlations between the UKPDS risk engine and blood vessel examinations in Korean type 2 diabetes patients. In the present study, in order to assess the risk of cardiovascular disease in patients who were just diagnosed with type 2 diabetes, we used the UKPDS risk engine among cardiovascular disease risk models and easy and non-invasive vascular examinations. The relationship between vascular tests and the UKPDS risk engine as well as the relationship between vascular tests and cardiovascular risk factors were investigated.
METHODS
Study subjects and anthropometric measurement
This study was performed between January 2003 and August 2007 at the Eulji General Hospital Diabetes Center, and employed the use of 75 g oral glucose tolerance test to diagnose diabetes. The diagnostic criteria set by the American Diabetes Association were used to diagnose type 2 diabetes patients that participated in this study [10]. Individuals who had previously used oral hypoglycemic agents for over 1 month, had previously received insulin treatment, or had a history of cardiovascular disease were excluded from this study.
Study participants answered a questionnaire regarding their disease history and were measured for height and weight. Participants then had their blood pressures measured using a mercury blood pressure monitor (Yamasu, Tokyo, Japan) after sitting for 10 minutes to stabilize their blood pressure. Waist circumference measurements were performed by the same examiner at the middle point between the bottom of the ribcage and the iliac crest. Body mass index (BMI) was calculated by dividing body weight by height squared (kg/m2).
Calculated risk of the UKPDS risk engine
Cardiovascular risk was scored for each participant using the UKPDS risk engine (version 2.0) by inputting risk factors such as age, gender, race, current smoking status, glycated hemoglobin, systolic blood pressure, total cholesterol, high density lipoprotein cholesterol (HDL-C), and presence or absence of atrial fibrillation, and the 10-year risk of coronary heart disease (CHD) and cerebrovascular disease were calculated [3,5,6].
Biochemical tests
Blood samples were taken after 8 hours of fasting, and blood glucose, cholesterol, triglycerides, and low density lipoprotein cholesterol (LDL-C) were measured on a Hitachi 7170 (Hitachi, Tokyo, Japan) using the enzymatic method. In addition, HDL-C and VLDL were measured after their precipitation.
Vascular examinations
Measuring CIMT
CIMT was measured using a high resolution B-mode ultrasound (LOGIC 400 CL PRO; General Electric, Milwaukee, WI, USA) by the same well-trained examiner. Ultrasonography of the common carotid artery, carotid bifurcation, and internal carotid artery of the left and right carotid arteries was performed. The distance between the leading edge of the first bright line of the far wall (lumen-intima interface) and the leading edge of the second bright line (media-adventitia interface) indicates the intima-media thickness. The distance between the first and second bright lines was measured in the separations of the low bright regions of the CIMT ultrasound. The CIMT values used for this study were the means of 6 measurements from different locations [11,12].
FMD
In order to verify the functionality of vascular endothelial cells, using a high resolution B-mode 11 MHz ultrasound, the FMD in the brachial artery was measured using the method of Raitakari and Celermajer [13]. Patients fasted and abstained from drugs, alcohol, caffeine, and tobacco for over 8 hours prior to testing.
Participants were stabilized in supine position, and the baseline diameter of the brachial artery was measured using a high-resolution B-mode ultrasonograph. A linear transducer was located 5 to 10 cm above the antecubital fossa, and the center of the brachial artery was set as a reference point. Under B-mode, the diameter was defined from one side of the interface (m-line) of the intima and media of the blood vessel to the same part on the opposite side. In order to reduce variation during diameter measurements, it was measured at arterial branching points and at the end of the diastolic phase just before the origin of an R wave using electrocardiogram (ECG). After baseline diameter measurements were completed, the transducer was removed, and a blood pressure cuff was attached to the middle of the upper arm and set at 250 mm Hg. After 5 minutes, we reduced the pressure on the cuff to 0 mm Hg and then removed the cuff. We then repositioned the transducer, and within the 50 seconds during which hyperemia sets in, we measured the maximum diameter of the brachial artery increased due to hyperemia. For baseline diameter, the measurements were calculated as percentages, and a flow mediated response was observed.
Brachial-ankle pulse wave velocity (BaPWV)
The measurements were taken in the morning after 8 hours of fasting and 10 minutes of resting using a VP1000 waveform analyzer (Omron, Tokyo, Japan). This instrument measured a pulse wave from both upper extremities and ankles, and the time difference was measured using ECG signals. The distance was obtained using a formula based on height and was used to calculate pulse wave velocity [14]. The blood pressure in limbs was measured using the vibratory method, and the left, right, and mean BaPWV were measured simultaneously. The mean BaPWV was used for statistical analysis.
