Abstract
Background
In patients over age 60 with acute myeloid leukemia (AML), cure rates are under 10% despite intensive chemotherapy. These patients often have comorbidities, and their treatment must be chosen with care. For those who are not candidates for intensive chemotherapy, one of the available options for palliative treatment should be chosen on the basis of an individual risk-benefit assessment.
Methods
Selective literature review.
Results
An evaluation of the patient’s general condition and comorbidities, a geriatric assessment, and specially designed risk scores are useful aids to the choice of an appropriate treatment. Some elderly patients with AML can benefit from intensive chemotherapy despite their age; for highly selected elderly patients, allogeneic stem-cell transplantation is an increasingly feasible option. Hypomethylating agents or low-dose cytarabine can be suitable for some patients. Further options include experimental treatment as part of a clinical trial, and supportive care alone. In the special case of acute promyelocytic leukemia, more than half of all patients can be cured with combination chemotherapy including all-trans retinoic acid.
Conclusion
The prognosis of elderly AML patients remains poor despite recent therapeutic advances. The appropriate treatment for each patient can be chosen on the basis of a risk-benefit assessment. Clinical trials evaluating new treatments are urgently needed.
Acute myeloid leukemia (AML) is a rare disease, with an overall incidence of 4 per 100 000 persons. It becomes more common with advancing age (1); thus, as the population ages, more cases of AML can be expected. The current five-year survival rates of patients under age 60 who receive intensive chemotherapy for AML range from 30% to over 40% (e1– e8). Age 60 is now internationally accepted as the dividing line between “younger” and “older” AML patients; this division is arbitrary, rather than evidence-based (2).
Older patients with AML who receive intensive chemotherapy have a markedly worse prognosis, with a 5-year survival rate of about 15% (3) (Figure 1a). There are multiple reasons for this. The toxicity of cytotoxic drugs is increased in the elderly. Moreover, older patients more commonly have particular risk factors for a bad outcome, including poor risk cytogenetic abnormalities (Table 1) and the overexpression of genes associated with drug resistance (2, 3). They also more often have medical contraindications to intensive chemotherapy. Finally, aside from all of these factors, age itself is an independent prognostic factor in AML (3). In this article, we provide an overview of the suitable treatment options for older patients with AML in the light of the recent introduction of hypomethylating chemotherapy as well as other new developments that affect the choice of treatment.
Table 1. Cytogenetic and molecular genetic risk classification in AML (from [4]).
| Risk class | Cytogenetic findings | Molecular genetic findings |
| Low*1 | t(8;21)(q22;q22) | RUNX1-RUNX1T1 |
| inv(16)(p13.1q22) or t(16;16)(p13.1;q22) | CBFB-MYH11 | |
| normal karyotype | NPM1 mutated and no FLT3-ITD | |
| normal karyotype | CEBPA mutated | |
| Intermediate(I) | normal karyotype | NPM1 mutated and FLT3-ITD |
| normal karyotype | NPM1 wild-type and FLT3-ITD | |
| normal karyotype | NPM1 wild-type and no FLT3-ITD | |
| Intermediate (II) | t(9;11)(p22;q23) | MLLT3-MLL |
| cytogenetic changes that are not associated with either low risk or high risk | ||
| High*1 | inv(3)(q21;q26.2) or | RPN1-EVI1 |
| t(3;3)(q21;q26.2) | ||
| t(6;9)(p23;q34) | DEK-NUP214 | |
| t(v;11)(v;q23)*2 | Fusion transcript with participation of MLL*2 | |
| −5 or del(5q); −7; abnl(17p); complex karyotype*3 |
*1The presence of low-risk changes entails classification in the low-risk group regardless of the possible presence of any further cytogenetic changes
*2Except the t (9;11)(p22;q23), harboring the MLLT3-MLL fusion transcript
*3A complex karyotype is defined by the presence of at least three cytogenetic changes, not including any of the following, which imply a special classification: t(15;17), t(8;21), inv(16) or t(16;16), t(9;11), t(v;11)(v;q23), t(6;9), inv(3) or t(3;3)
-: monosomy of the corresponding chromosome, abnl: abnormality, CBFB: core binding factor ß, CEBPA: CCAAT enhancer binding protein α, del: deletion, EVI1: ecotropic viral integration site 1, FLT3: FMS-like tyrosine kinase 3, inv: inversion, ITD: internal tandem duplication, MLL: mixed-lineage Leukemia, MLLT3: MLL translocation partner 3, MYH11: myosin heavy chain 11, NPM1: nucleophosmin 1, NUP214: nuclear pore complex protein 214, t: translocation, RPN1: ribophorin 1, RUNX1: runt-related transcription factor 1, RUNX1T1: RUNX1 translocation partner 1
Methods
The concrete treatment recommendations in this review are based on published studies in which patients were recruited from the year 2000 onward. Emphasis is laid on the findings of randomized controlled trials, as these provide scientific evidence of the highest level.
The outcome data presented here are based exclusively on studies published in the last two years that involved at least 200 unselected patients with AML.
Diagnostic evaluation
AML is diagnosed by bone marrow examination. The standard test battery for bone marrow samples currently includes cytomorphological and cytochemical studies, flow cytometry, cytogenetic analysis, and molecular studies for the detection of mutations that affect prognosis, including Flt3 and nucleophosmin mutations (4). Morphological and flow-cytometric studies mainly serve to distinguish AML from other elements of the differential diagnosis, such as lymphatic neoplasia; they also yield some prognostically relevant information. Cytogenetic analysis and testing for molecular abnormalities are done mainly for risk classification. The current ELN classification lists four risk groups (Table 1) (4).
