Abstract
Neuroinflammation and neuronal degeneration observed in Parkinson’s disease (PD) has been attributed in part to glial-mediated events. Increased expression of proinflammatory cytokines and abnormal accumulation of the neuronal protein, α-synuclein in the brain are also characteristic of PD. While increasing evidence suggests that astrocytes contribute to neuroinflammation and dopaminergic neuronal degeneration associated with PD, there remains much to learn about these astroglial-mediated events. Therefore, we investigated the in vitro effects of interleukin-1β (IL-1β) and α-synuclein on astroglial expression of interferon-γ inducible protein-10 (CXCL10), a proinflammatory and neurotoxic chemokine. IL-1β-induced CXCL10 protein expression was potentiated by co-exposure to α-synuclein. α-Synuclein did not significantly affect IL-1β-induced CXCL10 mRNA expression, but did mediate increased CXCL10 mRNA stability, which may explain, in part, the increased levels of secreted CXCL10 protein. Future investigations are warranted to more fully define the mechanism by which α-synuclein enhances IL-1β-induced astroglial CXCL10 expression. These findings highlight the importance of α-synuclein in modulating inflammatory events in astroglia. These events may be particularly relevant to the pathology of CNS disorders involving α-synuclein accumulation, including PD and HIV-1 associated dementia.
Keywords: Parkinson’s disease, alpha-synuclein, astrocyte, neuroinflammation, chemokine, NF-κB
Introduction
Increasing evidence suggests that neuroinflammation contributes to neuronal loss in Parkinson’s disease (PD) [11, 15]. Among the indicators of neuroinflammation in PD are glial activation and increased expression of inflammatory molecules in astroglia and microglia [10, 17, 30]. Microglia have been the predominant cell type implicated in neuroinflammation and neuronal degeneration in PD [12, 27]. However, astrocytes also contribute to dopaminergic neuronal degeneration [13, 34, 50]. One of the primary inflammatory molecules implicated in PD is interleukin-1β (IL-1β), which is elevated in the striatum and cerebrospinal fluid (CSF) of PD patients [3, 32]. Additionally, IL-1β mediates cell death in dopaminergic neurons and causes PD motor disabilities, further suggesting the involvement of IL-1β in PD [1, 21, 27].
The neuronal protein, α-synuclein is also implicated in the pathology of PD [45]. α-Synuclein is a small acidic protein (140 amino acid residues) predominately localized in presynaptic terminals in the CNS and loosely associates with synaptic vesicles [33]. Physiologically, α-synuclein seems to be involved in synaptic functions including neuronal plasticity and neurotransmission, particularly in dopaminergic neurons [4, 44]. Abnormal deposition of α-synuclein is involved in the neuropathogenesis of PD [2, 45]. Furthermore, α-synuclein deposition in the CNS is also elevated in multiple sclerosis [28] and HIV-1 infected patients [19]. An apparent prerequisite of α-synuclein neuropathy is its oligomerization into soluble protofibrils followed by their coalescence into insoluble fibrils, and accumulation into Lewy bodies, the neuropathologic hallmark of PD [25]. Extracellular α-synuclein can activate glia, resulting in production of reactive oxygen species, cytokines and chemokines, which contribute to neuroinflammation and neurodegeneration of dopaminergic neurons [20, 48, 51, 52]. Neuronal derived α-synuclein accumulated in astrocytes results in the induction of proinflammatory mediators, including CXCL10 [26]. Importantly, astroglial activation and CXCL10 have been implicated in numerous neuropathologies, including those involving abnormal α-synuclein deposition, such as PD [24, 26], multiple sclerosis [41], and HIV dementia [46, 47]. Yet, to date there is limited information on the direct effects of α-synuclein on glial chemokine expression. To our knowledge, this is the first report of α-synuclein effects on CXCL10 expression in human astroglial cells. We assessed the effects of α-synuclein and IL-1β on CXCL10 expression in human astroglial cells. CXCL10 expression in astroglial cells is dependent upon activation of the transcription factor NF-κB [7]; therefore, we also assessed the effects of these two inflammatory mediators on NF-κB activation.