AI
The peripheral arterial waveform was recorded from the radial artery using the Applanation tonometer aortic waveform analysis system (SphygmoCoR®; AtCor Medical Pty Ltd., Sydney, Australia), and then, using a generalized transfer function, the AI was obtained by calculating aortic waveforms [15]. In order to eliminate the effects of patient medication, measurements were taken after 8 hours of fasting. In addition, in order to eliminate the effects of heart rate during measurements, AI75 (augmentation index adjusted for 75 beats/min) was obtained and was used for statistical analysis.
Statistical analysis
Statistical analysis was performed on SPSS version 17.0 (SPSS Inc., Chicago, IL, USA) software. Data are expressed as the mean and standard deviation. The log values of variables that did not follow a normal distribution were calculated. In order to find a correlation between the vascular tests and the UKPDS risk engine which determined the 10-year CHD and 10-year stroke risk , after adjusting for age, a partial correlation analysis and multiple regression were tested. An ANCOVA was tested for correlation between the categorical variables of risk factors and vascular examinations, and a multiple regression analysis was used for continuous variables. P values less than 0.05 were considered to be statistically significant.
RESULTS
Clinical characteristics of the subjects
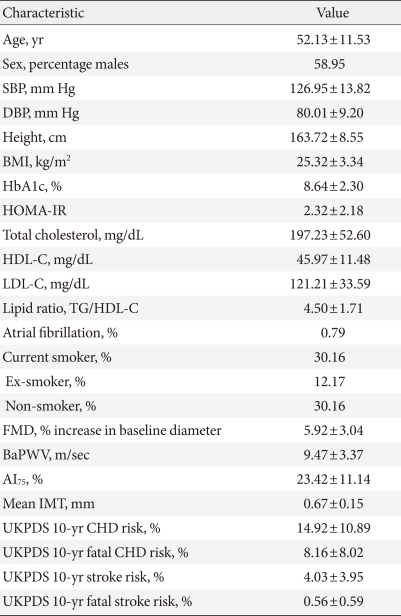
Among the 380 participants, the mean age was 52 years (range, 20 to 81 years). Three patients had atrial fibrillation and 114 were smokers (Table 1). Fifty percent of the patients were obese (BMI ≥25 kg/m2), 77% had high blood pressure (BP ≥130/80 mm Hg), and 34% were taking antihypertensive medication. Ninety-four percent of patients had hypercholesterolemia, which was classified as total cholesterol ≥200 mg/dL, triglyceride ≥150 mg/dL, LDL-C ≥100 mg/dL, or HDL-C (HDL-C; male, <40 mg/dL; female, <50 mg/dL).
Table 1.
Baseline characteristics of the study population
Values are presented as mean±standard deviation.
SBP, systolic blood pressure; DBP, diastolic blood pressure; BMI, body mass index; HOMA-IR, homeostasis model assessment of insulin resistance; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; TG, triglyceride; FMD, flow-mediated dilation; PWV, pulse wave velocity; AI, augmentation index; IMT, intima-media thickness; UKPDS, United Kingdom Prospective Diabetes Study; CHD, coronary heart disease.
The relevance of vascular tests and risk factors of cardiovascular disease
Among cardiovascular disease risk factors (age, gender, systolic blood pressure, lipid abnormalities, glycated hemoglobin, smoking, and atrial fibrillation), CIMT increased with age (P<0.001) and glycated hemoglobin (P=0.048), decreased with HDL-C (P=0.450), and was significantly different based on gender (P<0.001). FMD decreased significantly with increasing age (P<0.001) and systolic blood pressure (P=0.017). BaPWV increased with age (P<0.001) and total cholesterol (P<0.001). AI75 increased with HDL-C (P=0.034), and was different based on gender (P<0.001). No vascular tests were related to smoking or atrial fibrillation. The intraobserver coefficients of variation of FMD and AI were 47% and 51%, respectively. Since there was only one examiner in this study, the interobserver coefficient of variation could not be calculated.
