Abstract
Background
Alcohol withdrawal is associated with behavioral and chronobiological disturbances that may persist during protracted abstinence. We previously reported that C57BL/6J (B6) mice show marked but temporary reductions in running-wheel activity, and normal free-running circadian rhythms, following a 4-day chronic intermittent ethanol vapor (CIE) exposure (16 hours of ethanol vapor exposure alternating with 8 hours of withdrawal). In the present experiments, we extend these observations in two ways: (1) by examining post-CIE locomotor activity in C3H/HeJ (C3H) mice, an inbred strain characterized by high sensitivity to ethanol withdrawal, and (2) by directly comparing the responses of B6 and C3H mice to a longer-duration CIE protocol.
Methods
In Experiment 1, C3H mice were exposed to the same 4-day CIE protocol used in our previous study with B6 mice (referred to here as the 1-cycle CIE protocol). In Experiment 2, C3H and B6 mice were exposed to three successive 4-day CIE cycles, each separated by 2 days of withdrawal (the 3-cycle CIE protocol). Running-wheel activity was monitored prior to and following CIE, and post-CIE activity was recorded in constant darkness to allow assessment of free-running circadian period and phase.
Results
C3H mice displayed pronounced reductions in running-wheel activity that persisted for the duration of the recording period (up to 30 days) following both 1-cycle (Experiment 1) and 3-cycle (Experiment 2) CIE protocols. In contrast, B6 mice showed reductions in locomotor activity that persisted for about one week following the 3-cycle CIE protocol, similar to the results of our previous study using a 1-cycle protocol in this strain. Additionally, C3H mice showed significant shortening of free-running period following the 3-cycle, but not the 1-cycle, CIE protocol, while B6 mice showed normal free-running rhythms.
Conclusions
These results reveal genetic differences in the persistence of ethanol withdrawal-induced hypo-locomotion. In addition, chronobiological alterations during extended abstinence may depend on both genetic susceptibility and an extended prior withdrawal history. The present data establish a novel experimental model for long-term behavioral and circadian disruptions associated with ethanol withdrawal.
Keywords: Wheel Running, Circadian, Ethanol, Withdrawal, Inbred Mice
Introduction
Alcohol withdrawal is associated with a wide variety of neurobehavioral disturbances in both human alcoholics and in animal models of alcohol dependence. While these effects often resolve within a few days, withdrawal-related mood and sleep disruptions sometimes persist during protracted periods of abstinence (De Soto et al., 1985; Dackis et al., 1986; Drummond et al., 1998; Landolt and Gillin, 2001), and are associated with increased risk of relapse (Greenfield et al., 1998; Brower, 2001, 2003; Hasin et al., 2005). While studies of ethanol withdrawal in animal models have typically focused on the acute withdrawal phase, recent experiments have revealed more enduring withdrawal-related behavioral disturbances (Heilig et al., 2010). For example, rats and mice have been shown to display increases in anxiety- and depression-like behaviors several weeks after induction of ethanol dependence by exposure to ethanol vapor (Valdez et al., 2002; Zhao et al., 2007; Walker et al., 2010) or ethanol liquid diet (Valdez et al., 2003; Rylkova et al., 2007), or following an extended period of voluntary ethanol drinking (Stevenson et al., 2009). These long-term effects are believed to reflect neuroadaptive changes in multiple synaptic and intracellular signaling systems triggered by chronic ethanol exposure and/or withdrawal (Clapp et al., 2008; Koob and Le Moal, 2008).
Several studies have examined the effects of ethanol withdrawal on spontaneous locomotor activity, but the results of these studies have been inconsistent. Thus, ethanol-withdrawn rats and mice have been reported to display both increases (Griffiths et al., 1974; Mehta and Ticku, 1993; Uzbay and Kayaalp, 1995; Dahchour and De Witte, 1999) and decreases (Kliethermes et al., 2004; Slawecki and Roth, 2004; Getachew et al., 2008) in locomotor activity. These discrepant results may be due, in part, to differences in testing conditions. Thus, locomotor hyperactivity is seen most consistently when tests are conducted within about 6–8 hours following withdrawal, while locomotor hypoactivity is seen most consistently when activity is assessed at later time points and/or in novel test environments (i.e., in the elevated plus- or zero-maze, or in the open field). Indeed, little or no change in locomotor activity is seen when animals are tested under familiar, home-cage conditions (Kliethermes et al., 2004, 2005; Slawecki and Roth, 2004). In contrast, however, we recently reported that C57BL/6J (B6) mice display a period of reduced running-wheel activity for about one week following a 4-day chronic-intermittent ethanol vapor exposure (CIE) (Logan et al., 2010), despite the fact that locomotor activity was assessed under familiar, home-cage conditions. Therefore, it seems likely that the specific type of activity measured (e.g., running-wheel, open-field, home-cage) may be more important than environmental novelty in influencing post-withdrawal activity levels.
