Alterations of epigenetic marks have an important role in myeloid malignancies.1 Mutations have been found in several epigenetic regulators including ASXL1, DNMT3A, EZH2, IDH1/2 and TET2.2, 3 DNMT3A, IDH1/2 and TET2 are involved in the regulation of DNA methylation. EZH2 belongs to a complex of proteins called ‘polycomb repressor complex 2' (PRC2).4 ASXL1 is a regulator of PRC1, the other polycomb complex. Several other proteins belong to the two polycomb complexes; although alterations of ASXL15 and EZH26, 7 are probably prominent in myeloid diseases, we suspected that these other PRC components might also be involved in some cases.
We searched for mutations in SUZ12, a gene encoding a PCR2 protein, in 186 whole blood samples from 125 myeloproliferative neoplasms (MPNs), 14 MPNs at the blast-phase stage and 47 chronic myelomonocytic leukemias (CMML). The MPNs comprised 33 polycythemia vera (PV) and 4 post-PV myelofibrosis (MF), 51 essential thrombocythemia (ET) and 9 post-ET MF, 22 primary myelofibrosis, 3 unclassifiable and 3 myelodysplastic syndrome/MPN cases. SUZ12 is located on chromosome arm 17q (Figure 1a) and comprises 16 exons (Figure 1b). We determined the sequence of SUZ12 exons 10 to 16 (the existence of a highly conserved SUZ12 pseudogene prevented easy design of oligonucleotide primers for SUZ12 exons 1 to 9). In the MPN samples, we also searched for mutations of JAK2 (exon 14), ASXL1 (exons 12), DNMT3A (exons 15–23), IDH1 and IDH2 (exon 4), MPL (exon 10) and TET2 (all exons). Mutations were searched by Sanger DNA sequencing analysis except for the JAK2 mutation, which was detected by semi-quantitative PCR. We searched for SUZ12 deletions in 80 of the MPN samples and in the 47 CMML samples by using array-comparative genomic hybridization (aCGH) on high-density oligonucleotide microarrays (Hu-244A, Agilent Technologies, Massy, France), as described previously.8 All patients signed an informed consent and the study was approved by our ethics committee.
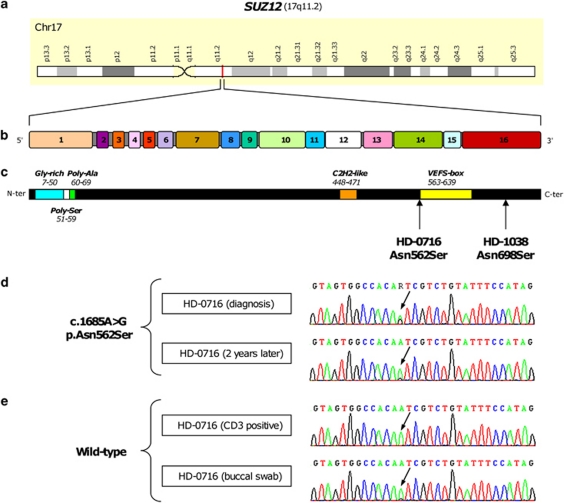
Figure 1.
Mutation of the SUZ12 gene in MPNs. (a) Localization of the SUZ12 gene in chromosomal region 17q11. (b) Representation of the 16 SUZ12 exons. (c) Representation of the 739-amino-acid-long SUZ12 protein with known motifs and domains. Localization of the mutations is indicated below by arrowheads. (d) Nucleotide sequences for HD-0716 PV case. Sequence of the mutated SUZ12 allele demonstrates change at the position indicated by an arrow in the sample taken at diagnosis. (e) The mutation was hardly detectable after 2 years of treatment and is absent from CD3 T-cells and buccal swab samples. The corresponding sequence is shown above the tracks.
We found mutations of SUZ12 in two MPN patients: a PV, HD-0716 and a blast phase of post-ET MF, HD-1038 (Table 1). The two mutations were missense and apparently heterozygous (Figures 1c and d). In the PV case, sequencing of DNA from buccal swab or CD3-purified T-cells showed the absence of variation compared with reference, thus showing that the mutation was acquired (Figure 1e). The HD-0716 PV case was JAK2 V617F-positive (the mutated allele burden was 15–30%) and TET2-mutated. After diagnosis, the patient, a 66-year-old man, was treated with hydroxyurea. After 2 years of treatment a blood sample processed and studied as the initial one showed that the SUZ12 mutation was barely detectable (Figure 1d). The HD-1038 blast phase of a post-ET MF was diagnosed in an 80-year-old woman. The chronic stage was not available for study. At the acute stage, the sample was JAK2 V617F-negative and TET2-mutated (c.3640C>T p.Arg1214Trp), the karyotype was complex and the aCGH profile showed, among other alterations, a heterozygous loss of TET2, 7q21-qter spanning EZH2, 12p12-p13 spanning AEBP2 and 17q11-q21 spanning NF1 and the other SUZ12 allele (Figure 2). No germline DNA was available. We did not detect any mutation of ASXL1 in the two cases, whereas 17 other MPN cases were mutated (Table 1). We found 2 mutations in DNMT3A,9 2 in IDH2, 1 in MPL and 15 in TET2. No SUZ12 mutation was found in the 47 CMML cases.