Material and methods
Human A172 astroglial cells (American Type Culture Collection, Manassas, VA) were maintained in Dulbecco's modified Eagle's medium containing 2 mM L-glutamine, 10% fetal bovine serum, 1% nonessential amino acids, 50 U/ml penicillin, 50 µg/ml streptomycin and 2 µg/ml amphotericin B [7]. To induce CXCL10 expression, growth medium in astroglial cultures was replaced with serum-free medium containing human recombinant IL-1β (250 pg/ml). The duration of cytokine exposure was 24 h for CXCL10 protein expression, 2–48 h for CXCL10 mRNA expression and 2–180 min for NF-κB activation. Purified human α-synuclein (r-Peptide, Bogart, GA) was agitated (“aged”) in phosphate buffered saline (10 ng/ml) for 14 d at 37°C to form an inflammatory-inducing conformation, followed by dilution to 480 pg/ml in serum-free medium and exposure to cells.
A standard dual-antibody solid phase immunoassay (ELISA Development Kit, Peprotech) was used for quantitation of CXCL10 secreted into cell culture supernatants as previously reported [7]. Absorbance of the end-product was read at 450 nm (λ correction set at 650 nm) on a BIO-TEK HT spectrophotometer. Total cell protein/well was determined using the bicinchoninic acid protein assay as previously described [8] in order to normalize data.
NF-κB activation, as determined by increased levels of active (DNA-binding) forms of NF-κB p65 and p50 in the nucleus, was assessed using the p65 and p50 Transcription Factor kits (Thermo Scientific, Rockford, IL) as previously described [43]. Nuclear protein was obtained using the Nuclear Extract Kit (version C4; Active Motif, Carlsbad, CA) and luminescence of the labeled NF-κB-DNA product was then measured.
Total RNA was isolated with TRIzol reagent (Invitrogen, Carlsbad, CA) and CXCL10 mRNA measured by real-time PCR using the SYBR Green detection method using the basic protocol previously described [6]. The RT-PCR primer pair sets were obtained from Invitrogen and amplification from first strand cDNA was performed as instructed by Invitrogen. The primer sequences for CXCL10 were AACCTCCAGTCTCAGCACCATGAA (forward) and AGGTACAGCGTAAGGTTCTAGAGAG (reverse); the primer pairs for GAPDH were GAGTCAACGGATTTGGTCGT (forward) and TTGATTTTGGAGGGATCTCG (reverse). PCR efficiency for GAPDH was similar to that of CXCL10 (>95%) and GAPDH was not affected by treatment. Assessment of CXCL10 mRNA stability was performed using the basic protocol previously described [29]. Briefly, cells were exposed to IL-1β (+/− α-synuclein) for 2.5 h, followed by the removal of media and the addition of fresh serum-free media with or without 10 µg/ml actinomycin D. Total RNA was collected after 3, 10, 30, 60, 90, and 180 min and CXCL10 mRNA levels determined as described above. Relative quantification of gene expression was evaluated using the comparative cycle threshold (CT) method [14] as previously described [6]. Expression of CXCL10 mRNA was normalized to the expression of the endogenous control, GAPDH mRNA. Relative expression was calculated using the comparative ΔΔCT method and fold change calculated using 2−ΔΔCT.
GraphPad Prism version 4 (Graph Pad Software Inc, San Diego, CA) and Statistica version 6 (StatSoft, Inc., Tulsa, OK) were used for statistical analyses. Results from independent experiments were combined for presentation as the mean ± S.E.M. One-way ANOVA, two-way ANOVA and Student-Neuman-Kuels-Keuls (SNK) pair-wise comparisons were used for parametric analyses. p < 0.05 was considered statistically significant.
Results
Constitutive expression of CXCL10 protein was very low to negligible (Fig. 1) and 24 h exposure to α-synuclein (480 pg/ml) did not induce expression of this chemokine (data not shown). However, IL-1β (250 pg/ml) induced CXCL10 protein expression and co-exposure with α-synuclein (480 pg/ml) resulted in a further increase in CXCL10 expression (Fig. 1). IL-1β-induced CXCL10 expression is NF-κB-dependent; therefore, we predicted that α-synuclein would potentiate CXCL10 expression pre-transcriptionally through modulation of NF-κB activity. Treatment of A172 cells with IL-1β increased nuclear levels of p65 and p50, which peaked at 60 min. post-stimulation (Fig. 2). However, co-exposure with α-synuclein did not alter IL-1β-induced NF-κB activation (Fig. 2).
Figure 1. α-Synuclein potentiated IL-1β-induced CXCL10 protein expression in human A172 astroglial cells.