In the present experiments, we first sought to extend our previous results by characterizing post-CIE running-wheel activity in the C3H/HeJ (C3H) inbred mouse, using an identical protocol to that employed previously in B6 mice (Experiment 1). In Experiment 2, both C3H and B6 mice were exposed to a more extended CIE protocol in which the original 4-day exposure regimen was repeated three times, interspersed with 2-day withdrawal periods. C3H mice were chosen for these experiments because this strain displays much greater sensitivity to the intoxicating effects of acute ethanol and to the seizure-potentiating effects of ethanol withdrawal, relative to B6 mice (Metten and Crabbe, 2005). Indeed, after accounting for strain differences in blood ethanol concentrations (BECs), C3H mice displayed the second-most severe handling-induced convulsions (HICs), while B6 mice displayed the least severe HICs, among the 15 inbred strains tested (Metten and Crabbe, 2005). The repeated CIE protocol was employed in the second experiment because of evidence that the effects of ethanol withdrawal can be potentiated by a history of prior withdrawals (Becker and Hale, 1993; Becker et al., 1997; Overstreet et al., 2002; O’Dell et al., 2004; Becker and Lopez, 2004; Lopez and Becker, 2005). Thus, we hypothesized that C3H mice would show more pronounced and/or more persistent withdrawal-induced reductions in running-wheel activity than B6 mice, and that the severity of these effects might be potentiated by a more extensive CIE history in both strains.
Another major aim of the present experiments was to explore the effects of ethanol withdrawal on circadian clock function. Accumulating evidence in both humans and experimental animals suggests reciprocal interactions between alcohol exposure (and/or alcohol withdrawal) and disruptions in sleep and circadian biological rhythms. In human alcoholics, chronobiological disruptions can persist during extended periods of abstinence (Drummond et al., 1998; Landolt and Gillin, 2001), and have been linked to increased risk of relapse (Brower, 2001, 2003). Recent animal experiments suggest that these effects are due in part to effects of ethanol on the circadian clock. Thus, chronic ethanol treatment alters fundamental circadian clock parameters, including free-running circadian period and responsiveness to phase-shifting stimuli, in rats (Rosenwasser et al., 2005a, b), hamsters (Mistlberger and Nadeau, 1992; Seggio et al., 2007; Ruby et al., 2009), and mice (Seggio et al., 2009; Brager et al., 2010). Nevertheless, we previously failed to detect any effect of CIE exposure on free-running period or phase in ethanol-withdrawn B6 mice under constant darkness (DD) (Logan et al., 2010). Since this negative result could reflect a generalized relative insensitivity of B6 mice to ethanol withdrawal, we speculated that circadian clock effects might be detected in the more withdrawal-sensitive C3H strain.
Material and methods
Subjects and apparatus
Upon arrival in the laboratory, 6–8 week old male C57BL/6J (B6) and C3H/HeJ (C3H) mice (Jackson Laboratories, Bar Harbor, ME), were weighed (range: 19–22 g) and housed individually in running-wheel cages (wheel diameter: 23 cm; Mini-Mitter Co., Bend, OR). Running-wheel cages were placed three per shelf in a light-shielded and sound-attenuating metal cabinet equipped with a standard fluorescent bulb on each shelf. Food (Prolab RMH 3000), and tap water were freely available throughout the experiment. Wheel-running activity was recorded and analyzed using the ClockLab interface system (Actimetrics, Co., Wilmette, IL).
Procedures
Initially, mice were maintained under a 12:12 light-dark (LD) cycle for 10–14 days to allow acclimation to running wheels and stabilization of activity levels. Following the acclimation period, mice were exposed to either a 1-cycle (Experiment 1) or a 3-cycle (Experiment 2) CIE protocol (see below), while controls were handled identically but exposed only to plain air. Experimental and control groups consisted of 10–15 animals per group; N’s for each group are available in Table 2. In Experiment 1, animals were exposed to CIE while housed in their individual running-wheel cages, while in Experiment 2 animals were removed from their running-wheel cages and exposed to CIE while group-housed in standard mouse cages, in order to increase the number of animals that could be placed concurrently in the inhalation chambers. The inhalation chambers consisted of large Plexiglas boxes (60 × 36 × 60 cm) constructed according to a design provided by Dr. Howard Becker, Medical University of South Carolina. Following CIE or plain air treatment, animals were returned to the activity recording cabinet and wheel-turns were recorded for an additional 2- (Experiment 1) or 4-week (Experiment 2) posttreatment period while maintained in constant darkness (DD) to evaluate the period and phase of free-running circadian activity rhythms. In addition, pre- and posttreatment water tube licking was recorded using contact-sensing drinkometer circuits in Experiment 1 only.
Table 2.
Mean (± SEM) wheel-running activity during the 5-day baseline period preceding 1-cycle (Experiment 1) and 3-cycle (Experiment 2) CIE and control treatments.
| Protocol | Strain | Group | N | Baseline Activity (Distance in meters) |
|---|---|---|---|---|
| 1-cycle | C3H | CONT | 12 | 1764 ± 821 |
| C3H | CIE | 11 | 1499 ± 278 | |
| 3-cycle | B6 | CONT | 15 | 7892 ± 473 |
| B6 | CIE | 10 | 7838 ± 829 | |
| C3H | CONT | 13 | 1001 ± 425* | |
| C3H | CIE | 12 | 1072 ± 272* |
Asterisk (*) indicates significant difference from B6 mice within the same treatment group (P < 0.001).