Choice of treatment
Once AML has been diagnosed, a decision must be made for intensive chemotherapy, non-intensive chemotherapy, or supportive care alone. With the exception of very few cases, intensive chemotherapy is the only option that provides a chance of long-term survival, but it also carries a high risk. A meta-analysis has shown that only 50% of older patients treated with intensive chemotherapy for remission induction actually do achieve a complete remission (CR), while 15% to 20% of them die as a complication of treatment (5). Intensive and non-intensive chemotherapy have been tested against each other in only one randomized comparative trial of the European Organisation for Research and Treatment of Cancer (EORTC), which included 60 patients aged 65 or older (6): In this trial, intensive chemotherapy was found to yield significantly better outcomes (evidence level Ib). Thus, the question of which elderly patients stand to benefit from intensive chemotherapy, and which do not, is a field of active research. The variables associated with increased mortality after intensive remission-inducing chemotherapy include poor general condition and the presence of relevant comorbidities. The Eastern Cooperative Oncology Group (ECOG) performance status is a reliable index of the patient’s general condition (Table 2) (e9). Patients over 75 with an ECOG performance status of 3 or 4 who undergo intensive chemotherapy have a 30-day mortality above 50% (7). The hematopoietic stem-cell transplantation comorbidity index (HCT-CI) is also correlated with the outcome after intensive remission-inducing chemotherapy (8, e10); this index is a measure of the presence and severity of associated diseases, summarized on a point scale (Table 3). Other prognosticating systems based on the same risk factors have not won general acceptance, mainly because they are impractical for routine use or have not been validated, among other reasons.
Table 2. Eastern Cooperative Oncology Group (ECOG) performance status (e9).
| ECOG Score | Definition |
| 0 | No impairment of general condition |
| 1 | Impairment of general condition without impairment of physical activity |
| 2 | Impaired physical activity, confined to bed for less than half of daylight hours |
| 3 | Impaired physical activity, confined to bed for more than half of daylight hours |
| 4 | Permanently confined to bed |
Table 3. Hematopoietic stem cell transplantation comorbidity index (HCT-CI) (from [e10])*1.
| Class of comorbidity | Definition | Points |
| Arrhythmia | Atrial flutter or fibrillation, sick sinus syndrome, or ventricular arrhythmias | 1 |
| Heart disease | Coronary heart disease requiring treatment with medication, stent, or bypass surgery; congestive heart failure; left ventricular ejection fraction 50% or less | 1 |
| Inflammatory bowel disease | Crohn’s disease or ulcerative colitis | 1 |
| Diabetes mellitus | Need for antidiabetic medication (insulin or oral antidiabetics) | 1 |
| Cerebrovascular disease | Transient ischemic attack or stroke | 1 |
| Mental illness | Depression or anxiety disorder requiring psychiatric treatment | 1 |
| Mild liver disease | Chronic hepatitis, bilirubin elevated to no more than 1.5 times the upper limit of normal or AST/ALT elevated to no more than 2.5 times the upper limit of normal | 1 |
| Overweight | Body mass index >35 kg/m2 | 1 |
| Infection | Need for antibiotics at the start of antileukemic treatment and beyond | 1 |
| Rheumatological disease | SLE, RA, polymyositis, soft-tissue rheumatism, or polymyalgia rheumatica | 2 |
| Gastric or duodenal ulcer | Need for medication | 2 |
| Renal disease | Serum creatinine >2 mg/dL, dialysis, or status post renal transplantation | 2 |
| Moderately severe lung disease | DLCO and/or FEV1 66%–80% or exertional dyspnea NYHA II | 2 |
| Neoplastic (tumor) disease | Any prior neoplastic disease requiring treatment, except non-melanotic skin tumors | 3 |
| Valvular heart disease | Any type except mitral valve prolapse | 3 |
| Severe lung disease | DLCO and/or FEV1 65% or dyspnea at rest or need for supplemental oxygen | 3 |
| Moderate or severe liver disease | Hepatic cirrhosis or bilirubin above 1.5 times the upper limit of normal or AST/ALT above 2.5 times the upper limit of normal | 3 |
| Low risk | Total | 0 |
| Intermediate risk | Total | 1–2 |
| High risk | Total | ≥3 |
1This score is correlated with mortality of AML patients after intensive remission-inducing chemotherapy. In the study of Giles et al. (8), the 28-day mortality was 3% in patients at low risk,11% in patients at intermediate risk, and 29% in patients at high risk
The authors, in collaboration with two study groups (the German AML Cooperative Group [AMLCG] and the Study Alliance Leukemia [SAL]), have developed a prognostic scoring system to estimate the probability of response to intensive chemotherapy, and of early death as a complication of treatment, in patients for whom intensive chemotherapy is a feasible option (9). This scoring system was designed on the basis of a retrospective analysis of the older AML patients who were intensively treated in the AMLCG99 trial, and was validated on an independent cohort consisting of the intensively treated patients in the AML96 trial of the SAL. Its predictive accuracy was assessed by the area under the curve (AUC) and the receiver operator characteristics, reflecting specificity versus (1 minus sensitivity). The calculated AUC values ranged from 0.63 to 0.71 (9).
The scoring system is based on the following variables:
Age
Hemoglobin concentration
Platelet count
Lactate dehydrogenase serum level
Fibrinogen
Fever
Leukemia type (de novo vs. secondary to radio- or chemotherapy)
Molecular and cytogenetic risk classification (Table 1).
The scoring system can be used to identify patients for whom intensive chemotherapy would be associated with a low chance of success and high mortality (Figure 1b). The prognostic score is easy to calculate with the aid of the website www.AML-Score.org. Patients for whom intensive chemotherapy is found to be unsuitable can be treated with non-intensive therapy of various types, supportive care alone, or experimental treatment with new drugs in a clinical trial. The possible utility of a geriatric assessment as an aid to decision-making in this situation is currently being studied. Deschler et al. (10) found that a limitation in the patient’s activities of daily living (ADL) with a Barthel index below 100, lessened performance status, and severe fatigue were all associated with a markedly lower survival rate after hypomethylating treatment with decitabine. This reported association is now being tested in a multicenter observational study under the direction of Prof. Lübbert (Freiburg, Germany). Though prognostic scores may well turn out to have clinical value, they should never be used as the sole basis of a recommendation for or against intensive chemotherapy; the clinical judgment of an experienced hematologist should remain decisive.
The whishes and expectations of the patient after thorough information about the risks and outlooks of various therapeutic strategies will ultimately guide the final therapy decision.