Correlation between the UKPDS engine and vascular tests
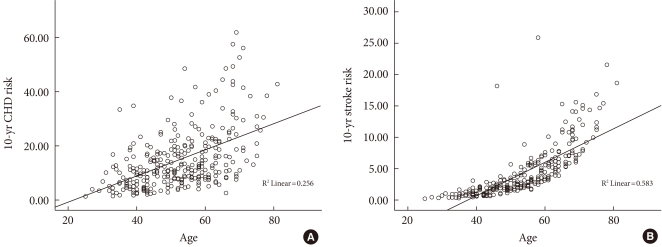
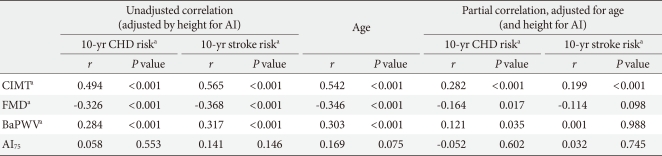
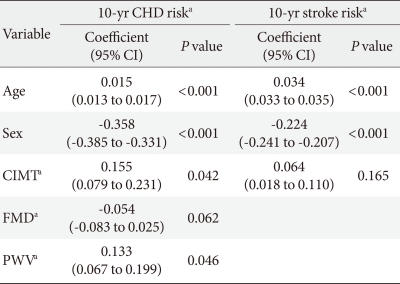
In the entire participants in the study, the 10-year CHD risk and 10-year stroke risk calculated using the UKPDS risk engine were 14.92% and 4.03%, respectively. The 10-year stroke risk was lower than 10-year CHD risk because there were only 3 patients with atrial fibrillation in this study. The mean values of study population were 50 years old, smoker, a systolic blood pressure of 130 mm Hg, glycated hemoglobin of 8.64%, total cholesterol of 200 mg/dL, and HDL-C of 45 mg/dL. With these mean values, the 10-year stroke risk calculated using the UKPDS risk engine, in a male patient with and without a history of atrial fibrillation was 16.7% and 2.1%, respectively, which shows that atrial fibrillation has a large effect on the 10-year stroke risk. Because of few atrial fibrillation patients, there was a difference between 10-year CHD risk and 10-year stroke risk in the present study. Prior to adjusting for age, the 10-year CHD risk and 10-year stroke risk were significantly correlated with FMD, BaPWV, and CIMT. However, there was no correlation with AI75. The 10-year CHD and 10-year stroke risks both increased with age (Fig. 1). In addition, age was significantly correlated with the all four vascular tests. After adjusting for age, there was a significant correlation between the 10-year CHD risk and CIMT (P<0.001), FMD (P<0.017), and BaPWV (P<0.035) and a significant correlation between the 10-year stroke risk and CIMT (P<0.001) (Table 2). CIMT, FMD, BaPWV, age, and gender values were treated as independent variables, and 10-year CHD risk was treated as a dependent variable. Therefore, a multiple regression analysis confirmed age, gender, CIMT and BaPWV independently affect 10-year CHD. The 10-year stroke risk was treated as a dependent variable, age, gender, and CIMT were treated as independent variables. Therefore, a multiple regression analysis confirmed age and gender affecting 10-year stroke risk (Table 3).
Fig. 1.
Plot of 10-year coronary heart disease (CHD) and 10-year stroke risks according to age. (A) 10-year CHD risk. (B) 10-year stroke risk.
Table 2.
Correlation of vascular function with the UKPDS risk engine and age in newly diagnosed type 2 diabetics
UKPDS, United Kingdom Prospective Diabetes Study; AI, augmentation index; CHD, coronary heart disease; CIMT, carotid intima-media thickness; FMD, flow-mediated dilation; PWV, pulse wave velocity.
aUsing log transformed data.
Table 3.
Multiple regression analysis with 10-year CHD risk or 10-year stroke risk as the dependent variable
Dependent variable: 10-year CHD risk, R2=0.648, P<0.001 or 10-year stroke risk, R2=0.878, P<0.001.
CHD, coronary heart disease; CI, confidence interval; CIMT, carotid intima-media thickness; FMD, flow-mediated dilation; PWV, pulse wave velocity.
aUsing log transformed data.
DISCUSSION
Risk of cardiovascular disease calculated by the UKPDS risk engine
The Framingham risk score and SCORE [16] are risk models used to assess the risk of cardiovascular disease in the general population. The UKPDS risk engine is a risk model used only for diabetes patients. van der Heijden et al. [17] found that the UKPDS risk function was more accurate in predicting coronary artery disease in a Hoorn study cohort of newly diagnosed type 2 diabetes patients than were the Framingham and SCORE methods. The UKPDS studies only included newly diagnosed type 2 diabetes patients, and the UKPDS risk engine used values of patients diagnosed within 1 to 2 years [17]. In the present study, performed on patients who were recently diagnosed with diabetes, the 10-year CHD risk and 10-year stroke risk were 14.92% and 4.03%, respectively, calculated using the UKPDS risk engine. The 10-year CHD risk was relatively high, and the risk of coronary heart disease tended to increase after onset of type 2 diabetes.