Chronic intermittent ethanol (CIE) protocols
Based on the prior work of Becker and coworkers (e.g., Becker & Hale, 1993; Becker et al., 1997) and on our own previous study (Logan et al., 2010), the CIE protocol employed in Experiment 1 consisted of a 4-day intermittent exposure to ethanol vapor (i.e., 16 hours of ethanol vapor alternating with 8 hours of plain air, with each vapor exposure period beginning at dark onset). Because previous studies indicated that responses to ethanol withdrawal following CIE may be potentiated by increasing the number of prior withdrawals (Becker et al., 1997; Becker and Lopez, 2004; Lopez and Becker, 2005), Experiment 2 employed a CIE protocol in which the basic 4-day procedure was repeated 3 times, each separated by a 2-day period of withdrawal. Thus, the animals in Experiment 2 spent a total of 16 days in the inhalation chambers, and were exposed to ethanol vapor on 12 of the 16 days. We refer to these procedures as the 1-cycle CIE and the 3-cycle CIE protocol, respectively. Control animals were handled identically, but exposed only to plain air. Immediately prior to each ethanol vapor exposure, CIE animals were administered a priming injection containing 1.6 g/kg ethanol and 68.1 mg/kg pyrazole HCl, an alcohol dehydrogenase inhibitor used to rapidly increase and stabilize blood ethanol concentrations (e.g., Becker & Hale, 1993). Pyrazole was dissolved in 20% v/v ethanol solution and injected i.p. in a volume of 10 ml/kg, while control animals were administered an identical dose of pyrazole in 0.9% saline solution at the same volume. All animals were weighed prior to and following each 4-day CIE cycle to ensure appropriate injection volumes, and to monitor possible CIE-induced changes in body weight.
Air and ethanol vapor were delivered to the exposure chambers at a rate of 10–12 liters per minute, ensuring adequate air-flow to meet the animals’ respiratory requirements. Ethanol was vaporized using a pressurized pump to push air through a porous diffusing stone submerged in a 1.0 liter bottle filled with 95% ethanol. To ensure ethanol vapor concentrations were within an appropriate range (10–12 mg/L) and stable across treatment days, 5.0 ml air samples were extracted from the ethanol chambers using a 60 ml syringe and mixed with 55.0 ml of ambient air. The diluted sample was injected into a breathalyzer (Lifeloc FC-10; Wheat Ridge, CO) and the resultant readings were compared to a standardized calibration curve of known ethanol concentration to determine chamber ethanol concentration.
Measurement of ethanol concentrations in tail blood
Blood ethanol concentrations (BECs) were measured in experimental animals at the termination of each 4-day CIE exposure. Briefly, each mouse was removed from the cage and gently placed in a plastic restraining tube, and a small (approximately 10 µl) blood sample was collected from the tip of the tail. Blood was collected directly into a heparinized capillary tube and centrifuged for 2 minutes to separate plasma from serum. BECs were determined from 5 µl plasma samples using an AM-1 alcohol analyzer (Analox Instruments, Lunenburg, MA).
Data analysis
Activity levels
Daily wheel-turns were recorded under both pre- and posttreatment conditions. The mean number of wheel-turns per day over the final five days prior to treatment was converted to distance traveled based on the diameter of the running wheel and used as a measure of baseline activity level for each animal. Daily posttreatment activity levels were first expressed as a percentage of an individual’s own baseline activity prior to averaging across animals, in order to reduce the effects of individual differences in activity level. An identical procedure was used to analyze daily water-tube licks in Experiment 1.
Circadian activity rhythms
Standard raster-style circadian actograms were generated using ClockLab software for visual inspection of activity patterns (cf. Figs. 1 and 2), and free-running periods were determined by the slope of straight lines fit to activity onsets with the assistance of ClockLab’s automated onset-detection feature. In addition, the initial phase of the free-running activity rhythm was determined by back-extrapolating the fitted line to the first day of DD following treatment. This method was utilized because mice exposed to ethanol treatment displayed significantly reduced running-wheel activity on the first few posttreatment days, and reliable activity onsets could not be determined.
Fig. 1.

Standard raster-style circadian actograms (double-plotted) of wheel-running (left) and water-tube licking activity (right) of representative individual C3H mice exposed to 1-cycle control (CONT) and CIE treatments (top and bottom panels, respectively). Arrows indicate beginning and end of treatment protocols. Shaded region denotes lights off. Note that wheel-running was recorded during treatment but that licking was not.
Fig. 2.
