Abstract
Purpose
Positive surgical margin (PSM) status following radical prostatectomy (RP) is a well-established prognostic factor. The aim of the present study is to evaluate whether number of PSMs or bilaterality of PSMs might have prognostic significance for biochemical recurrence (BCR) in the population with a PSM status following RP.
Methods
We evaluated 1,395 RP pathology reports from our center between 1980 and 2006. All patients who underwent (neo)-adjuvant therapy were excluded, leaving a cohort of 1,009 patients, with 249 (24.7%) subjects having a PSM at RP of whom 29.4% had multiple PSMs (≥ 2 sites), while 13.6% had bilateral PSMs. Median follow-up was 40 months (range 0–258 months). We used BCR-free survival as the primary study outcome. BCR was defined as any rise in PSA above or equal to 0.2 ng/ml.
Results
Of patients with a PSM status, 41% (95% CI: 33–49%) developed BCR within 5 years, compared to 12% (95% CI: 9–15%) in the population without a PSM. Multivariable analysis identified PSA at diagnosis and RP Gleason score as independent predictive factors for BCR. Increasing number and/or bilaterality of PSM did not lead to significant higher rates of BCR.
Conclusion
In patients with a PSM, the number of positive sites or bilaterality of PSM status does not add prognostic information for risk of BCR. Survival curve slopes were different for patients with bilateral PSM, showing a significant tendency to progress to BCR earlier during follow-up than patients with unilateral PSM.
Keywords: Prostate cancer, Surgical margin, Biochemical recurrence, Bilateral, Number, Prognosis
Introduction
The 5- and 10-year overall survival rates of radical prostatectomy (RP) are excellent [1], leading to significant survival benefit compared to watchful waiting [2]. In the absence of extraprostatic extension and positive surgical margins (PSM), the rates of biochemical recurrence (BCR) are low [3]. This does not stand true for patients with a PSM, a common pathological feature following RP, with a prevalence varying between 5 and 43% in different series [4, 5]. Several studies have shown PSM to be one of the most important prognostic factors for BCR following radical prostatectomy [4–13].
The EORTC 22911 trial [14] established that adjuvant external irradiation after RP improves biochemical recurrence (BCR)-free survival in patients with a PSM or pathological T3 stage. Whether this translates into an overall survival benefit could not be detected due to a relative short follow-up. In this trial, 43.7% of the patients in the wait-and-see arm experienced biochemical or clinical progression or death. This percentage was reduced to 26.1% in the irradiation arm. However, if all patients in this series would have received immediate radiotherapy, over fifty percent would have received intervention without ever progressing to BCR at the cost of radiotherapeutic toxicity. Therefore, characterization of patients at high risk of BCR after RP would be of great help to identify those patients benefiting most of immediate postoperative radiotherapy. Reevaluation of the EORTC 22911 data by van der Kwast et al. [15] stressed that among patients with adverse pathological features on prostatectomy, those with PSM benefit most from immediate radiotherapy, preventing 291 BCR events for every 1,000 treated. We hypothesized that the number of PSMs or bilaterality of PSMs is an additional risk factor of BCR in patients with a PSM and may indicate who will be ideal candidates for adjuvant postoperative radiotherapy.
Patients and methods
Study population and data retrieval
The pathology reports of 1,395 open retropubic RP procedures performed at the Department of Urology, Université Catholique de Louvain, Brussels, between 1980 and 2006 by two surgeons were retrospectively evaluated for PSM status. Since 1999, intra-operative frozen sections were used to avoid PSMs. Patient and tumor characteristics were retrospectively reviewed for all subjects with a PSM. All subjects who were treated with neo-adjuvant and/or adjuvant hormonal therapy or immediate postoperative radiotherapy were excluded. Data on number and bilaterality of PSM, as well as pathological stage, RP Gleason score and the presence of perineural invasion (PNI) were obtained from the original pathology reports. Charts were retrospectively reviewed for PSA at diagnosis and follow-up data. BCR was defined as any rise in PSA (ng/ml) above or equal to 0.2 ng/ml.