CIMT
CIMT increases from the onset of atherosclerosis. The Atherosclerosis Risk in Communities (ARIC) study examined CIMT in 5,552 males and 7,289 females, ages 45 to 64, followed for an average of 5.2 years between 1987 and 1993. Coronary heart disease was 1.85 times more prevalent in males and 5.07 times more prevalent in females who had a CIMT greater than 1 mm compared to those with a CIMT less than 1 mm [18]. In another ARIC study that examined the relevance of CIMT and cerebrovascular disease over an average of 7.2 years, ischemic cerebrovascular disease was 1.78 times more prevalent in males and 2.02 times more prevalent in females with a CIMT greater than 1 mm compared to that of those with a CIMT less than 1 mm [11]. One meta-analysis showed that type 2 diabetes patients had an average CIMT that was 0.13 mm greater than normal individuals, which was associated with a 40% greater probability of type 2 diabetics developing cardiovascular disease compared to healthy individuals [19].
In a Korean study, Bae et al. [20] showed that the mean CIMT of healthy individuals with no risk factors for cardiovascular disease was 0.63±0.11 mm, and the mean CIMT of the diabetic study subjects was 0.75±0.17 mm. The mean CIMT of our patients (0.67±0.15 mm) was higher than that reported by Bae et al. Thus, even recently diagnosed type 2 diabetes patients have higher CIMT values than healthy individuals.
In this study, the 10-year CHD risk and the 10-year stroke risk calculated using the UKPDS risk engine and CIMT were positively correlated (P<0.001, respectively). Further, the CIMT and UKPDS risk engine were both effective methods to predict cardiovascular disease in recently diagnosed type 2 diabetes patients. In multiple regression analyses, however, CIMT was not an independent influencing factor of 10-year stroke risk.
A study performed by Temelkova-Kurktschiev et al. [21] on newly diagnosed type 2 diabetes patients found no cardiovascular risk factors that were associated with CIMT, despite few enrolled patients. Matsumoto et al. [22] showed that age, systolic blood pressure, glycated hemoglobin, and HDL-C were all associated with increases in CIMT in 438 type 2 diabetes patients. A study of Korean type 2 diabetes patients found that gender, smoking, systolic blood pressure, LDL, and glycated hemoglobin were significantly correlated with CIMT [12]. The differences in these results likely result from differences in the study populations. In our study, among the variables that compose the risk score from the UKPDS risk engine, age (P<0.001), gender (P<0.001), and glycated hemoglobin (P=0.048) were all independent risk factors for CIMT, and a decrease in CIMT was associated with an increase in HDL-C (P=0.045). Therefore, in recently diagnosed Korean type 2 diabetes patients, age, gender, glycated hemoglobin, and HDL-C are closely related.
FMD
Endothelial cell dysfunction is associated with multiple cardiovascular risk factors [23] and occurs early in atherosclerosis. Henry et al. [24] found a meaningful decrease in FMD in type 2 diabetes patients compared to healthy controls and an impaired glucose tolerance (IGT) group. Su et al. [25] assessed endothelial cell dysfunction in FMD in 46 control patients, 30 impaired fasting glucose (IFG) patients, 38 IGT patients, and 44 type 2 diabetes patients. The IGT group had a significantly lower FMD compared to the control group. Ravikumar et al. [26] reported an FMD of 6.64±4.38% in 50 control patients and 1.72±2.82% in 50 diabetic patients. A study of Korean patients by Shin et al. [27] showed that the FMD of type 2 diabetes patients who had no history of cardiovascular complications was 7.6±0.2%, and the FMD of non-diabetics of similar age and gender was 8.9±0.4%. Our mean FMD was 5.92±3.04%, which is lower than the mean FMD value from another Korean study of type 2 diabetes patients. Thus, the endothelial cell function evaluated by FMD decreases in patients recently diagnosed with type 2 diabetes.
Until now, there have been very few studies on the relationship between the calculated 10-year CHD risk using the UKPDS risk engine and FMD. In this study, only 10-year CHD risk was negative correlated with FMD (P=0.022). This is the first study that FMD and 10-year CHD risk has a significant correlation in newly diagnosed Korean type 2 diabetes patients. This result suggests that FMD can predict coronary heart disease in newly diagnosed type 2 diabetes patients.