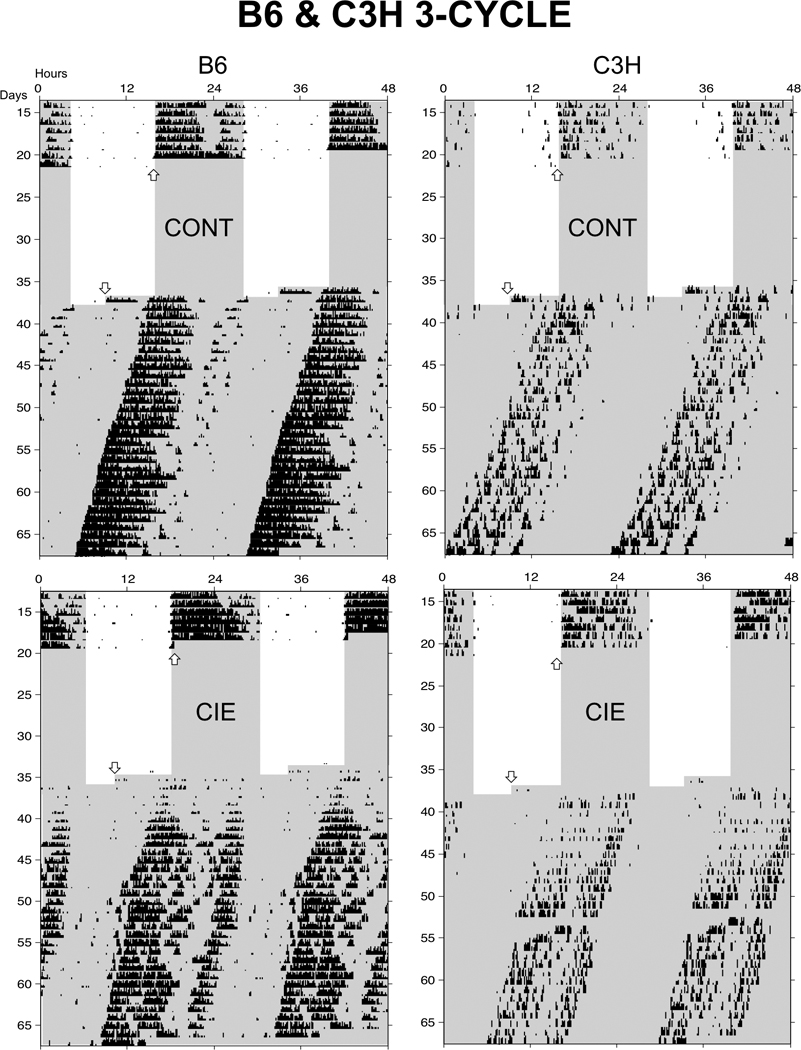
Standard raster-style circadian actograms (double-plotted) of representative B6 and C3H mice exposed to 3-cycle control (CONT; top) and CIE (bottom) treatments. Arrows indicate beginning and end of treatment protocols. Shaded region denotes lights off. Note that wheel-running activity was not recorded during treatment.
Body weights
Body weights were obtained in CIE and control animals at the beginning of the CIE protocol, after each 4-day CIE cycle, and several weeks later at the termination of behavioral testing. The effects of CIE on body weight were evaluated by computing percent body weight change from the beginning to the end of the protocol.
Statistics
Baseline activity levels were compared across strains, treatments (CIE vs. control) and experimental protocols (1-cycle vs. 3-cycle) using two separate 2-factor ANOVAs. The first analysis examined treatment and protocol effects in C3H mice and the second examined strain and treatment effects for the 3-cycle protocol. Posttreatment wheel-running and water-tube licking activities were analyzed using a 2-factor (groups by days) repeated-measures ANOVA, and Bonferroni-protected t-tests were used to compare CIE and control animals on individual posttreatment days when a groups-by-days interaction was detected. In addition, a series of Bonferroni-protected t-tests were conducted to determine which posttreatment days differed from the mean of the 5-day baseline for both CIE-exposed and control groups. Between-groups t-tests were used to compare free-running circadian period and initial phase between CIE and control animals within each experiment. Changes in BEC over the course of the 3-cycle CIE protocol (Experiment 2) were analyzed by 2-factor (strain by cycle number) ANOVA and Bonferonni protected t-tests were used to compare BEC between strains for each CIE cycle. Finally, pretreatment and terminal body weights, and posttreatment body weight change, were analyzed using 2-factor (strain by treatment) ANOVA at each measurement point.
Ethical considerations
The experimental procedures were approved by the University of Maine Institutional Animal Case and Use Committee (IACUC).
Results
Blood ethanol concentrations
Mean BEC for C3H mice following the 1-cycle CIE protocol (Experiment 1) was 231.39 ± 20.60 mg/dl. For Experiment 2, repeated-measures ANOVA revealed a strain by CIE cycle interaction on BECs (F2,60 = 5.967, P = 0.004) (Table 1). This interaction reflects the fact that BECs progressively increased over the 3 CIE cycles in B6 mice while C3H mice showed a drop in BECs between CIE cycles 2 and 3. Despite this significant interaction, Bonferroni-protected t-tests failed to detect significant strain differences following any of the 3 CIE cycles or in mean CIE levels averaged across CIE cycles.
Table 1.