Pathology processing
The left and right sides of the prostate gland were identified by a longitudinal incision into the right anterior half. Following fixation in buffered 10% formaldehyde, the 5-mm-thick proximal and distal transections of the prostate were serially sectioned at 2-mm intervals parallel to the urethra. The tips of the vasa deferentia were transected, and the seminal vesicles were longitudinally sectioned up to their junction with the prostate. The remaining prostate gland was then serially sectioned perpendicularly to the apical–basal axis at 5-mm intervals to perform whole mount sections. The external surface of surgical radical prostatectomy specimens was covered with ink since 1990. Surgical margins were considered as positive or negative when the malignant cells were separated without or with any amount of benign tissue from the inked edge of the surgical resection of the gland, respectively. The paraffin-embedded tissues were recut if necessary until visualization of the inked margin. When the margins had not been inked, the paraffin blocks were recut until the whole circumference of the tissue sample was mounted on the slide. The edges of the artifactual disruptions of prostatic or extraprostatic tissue were not considered as surgical margins.
Statistical analysis
For statistical analysis, we used SPSS software (SPSS, version 16.0.01, Chicago, Illinois, USA). Kaplan–Meier survival analysis was performed with BCR-free survival as outcome for both single versus multiple PSM and unilateral versus bilateral PSM. The Wilcoxon test was used to detect significant differences in BCR-free survival rates between groups. Multivariable Cox proportional hazards regression models were composed to determine prognostic factors for BCR. As multiple PSMs will represent a large subgroup of bilateral PSMs, we performed multivariable analysis for both factors separately. Statistical significance in our study was set at P < 0.05. With the numbers of single and multiple PSM cases in our series, and an assumed 40% 5-year risk of BCR, we had an 80% power to detect a hazard ratio (HR) of 1.5. Likewise, we had a power of 80% of detecting a HR of 1.7 for the comparison between unilateral and bilateral PSM.
Results
We found sufficient data on surgical margin status for 1,314 patients to include them in our study. Of these, 378 (27.1%) had one or more PSMs. Those who received immediate postoperative radiotherapy (n = 181), neo-adjuvant (n = 171), or adjuvant hormonal therapy (n = 18) were excluded, leaving a cohort of 1,009 patients, with 249 (24.7%) subjects having one or more PSMs. Of 723 patients with a pT2 status, 122 (16.9%) had a PSM, compared to 123 of 266 (46.2%) patients with a pT3 status, 8 patients had pT4 tumor, of which 4 had PSM (50.0%). Seven patients had pT0 status upon RP, and in 5 patients, the pathological stage could not be established. The median follow-up was 40 months (range 0–258 months). We found sufficient data on number of PSMs in 218/249 pathology reports; of these, 70.6% had a single PSM, while the remaining 29.4% had two or more PSMs. In 191/249 subjects, sufficient data were reported to determine PSM bilaterality; of these, 86.4% of the PSMs were unilateral and the remaining 13.6% bilateral. Pathological stage differed significantly between single and multiple PSM status (P = 0.04) and unilateral versus bilateral PSM status (P = 0.03), with patients with multiple or bilateral PSMs having higher pathological stages. Age, PSA at diagnosis, and RP Gleason score did not differ significantly among groups. Table 1 summarizes all relevant patient characteristics as identified in our series.
Table 1.