According to Celermajer et al. [28], endothelial cell function measured using FMD and cardiovascular risk factors such as smoking, hyperlipidemia, hypertension, gender, and age are all related. In a Korean study of 57 diabetes patients at an average of 10 years after diagnosis, there was no correlation between FMD and cardiovascular risk factors such as age, glycated hemoglobin, or hyperlipidemia [27]. In our study, of cardiovascular risk factors included in the UKPDS risk engine, age (P<0.001) and systolic blood pressure (P=0.017) were significantly correlated with FMD (Table 2). Smoking, hyperlipidemia, gender, and glycated hemoglobin had no association with FMD. These results confirmed that vascular function, as assessed by FMD, is closely related with age and systolic blood pressure in newly diagnosed type 2 diabetics.
PWV
Arterial stiffness measured by PWV increases as the risk of developing cardiovascular disease increases [29], and central arterial stiffness is more closely related to cardiovascular disease than is peripheral arterial stiffness [8]. Thus, carotid-femoral PWV, which reflects central arterial stiffness, is frequently used in large-scale prospective studies. In Japan, the BaPWV, which is easy to measure and is far from the inguinal region, is commonly used [30]. However, BaPWV only reflects about 60% of the central arterial stiffness and about 20% of the peripheral arteries [31].
Although Blacher et al. [32] used a different PWV method, they reported a close association between cardiovascular disease and a carotid-femoral PWV greater than 13 m/sec. Our study reported a lower mean BaPWV of 9.47±3.37 m/sec in patients with a mean age of 52 years. However, in a study of healthy Koreans with an age of 50 to 60 years, the mean BaPWV was 7.84±2.37 m/sec and it was lower than our result, supporting that arterial stiffness increases in the initial stages of type 2 diabetes [33]. Additionally, our PWV results are consistent with results reported by Cruickshank et al. [34] that PWV tends to increase more in type 2 diabetics than in a normal control group. Additionally, BaPWV correlated with the 10-year CHD risk (P=0.035) calculated by the UKPDS risk engine. A correlation between only 10 year risk calculated by the UKPDS risk engine and BaPWV are likely because BaPWV does not reflect the stiffness of the central artery as well as carotid-femoral PWV. Therefore, further prospective studies are required to correlate cardiovascular disease and arterial stiffness.
AI
The interaction between the incident pulse wave (from the heart to the periphery) and the reflected pulse wave (from the periphery to the central region) is assessed by pulse wave analysis and expressed as the AI. Systemic arterial stiffness and changes in central arterial pressure are included as well, thus AI reflects both micro- and macrovascular function [8].
In a study of 2,232 individuals from the Framingham heart study cohort who participated in the study for an average of 7.8 years, Mitchell et al. [29] reported that there was no association between AI and cardiovascular disease. This was consistent with a study of hypertensive elderly women that was unable to predict cardiovascular disease using AI [35]. In a patient group that included 20% diabetics, however, an increase in AI reflected an increase in the prevalence of cardiovascular disease [15]. In a Hoorn study, AI increased in type 2 diabetics, but there was no difference between the IGT and normal control groups [36]. We observed no correlation between the UKPDS risk engine and AI. Since there are conflicting results on the association between AI and cardiovascular disease, additional research is required on Korean type 2 diabetics before using AI clinically in these patients.
This study has several limitations. First, it was a cross-sectional study so there was no way to confirm causal relationships. Second, there was no control group for comparing vascular tests. Although we cannot confirm the extent of vascular dysfunction or atherosclerosis by comparing to other studies, to evaluate our results, we compared our results to those of a domestic study that used the same examination methods on patients from the same age group. Third, the intraobserver coefficient of variation for AI and PVW were high, and the reliability of these tests was low.
In conclusion, the increased risk of cardiovascular disease at the onset of type 2 diabetes observed in this study is consistent with previous studies. In our study of the relationships between the UKPDS risk engine and vascular tests, the 10-year CHD risk was correlated with CIMT, FMD, and PWV, and the 10-year stroke risk was correlated with CIMT. Therefore, in order to accurately predict the risk of cardiovascular disease in Korean type 2 diabetics, future studies should focus on an additional prospective studies using the UKPDS risk engine with CIMT, FMD, and PWV.
Footnotes
No potential conflict of interest relevant to this article was reported.