Cells were either unstimulated or exposed to IL-1β (250 pg/ml) in the presence or absence of α-synuclein (480 pg/ml) in serum-free medium for 24 h. CXCL10 protein in the media was quantitated by ELISA. Data represent the mean ± S.E.M. (n = 3–8). Significant differences were determined by one-way ANOVA with SNK pair-wise comparisons. ***p < 0.001 vs IL-1β, **p < 0.01 vs IL-1β.
Figure 2. α-Synuclein did not alter IL-1β-induced NF-κB activation in human A172 astroglial cells.
Cells were exposed to IL-1β (250 pg/ml) in the presence or absence of α-synuclein (480 pg/ml) in serum-free medium for 2–120 minutes. The levels of active (A) NF-κB p65 and (B) p50 in the nucleus were determined using the Transcription Factor kits (Thermo Scientific, Rockford, IL). Luminescence of the labeled NF-κB-DNA product was then measured. Data represent the mean ± S.E.M. (n = 2–4). Two-way ANOVA (treatment × time) with SNK pair-wise comparisons indicated a significant effect of time (p65: **p < 0.009 vs. 2 min, #p < 0.002 vs. 60 min; p50: **p < 0.002 vs. 2 min, *p < 0.05 vs. 2 min, #p < 0.03 vs. 60 min), whereas, α-synuclein did not significantly (p > 0.89) affect IL-1β-induced increases in nuclear levels of NF-κB.
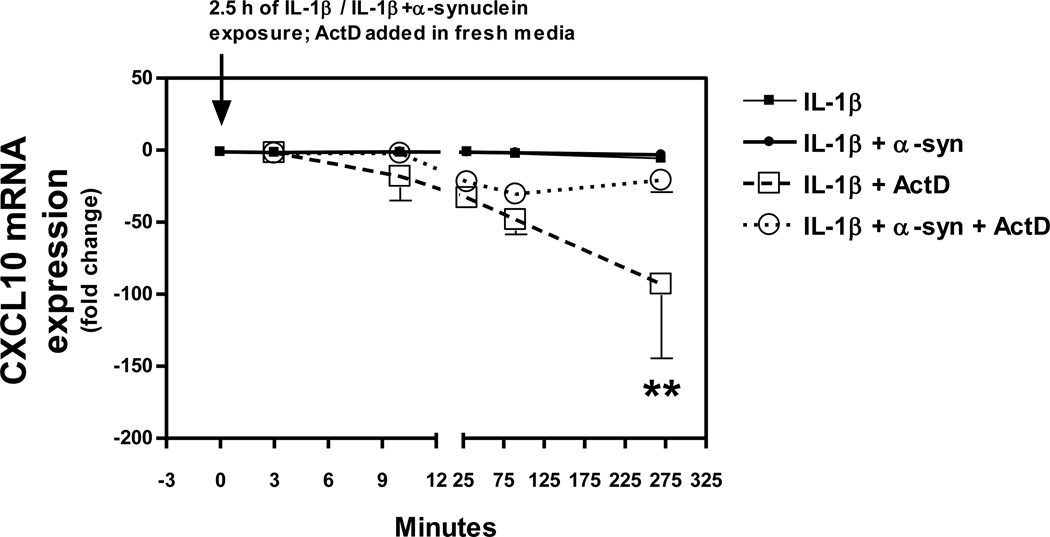
IL-1β-induced CXCL10 mRNA expression peaked at 8 h and returned to baseline by 16 h (Fig. 3). Co-exposure of cells to IL-1β and α-synulcein did not alter levels of CXCL10 mRNA expression (Fig. 4). However, the stability of CXCL10 mRNA was enhanced in cells co-exposed to IL-1β and α-synuclein compared to those treated with IL-1β alone (Fig. 4).
Figure 3. α-Synuclein did not affect IL-1β-stimulated CXCL10 mRNA expression in human A172 astroglial cells.
Cells were exposed to IL-1β (250 pg/ml) in the presence or absence of α-synuclein (480 pg/ml) in serum-free medium for 2–48 h. CXCL10 mRNA levels were assessed using real time RT-PCR. Data were normalized to GAPDH mRNA within each independent experiment and expressed as fold change (compared to IL-1β-stimulated group at 2 h). Data represent the mean ± S.E.M. (n = 7–15). Two-way ANOVA (treatment × time) with SNK pair-wise comparisons indicated a significant effect of time (*p < 0.04, vs. 2 min), whereas, α-synuclein did not significantly (p = 0.72) affect IL-1β-induced increases in CXCL10 mRNA expression.