Patient characteristics
| Variable | PSM | Single PSM | Multiple PSM | Unilateral PSM | Bilateral PSM |
|---|---|---|---|---|---|
| Number of subjects | 249 | 154/218 (70.6%) | 64/218 (29.4%) | 165/191 (86.4%) | 26/191 (13.6%) |
| Age (mean ± SD) | 63.8 ± 6.9 | 63.6 ± 6.9 | 64.7 ± 6.1 | 63.8 ± 6.5 | 64.3 ± 7.0 |
| PSA at diagnosis (mean ± SD) | 11.7 ± 9.7 | 10.7 ± 7.8 | 12.9 ± 12.8 | 11.4 ± 8.4 | 11.8 ± 13.1 |
| Pathological stage | |||||
| pT2 | 122 (49.8%) | 82 (53.9%) | 25 (40.3%) | 80 (49.7%) | 10 (38.5%) |
| pT3 | 123 (50.2%) | 70 (46.1%) | 37 (59.7%) | 81 (50.3%) | 16 (61.5%) |
| Missing | 4 | 2 | 2 | 4 | 0 |
| RP Gleason score | |||||
| ≤6 | 173 (69.8%) | 110 (71.4%) | 41 (64.1%) | 118 (71.5%) | 15 (57.7%) |
| 7 | 66 (26.6%) | 40 (26.0%) | 18 (28.1%) | 41 (24.8%) | 9 (34.6%) |
| 8–10 | 9 (3.6%) | 4 (2.6%) | 5 (7.8%) | 6 (3.6%) | 2 (7.7%) |
| Missing | 1 | 0 | 0 | 0 | 0 |
| 5-year BCR (95% CI) | 41% (33–49%) | 43% (32–54%) | 46% (31–61%) | 45% (35–55%) | 46% (24–68%) |
PSM positive surgical margin, SD standard deviation, CI confidence interval, RP radical prostatectomy, BCR biochemical recurrence rate
Overall, 41% (95% CI: 33–49%) of subjects with a PSM developed a BCR within 5 years, compared to 12% (95% CI: 9–15%) in the population without a PSM. When subdivided into single and multiple PSMs, these percentages were 43% (95% CI: 32–54%) versus 46% (95% CI: 31–61%), respectively. Unilateral versus bilateral PSM subjects had 5-year BCR rates of 45% (95% CI: 35–55%) versus 46% (95% CI: 24–68%), respectively. Patients with data missing on number of PSMs had a 5-year BCR rate of 24% (95% CI: 6–42%), while those with missing data on bilaterality had a 5-year BCR rate of 30% (95% CI: 16–44%). When we used the Wilcoxon test to detect differences in the slope of the survival curves, we found that survival curve slopes for unilateral versus bilateral PSM did differ significantly (P = 0.029), as the bilateral PSM cohort did progress to BCR earlier during follow-up, with the curves closing in later on, see Fig. 1.
Fig. 1.
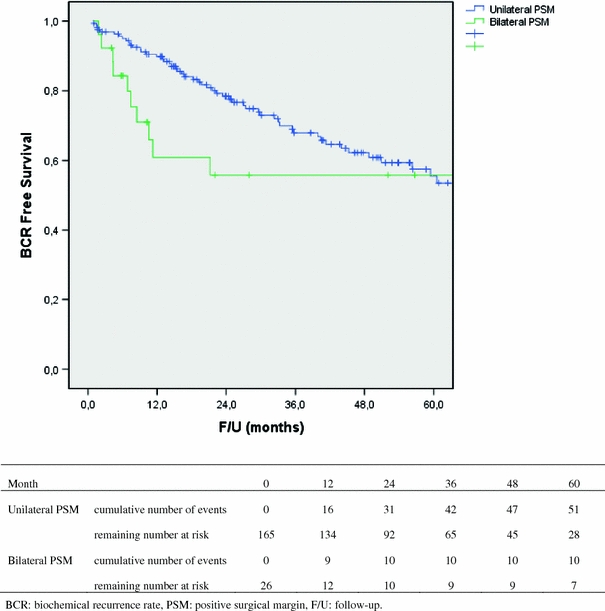
Kaplan–Meier BCR-free survival curves for unilateral and bilateral PSM
Univariable analysis identified PSA at diagnosis, pathological stage, RP Gleason score, and perineural invasion (PNI) as possible predictors of BCR after RP in this PSM series, and these factors were consequently included in the multivariable analysis. Bilateral PSM status and multiple PSM status had no prognostic value for BCR on univariable analysis. Multivariable analysis identified PSA at diagnosis and RP Gleason score as independent prognostic factors for BCR. Bilateral PSM status and number of PSMs did not add prognostic information, see Table 2.