Table 1. Mutations and deletions of SUZ12 in patients with myeloproliferative neoplasms.
| SUZ12 mutation | SUZ12 loss | ASXL1 mutation | ASXL1 loss | DNMT3A mutation | IDH1/2 mutation | JAK2 mutation | MPL mutation | TET2 mutation | TET2 loss | |
|---|---|---|---|---|---|---|---|---|---|---|
| Polycythemia vera | 1/33 | 0/7 | 3/33 | 0/7 | 1/33 | 0/33 | 32/33 | 0/33 | 4/33 | 0/7 |
| Essential thrombocythemia | 0/51 | 0/19 | 2/51 | 0/19 | 0/51 | 0/51 | 32/51 | 0/51 | 3/51 | 0/19 |
| Myelofibrosis | 0/35 | 1/35 | 8/35 | 1/35 | 1/35 | 0/35 | 24/35 | 0/35 | 4/35 | 0/35 |
| Other MPN | 0/6 | 0/6 | 2/6 | 0/6 | 0/6 | 0/6 | 1/6 | 0/6 | 2/6 | 0/6 |
| Blast phase MPN | 1/14a | 2/13a | 2/14 | 0/13 | 0/14 | 2/14 | 4/14 | 1/14 | 2/14a | 2/13a |
| Total MPNs | 2/139a | 3/80a | 17/139 | 1/80 | 2/139 | 2/139 | 93/139 | 1/139 | 15/139a | 2/80a |
Abbreviation: MPN, myeloproliferative neoplasm.
HD-1038 sample in common.
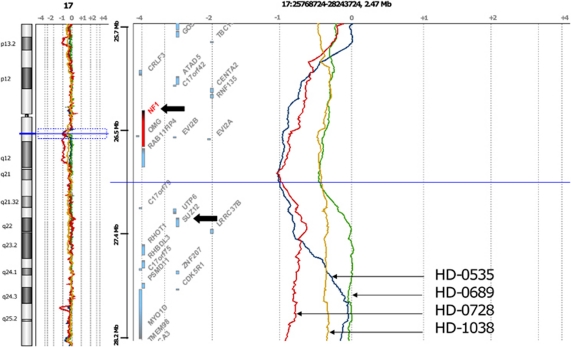
Figure 2.
aCGH profile of chromosome 17 in four MPN cases showing loss of the NF1-SUZ12 region. HD-0535 (blue profile), a blast phase primary myelofibrosis (PMF), HD-0728 (red profile), a PMF and HD-1038 (yellow profile), a blast phase post-ET MF show loss, among other genes, of NF1 and SUZ12, whereas in HD-0689 (green profile), a PMF, no loss of SUZ12 is observed. Deletion of NF1 but not SUZ12 in HD-0689, and mutation of SUZ12 in HD-1038 suggest both genes participate in leukemogenesis.
Deletions of the NF1 gene at 17q11 are frequent in MPNs.10 By using aCGH we found that the SUZ12 gene, which lies close to NF1, is often included in these deletions. We found SUZ12 deletions in three MPNs and in two CMML cases (Figure 2). This was the case for HD-1038. No aCGH profile was available for HD-0716. Two other MPN cases (HD-0535 and HD-0728) showed a deletion of a SUZ12 allele but were not mutated (Table 1); however, as mentioned above, the first nine exons of the gene were not studied. In another MPN case (HD-0689), the deletion encompassed NF1 but not SUZ12 (Figure 2), suggesting that NF1 was the actual targeted gene of the deletion.
Our study shows that a mutation can affect a PRC2 component other than EZH2 in few cases of MPN. This mutation of SUZ12 may substitute for or cooperate with a mutation of EZH2 to compromise PRC2. This reinforces the current view that position epigenetic regulators as major players of leukemogenesis, together with signaling molecules and transcription factors. SUZ12 mutations are rare but are actually in the same range as DNMT3A,9 IDH2 and MPL. Because we tested only the last exons of the gene our study may have underestimated the frequency of SUZ12 mutations. Interestingly, in addition to mutations, SUZ12 function could be affected by gene loss. The 17q11 region encompassing NF1 and SUZ12 is deleted in MPNs10 and CMMLs. Our study suggests that both genes may participate to leukemogenesis, explaining why the NF1-SUZ12 17q11 region is often lost en bloc. It is also possible that other genes are involved; we sequenced the exons of RAB11FIP4, located between the NF1 and SUZ12, in our series of MPN cases but found no mutation.