Mean (± SEM) cycle-by-cycle and overall mean BECs for B6 and C3H mice exposed to a 3-cycle CIE protocol.
| Strain | Cycle 1 | Cycle 2 | Cycle 3 | Mean |
|---|---|---|---|---|
| B6 | 129.88 ± 18.67 | 140.51 ± 28.2 | 191.44 ± 15.46 | 153.94 ± 18.90 |
| C3H | 177.46 ± 18.53 | 194.27 ± 12.71 | 126.83 ± 19.92 | 166.18 ± 11.87 |
Wheel-running activity
Baseline activity
While C3H mice showed higher baseline activity levels in Experiment 1 than in Experiment 2 (F1,44 = 16.68, P < 0.001), there were no differences in baseline activity between treatment groups (Table 2). Similarly, while C3H mice were substantially less active at baseline than were B6 mice in Experiment 2 (F1,46 = 178.80, P < 0.001), there were no effects of treatment group (Table 2). Thus, CIE and control animals were well-matched for baseline activity levels in both experiments.
Experiment 1
Inspection of circadian actograms (Fig. 1) indicated that C3H mice showed substantial reductions in wheel-running activity both during and following 1-cycle CIE treatment, and that similar changes were not seen in air-exposed controls. Indeed, quantitative analysis of relative activity levels showed that CIE-exposed C3H mice were less active than controls for the duration of posttreatment activity recording, and that controls actually showed increased posttreatment activity (Fig. 3). Two-factor repeated measures ANOVA conducted on posttreatment activity revealed a significant main effect of treatment group (F1,16 = 12.180, P < 0.001), and a significant group by day interaction (F19,304= 2.627, P < 0.001), while Bonferroni-protected t-tests showed significant CIE-related activity reductions relative to air-exposed controls for days 1–7, 14 and 16. Further, activity levels in CIE animals were significantly reduced from baseline levels on all posttreatment days except days 12–14, while increases in activity levels seen in control animals were not significant for any posttreatment day. Taken together, these results indicate that activity levels in CIE-exposed animals gradually returned towards baseline (and control) levels over the 16 days of posttreatment recording. In contrast, inspection of actograms revealed no evidence for posttreatment alterations in water-tube contacts in either CIE or control groups (Fig. 1), and while repeated-measures ANOVA of posttreatment licking showed a significant main effect of posttreatment day (F19,304= 4.571, P < 0.001), there were no effects of treatment group or any group by day interaction (Fig. 3). Thus, CIE-induced reductions in locomotor activity were not accompanied by corresponding reductions in water-tube contacts.
Fig. 3.
Mean (±SEM) wheel-running (left) and water-tube licking (right), plotted relative to baseline levels (see text), following 1-cycle control and CIE treatments in C3H mice (Experiment 1). Dotted lines represent 100% baseline activity level. Asterisks (*, P < 0.05; **, P < 0.01) indicate days on which significant group differences were detected by Bonferroni-protected post-hoc tests.
Experiment 2
Inspection of circadian actograms indicated that B6 mice showed generally transient reductions in wheel-running activity, while C3H mice displayed more enduring activity reductions, following 3-cycle CIE treatment (Fig. 2). In B6 mice, two-factor repeated measures ANOVA revealed significant main effects of posttreatment day (F34,782= 5.094, P < 0.001) and treatment group (F1,23 = 2.888, P < 0.001), and a significant group by day interaction (F34,782= 1.508, P < 0.05), while Bonferroni-protected t-tests showed reduced activity in CIE-exposed relative to air-exposed B6 mice only for posttreatment days 1 and 2 (Fig. 4). Despite the fact that group differences were only detected for the first 2 posttreatment days, post-hoc comparisons showed that activity levels were significantly reduced in CIE-exposed animals relative to baseline on days 1–8, 10, 13, 19, 22 and 30, while activity levels were reduced in air-exposed animals relative to baseline on posttreatment days 4, 5 and 17.
Fig. 4.
Mean (±SEM) levels of wheel-running activity plotted relative to baseline levels following 3-cycle control and CIE treatments in B6 (left) and C3H (right) mice. Other conventions as in Fig. 3.
Analysis of posttreatment activity in C3H mice also revealed significant main effects of posttreatment day (F34,646 = 1.728, P < 0.01) and treatment group (F1,19 = 17.260, P < 0.001) and a significant group by day interaction (F34,646 = 1.473, P < 0.05) (Fig. 4). In contrast to the results for B6 mice, however, Bonferroni-protected post-hoc comparisons showed that activity levels in CIE-exposed and air-exposed C3H mice differed on posttreatment days 1–8, 15, 19–22, 26 and 28–30. Finally, post-hoc comparisons showed that activity levels in CIE-exposed C3H mice differed from baseline levels on all posttreatment days, while no changes from baseline activity were seen in control animals. Overall, these results indicate that both B6 and C3H mice showed reduced activity following the 3-cycle CIE protocol, but that these effects were generally more persistent in C3H than B6 animals.Circadian period and phase
While there were no significant effects on free-running circadian period following 1-cycle CIE treatment (Experiment 1), C3H mice displayed significantly shorter freerunning periods relative to air controls following the 3-cycle CIE protocol (Experiment 2) (t15 = 4.132, P < 0.001); similar effects were not seen in B6 mice (Fig. 5). Additionally, no differences were found for initial phase of activity onset in either experiment.