Table 2.
Univariable and multivariable analysis of prognostic factors for BCR in a PSM cohort
| Variable | Univariable analysis HR (95% CI) | Multivariable analysis HR (95% CI) | P-value | Multivariable analysis HR (95% CI) | P-value |
|---|---|---|---|---|---|
| Age at RP | 1.00 (0.97–1.03) | N/A | N/A | N/A | N/A |
| PSA at diagnosis (ng/ml) | 1.04 (1.02–1.05) | 1.03 (1.01–1.05) | P = 0.002 | 1.04 (1.02–1.06) | P = 0.000 |
| Pathological stage (pT3 vs. pT2) | 2.36 (1.47–3.79) | 1.45 (0.82–2.58) | P > 0.05 | 1.42 (0.77–2.63) | P > 0.05 |
| RP Gleason score (per point) | 1.42 (1.15–1.76) | 1.34 (1.07–1.68) | P = 0.01 | 1.32 (1.04–1.66) | P = 0.02 |
| PNI | 2.20 (1.40–3.44) | 1.22 (0.71–2.09) | P > 0.05 | 1.43 (0.82–2.50) | P > 0.05 |
| Multiple vs. single PSM | 1.37 (0.72–2.64) | 1.05 (0.63–1.74) | P > 0.05 | N/A | N/A |
| Bilateral vs. unilateral PSM | 1.28 (0.79–2.07) | N/A | N/A | 1.32 (0.66-2.62) | P > 0.05 |
Values in italics are statistically signifiicant at P < 0.05
BCR biochemical recurrence, PSM positive surgical margin, HR hazard ratio, CI confidence interval, RP radical prostatectomy, PNI perineural invasion, N/A not applicable
Discussion
Data on 5-year risk of BCR for PSM patients following RP are reported between 25 and 47% [5, 10–12]. No effect on prostate cancer-specific survival or overall survival has been determined for PSM status, probably because the available follow-up does not suffice to detect these differences if present. Preoperative PSA, RP Gleason score, and pathological stage are well-established predictors of BCR following RP [3, 16–19]. In correspondence with other series, we identified RP Gleason score as a prognostic factor for BCR among patients with a PSM [5, 7, 9, 11]. All these earlier reports identified pathological stage as a prognostic factor for BCR as well, which we could only confirm in univariable analysis. This might be contributed to the 370 patients that were excluded from our analysis because of (neo)-adjuvant therapy leading to a disproportional exclusion of poor-risk subjects with higher pT stages. Furthermore, pT3 subjects were more likely to have PSM, which was included in multivariable analysis, and might, due to its profound effect on BCR rates, diminish the effect of pT3 stage on BCR rates. Much debate remains over the prognostic value of PNI in RP specimens [20, 21]. In our subset of patients with PSMs, we could not identify PNI as an independent prognostic factor for BCR on multivariable analysis.