Intensive chemotherapy
Intensive chemotherapy begins with one or two courses of remission-inducing chemotherapy, usually based on cytarabine and an anthracycline. The various types of induction therapy that are now in use all yield comparable results; in a meta-analysis incorporating the findings of 65 randomized controlled studies, all of the induction schemes tested were found to be equally effective with regard to overall survival, compared to reference treatment with standard doses of cytarabine and daunorubicin (11). The so-called Intergroup Protocol, which has been accepted as a common standard arm for therapeutic trials by various German study groups, can be a possible reference therapy (12). The typical risks of remission-inducing chemotherapy for AML include possibly lethal infections due to neutropenia, which are more common in older patients; mucositis; Cytarabine-induced skin rash; and cardiac side effects of anthracyclines (cardiomyopathy, arrhythmia). Thus, remission-inducing chemotherapy for AML should only be performed in experienced hematological centers. We have shown, in a non-randomized study, that post-remission therapy is a prerequisite for long-term disease-free survival (evidence level IIa) (13), although the optimal post-remission therapy for older patients remains unclear and is a question of active research (e11). In a randomized trial conducted by the Cancer and Leukemia Group B, an increased dose of cytarabine in older patients was associated with more severe side effects, particularly affecting the central nervous system (CNS), and did not prolong survival, as opposed to younger patients (14). Patients who do achieve a complete remission (CR; for the definition of treatment responses, cf. Table 4 from [15]) usually receive one to several cycles of consolidation therapy, often likewise based on cytarabine in varying doses among different protocols, with older patients receiving fewer courses and lower doses in each course (e.g., 5–6 g/m2 instead of 36 g/m2 per course) because of CNS toxicity. As specified by the trial protocol, some patients then receive maintenance treatment based on either classic cytotoxic drugs or experimental drugs. There is no adequate evidence for the use of maintenance treatment outside of clinical trials (e12). Considering the unsatisfactory results of intensive chemotherapy, with a long-term survival rate below 15%, the inclusion of older patients in clinical trials is to be welcomed (evidence level IV) (e13).
Stem-cell transplantation
The treatment of younger AML patients with allogeneic stem-cell transplantation is becoming increasingly common (e14), as meta-analyses have revealed a survival advantage for AML patients with an available donor compared to those without a donor (16). According to current data from the German AML Intergroup, allogeneic stem-cell transplantation is performed in 20% to 30% of younger patients in their first complete remission, depending on the study group (T. Büchner, manuscript in preparation). This form of treatment is associated with a substantially increased morbidity and mortality in older patients, mainly due to infectious complications and graft-versus-host disease (GvHD). A retrospective analysis of 52 patients aged 60 or above who underwent allogeneic stem-cell transplantation with classic myeloablative conditioning for hematological diseases revealed a 3-year treatment-related mortality of 42%, a 20% rate of severe (grade III or IV) acute GvHD, and a 53% rate of extensive chronic GvHD (e15). Nonetheless, advances in tissue typing, the increasing availability of unrelated donors, and modern, reduced-intensity conditioning (RIC) protocols with decreased toxicity have now made stem-cell transplantation a feasible therapeutic option for older patients as well (17). Currently, only highly selected elderly patients are being offered allogeneic stem-cell transplantation in first CR outside of clinical trials. In a recently published, non-randomized comparative study, the 3-year survival rate of patients aged 60 to 70 who underwent allogeneic stem-cell transplantation in their first remission was higher after RIC than after classic myeloablative conditioning treatment (37% vs. 25%), but this difference was not statistically significant (evidence level III) (e16). The putative benefit of an allogeneic stem-cell transplantation with RIC compared to classical consolidation chemotherapy for older AML patients in first CR is currently being studied in an international randomized trial under the direction of Prof. Niederwieser (Leipzig).
Palliative chemotherapy
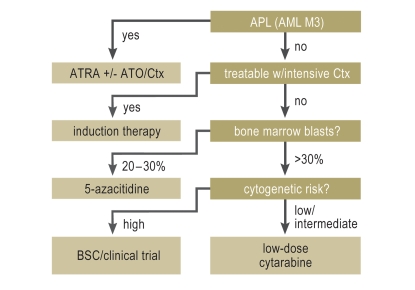
It has recently been discovered that patients with less proliferative AML (defined as a bone marrow blast percentage of 30% or less) stand to benefit from a palliative treatment with hypomethylating drugs such as 5-azacitidine and decitabine, which partially revert the aberrant methylation of cytosine remnants in the DNA of leukemic cells (for a review, see [18]). Data from recently published randomized trials suggest that the efficacy of treatment with these drugs may be comparable to that of intensive chemotherapy (19) und superior to that of other palliative treatment approaches (19, 20). They can be given on an outpatient basis, as their side effects (e.g., altered blood counts, skin irritation, infections and abscesses at the injection site) are much less severe than those of intensive chemotherapy (evidence level Ib) (19, 20). The median survival under treatment with 5-azacitidine was found to be 24 months (19). Interestingly, patients with high-risk cytogenetic aberrations also responded to hypomethylating drugs. Treatment with hypomethylating drugs, unlike treatment with low-dose cytarabine, was also beneficial for patients achieving only an incomplete (CRi, CRp) or partial remission (PR) as best response (Table 4). In view of the poor results of other treatments, treatment with 5-azacitidine (75 mg/m2 SC qd for seven days every four weeks until disease progression) is now considered standard for elderly AML patients who are not suitable candidates for intensive chemotherapy, with a bone marrow blast percentage of 30% or less and multiline dysplasia (Figure 2), and has been approved for this indication in Germany (evidence level Ib). Decitabine, on the other hand, is currently not approved in Germany. Randomized trials (including those of the German SAL, AMLSG, and OSHO study groups) are now underway to determine whether treatment with 5-azacitidine is also effective in patients with more than 30% blasts in their bone marrow, and whether it has an additional effect when given in combination with intensive chemotherapy.
Table 4. Definitions of the response to treatment, modified from the criteria of the International Working Group (IWG) and the European Leukemia Net (ELN) (4, 15).
| Type of response | Definition |
| Complete remission (CR) | Disappearance of blasts from peripheral blood, reduction of bone marrow blasts to <5% with complete recovery of peripheral blood counts with no need for red-cell transfusion, a granulocyte count ≥ 1000/µl, and a platelet count ≥ 100 000/µl |
| Complete remission with incomplete platelet regeneration (CRp) | Disappearance of blasts from peripheral blood, reduction of bone marrow blasts to <5% with granulocyte regeneration (≥≥ 1 000/µl) and incomplete platelet regeneration (< 100 000/µl) |
| Complete remission with incomplete neutrophil regeneration (CRi) | Disappearance of blasts from peripheral blood, reduction of bone marrow blasts to <5% with incomplete granulocyte regeneration (< 1000/µl), regardless of the degree of platelet regeneration |
| Partial remission (PR) | More than 50% reduction of bone marrow blast fraction to a value between 5% and 25% with disappearance of blasts in peripheral blood, with or without recovery of peripheral blood counts |
| Treatment failure | Persistence of blasts in blood, or persistence of >25% blasts in bone marrow, or less than 50% reduction of bone marrow blast fraction |
Figure 2.