Fig. 5.
Mean (±SEM) free-running circadian period and initial phase in constant darkness (DD) following CIE protocols in B6 and C3H mice. Asterisks (***) indicate significant difference between CIE and CONT mice (P < 0.0001).
Body weights
There were no significant differences in pretreatment body weights between treatment groups in either Experiment 1 or 2 (Table 3). Further, there was no significant difference between the effects of 1-cycle CIE and control treatments on body weight change in C3H mice (Experiment 1). In contrast, there were significant changes in body weight in both C3H and B6 mice relative to air controls following 3-cycle CIE treatment (Experiment 2; main effect of treatment: F1,46 = 11.55, P = 0.001). While this treatment effect was no longer detectable at the end of the experiment, C3H mice were somewhat heavier than B6 mice at this time (F1,46 = 12.80, P < 0.001).
Table 3.
Mean (± SEM) initial body weight (BW) immediately prior to CIE or control (CON) treatment, percent change in body weight immediately following treatment, and body weight at the termination of behavioral recording 2–4 weeks later.
| Protocol | Strain | Group | Initial BW (g) | % BW Change | Terminal BW (g) |
|---|---|---|---|---|---|
| 1-cycle | C3H | CONT | 28.9 ± 0.92 | +0.78 ± 0.90 | 31.2 ± 0.40 |
| C3H | CIE | 29.4 ± 0.87 | − 0.33 ± 0.50 | 31.7 ± 0.87 | |
| 3-cycle | B6 | CONT | 27.0 ± 0.70 | −0.45 ± 0.45 | 28.9 ± 0.55 |
| B6 | CIE | 26.8 ± 0.97 | −5.49 ± 2.38* | 28.6 ± 0.51 | |
| C3H | CONT | 28.2 ± 0.89 | +2.23 ± 1.18 | 30.9 ± 0.99# | |
| C3H | CIE | 27.9 ± 0.74 | −3.80 ± 2.34* | 32.9 ± 1.22# |
Asterisk (*) indicates significant differences between CIE and CONT animals (P < 0.05), whereas pound sign (#) indicates significant differences between C3H and B6 mice (P < 0.001).
Discussion
The present results indicate that mice exhibit strain-dependent alterations in running-wheel activity following exposure to chronic intermittent ethanol vapor. As in our previous report employing a 1-cycle CIE protocol (Logan et al., 2010), B6 mice displayed a relatively transient period of reduced running-wheel activity following the 3-cycle CIE exposure (Experiment 2). In marked contrast, C3H mice displayed enduring locomotor hypoactivity that persisted throughout posttreatment recording (16 days in Experiment 1, 30 days in Experiment 2) following both 1-cycle and 3-cycle CIE treatments.. In addition, C3H mice displayed a significant shortening of free-running circadian period following the 3-cycle CIE protocol that was not observed in B6 mice. In principle, strain differences in these or other withdrawal-related responses could be related to genetic differences in initial ethanol metabolism and/or development of metabolic tolerance. Indeed, B6 and C3H mice in the present study showed opposite changes in BECs across repeated treatment cycles, and C3H mice displayed somewhat lower BECs than B6 mice at the time of final withdrawal. Nevertheless, since BECs are generally positively correlated with withdrawal severity while tolerance is negatively correlated with withdrawal severity (Crabbe et al., 1983; Metten and Crabbe, 2005), pharmacokinetic differences are unlikely to account for the strain differences in locomotor and circadian responses to ethanol withdrawal observed here. Thus, while additional genotypes will need to be examined in future experiments, these results indicate that locomotor and circadian responses to ethanol withdrawal, like many other ethanol-related phenotypes (Crabbe, 2008), are subject to significant genetic influence.
As described above, previous studies have established that various responses to ethanol withdrawal are potentiated by an extended history of prior withdrawals. While our studies have revealed qualitatively similar effects on locomotor activity following 1-cycle and 3-cycle CIE protocols (Logan et al., 2010; present data), careful analysis of the present data suggests that more enduring effects may occur following the more extensive 3-cycle CIE treatment. Thus, C3H mice exhibited about 80% of baseline activity levels by 2 weeks following the 1-cycle CIE treatment, but only about 50% of baseline activity levels at 4 weeks following the 3-cycle treatment. It must be noted, however, that while not significant for the group as a whole, several air-exposed animals showed dramatic increases in relative activity following the control procedure, especially in Experiment 1, seriously complicating this comparison. While the reasons for increased activity in air-only controls cannot be discerned at present, control animals in these experiments are maintained for several days in a novel environment (i.e., the vapor chambers) and subjected to repeated daily pyrazole injections, procedures that cannot be assumed to be behaviorally “neutral”. Whatever the ultimate explanation, further research will be required to clarify the possible effects of CIE exposure history on withdrawal-induced hypolocomotion.