Five studies address the number of PSMs in detail in populations that did not receive immediate postoperative therapy and yielded contradictory results. In a subset of 80 PSM patients analyzed by Lowe and Lieberman, a significant increase in BCR for patients with multiple PSMs compared with single PSM was found [22], a finding confirmed in another series by Sofer et al. with 210 patients with PSM [23]. Both series did not include pathological stage in their multivariable analyses which may have led to biased results as one might hypothesize that extraprostatic extension (pT3) is far more common in the subgroup with multiple PSM status. In our series, we could confirm that pathological T3 stage was significantly more common in the multiple and bilateral PSM cohorts compared with the single PSM cohort. This hypothesis is supported by a larger series by Blute et al. of 697 pT2 patients with a PSM in which only a slightly higher rate of BCR was found for patients with multiple PSM when compared with those with a single PSM [24]. Also, a more recent report on PSM status in 354 patients with extraprostatic carcinoma (pT3a/b) on RP could also not detect a significant difference in BCR between patients with single and multiple PSMs [11]. Therefore, we think that any report on PSM status should include pathological stage in the multivariable analysis in order to be able to assess the independent prognostic value of PSM status properly. Furthermore, as patients with multiple or bilateral PSMs tend to experience BCR earlier during follow-up with Kaplan–Meier curves closing in later on, the relative short follow-up of 22 months by Sofer et al. [23] could lead to a false impression of increased BCR for multiple PSMs when processing Kaplan–Meier curves on these data. This is supported by the difference in calculated 5-year BCR rates for multiple PSMs between ours and their series, 41% versus approximately 60% (read from the Kaplan–Meier curve), respectively. A series by Jayachandran et al. reporting on 902 patients with PSM and/or pT3 disease could not identify pathological stage as an independent predictor of BCR on univariable analysis, and thus, did not incorporate this factor in their multivariable analysis [25]. Consequently, they found number of PSM to be significantly associated with BCR on univariable and multivariable analysis, a finding we could not confirm. In their series, they did however exclude a substantial number of patients (n = 205) with seminal vesicle invasion, which could account for a large number of BCR subjects in our series as these are relative poor-risk subjects within the pT3 subgroup.
We did not take the extent or length and site of PSM into account, and contradictory reports have been published on this issue. Some finding length of PSM as a prognostic marker for BCR [26], whereas others could not identify extent of PSM as a prognostic marker [8, 27]. Most series did not find site of PSM to be predictive of BCR [23, 27], while Blute et al. [24] identified the prostate base as the only anatomic site of PSM predictive for recurrence at that specific anatomic site with a significant effect on 5-year risk of BCR, which increased from 15 to 44% in case of a PSM at the prostate base.
To our knowledge, no study on bilaterality of PSM status has been published before. Nevertheless, one might hypothesize that bilateral PSM status might influence BCR-free survival, as it might express more extensive tumor involvement of the prostate bed after RP. Nevertheless, we could not identify an independent prognostic value of bilateral PSM status on multivariable analysis in our series. We found patients with bilateral PSM to progress to BCR earlier during follow-up, with survival curves closing in at about 5-years of follow-up.
The main limitation of our series is the retrospective data collection, which led to a substantial number of missing data in which we could not establish the details of PSM status on number and bilaterality. Interestingly, patients with missing data did better as far as BCR rates are concerned, which might be attributed to less detailed pathological reporting on number and bilaterality of PSM in case of a single or limited multiple PSM status. Another concern is the relatively low power of our series to detect differences between groups; this is mainly an issue in the comparison of unilateral versus bilateral PSM, as only 26 subjects were documented to have bilateral PSM. Furthermore, the median follow-up of approximately 40 months could be insufficient to detect differences in long-term BCR in our series.
We conclude that number or bilaterality of PSM is not independent predictors of BCR, although patients with bilateral PSMs did show a significant tendency to progress to BCR earlier during follow-up compared to patients with unilateral PSMs. For conclusive evidence, future prospective series with longer median follow-up addressing this issue are needed. The search for more valid prognostic markers for disease recurrence following RP continues for better risk stratification and decision making regarding timing of adjuvant radiotherapy in postprostatectomy subjects.