Figure 4. Co-exposure to α-synuclein results in enhanced CXCL10 mRNA stability in IL-1β-stimulated human A172 astroglial cells.
Cells were exposed to IL-1β (250 pg/ml) in the presence or absence of α-synuclein (480 pg/ml) in serum-free medium for 2.5 h. Cultures were replenished with media alone or media containing 10 µg/ml actinomycin D (ActD). The time-course (3–270 min) of CXCL10 and GAPDH mRNA expression was assessed using quantitative real time RT-PCR. Data were normalized to GAPDH within each independent experiment and expressed as fold change (relative to IL-1β treated cells at time 0). Data represent the mean ± S.E.M. (n = 2–6). Two-way ANOVA (treatment × time) indicated that treatment, time and the interaction were each highly significant (p < 0.001). **p < 0.01 vs. IL-1β + α-Syn + ActD as determined by SNK pair-wise comparisons.
Discussion
α-Synuclein is implicated in the pathology of PD and may also contribute to other neuropathologies [5, 11, 19, 28, 42]. A number of in vivo and in vitro studies have reported the toxic actions of α-synuclein on oligodendroglial and neuronal cells [11, 23, 51]. For example, aggregated α-synuclein at concentrations similar to the concentrations used in the current study resulted in dopaminergic neurotoxicity in rat primary mesencephalic neuronal cultures [51]. α-Synuclein can also induce glial degeneration [22, 39], yet, under the experimental conditions of the present study, viability of A172 astroglial cells was not affected by α-synuclein, as determined by the MTT reduction assay (data not shown).
PD, MS and HIV-1 associated dementia are all associated with neuroinflammation as evidenced by increased levels of proinflammatory cytokines and chemokines [24, 32, 36, 41]. Astrogliosis is also evident in these neuropathologies [16, 31] and astrocytes are a major source of chemokine production in the CNS [9]. Together, these findings underscore the importance in gaining a better understanding of the effects of α-synuclein on astrocytes in the presence of on-going inflammation.
IL-1β is known to induce CXCL10 in human astroglial cells, however, while 10 ng/ml IL-1β is a common concentration used [35, 38], we have demonstrated that a relatively low concentration of IL-1β (250 pg/ml) induces CXCL10. Furthermore, this is the first report on the combined effects of α-synuclein and IL-β on CXCL10 expression in human astroglial cells. Cytokine-induced CXCL10 expression is NF-κB-dependent [7] and α-synuclein induces NF-κB activation [20]. Therefore, we predicted that the potentiation of IL-1β-induced CXCL10 expression by α-synuclein would involve modulation of NF-κB activation. However, our findings suggested that the potentiation of IL-1β-induced CXCL10 expression by α-synuclein was not due to enhanced activation of NF-κB.
Post-transcriptional control of gene expression through mRNA transcript stability is important in the regulation of inflammatory genes, including those encoding cytokines and chemokines [40]. CXCL10, in particular, has a transcript half-life of about 30 min under resting conditions [37]. To determine the effects of α-synuclein on CXCL10 mRNA stability we utilized the addition of a de novo transcription inhibitor, actinomycin D, an approach similar to that used by others [49]. Interestingly, we found that CXCL10 mRNA degradation was delayed in cells co-exposed to α-synuclein and IL-1β, relative to cells treated with IL-1β alone. While these findings indicate that α-synuclein facilitates the stability of this CXCL10 mRNA in A172 cells, the molecular mechanism involved remains to be defined. Among the proteins reported to interact with mRNA transcripts to regulate mRNA degradation, only nucleoli has been shown to interact with α-synuclein [18]. Yet, further investigation into the direct and indirect effects of α-synuclein on CXCL10 mRNA transcripts is warranted.
Gaining further insights into the mechanism by which α-synuclein potentiates IL-1β-induced chemokine expression in astroglia is expected to broaden our understanding of the cellular events in certain neuropathologies. Additionally, these findings may foster the development of therapeutic strategies to manipulate neuroinflammatory conditions.
Highlights.
A low concentration of IL-1β (250 pg/ml) induces NF-κB activation and CXCL10 expression in human astroglial cells.