Post-withdrawal reductions in locomotor activity could be secondary to general withdrawal-induced malaise or lethargy. Indeed, ethanol vapor-exposed mice generally display modest reductions in food intake (Kliethermes et al., 2005) and body weight; Kliethermes et al., 2005; present study). In the present study, significant weight loss was seen following the 3-cycle but not the 1-cycle protocol, and while assessment of post-CIE water-tube contacts in Experiment 1 showed no evidence for a generalized reduction in ingestive behavior, we did not measure this parameter in Experiment 2. On the other hand, terminal body weights did not differ between CIE and control animals even in Experiment 2, in which significant reductions in locomotor activity persisted throughout the duration of the experiment. Thus, while we cannot rule out a possible role for general malaise or lethargy, we do not believe that CIE-induced reductions in running-wheel activity reflect a persisting illness-like state.
Instead, we believe that reduced running-wheel activity in CIE-exposed mice may reflect the specific motivational significance of this behavior. Running-wheel activity is intrinsically rewarding (Sherwin, 1998; Lett et al., 2000; de Visser et al., 2007), has antidepressant and anxiolytic effects (Duman et al., 2008; Salam et al., 2009), and is genetically distinct from other forms of locomotor activity (Rosenwasser et al., 1996; Bronikowski et al., 2001). Further, Kliethermes et al. (2005) previously showed that ethanol vapor withdrawal produces only very transient effects on general home-cage ambulation, even in a line of mice selectively bred for high levels of ethanol withdrawal sensitivity. Thus, the long-term reductions in locomotor activity seen in the present study may be specific to running-wheel activity and possibly other forms of motivated reward-seeking. Thus, assessment of running-wheel activity may provide a simple and non-invasive alternative to the use of electrical brain stimulation as a measure of reward-seeking behavior in animal studies of ethanol and drug withdrawal (e.g., Schulteis et al., 1995; Koob et al., 2004; Schulteis and Liu, 2006; Rylkova et al., 2009).
The present results also contribute to a rapidly growing literature on the chronobiology of ethanol. Recent studies demonstrate that chronic ethanol intake alters free-running circadian period and responsiveness to phase-shifting stimuli in rats, mice and hamsters (Mistlberger and Nadeau, 1992; Rosenwasser et al., 2005a, 2005b; Seggio et al., 2007, 2009; Ruby et al., 2009; Brager et al., 2010), and that these findings are mediated in part by direct pharmacological effects on the suprachiasmatic nucleus (SCN) circadian pacemaker (Prosser et al., 2008; Prosser and Glass, 2009; Ruby et al., 2009; McElroy et al., 2009). Previous experiments also indicate that effects of ethanol on the circadian pacemaker persist during acute withdrawal, and may show longer-term persistence in some animals following voluntary or forced ethanol drinking (Rosenwasser et al., 2005a; Seggio et al., 2009; Brager et al., 2010). The present results provide the first evidence for persisting effects of ethanol withdrawal on free-running circadian period. Nevertheless, since such effects were observed only in the C3H strain, and only following the repeated CIE protocol, the occurrence of chronobiological alterations during extended abstinence may be dependent on both genetic susceptibility and an extended history of ethanol exposure and/or withdrawal.
Little is known concerning possible relationships between the physiological disruptions associated with acute withdrawal (such as seizures) and the more persisting disturbances sometimes seen during protracted abstinence (Heilig et al., 2010). This is an important question with potential implications for the clinical management of both acute and persisting withdrawal symptoms, and possibly for the prevention of relapse. Previous studies of withdrawal-induced seizures have commonly employed the C3H strain (Becker and Hale, 1993; Becker et al., 1997), and indeed, studies of strain differences in HIC susceptibility during acute ethanol withdrawal indicate that B6 mice are rather resistant to ethanol-withdrawal seizures relative to the more susceptible C3H mice (Metten and Crabbe, 2005; but see Metten et al., 2010). Our results indicate that C3H and B6 mice also exhibit high and low sensitivity to withdrawal-induced hypolocomotion, suggesting a possible genetic correlation between these responses. Of course, additional studies testing a wider range of genotypes would be required to determine whether significant genetic correlations exist between acute and protracted responses to ethanol withdrawal across disparate behavioral domains.
While most alcoholics exhibit relatively rapid resolution of withdrawal-associated behavioral and chronobiological disturbances, some patients can display persistent alterations in these functions over the course of protracted abstinence (De Soto et al., 1985; Dackis et al., 1986; Drummond et al., 1998; Landolt and Gillin, 2001). Further, persistent affective (Greenfield et al., 1998; Hasin et al., 2002) and chronobiological (Brower, 2001, 2003) disturbances have been linked to increased risk of relapse. Therefore, the model for long-term withdrawal-induced behavioral disruption developed in the present study may prove useful for elucidating risk factors for relapse, and possibly for development of new chronobiological and/or exercise-based interventions for mitigation of these risk factors.
To conclude, we have developed a novel experimental model of long-term behavioral and chronobiological disruption associated with alcohol dependence. We believe that this model could serve as a useful platform for subsequent genetic, pharmacological and neurobiological studies of the long-term consequences of ethanol dependence and withdrawal.