Acknowledgments
Conflict of interest
The authors declare that they have no conflict of interest.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
References
- 1.Zincke H, Oesteling JE, Blute ML, Bergstrahl EJ, Myers RP, Barrett DM. Long-term results after radical prostatectomy for clinically localized prostate cancer. J Urol. 1994;152:1850–1857. doi: 10.1016/s0022-5347(17)32399-6. [DOI] [PubMed] [Google Scholar]
- 2.Bill-Axelson A, Holmberg L, Filén F, et al. Radical prostatectomy versus watchful waiting in localized prostate cancer: the Scandinavian prostate cancer group-4 randomized trial. J Natl Cancer Inst. 2008;100(16):1144–1154. doi: 10.1093/jnci/djn255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Partin AW, Piantadosi S, Sanda MG, et al. Selection of men at high risk for disease recurrence for experimental adjuvant therapy following radical prostatectomy. Urology. 1995;45:831–838. doi: 10.1016/S0090-4295(99)80091-0. [DOI] [PubMed] [Google Scholar]
- 4.Epstein JI. Incidence and significance of positive margins in radical prostatectomy specimens. Urol Clin North Am. 1996;23:651–663. doi: 10.1016/S0094-0143(05)70343-8. [DOI] [PubMed] [Google Scholar]
- 5.Swindle P, Eastham JA, Ohori M, et al. Do margins matter? The prognostic significance of positive surgical margins in radical prostatectomy specimens. J Urol. 2005;174:903–907. doi: 10.1097/01.ju.0000169475.00949.78. [DOI] [PubMed] [Google Scholar]
- 6.Grossfeld GD, Chang JJ, Broering JM, et al. Impact of positive surgical margins on prostate cancer recurrence and the use of secondary cancer treatment: data from the CaPSURE database. J Urol. 2000;163:1171–1177. doi: 10.1016/S0022-5347(05)67716-6. [DOI] [PubMed] [Google Scholar]
- 7.Simon MA, Kim S, Soloway MS. Prostate specific antigen recurrence rates are low after radical retropubic prostatectomy and positive margins. J Urol. 2006;175:140–145. doi: 10.1016/S0022-5347(05)00050-9. [DOI] [PubMed] [Google Scholar]
- 8.Vis AN, Schröder FH, Van der Kwast TH. The actual value of the surgical margin status as a predictor of disease progression in men with early prostate cancer. Eur Urol. 2006;50:258–265. doi: 10.1016/j.eururo.2005.11.030. [DOI] [PubMed] [Google Scholar]
- 9.Pfitzenmaier J, Pahernik S, Tremmel T, Haferkamp A, Buse S, Hohenfellner M. Positive surgical margins after radical prostatectomy: do they have an impact on biochemical or clinical progression. BJU Int. 2008;102:1413–1418. doi: 10.1111/j.1464-410X.2008.07563.x. [DOI] [PubMed] [Google Scholar]
- 10.Blute ML, Bostwick DG, Seay TM, Martin SK, Slezak JM, Bergstralh EJ, Zincke H. Pathologic classification of prostate carcinoma. The impact of margin status. Cancer. 1998;82:902–908. doi: 10.1002/(SICI)1097-0142(19980301)82:5<902::AID-CNCR15>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
- 11.Kausik SJ, Blute ML, Sebo TJ, Leibovich BC, Bergstralh EJ, Slezak J, Zincke H. Prognostic significance of positive surgical margins in patients with extraprostatic carcinoma after radical prostatectomy. Cancer. 2002;95:1215–1219. doi: 10.1002/cncr.10871. [DOI] [PubMed] [Google Scholar]
- 12.Karakiewicz PI, Eastham JA, Graefen M, et al. Prognostic impact of positive surgical margins in surgically treated prostate cancer: multi-institutional assessment of 5831 patients. Urology. 2005;66:1245–1250. doi: 10.1016/j.urology.2005.06.108. [DOI] [PubMed] [Google Scholar]
- 13.Yossepowitch O, Bjartell A, Eastham JA, Graefen M, Guillonneau BD, Karakiewicz PI, Montironi R, Montorsi F. Positive surgical margins in radical prostatectomy: outlining the problem and its long-term consequences. Eur Urol. 2009;55:87–99. doi: 10.1016/j.eururo.2008.09.051. [DOI] [PubMed] [Google Scholar]