Flowchart for the stratification of primary treatment in older patients with acute myelocytic leukemia (AML)
For patients with acute promyelocytic leukemia (APL), aggressive substitution therapy in case of coagulopathy and the rapid initiation of combination therapy including ATRA can be life-saving. For all other AML patients, intensive chemotherapy is recommended if the patient is a suitable candidate, possibly supplemented by allogeneic stem-cell transplantation in selected patients, once complete remission has been attained. Patients for whom intensive chemotherapy is not feasible and have 30% or fewer blasts in their bone marrow can benefit from hypomethylating therapy with 5-azacitidine, while those with more than 30% blasts can benefit from treatment with low-dose cytarabine. The notion that patients with cytogenetic changes signifying high risk do not benefit from low-dose cytarabine is derived from a subgroup analysis and has not been further tested; this issue must be addressed in a prospective study. No other specific treatments are available for patients for whom the above treatments are not feasible. They should receive supportive care or be enrolled in clinical trials of new drugs according to their wishes.
APL, acute promyelocytic leukemia; ATO, arsenic trioxide; ATRA, all-trans retinoic acid; BSC, best supportive care.
Treatment with low-dose cytarabine (10 mg/m2 SC b.i.d. for 10 days, repeated every four weeks) was compared to treatment with hydroxyurea (HU) in a randomized trial; the resulting CR rates were 18% for cytarabine and 1% for HU, and the one-year survival after cytarabine treatment was about 25% (21), significantly better than that after HU. This treatment is well tolerated and can be provided on an outpatient basis. Its main side effect is an alteration of the blood counts. The survival advantage was only present among patients who attained a complete remission, and subgroup analysis revealed no benefit among patients with high-risk cytogenetic changes. Thus, this treatment can be recommended for patients who are not suitable candidates for either intensive remission-inducing therapy or hypomethylating therapy (evidence level Ib) (Figure 2). The subgroup analysis in the study of Burnett et al. does not provide enough evidence to settle the question whether patients with high-risk cytogenetic changes might benefit from this treatment; since relevant data from prospective trials are lacking, treating such patients in this way is not recommended in routine clinical practice (Figure 2). There is no standard treatment at present for patients who are not suitable candidates for intensive chemotherapy with more than 30% blasts in their bone marrow and high-risk cytogenetic changes (Figure 2). In case of marked leukocytosis, cytoreductive therapy may be indicated to control the white blood count, with mild leukocytosis (15–20 000/µL) as the target (evidence level IV). Meticulous prevention of infections, empirical antibiotic treatment (e17), and substitution of blood products can prolong the time to the development of leukemia-associated complications (evidence level IV); nevertheless, the median survival time among the patients in the trial cited in (21) was only 35 days. In this situation, therefore, the patient’s personal wishes with regard to further treatment (after adequate information from the physician) are paramount. One option is to participate in a clinical trial of a new drug (evidence level IV) (e13).
Acute promyelocytic leukemia
Acute promyelocytic leukemia (APL) is a special type of AML that is regularly associated with a t (15; 17) translocation. In Germany, APL accounts for about 10% of older patients with AML. APL in older patients has a relatively good prognosis (22 and Figure 1c). Treatment with all-trans retinoic acid (ATRA, also called tretinoin, the acid form of vitamin A) induces differentiation of the malignant promyelocytes, which then lose their proliferative ability and die. To prevent the development of drug resistance and to avoid differentiation syndrome, ATRA must be given in combination, usually with a chemotherapeutic drug, in most cases an anthracycline. This can be considered the current standard treatment for APL (23). Historically, the introduction of ATRA for APL led to a dramatic improvement in survival; thus, at present, a controlled trial of ATRA for this indication is no longer ethically permissible (evidence level III). Trials are currently addressing the efficacy of ATRA combined with arsenic trioxide (ATO), compared to ATRA combined with a chemotherapeutic drug. The established combinations, followed by consolidation therapy if CR is attained and sometimes by maintenance therapy containing ATRA, have led to cure rates above 50%, even among older patients (Figure 1c). APL is regularly associated with disseminated intravascular coagulation at the time of diagnosis, resulting in a substantial early death rate due to fatal hemorrhage. A recommendation based on expert consensus has therefore been issued stating that patients in whom APL is suspected on morphological grounds should receive aggressive substitution of platelets and clotting factors and should be treated for APL immediately, even before the diagnosis is confirmed with molecular-genetic techniques (evidence level IV) (24). Even patients with recurrent APL and those who fail to respond to treatment including ATRA still have a chance of attaining lasting remission through treatment with ATO, either as the sole agent or as a component of combination therapy (e18– e25); ATO has been approved for the treatment of refractory or recurrent APL in Germany (evidence level IIb). Once complete remission has been achieved, regular molecular monitoring is recommended, with the use of a quantitative polymerase chain reaction (PCR) to determine the degree of expression of the leukemia-specific PML-RARα fusion transcript that results from the t (15;17) translocation. Pre-emptive therapy should be initiated promptly if a molecular recurrence is detected (evidence level III) (25).
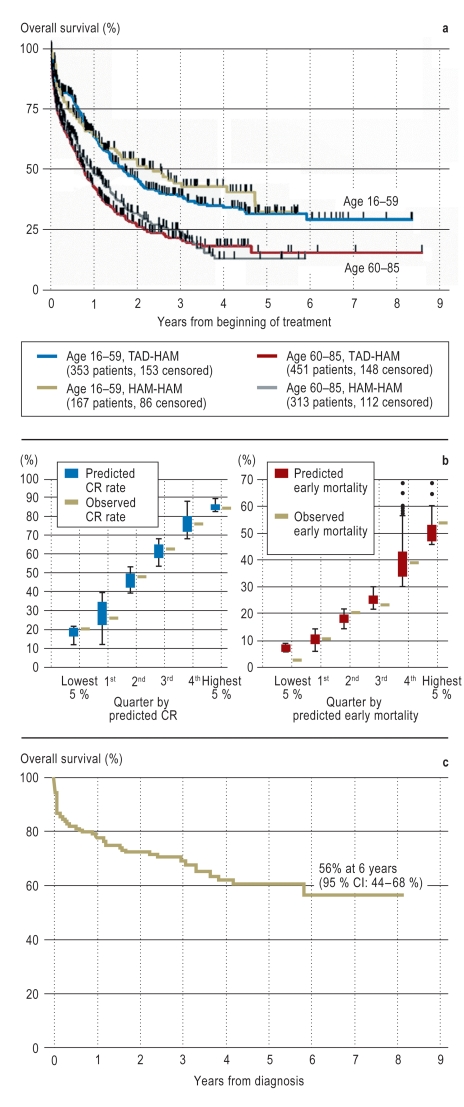
Figure 1.