Co-exposure to α-synuclein potentiates IL-1β -induced CXCL10 protein expression.
α-Synuclein does not alter IL-1β -induced NF-κB activation.
α-Synuclein mediates an increase in CXCL10 mRNA stability, which may account for increased CXCL10 protein expression.
Acknowledgements
This project was supported in part by OSU-CHS Intramural funding (RLD, JTC), NIH Grant NS062664 (RLD) and NIH Grant HD48462 (JTC).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Barnum CJ, Eskow KL, Dupre K, Blandino P, Jr, Deak T, Bishop C. Exogenous corticosterone reduces L-DOPA-induced dyskinesia in the hemi-parkinsonian rat: role for interleukin-1beta. Neuroscience. 2008;156:30–41. doi: 10.1016/j.neuroscience.2008.07.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Beyer K, Ispierto L, Latorre P, Tolosa E, Ariza A. Alpha- and beta-synuclein expression in Parkinson disease with and without dementia. J Neurol Sci. 2011 doi: 10.1016/j.jns.2011.05.049. [DOI] [PubMed] [Google Scholar]
- 3.Blum-Degen D, Muller T, Kuhn W, Gerlach M, Przuntek H, Riederer P. Interleukin-1 beta and interleukin-6 are elevated in the cerebrospinal fluid of Alzheimer's and de novo Parkinson's disease patients. Neurosci Lett. 1995;202:17–20. doi: 10.1016/0304-3940(95)12192-7. [DOI] [PubMed] [Google Scholar]
- 4.Clayton DF, George JM. The synucleins: a family of proteins involved in synaptic function, plasticity, neurodegeneration and disease. Trends Neurosci. 1998;21:249–254. doi: 10.1016/s0166-2236(97)01213-7. [DOI] [PubMed] [Google Scholar]
- 5.Croisier E, Graeber MB. Glial degeneration and reactive gliosis in alpha-synucleinopathies: the emerging concept of primary gliodegeneration. Acta Neuropathol. 2006;112:517–530. doi: 10.1007/s00401-006-0119-z. [DOI] [PubMed] [Google Scholar]
- 6.Davis RL, Buck DJ, Saffarian N, Mohan S, DeSilva U, Fernando SC, Stevens CW. Beta-funaltrexamine inhibits inducible nitric-oxide synthase expression in human astroglial cells. J Neuroimmune Pharmacol. 2008;3:150–153. doi: 10.1007/s11481-008-9102-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Davis RL, Buck DJ, Saffarian N, Stevens CW. The opioid antagonist, beta-funaltrexamine, inhibits chemokine expression in human astroglial cells. J Neuroimmunol. 2007;186:141–149. doi: 10.1016/j.jneuroim.2007.03.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Davis RL, Dertien J, Syapin PJ. Ethanol-induced modulation of inducible nitric-oxide synthase activity in human A172 astrocytoma cells. Alcohol Clin Exp Res. 2002;26:1404–1411. doi: 10.1097/01.ALC.0000030841.92766.80. [DOI] [PubMed] [Google Scholar]
- 9.Dong Y, Benveniste EN. Immune function of astrocytes. Glia. 2001;36:180–190. doi: 10.1002/glia.1107. [DOI] [PubMed] [Google Scholar]
- 10.Forno LS, DeLanney LE, Irwin I, Di Monte D, Langston JW. Astrocytes and Parkinson's disease. Prog Brain Res. 1992;94:429–436. doi: 10.1016/s0079-6123(08)61770-7. [DOI] [PubMed] [Google Scholar]
- 11.Gao HM, Zhang F, Zhou H, Kam W, Wilson B, Hong JS. Neuroinflammation and alpha-synuclein dysfunction potentiate each other, driving chronic progression of neurodegeneration in a mouse model of Parkinson's disease. Environ Health Perspect. 2010;119:807–814. doi: 10.1289/ehp.1003013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gerhard A, Pavese N, Hotton G, Turkheimer F, Es M, Hammers A, Eggert K, Oertel W, Banati RB, Brooks DJ. In vivo imaging of microglial activation with [11C](R)-PK11195 PET in idiopathic Parkinson's disease. Neurobiol Dis. 2006;21:404–412. doi: 10.1016/j.nbd.2005.08.002. [DOI] [PubMed] [Google Scholar]