PRC2 is the major methyltransferase for H3K27 methylation, a modification of histone H3 that represses gene expression programs throughout development.4 Mice with loss of function mutations in PRC2 components display enhanced activity of their hematopoietic stem cell/progenitor population and loss of SUZ12 function in particular enhances hematopoietic stem cell activity.11 It remains to be determined whether the mutations we have identified lead to a loss of function of the protein. In the HD-0716 case, the c.1685A>G p.Asn562Ser mutation may affect the VEFS-box known to be critical for EZH2 interaction,12 consequently disrupting PRC2 function. In the HD-1038 case, the deletion of an allele combined with the mutation of the other allele suggests a two-hit process associated with a tumor suppressor. Whether SUZ12 mutations are present in other hematopoietic diseases should be determined. Whether other PRC2 components, such as AEBP2, EED and JARID2, are mutated and how they combined with other mutations in hematopoietic diseases should also be investigated.
Many genes contribute to leukemogenesis through mutations of their sequence; our study also shows that, beside the major contributors, most of which may be known already, there may be a number of rarely mutated contributors. This could render the molecular diagnosis of myeloid diseases more difficult to establish. Many of these rarely mutated genes (for example, SUZ12) may affect the same functions and pathways (for example, polycomb repression) as the frequently mutated ones (for example, EZH2).
Acknowledgments
This work was supported by Inserm, Institut Paoli-Calmettes, and grants from Association pour la Recherche contre le Cancer (DB, no. 4992, 2010) and Association Laurette Fugain (MJM, 2010).
The authors declare no conflict of interest.
References
- Acquaviva C, Gelsi-Boyer V, Birnbaum D. Myelodysplastic syndromes: lost between two states. Leukemia. 2010;24:1–5. doi: 10.1038/leu.2009.157. [DOI] [PubMed] [Google Scholar]
- Tefferi A. Novel mutations and their functional and clinical relevance in myeloproliferative neoplasms: JAK2, MPL, TET2, ASXL1, CBL, IDH and IKZF1. Leukemia. 2010;24:1128–1138. doi: 10.1038/leu.2010.69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vainchenker W, Delhommeau F, Constantinescu SN, Bernard OA.New mutations and pathogenesis of myeloproliferative neoplasms Blood 2011(in press). [DOI] [PubMed]
- Margueron R, Reinberg D. The polycomb complex PRC2 and its mark in life. Nature. 2011;469:343–349. doi: 10.1038/nature09784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gelsi-Boyer V, Trouplin V, Adelaide J, Bonansea J, Cervera N, Carbuccia N, et al. Mutations of polycomb-associated gene ASXL1 in myelodysplastic syndromes and chronic myelomonocytic leukaemia. Br J Haematol. 2009;145:788–800. doi: 10.1111/j.1365-2141.2009.07697.x. [DOI] [PubMed] [Google Scholar]
- Ernst T, Chase AJ, Score J, Hidalgo-Curtis CE, Bryant C, Jones AV, et al. Inactivating mutations of the histone methyltransferase gene EZH2 in myeloid disorders. Nat Genet. 2010;42:722–726. doi: 10.1038/ng.621. [DOI] [PubMed] [Google Scholar]
- Nikoloski G, Langemeijer SM, Kuiper RP, Knops R, Massop M, Tonnissen ER, et al. Somatic mutations of the histone methyltransferase gene EZH2 in myelodysplastic syndromes. Nat Genet. 2010;42:665–667. doi: 10.1038/ng.620. [DOI] [PubMed] [Google Scholar]
- Gelsi-Boyer V, Trouplin V, Adélaïde J, Aceto N, Remy V, Pinson S, et al. Genome profiling of chronic myelomonocytic leukemia: frequent alterations of RAS and RUNX1 genes. BMC Cancer. 2008;8:299–314. doi: 10.1186/1471-2407-8-299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brecqueville M, Cervera N, Gelsi-Boyer V, Murati A, Adélaïde J, Chaffanet M, et al. Rare mutations in DNMT3A in myeloproliferative neoplasms and myelodysplastic syndromes. Blood Cancer J. 2011;1:e18. doi: 10.1038/bcj.2011.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klampfl T, Harutyunyan A, Berg T, Gisslinger B, Schalling M, Bagienski K, et al. Genome integrity of myeloproliferative neoplasms in chronic phase and during disease progression. Blood. 2011;118:167–176. doi: 10.1182/blood-2011-01-331678. [DOI] [PubMed] [Google Scholar]
- Majewski IJ, Blewitt ME, de Graaf CA, McManus EJ, Bahlo M, Hilton AA, et al. Polycomb repressive complex 2 (PRC2) restricts hematopoietic stem cell activity. PLoS Biol. 2008;6:e93. doi: 10.1371/journal.pbio.0060093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto K, Sonoda M, Inokuchi J, Shirasawa S, Sasazuki T. Polycomb group suppressor of Zeste 12 links heterochromatin protein 1α and enhancer of Zeste 2. J Biol Chem. 2004;279:401–406. doi: 10.1074/jbc.M307344200. [DOI] [PubMed] [Google Scholar]