Prognosis of older patients with AML
Overall survival curves of older and younger patients with AML (not APL) in the AMLCG1999 therapeutic trial. Age had a marked effect on survival. It made no appreciable difference which of two chemotherapeutic regimens was used (TAD-HAM or HAM-HAM).
Comparison of observed rates of complete remission (CR, left) and early mortality (i.e., defined as death within 60 days of the initiation of treatment; right) with predicted values derived from AML scores. These scores enable the identification before treatment of patients for whom intensive remission-inducing chemotherapy would be associated with high early mortality and a low probability of CR. The predicted values are very close to the observed values. Patients whose predicted early mortality is above 50% or who have a low probability of attaining CR generally do not benefit from intensive chemotherapy, even if they are considered candidates for it in principle.
Overall survival curve of older patients with acute promyelocytic leukemia (APL) treated with ATRA and chemotherapy. Noteworthy features are both the high early mortality and the 6-year survival rate of 56%, which is much better than the less than 20% survival of older patients with other subtypes of AML (Figure 1a).
TAD: thioguanine 100 mg/m2 p.o. b.i.d. on days 3–9, cytarabine 100 mg/ m2 IV as a continuous 24-hour infusion on days 1 and 2 and IV b.i.d. on days 3–7, and daunorubicin 60 mg/ m2 IV qd on days 3–5. HAM: cytarabine 1 g/ m2 IV b.i.d. on days 1–3 and mitoxantrone 12 mg/ m2 IV qd on days 3–5.
Büchner T et al.: J Clin Oncol 2009, 27: 61–9; with the kind permission of the American Society of Clinical Oncology
Krug U et al.: Lancet 2010; 376: 2000–9; with the kind permission of Elsevier Ltd.
Mandelli F et al.: Leukemia 2003; 17: 1085–90; with the kind permission of the Nature Publishing Group
Key Messages.
For older patients with acute myeloid leukemia, the choice of the suitable treatments is of utmost importance.
Treatment is chosen on the basis of risk factors that are easy to assess, which are derived from a standard diagnostic evaluation, specific prognostic scores, and a geriatric assessment.
Selected older patients are suitable candidates for intensive chemotherapy of the same type offered to younger patients, possibly including allogeneic stem-cell transplantation with reduced-dose conditioning treatment.
Certain groups of patients who are not candidates for intensive chemotherapy can be treated non-intensively with chemotherapeutic drugs such as azacitidine or low-dose cytarabine.
For some older patients with AML, no good therapeutic options are available. Such patients have a very poor prognosis; they can be given purely supportive care, or they can be offered the chance to participate in a therapeutic trial of a new drug. The wishes of the well-informed patient are paramount.
eBOX. Addresses of Study Groups in Germany.
German AML Cooperative Group (AMLCG)
Munich Center
Prof. W. E. Hiddemann / PD Dr. K. Spiekermann
Medizinische Klinik und Poliklinik III
Klinikum der Universität München – Grosshadern
Marchioninistr. 15, D-81377 Munich, Germany
Tel.: +49 (0)89 7095-2551
sekrmed3@med.uni-muenchen.de
med3.klinikum.uni-muenchen.de
Regensburg Center
Prof. J. Braess
Krankenhaus Barmherzige Brüder
Prüfeninger Str. 86, D-93049 Regensburg, Germany
Tel.: +40 (0)941 369-2151
onkologie@barmherzige-regensburg.de
German-Austrian AML Study Group (AMLSG)
Ulm Center
Prof. Döhner / PD Dr. R.F. Schlenk
Klinik für Innere Medizin III
Universitätsklinikum Ulm
Albert-Einstein-Allee 23, D-89081 Ulm, Germany
Tel.: +49 (0)731 500-45911/-45912
aml.sekretariat@uniklinik-ulm.de
www.uni-ulm.de/onkologie/AMLSG/
Hanover Center
Prof. A. Ganser / Prof. J. Krauter
Klinik für Hämatologie, Hämostaseologie, Onkologie und Stammzelltransplantation
Medizinische Hochschule Hannover
Carl-Neuberg-Str. 1, D-30625 Hanover, Germany
Tel.: +49 (0)511 532-3020
haematologie.onkologie@mh-hannover.de
www.mh-hannover.de/250.html
Study Alliance Leukemia (SAL)
Dresden Center
Prof. G. Ehninger / Prof. M. Schaich / Dr. Ch. Röllig
Medizinische Klinik und Poliklinik I
Universitätsklinikum Carl Gustav Carus
Fetscherstr. 74, D-01307 Dresden, Germany
Tel.: +49 (0)351 458-4671
kerstin.wirth@uniklinikum-dresden.de
www.mk1dd.de
Münster Center
Prof. W.E. Berdel / Prof. C. Müller-Tidow / PD Dr. U. Krug
Medizinische Klinik und Poliklinik A
Universitätsklinikum Münster
Albert-Schweitzer-Str. 33, D-48149 Münster, Germany
Tel.: 0251 83-47587/-52995/-46012
berdsekr@uni-muenster.de; Claudia.Tuschen@ukmuenster.de
www.meda-muenster.de
Frankfurt am Main Center
Prof. H. Serve / Dr. B. Steffen / Dr. C. Brandts
Medizinische Klinik II
Klinikum der Johann Wolfgang Goethe-Universität
Theodor-Stern-Kai 7, D-60590 Frankfurt am Main, Germany
Tel.: +49 (0)69 6301-4634
Ute.Eilers@kgu.de
www.kgu.de
East German Study Group of Hematology and Oncology (OSHO):
Leipzig Center
Prof. D. Niederwieser
Abteilung Hämatologie, internistische Onkologie und Hämostaseologie
Universitätsklinikum Leipzig AöR
Johannisallee 32 A, D-04103 Leipzig, Germany
Tel.: +49 (0)341 97-13050
meyer.claudia@medizin.uni-leipzig.de
http://haemonko.uniklinikum-leipzig.de/
German AML Intergroup Study
Münster Center
Prof. Th. Büchner / PD Dr. U. Krug
Medizinische Klinik und Poliklinik A
Universitätsklinikum Münster
Albert-Schweitzer-Str. 33, D-48149 Münster, Germany
Tel.: +49 (0)251 83-47597/-46012
buechnr@uni-muenster.de, utz.krug@ukmuenster.de
www.meda-muenster.de/
Munich Center (Biometrics)
Prof. J. Hasford / Dr. M. Pfirrmann
IBE, Institut für Medizinische Informationsverarbeitung, Biometrie und Epidemiologie
Ludwig-Maximilian-Universität
Marchioninistr. 15, D-81377 Munich, Germany
Tel.: +49 (0)89 7095-7489
markus.pfirrmann@ibe.med.uni-muenchen.de
www.ibe.med.uni-muenchen.de/
Useful Links
German Competence Network for Acute and Chronic Leukemia:
www.kompetenznetz-leukaemie.de
Website for calculating the probabilities of complete remission and of treatment-related death in older patients treated with intensive chemotherapy:
www.AML-Score.org
Acknowledgments
Translated from the original German by Ethan Taub, M.D.