- 13.Henning J, Strauss U, Wree A, Gimsa J, Rolfs A, Benecke R, Gimsa U. Differential astroglial activation in 6-hydroxydopamine models of Parkinson's disease. Neurosci Res. 2008;62:246–253. doi: 10.1016/j.neures.2008.09.001. [DOI] [PubMed] [Google Scholar]
- 14.Hettinger AM, Allen MR, Zhang BR, Goad DW, Malayer JR, Geisert RD. Presence of the acute phase protein, bikunin, in the endometrium of gilts during estrous cycle and early pregnancy. Biol Reprod. 2001;65:507–513. doi: 10.1095/biolreprod65.2.507. [DOI] [PubMed] [Google Scholar]
- 15.Hirsch EC, Hunot S. Neuroinflammation in Parkinson's disease: a target for neuroprotection? Lancet Neurol. 2009;8:382–397. doi: 10.1016/S1474-4422(09)70062-6. [DOI] [PubMed] [Google Scholar]
- 16.Hirsch EC, Hunot S, Hartmann A. Neuroinflammatory processes in Parkinson's disease. Parkinsonism Relat Disord. 2005;11 Suppl 1:S9–S15. doi: 10.1016/j.parkreldis.2004.10.013. [DOI] [PubMed] [Google Scholar]
- 17.Hunot S, Dugas N, Faucheux B, Hartmann A, Tardieu M, Debre P, Agid Y, Dugas B, Hirsch EC. FcepsilonRII/CD23 is expressed in Parkinson's disease and induces, in vitro, production of nitric oxide and tumor necrosis factor-alpha in glial cells. Journal of Neuroscience. 1999;19:3440–3447. doi: 10.1523/JNEUROSCI.19-09-03440.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jin J, Li GJ, Davis J, Zhu D, Wang Y, Pan C, Zhang J. Identification of novel proteins associated with both alpha-synuclein and DJ-1. Mol Cell Proteomics. 2007;6:845–859. doi: 10.1074/mcp.M600182-MCP200. [DOI] [PubMed] [Google Scholar]
- 19.Khanlou N, Moore DJ, Chana G, Cherner M, Lazzaretto D, Dawes S, Grant I, Masliah E, Everall IP. Increased frequency of alpha-synuclein in the substantia nigra in human immunodeficiency virus infection. J Neurovirol. 2009;15:131–138. doi: 10.1080/13550280802578075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Klegeris A, Giasson BI, Zhang H, Maguire J, Pelech S, McGeer PL. Alpha-synuclein and its disease-causing mutants induce ICAM-1 and IL-6 in human astrocytes and astrocytoma cells. Faseb J. 2006;20:2000–2008. doi: 10.1096/fj.06-6183com. [DOI] [PubMed] [Google Scholar]
- 21.Koprich JB, Reske-Nielsen C, Mithal P, Isacson O. Neuroinflammation mediated by IL-1beta increases susceptibility of dopamine neurons to degeneration in an animal model of Parkinson's disease. J Neuroinflammation. 2008;5:8. doi: 10.1186/1742-2094-5-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kosel S, Egensperger R, von Eitzen U, Mehraein P, Graeber MB. On the question of apoptosis in the parkinsonian substantia nigra. Acta Neuropathol. 1997;93:105–108. doi: 10.1007/s004010050590. [DOI] [PubMed] [Google Scholar]
- 23.Kragh CL, Lund LB, Febbraro F, Hansen HD, Gai WP, El-Agnaf O, Richter-Landsberg C, Jensen PH. {alpha}-Synuclein Aggregation and Ser-129 Phosphorylation-dependent Cell Death in Oligodendroglial Cells. J Biol Chem. 2009;284:10211–10222. doi: 10.1074/jbc.M809671200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lee HJ, Kim C, Lee SJ. Alpha-synuclein stimulation of astrocytes: Potential role for neuroinflammation and neuroprotection. Oxid Med Cell Longev. 2010;3:283–287. doi: 10.4161/oxim.3.4.12809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lee HJ, Lee SJ. Characterization of cytoplasmic alpha-synuclein aggregates. Fibril formation is tightly linked to the inclusion-forming process in cells. J Biol Chem. 2002;277:48976–48983. doi: 10.1074/jbc.M208192200. [DOI] [PubMed] [Google Scholar]