References
- 1.Barrett-Connor EL, Cohn BA, Wingard DL, Edelstein SL. Why is diabetes mellitus a stronger risk factor for fatal ischemic heart disease in women than in men? The Rancho Bernardo Study. JAMA. 1991;265:627–631. [PubMed] [Google Scholar]
- 2.Koskinen P, Manttari M, Manninen V, Huttunen JK, Heinonen OP, Frick MH. Coronary heart disease incidence in NIDDM patients in the Helsinki Heart Study. Diabetes Care. 1992;15:820–825. doi: 10.2337/diacare.15.7.820. [DOI] [PubMed] [Google Scholar]
- 3.Stevens RJ, Coleman RL, Adler AI, Stratton IM, Matthews DR, Holman RR. Risk factors for myocardial infarction case fatality and stroke case fatality in type 2 diabetes: UKPDS 66. Diabetes Care. 2004;27:201–207. doi: 10.2337/diacare.27.1.201. [DOI] [PubMed] [Google Scholar]
- 4.Haffner SM, Lehto S, Ronnemaa T, Pyorala K, Laakso M. Mortality from coronary heart disease in subjects with type 2 diabetes and in nondiabetic subjects with and without prior myocardial infarction. N Engl J Med. 1998;339:229–234. doi: 10.1056/NEJM199807233390404. [DOI] [PubMed] [Google Scholar]
- 5.Stevens RJ, Kothari V, Adler AI, Stratton IM United Kingdom Prospective Diabetes Study (UKPDS) Group. The UKPDS risk engine: a model for the risk of coronary heart disease in type II diabetes (UKPDS 56) Clin Sci (Lond) 2001;101:671–679. [PubMed] [Google Scholar]
- 6.Kothari V, Stevens RJ, Adler AI, Stratton IM, Manley SE, Neil HA, Holman RR. UKPDS 60: risk of stroke in type 2 diabetes estimated by the UK Prospective Diabetes Study risk engine. Stroke. 2002;33:1776–1781. doi: 10.1161/01.str.0000020091.07144.c7. [DOI] [PubMed] [Google Scholar]
- 7.UK Prospective Diabetes Study (UKPDS). VIII. Study design, progress and performance. Diabetologia. 1991;34:877–890. [PubMed] [Google Scholar]
- 8.Tomiyama H, Yamashina A. Non-invasive vascular function tests: their pathophysiological background and clinical application. Circ J. 2010;74:24–33. doi: 10.1253/circj.cj-09-0534. [DOI] [PubMed] [Google Scholar]
- 9.Kullo IJ, Malik AR. Arterial ultrasonography and tonometry as adjuncts to cardiovascular risk stratification. J Am Coll Cardiol. 2007;49:1413–1426. doi: 10.1016/j.jacc.2006.11.039. [DOI] [PubMed] [Google Scholar]
- 10.American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2006;29(Suppl 1):S43–S48. [PubMed] [Google Scholar]
- 11.Chambless LE, Folsom AR, Clegg LX, Sharrett AR, Shahar E, Nieto FJ, Rosamond WD, Evans G. Carotid wall thickness is predictive of incident clinical stroke: the Atherosclerosis Risk in Communities (ARIC) study. Am J Epidemiol. 2000;151:478–487. doi: 10.1093/oxfordjournals.aje.a010233. [DOI] [PubMed] [Google Scholar]
- 12.Lee HC, Nam JH, Lim SK, Huh KB, Kim KR, Nam SY, Park SW, Ahn CW, Song YD, Kim DJ, Ko YG. Associations of carotid intinma-media thickness measured by high resolution b-mode ultrasonography and atherosclerotic risk factors in NIDDM patients. J Korean Diabetes Assoc. 1999;23:234–242. [Google Scholar]
- 13.Raitakari OT, Celermajer DS. Flow-mediated dilatation. Br J Clin Pharmacol. 2000;50:397–404. doi: 10.1046/j.1365-2125.2000.00277.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yamashina A, Tomiyama H, Takeda K, Tsuda H, Arai T, Hirose K, Koji Y, Hori S, Yamamoto Y. Validity, reproducibility, and clinical significance of noninvasive brachial-ankle pulse wave velocity measurement. Hypertens Res. 2002;25:359–364. doi: 10.1291/hypres.25.359. [DOI] [PubMed] [Google Scholar]