Footnotes
Conflict of interest statement
PD Dr. Krug has been a paid consultant for, and/or received reimbursement for meeting participation fees and costs of travel and lodging from, the following companies: MedA Pharma, Sunesis Pharmaceuticals, Amgen, Mundipharma, and Celgene. He has received lecture honoraria from MedA Pharma, Sunesis Pharmaceuticals, and Alexion. He has received payment from Celgene and Bristol Myers Squibb for preparing scientific continuing education events, as well as from Clavis Pharma, Genzyme and Glaxo Smith Kline for performing clinical trials on behalf of these companies. He has also received money from Boehringer Ingelheim for a research project that has been initiated.
Prof. Berdel has been a paid consultant for, and/or received reimbursement for meeting participation fees and costs of travel and lodging from, the following companies: Wyeth, Amgen, Novartis, Celgene, and Roche. He has received lecture honoraria from Physicians World Thomson and Merck-Deutschland. He has been paid by Wyeth for performing clinical trials on its behalf. He has also received money from Wyeth, Novartis, Roche, and Amgen for a research project that has been initiated.
Prof. Müller Tidow is the holder of a patent on delocalization molecules for tumor treatment. He has received reimbursement for participation fees for meetings/educational events and costs of travel and lodging from Celgene, Bayer, and Novartis and lecture honoraria from Celgene, Pfizer, Novartis, Bayer, and Roche. He has received third-party funding for the performance of clinical trials on behalf of Boehringer Ingelheim, Ambit, and Novartis. He has also received money from Celgene, Amgen, and Novartis for a research project that has been initiated.
Prof. Büchner has been a paid consultant for Meda, from which he has also received reimbursement for meeting participation fees and costs of travel and lodging, as well as payment for preparing scientific continuing education events.
References
- 1.National Cancer Institute. Acute myeloid leukemia. http://www.seer.cancer.gov/statfacts/html/amyl.htm?statfacts_page=amyl.html&x=15&y=17.
- 2.Estey E. AML in older patients: are we making progress? Best Pract Res Clin Haematol. 2009;22:529–536. doi: 10.1016/j.beha.2009.08.007. [DOI] [PubMed] [Google Scholar]
- 3.Büchner T, Berdel WE, Haferlach C, Haferlach T, Schnittger S, Müller-Tidow C, et al. Age-related risk profile and chemotherapy dose response in acute myeloid leukemia: a study by the German Acute Myeloid Leukemia Cooperative Group. J Clin Oncol. 2009;27:61–69. doi: 10.1200/JCO.2007.15.4245. [DOI] [PubMed] [Google Scholar]
- 4.Döhner H, Estey EH, Amadori S, Appelbaum FR, Büchner T, Burnett AK, et al. Diagnosis and management of acute myeloid leukemia in adults: recommendations from an international expert panel, on behalf of the European LeukemiaNet. Blood. 2010;115:453–474. doi: 10.1182/blood-2009-07-235358. [DOI] [PubMed] [Google Scholar]
- 5.Estey EH. General approach to and perspectives on clinical research in older patients with newly diagnosed acute myeloid leukemia. Seminars in Hematology. 2006;43:89–95. doi: 10.1053/j.seminhematol.2006.01.002. [DOI] [PubMed] [Google Scholar]
- 6.Löwenberg B, Zittoun R, Kerkhofs H, et al. On the value of intensive remission-induction chemotherapy in elderly patients of 65+ years with acute myeloid leukemia: a randomized phase III study of the European Organization for Research and Treatment of Cancer Leukemia Group. J Clin Oncol. 1989;7:1268–1274. doi: 10.1200/JCO.1989.7.9.1268. [DOI] [PubMed] [Google Scholar]
- 7.Appelbaum FR, Gundacker H, Head DR, et al. Age and acute myeloid leukemia. Blood. 2006;107:3481–3485. doi: 10.1182/blood-2005-09-3724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Giles FJ, Borthakur G, Ravandi F, et al. The haematopoietic cell transplantation comorbidity index score is predictive of early death and survival in patients over 60 years of age receiving induction therapy for acute myeloid leukaemia. Br J Haematol. 2007;136:624–627. doi: 10.1111/j.1365-2141.2006.06476.x. [DOI] [PubMed] [Google Scholar]
- 9.Krug U, Röllig C, Koschmieder A, et al. Complete remission and early death after intensive chemotherapy in patients aged 60 years or older with acute myeloid leukaemia: a web-based application for prediction of outcomes. Lancet. 2010;376:2000–2008. doi: 10.1016/S0140-6736(10)62105-8. [DOI] [PubMed] [Google Scholar]
- 10.Deschler B, Ihorst G, Platzbecker U, Germing U, Lübbert M. Development of a Frailty Score for Older Patients with Myelodysplastic Syndromes and Acute Myeloid Leukemia. Blood. 2009;114 [Google Scholar]
- 11.Ziogas DC, Voulgarelis M, Zintzaras E. A Network Meta-analysis of Randomized Controlled Trials of Induction Treatments in Acute Myeloid Leukemia in the Elderly. Clin Ther. 2011;33:254–279. doi: 10.1016/j.clinthera.2011.04.004. [DOI] [PubMed] [Google Scholar]
- 12.Büchner T, Döhner H, Ehninger G, Ganser A, Niederwieser D, Hasford J. Cross-trial networking in AML: a step forward rather than corner cutting. Leuk Res. 2004;28:649–650. doi: 10.1016/j.leukres.2003.10.031. [DOI] [PubMed] [Google Scholar]