- 26.Lee HJ, Suk JE, Patrick C, Bae EJ, Cho JH, Rho S, Hwang D, Masliah E, Lee SJ. Direct transfer of alpha-synuclein from neuron to astroglia causes inflammatory responses in synucleinopathies. J Biol Chem. 2010;285:9262–9272. doi: 10.1074/jbc.M109.081125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Long-Smith CM, Sullivan AM, Nolan YM. The influence of microglia on the pathogenesis of Parkinson's disease. Prog Neurobiol. 2009;89:277–287. doi: 10.1016/j.pneurobio.2009.08.001. [DOI] [PubMed] [Google Scholar]
- 28.Lu JQ, Fan Y, Mitha AP, Bell R, Metz L, Moore GR, Yong VW. Association of alpha-synuclein immunoreactivity with inflammatory activity in multiple sclerosis lesions. J Neuropathol Exp Neurol. 2009;68:179–189. doi: 10.1097/NEN.0b013e318196e905. [DOI] [PubMed] [Google Scholar]
- 29.Maier A, Zimmermann C, Beglinger C, Drewe J, Gutmann H. Effects of budesonide on P-glycoprotein expression in intestinal cell lines. Br J Pharmacol. 2007;150:361–368. doi: 10.1038/sj.bjp.0706992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Miklossy J, Doudet DD, Schwab C, Yu S, McGeer EG, McGeer PL. Role of ICAM-1 in persisting inflammation in Parkinson disease and MPTP monkeys. Exp Neurol. 2006;197:275–283. doi: 10.1016/j.expneurol.2005.10.034. [DOI] [PubMed] [Google Scholar]
- 31.Minagar A, Shapshak P, Fujimura R, Ownby R, Heyes M, Eisdorfer C. The role of macrophage/microglia and astrocytes in the pathogenesis of three neurologic disorders: HIV-associated dementia, Alzheimer disease, and multiple sclerosis. J Neurol Sci. 2002;202:13–23. doi: 10.1016/s0022-510x(02)00207-1. [DOI] [PubMed] [Google Scholar]
- 32.Mogi M, Harada M, Kondo T, Riederer P, Inagaki H, Minami M, Nagatsu T. Interleukin-1 beta, interleukin-6, epidermal growth factor and transforming growth factor-alpha are elevated in the brain from parkinsonian patients. Neurosci Lett. 1994;180:147–150. doi: 10.1016/0304-3940(94)90508-8. [DOI] [PubMed] [Google Scholar]
- 33.Norris EH, Giasson BI, Lee VM. Alpha-synuclein: normal function and role in neurodegenerative diseases. Curr Top Dev Biol. 2004;60:17–54. doi: 10.1016/S0070-2153(04)60002-0. [DOI] [PubMed] [Google Scholar]
- 34.Rappold PM, Tieu K. Astrocytes and therapeutics for Parkinson's disease. Neurotherapeutics. 7:413–423. doi: 10.1016/j.nurt.2010.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rivieccio MA, John GR, Song X, Suh HS, Zhao Y, Lee SC, Brosnan CF. The cytokine IL-1beta activates IFN response factor 3 in human fetal astrocytes in culture. J Immunol. 2005;174:3719–3726. doi: 10.4049/jimmunol.174.6.3719. [DOI] [PubMed] [Google Scholar]
- 36.Sanders VJ, Pittman CA, White MG, Wang G, Wiley CA, Achim CL. Chemokines and receptors in HIV encephalitis. Aids. 1998;12:1021–1026. [PubMed] [Google Scholar]
- 37.Shanmugam N, Ransohoff RM, Natarajan R. Interferon-gamma-inducible protein (IP)-10 mRNA stabilized by RNA-binding proteins in monocytes treated with S100b. J Biol Chem. 2006;281:31212–31221. doi: 10.1074/jbc.M602445200. [DOI] [PubMed] [Google Scholar]
- 38.Sheng WS, Hu S, Min X, Cabral GA, Lokensgard JR, Peterson PK. Synthetic cannabinoid WIN55,212-2 inhibits generation of inflammatory mediators by IL-1beta-stimulated human astrocytes. Glia. 2005;49:211–219. doi: 10.1002/glia.20108. [DOI] [PubMed] [Google Scholar]
- 39.Stefanova N, Klimaschewski L, Poewe W, Wenning GK, Reindl M. Glial cell death induced by overexpression of alpha-synuclein. J Neurosci Res. 2001;65:432–438. doi: 10.1002/jnr.1171. [DOI] [PubMed] [Google Scholar]