- 15.Weber T, Auer J, O'Rourke MF, Kvas E, Lassnig E, Berent R, Eber B. Arterial stiffness, wave reflections, and the risk of coronary artery disease. Circulation. 2004;109:184–189. doi: 10.1161/01.CIR.0000105767.94169.E3. [DOI] [PubMed] [Google Scholar]
- 16.Conroy RM, Pyorala K, Fitzgerald AP, Sans S, Menotti A, De Backer G, De Bacquer D, Ducimetiere P, Jousilahti P, Keil U, Njolstad I, Oganov RG, Thomsen T, Tunstall-Pedoe H, Tverdal A, Wedel H, Whincup P, Wilhelmsen L, Graham IM SCORE project group. Estimation of ten-year risk of fatal cardiovascular disease in Europe: the SCORE project. Eur Heart J. 2003;24:987–1003. doi: 10.1016/s0195-668x(03)00114-3. [DOI] [PubMed] [Google Scholar]
- 17.van der Heijden AA, Ortegon MM, Niessen LW, Nijpels G, Dekker JM. Prediction of coronary heart disease risk in a general, pre-diabetic, and diabetic population during 10 years of follow-up: accuracy of the Framingham, SCORE, and UKPDS risk functions: The Hoorn Study. Diabetes Care. 2009;32:2094–2098. doi: 10.2337/dc09-0745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Chambless LE, Heiss G, Folsom AR, Rosamond W, Szklo M, Sharrett AR, Clegg LX. Association of coronary heart disease incidence with carotid arterial wall thickness and major risk factors: the Atherosclerosis Risk in Communities (ARIC) Study, 1987-1993. Am J Epidemiol. 1997;146:483–494. doi: 10.1093/oxfordjournals.aje.a009302. [DOI] [PubMed] [Google Scholar]
- 19.Brohall G, Oden A, Fagerberg B. Carotid artery intima-media thickness in patients with type 2 diabetes mellitus and impaired glucose tolerance: a systematic review. Diabet Med. 2006;23:609–616. doi: 10.1111/j.1464-5491.2005.01725.x. [DOI] [PubMed] [Google Scholar]
- 20.Bae JH, Seung KB, Jung HO, Kim KY, Yoo KD, Kim CM, Cho SW, Cho SK, Kim YK, Rhee MY, Cho MC, Kim KS, Jin SW, Lee JM, Kim KS, Hyun DW, Cho YK, Seong IW, Jeong JO, Park SC, Jeong JY, Woo JT, Koh G, Lim SW. Analysis of Korean carotid intima-media thickness in Korean healthy subjects and patients with risk factors: Korea multi-center epidemiological study. Korean Circ J. 2005;35:513–524. [Google Scholar]
- 21.Temelkova-Kurktschiev TS, Koehler C, Leonhardt W, Schaper F, Henkel E, Siegert G, Hanefeld M. Increased intimal-medial thickness in newly detected type 2 diabetes: risk factors. Diabetes Care. 1999;22:333–338. doi: 10.2337/diacare.22.2.333. [DOI] [PubMed] [Google Scholar]
- 22.Matsumoto K, Sera Y, Nakamura H, Ueki Y, Miyake S. Correlation between common carotid arterial wall thickness and ischemic stroke in patients with type 2 diabetes mellitus. Metabolism. 2002;51:244–247. doi: 10.1053/meta.2002.28971. [DOI] [PubMed] [Google Scholar]
- 23.Benjamin EJ, Larson MG, Keyes MJ, Mitchell GF, Vasan RS, Keaney JF, Jr, Lehman BT, Fan S, Osypiuk E, Vita JA. Clinical correlates and heritability of flow-mediated dilation in the community: the Framingham Heart Study. Circulation. 2004;109:613–619. doi: 10.1161/01.CIR.0000112565.60887.1E. [DOI] [PubMed] [Google Scholar]
- 24.Henry RM, Ferreira I, Kostense PJ, Dekker JM, Nijpels G, Heine RJ, Kamp O, Bouter LM, Stehouwer CD. Type 2 diabetes is associated with impaired endothelium-dependent, flow-mediated dilation, but impaired glucose metabolism is not: The Hoorn Study. Atherosclerosis. 2004;174:49–56. doi: 10.1016/j.atherosclerosis.2004.01.002. [DOI] [PubMed] [Google Scholar]
- 25.Su Y, Liu XM, Sun YM, Wang YY, Luan Y, Wu Y. Endothelial dysfunction in impaired fasting glycemia, impaired glucose tolerance, and type 2 diabetes mellitus. Am J Cardiol. 2008;102:497–498. doi: 10.1016/j.amjcard.2008.03.087. [DOI] [PubMed] [Google Scholar]