- 13.Büchner T, Urbanitz D, Hiddemann W, et al. Intensified induction and consolidation with or without maintenance chemotherapy for acute myeloid leukemia (AML): two multicenter studies of the German AML Cooperative Group. J Clin Oncol. 1985;3:1583–1589. doi: 10.1200/JCO.1985.3.12.1583. [DOI] [PubMed] [Google Scholar]
- 14.Mayer RJ, Davis RB, Schiffer CA, et al. Intensive postremission chemotherapy in adults with acute myeloid leukemia. Cancer and Leukemia Group B. N Engl J Med. 1994;331:896–903. doi: 10.1056/NEJM199410063311402. [DOI] [PubMed] [Google Scholar]
- 15.Cheson BD, Bennett JM, Kopecky KJ, et al. Revised recommendations of the International Working Group for Diagnosis, Standardization of Response Criteria, Treatment Outcomes, and Reporting Standards for Therapeutic Trials in Acute Myeloid Leukemia. J Clin Oncol. 2003;21:4642–4649. doi: 10.1200/JCO.2003.04.036. [DOI] [PubMed] [Google Scholar]
- 16.Cornelissen JJ, van Putten WL, Verdonck LF, et al. Results of a HOVON/SAKK donor versus no-donor analysis of myeloablative HLA-identical sibling stem cell transplantation in first remission acute myeloid leukemia in young and middle-aged adults: benefits for whom? Blood. 2007;109:3658–3866. doi: 10.1182/blood-2006-06-025627. [DOI] [PubMed] [Google Scholar]
- 17.Zander AR, Bacher U, Finke J. Allogeneic Stem Cell Transplantation in Acute Myeloid Leukemia. Dtsch Arztebl Int. 2008;105:663–669. doi: 10.3238/arztebl.2008.0663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Krug U, Lübbert M, Büchner T. Maintenance therapy in acute myeloid leukemia revisited: will new agents rekindle an old interest? Curr Opin Hematol. 2010;17:85–90. doi: 10.1097/MOH.0b013e3283366bf4. [DOI] [PubMed] [Google Scholar]
- 19.Fenaux P, Mufti GJ, Hellström-Lindberg E, et al. Azacitidine prolongs overall survival compared with conventional care regimes in elderly patients with low bone marrow blast count acute myeloid leukemia. J Clin Oncol. 2010;28:562–569. doi: 10.1200/JCO.2009.23.8329. [DOI] [PubMed] [Google Scholar]
- 20.Thomas X, Dmoszynska A, Wierzbowska A, et al. Results from a randomized phase III trial of decitabine versus supportive care or low-dose cytarabine for the treatment of older patients with newly diagnosed AML. J Clin Oncol. 2011;29 doi: 10.1200/JCO.2011.38.9429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Burnett AK, Milligan D, Prentice AG, et al. A comparison of low-dose cytarabine and hydroxyurea with or without all-trans retinoic acid for acute myeloid leukemia and high-risk myelodysplastic syndrome in patients not considered fit for intensive treatment. Cancer. 2007;109:1114–1124. doi: 10.1002/cncr.22496. [DOI] [PubMed] [Google Scholar]
- 22.Tsimberidou AM, Kantarjian H, Keating MJ, Estey E. Optimizing treatment for elderly patients with acute promyelocytic leukemia: is it time to replace chemotherapy with all-trans retinoic acid and arsenic trioxide? Leuk Lymphoma. 2006;47:2282–2288. doi: 10.1080/10428190600807178. [DOI] [PubMed] [Google Scholar]
- 23.Sanz MA, Grimwade D, Tallman MS, et al. Management of acute promyelocytic leukemia: recommendations from an expert panel on behalf of the European LeukemiaNet. Blood. 2009;113:1875–1891. doi: 10.1182/blood-2008-04-150250. [DOI] [PubMed] [Google Scholar]
- 24.Degos L, Dombret H, Chomienne C, et al. All-trans-retinoic acid as a differentiating agent in the treatment of acute promyelocytic leukemia. Blood. 1995;85:2643–2653. [PubMed] [Google Scholar]
- 25.Grimwade D, Lo Coco F. Acute promyelocytic leukemia: a model for the role of molecular diagnosis and residual disease monitoring in directing treatment approach in acute myeloid leukemia. Leukemia. 2002;16:1959–1973. doi: 10.1038/sj.leu.2402721. [DOI] [PubMed] [Google Scholar]
- e1.Kolitz JE, George SL, Marcucci G, et al. P-glycoprotein inhibition using valspodar (PSC-833) does not improve outcomes for patients under age 60 years with newly diagnosed acute myeloid leukemia: Cancer and Leukemia Group B study 19808. Blood. 2010;116:1413–1421. doi: 10.1182/blood-2009-07-229492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e2.Fernandez HF, Sun Z, Yao X, Litzow MR, et al. Anthracycline dose intensification in acute myeloid leukemia. N Engl J Med. 2009;361:1249–1259. doi: 10.1056/NEJMoa0904544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e3.Mandelli F, Vignetti M, Suciu S, et al. Daunorubicin versus mitoxantrone versus idarubicin as induction and consolidation chemotherapy for adults with acute myeloid leukemia: the EORTC and GIMEMA Groups Study AML-10. J Clin Oncol. 2009;27:5397–5403. doi: 10.1200/JCO.2008.20.6490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e4.Ohtake S, Miyawaki S, Fujita H, et al. Randomized study of induction therapy comparing standard-dose idarubicin with high-dose daunorubicin in adult patients with previously untreated acute myeloid leukemia: the JALSG AML201 Study. Blood. 2011;117:2358–2365. doi: 10.1182/blood-2010-03-273243. [DOI] [PubMed] [Google Scholar]