- 40.Sun D, Ding A. MyD88-mediated stabilization of interferon-gamma-induced cytokine and chemokine mRNA. Nat Immunol. 2006;7:375–381. doi: 10.1038/ni1308. [DOI] [PubMed] [Google Scholar]
- 41.Tanuma N, Sakuma H, Sasaki A, Matsumoto Y. Chemokine expression by astrocytes plays a role in microglia/macrophage activation and subsequent neurodegeneration in secondary progressive multiple sclerosis. Acta Neuropathol. 2006;112:195–204. doi: 10.1007/s00401-006-0083-7. [DOI] [PubMed] [Google Scholar]
- 42.Tokuda T, Qureshi MM, Ardah MT, Varghese S, Shehab SA, Kasai T, Ishigami N, Tamaoka A, Nakagawa M, El-Agnaf OM. Detection of elevated levels of alpha-synuclein oligomers in CSF from patients with Parkinson disease. Neurology. 75:1766–1772. doi: 10.1212/WNL.0b013e3181fd613b. [DOI] [PubMed] [Google Scholar]
- 43.Tousi NS, Buck DJ, Zecca L, Davis RL. Neuromelanin inhibits CXCL10 expression in human astroglial cells. Neurosci Lett. 2010;486:47–50. doi: 10.1016/j.neulet.2010.09.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Vekrellis K, Rideout HJ, Stefanis L. Neurobiology of alpha-synuclein. Mol Neurobiol. 2004;30:1–21. doi: 10.1385/MN:30:1:001. [DOI] [PubMed] [Google Scholar]
- 45.Venda LL, Cragg SJ, Buchman VL, Wade-Martins R. alpha-Synuclein and dopamine at the crossroads of Parkinson's disease. Trends Neurosci. 2004;33:559–568. doi: 10.1016/j.tins.2010.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Williams R, Dhillon NK, Hegde ST, Yao H, Peng F, Callen S, Chebloune Y, Davis RL, Buch SJ. Proinflammatory cytokines and HIV-1 synergistically enhance CXCL10 expression in human astrocytes. Glia. 2009;57:734–743. doi: 10.1002/glia.20801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Williams R, Yao H, Peng F, Yang Y, Bethel-Brown C, Buch S. Cooperative induction of CXCL10 involves NADPH oxidase: Implications for HIV dementia. Glia. 2010;58:611–621. doi: 10.1002/glia.20949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Xu J, Kao SY, Lee FJ, Song W, Jin LW, Yankner BA. Dopamine-dependent neurotoxicity of alpha-synuclein: a mechanism for selective neurodegeneration in Parkinson disease. Nat Med. 2002;8:600–606. doi: 10.1038/nm0602-600. [DOI] [PubMed] [Google Scholar]
- 49.Yao Y, Kubota T, Sato K, Takeuchi H, Kitai R, Matsukawa S. Interferons upregulate thymidine phosphorylase expression via JAK-STAT-dependent transcriptional activation and mRNA stabilization in human glioblastoma cells. J Neurooncol. 2005;72:217–223. doi: 10.1007/s11060-004-3012-4. [DOI] [PubMed] [Google Scholar]
- 50.Yasuda Y, Shimoda T, Uno K, Tateishi N, Furuya S, Yagi K, Suzuki K, Fujita S. The effects of MPTP on the activation of microglia/astrocytes and cytokine/chemokine levels in different mice strains. J Neuroimmunol. 2008;204:43–51. doi: 10.1016/j.jneuroim.2008.08.003. [DOI] [PubMed] [Google Scholar]
- 51.Zhang W, Wang T, Pei Z, Miller DS, Wu X, Block ML, Wilson B, Zhang W, Zhou Y, Hong JS, Zhang J. Aggregated alpha-synuclein activates microglia: a process leading to disease progression in Parkinson's disease. Faseb J. 2005;19:533–542. doi: 10.1096/fj.04-2751com. [DOI] [PubMed] [Google Scholar]
- 52.Zhou W, Schaack J, Zawada WM, Freed CR. Overexpression of human alpha-synuclein causes dopamine neuron death in primary human mesencephalic culture. Brain Res. 2002;926:42–50. doi: 10.1016/s0006-8993(01)03292-9. [DOI] [PubMed] [Google Scholar]