- 26.Ravikumar R, Deepa R, Shanthirani C, Mohan V. Comparison of carotid intima-media thickness, arterial stiffness, and brachial artery flow mediated dilatation in diabetic and nondiabetic subjects (The Chennai Urban Population Study [CUPS-9]) Am J Cardiol. 2002;90:702–707. doi: 10.1016/s0002-9149(02)02593-6. [DOI] [PubMed] [Google Scholar]
- 27.Shin JY, Lee MY, Koh JH, Kim JY, Shin YG, Chung CH. Association between impaired vascular endothelial function and high sensitivity C-reactive protein, a chronic inflammatory marker, in patients with type 2 diabetes mellitus. J Korean Diabetes Assoc. 2005;29:469–478. [Google Scholar]
- 28.Celermajer DS, Sorensen KE, Bull C, Robinson J, Deanfield JE. Endothelium-dependent dilation in the systemic arteries of asymptomatic subjects relates to coronary risk factors and their interaction. J Am Coll Cardiol. 1994;24:1468–1474. doi: 10.1016/0735-1097(94)90141-4. [DOI] [PubMed] [Google Scholar]
- 29.Mitchell GF, Hwang SJ, Vasan RS, Larson MG, Pencina MJ, Hamburg NM, Vita JA, Levy D, Benjamin EJ. Arterial stiffness and cardiovascular events: the Framingham Heart Study. Circulation. 2010;121:505–511. doi: 10.1161/CIRCULATIONAHA.109.886655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sugawara J, Hayashi K, Yokoi T, Cortez-Cooper MY, DeVan AE, Anton MA, Tanaka H. Brachial-ankle pulse wave velocity: an index of central arterial stiffness? J Hum Hypertens. 2005;19:401–406. doi: 10.1038/sj.jhh.1001838. [DOI] [PubMed] [Google Scholar]
- 31.Laurent S, Cockcroft J, Van Bortel L, Boutouyrie P, Giannattasio C, Hayoz D, Pannier B, Vlachopoulos C, Wilkinson I, Struijker-Boudier H European Network for Non-invasive Investigation of Large Arteries. Expert consensus document on arterial stiffness: methodological issues and clinical applications. Eur Heart J. 2006;27:2588–2605. doi: 10.1093/eurheartj/ehl254. [DOI] [PubMed] [Google Scholar]
- 32.Blacher J, Asmar R, Djane S, London GM, Safar ME. Aortic pulse wave velocity as a marker of cardiovascular risk in hypertensive patients. Hypertension. 1999;33:1111–1117. doi: 10.1161/01.hyp.33.5.1111. [DOI] [PubMed] [Google Scholar]
- 33.Park HY, Lee SY, Cho SW, Kwon SU, Namgung J, Hyung J, Lee WR. Correlations between the left ventricular diastolic function and aortic stiffness in healthy aged subjects. Korean Circ J. 2006;36:393–399. [Google Scholar]
- 34.Cruickshank K, Riste L, Anderson SG, Wright JS, Dunn G, Gosling RG. Aortic pulse-wave velocity and its relationship to mortality in diabetes and glucose intolerance: an integrated index of vascular function? Circulation. 2002;106:2085–2090. doi: 10.1161/01.cir.0000033824.02722.f7. [DOI] [PubMed] [Google Scholar]
- 35.Dart AM, Gatzka CD, Kingwell BA, Willson K, Cameron JD, Liang YL, Berry KL, Wing LM, Reid CM, Ryan P, Beilin LJ, Jennings GL, Johnston CI, McNeil JJ, Macdonald GJ, Morgan TO, West MJ. Brachial blood pressure but not carotid arterial waveforms predict cardiovascular events in elderly female hypertensives. Hypertension. 2006;47:785–790. doi: 10.1161/01.HYP.0000209340.33592.50. [DOI] [PubMed] [Google Scholar]
- 36.Schram MT, Henry RM, van Dijk RA, Kostense PJ, Dekker JM, Nijpels G, Heine RJ, Bouter LM, Westerhof N, Stehouwer CD. Increased central artery stiffness in impaired glucose metabolism and type 2 diabetes: the Hoorn Study. Hypertension. 2004;43:176–181. doi: 10.1161/01.HYP.0000111829.46090.92. [DOI] [PubMed] [Google Scholar]