- e5.Burnett AK, Hills RK, Milligan DW, et al. Attempts to optimize induction and consolidation treatment in acute myeloid leukemia: results of the MRC AML12 trial. J Clin Oncol. 2010;28:586–595. doi: 10.1200/JCO.2009.22.9088. [DOI] [PubMed] [Google Scholar]
- e6.Thomas X, Elhamri M, Raffoux E, et al. Comparison of high-dose cytarabine and timed-sequential chemotherapy as consolidation for younger adults with AML in first remission: the ALFA-9802 study. Blood. 2011;118:1754–1762. doi: 10.1182/blood-2011-04-349258. [DOI] [PubMed] [Google Scholar]
- e7.Schaich M, Röllig C, Soucek S, et al. Cytarabine Dose of 36 g/m2 Compared With 12 g/m2 Within First Consolidation in Acute Myeloid Leukemia: Results of Patients Enrolled Onto the Prospective Randomized AML96 Study. J Clin Oncol. 2011;29:2696–2702. doi: 10.1200/JCO.2010.33.7303. [DOI] [PubMed] [Google Scholar]
- e8.Löwenberg B, Pabst T, Vellenga E, et al. Cytarabine dose for acute myeloid leukemia. N Engl J Med. 2011;364:1027–1036. doi: 10.1056/NEJMoa1010222. [DOI] [PubMed] [Google Scholar]
- e9.Oken MM, Creech RH, Tormey DC, et al. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol. 1982;5:649–655. [PubMed] [Google Scholar]
- e10.Sorror ML, Maris MB, Storb R, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood. 2005;106:2912–2919. doi: 10.1182/blood-2005-05-2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e11.Löwenberg B, Beck J, Graux C, et al. Gemtuzumab ozogamicin as postremission treatment in AML at 60 years of age or more: results of a multicenter phase 3 study. Blood. 2010;115:2586–2591. doi: 10.1182/blood-2009-10-246470. [DOI] [PubMed] [Google Scholar]
- e12.Deutsche Gesellschaft für Hämatologie und Onkologie. Leitlinien der DGHO für die Diagnostik und Therapie der Akuten Myeloischen Leukämie (AML) www.dgho-onkopedia.de/onkopedia/leitlinien/aml.
- e13.National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Acute Myeloid Leukemia. www.nccn.org/professionals/physician_gls/pdf/aml.pdf.
- e14.Gooley TA, Chien JW, Pergam SA, et al. Reduced mortality after allogeneic hematopoietic-cell transplantation. N Engl J Med. 2010;363:2091–2101. doi: 10.1056/NEJMoa1004383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e15.Wallen H, Gooley TA, Deeg HJ, et al. Ablative Allogeneic Hematopoietic Cell Transplantation in Adults 60 Years of Age and Older. J Clin Oncol. 2005;23:3439–3446. doi: 10.1200/JCO.2005.05.694. [DOI] [PubMed] [Google Scholar]
- e16.Farag SS, Maharry K, Zhang MJ, et al. Comparison of reduced intensity hematopoietic cell transplantation with chemotherapy in patients aged 60-70 years with acute myeloid leukemia in first remission. Biol Blood Marrow Transplant. 2011;17:1796–1803. doi: 10.1016/j.bbmt.2011.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- e17.Deutsche Krebsgesellschaft. Kurzgefasste interdisziplinäre Leitlinien 2008. Leitlinie O6, Infektionen bei Neutropenie. www.krebsgesellschaft.de/download/ll_o_06.pdf.
- e18.Shen ZX, Chen GQ, Ni JH, et al. Use of arsenic trioxide (As2O3) in the treatment of acute promyelocytic leukemia (APL): II. Clinical efficacy and pharmacokinetics in relapsed patients. Blood. 1997;89:3354–3360. [PubMed] [Google Scholar]
- e19.Soignet SL, Maslak P, Wang ZG, et al. Complete remission after treatment of acute promyelocytic leukemia with arsenic trioxide. N Engl J Med. 1998;339:1341–1348. doi: 10.1056/NEJM199811053391901. [DOI] [PubMed] [Google Scholar]
- e20.Niu C, Yan H, Yu T, et al. Studies on treatment of acute promyelocytic leukemia with arsenic trioxide: remission induction, follow-up, and molecular monitoring in 11 newly diagnosed and 47 relapsed acute promyelocytic leukemia patients. Blood. 1999;94:3315–3324. [PubMed] [Google Scholar]
- e21.Soignet SL, Frankel SR, Douer D, et al. United States multicenter study of arsenic trioxide in relapsed acute promyelocytic leukemia. J Clin Oncol. 2001;19:3852–3860. doi: 10.1200/JCO.2001.19.18.3852. [DOI] [PubMed] [Google Scholar]
- e22.Au WY, Lie AK, Chim CS, et al. Arsenic trioxide in comparison with chemotherapy and bone marrow transplantation for the treatment of relapsed acute promyelocytic leukaemia. Ann Oncol. 2003;14:752–757. doi: 10.1093/annonc/mdg208. [DOI] [PubMed] [Google Scholar]
- e23.Lazo G, Kantarjian H, Estey E, Thomas D, O’Brien S, Cortes J. Use of arsenic trioxide (As2O3) in the treatment of patients with acute promyelocytic leukemia: the M. D. Anderson experience. Cancer. 2003;97:2218–2224. doi: 10.1002/cncr.11314. [DOI] [PubMed] [Google Scholar]
- e24.Raffoux E, Rousselot P, Poupon J, et al. Combined treatment with arsenic trioxide and all-trans-retinoic acid in patients with relapsed acute promyelocytic leukemia. J Clin Oncol. 2003;21:2326–2334. doi: 10.1200/JCO.2003.01.149. [DOI] [PubMed] [Google Scholar]
- e25.Shigeno K, Naito K, Sahara N, et al. Arsenic trioxide therapy in relapsed or refractory Japanese patients with acute promyelocytic leukemia: updated outcomes of the phase II study and postremission therapies. Int J Hematol. 2005;82:224–229. doi: 10.1532/IJH97.05044. [DOI] [PubMed] [Google Scholar]