- 14.Bolla M, van Poppel H, Collette L, et al. Postoperative radiotherapy after radical prostatectomy: a randomized controlled trial (EORTC trial 22911) Lancet. 2005;366:572–578. doi: 10.1016/S0140-6736(05)67101-2. [DOI] [PubMed] [Google Scholar]
- 15.Van der Kwast TH, Bolla M, van Poppel H, et al. Identification of patients with prostate cancer who benefit from immediate postoperative radiotherapy: EORTC 22911. J Clin Oncol. 2007;25:4178–4186. doi: 10.1200/JCO.2006.10.4067. [DOI] [PubMed] [Google Scholar]
- 16.Bostwick DG, Grignon DJ, Hammond EH, et al. Prognostic factors in prostate cancer. College of American Pathologists consensus statement 1999. Arch Pathol Lab Med. 2000;124:995–1000. doi: 10.5858/2000-124-0995-PFIPC. [DOI] [PubMed] [Google Scholar]
- 17.D’Amico A, Whittington R, Malkowicz SB, et al. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA. 1998;280:969–974. doi: 10.1001/jama.280.11.969. [DOI] [PubMed] [Google Scholar]
- 18.Kattan MW, Wheeler TM, Scardino PT. Postoperative nomogram for disease recurrence after radical prostatectomy for prostate cancer. J Clin Oncol. 1999;17:1499–1507. doi: 10.1200/JCO.1999.17.5.1499. [DOI] [PubMed] [Google Scholar]
- 19.Budäus L, Isbarn H, Eichelberg C, et al. Biochemical recurrence after radical prostatectomy: multiplicative interaction between surgical margin status and pathological stage. J Urol. 2010;184:1341–1346. doi: 10.1016/j.juro.2010.06.018. [DOI] [PubMed] [Google Scholar]
- 20.Merrilees AD, Bethwaite PB, Russell GL, Robinson RG, Delahunt B. Parameters of perineural invasion in radical prostatectomy specimen lack prognostic significance. Mod Pathol. 2008;21:1095–1100. doi: 10.1038/modpathol.2008.81. [DOI] [PubMed] [Google Scholar]
- 21.D’Amico AV, Wu Y, Chen M-H, et al. Perineural invasion as a predictor of biochemical recurrence outcome following radical prostatectomy for select men with clinically localized prostate cancer. J Urol. 2001;165:126–129. doi: 10.1097/00005392-200101000-00031. [DOI] [PubMed] [Google Scholar]
- 22.Lowe BA, Lieberman SF. Disease recurrence and progression in untreated pathologic stage T3 prostate cancer: selecting the patient for adjuvant therapy. J Urol. 1997;158:1452–1456. doi: 10.1016/S0022-5347(01)64240-X. [DOI] [PubMed] [Google Scholar]
- 23.Sofer M, Hamilton-Nelson KL, Civantos F, Soloway MS. Positive surgical margins after radical retropubic prostatectomy: the influence of site and number on progression. J Urol. 2002;167:2453–2456. doi: 10.1016/S0022-5347(05)65003-3. [DOI] [PubMed] [Google Scholar]
- 24.Blute ML, Bostwick DG, Bergstralh EJ, Slezak JM, Martin SK, Amling CL, Zincke H. Anatomic site-specific positive margins in organ-confined prostate cancer and its impact on outcome after radical prostatectomy. Urology. 1997;50:733–739. doi: 10.1016/S0090-4295(97)00450-0. [DOI] [PubMed] [Google Scholar]
- 25.Jayachandran J, Bañez LL, Levy DE, Aronson WJ, Terris MK, Presti JC, Jr, Amling CL, Kane CJ, Freedland SJ. Risk stratification for biochemical recurrence in men with positive surgical margins or extracapsular disease after radical prostatectomy: results from the SEARCH database. J Urol. 2008;179:1791–1796. doi: 10.1016/j.juro.2008.01.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ochiai A, Sotelo T, Troncoso P, Bhadkamkar V, Babaian RJ. Natural history of biochemical progression after radical prostatectomy based on length of positive margin. Urology. 2008;71:308–312. doi: 10.1016/j.urology.2007.08.042. [DOI] [PubMed] [Google Scholar]
- 27.Watson RB, Civantos F, Soloway MS. Positive surgical margins with radical prostatectomy: detailed pathological analysis and prognosis. Urology. 1996;48:80–90. doi: 10.1016/S0090-4295(96)00092-1. [DOI] [PubMed] [Google Scholar]