Acknowledgement
Supported by a subcontract to AMR from the Integrative Neuroscience Initiative on Alcoholism (INIA-Stress), NIAAA U01 AA13641, Kathy Grant, Principal Investigator.
References
- Becker HC, Diaz-Granados JL, Weathersby RT. Repeated ethanol withdrawal experience increases the severity and duration of subsequent withdrawal seizures in mice. Alcohol. 1997;14(4):319–326. doi: 10.1016/s0741-8329(97)87949-9. [DOI] [PubMed] [Google Scholar]
- Becker HC, Hale RL. Repeated episodes of ethanol withdrawal potentiates the severity of subsequent withdrawal seizures: an animal model of alcohol withdrawal "kindling". Alcohol Clin Exp Res. 1993;17(1):94–98. doi: 10.1111/j.1530-0277.1993.tb00731.x. [DOI] [PubMed] [Google Scholar]
- Becker HC, Lopez MF. Increased ethanol drinking after repeated chronic ethanol exposure and withdrawal experience in C57BL/6 mice. Alcohol Clin Exp Res. 2004;28(12):1829–1838. doi: 10.1097/01.alc.0000149977.95306.3a. [DOI] [PubMed] [Google Scholar]
- Brager AJ, Ruby CL, Prosser RA, Glass JD. Chronic ethanol disrupts circadian photic entrainment and daily locomotor activity in the mouse. Alcohol Clin Exp Res. 2010;34(7):1266–1273. doi: 10.1111/j.1530-0277.2010.01204.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bronikowski AM, Carter PA, Swallow JG, Girard IA, Rhodes JS, Garland T., Jr Open-field behavior in house mice selectively bred for high voluntary wheel-running. Behav Genet. 2001;31(3):309–316. doi: 10.1023/a:1012283426530. [DOI] [PubMed] [Google Scholar]
- Brower KJ. Insomnia, alcoholism and relapse. Sleep Med Rev. 2003;7(6):523–539. doi: 10.1016/s1087-0792(03)90005-0. [DOI] [PubMed] [Google Scholar]
- Brower KJ, Aldrich MS, Robinson EA, Zucker RA, Greden JF. Insomnia, self-medication, and relapse to alcoholism. Am J Psychiat. 2001;158(3):399–404. doi: 10.1176/appi.ajp.158.3.399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clapp P, Bhave SV, Hoffman PL. How adaptation of the brain to alcohol leads to dependence: a pharmacological perspective. Alcohol Res Health. 2008;31(4):310–339. [PMC free article] [PubMed] [Google Scholar]
- Crabbe JC. Neurogenetic studies of alcohol addiction. Phil Trans Royal Soc B. 2008;363:3201–3211. doi: 10.1098/rstb.2008.0101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crabbe JC, Young ER, Kosobud A. Genetic correlations with ethanol withdrawal severity. Pharmacol Biochem Behav. 1983;18(S1):541–547. doi: 10.1016/0091-3057(83)90233-2. [DOI] [PubMed] [Google Scholar]
- Dackis CA, Gold MS, Pottash AL, Sweeney DR. Evaluating depression in alcoholics. Psychiatry Res. 1986;17(2):105–109. doi: 10.1016/0165-1781(86)90065-x. [DOI] [PubMed] [Google Scholar]
- Dahchour A, De Witte P. Acamprosate decreases the hypermotility during repeated ethanol withdrawal. Alcohol. 1999;18(1):77–81. doi: 10.1016/s0741-8329(98)00071-8. [DOI] [PubMed] [Google Scholar]
- de Visser L, van den Bos R, Stoker AK, Kas MJ, Spruijt BM. Effects of genetic background and environmental novelty on wheel running as a rewarding behaviour in mice. Behav Brain Res. 2007;177(2):290–297. doi: 10.1016/j.bbr.2006.11.019. [DOI] [PubMed] [Google Scholar]
- De Soto CB, O'Donnell WE, Allred LJ, Lopes CE. Symptomatology in alcoholics at various stages of abstinence. Alcohol Clin Exp Res. 1985;9(6):505–512. doi: 10.1111/j.1530-0277.1985.tb05592.x. [DOI] [PubMed] [Google Scholar]
- Drummond SP, Gillin JC, Smith TL, DeModena A. The sleep of abstinent pure primary alcoholic patients: natural course and relationship to relapse. Alcohol Clin Exp Res. 1998;22(8):1796–1802. [PubMed] [Google Scholar]
- Duman CH, Schlesinger L, Russell DS, Duman RS. Voluntary exercise produces antidepressant and anxiolytic behavioral effects in mice. Brain Res. 2008;1199:148–158. doi: 10.1016/j.brainres.2007.12.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Getachew B, hauser SR, Taylor RE, Tizabi Y. Desipramine blocks alcohol-induced anxiety- and depressive-like behaviors in two rat strains. Pharmacol Biochem Behav. 2008;91:97–103. doi: 10.1016/j.pbb.2008.06.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greenfield SF, Weiss RD, Muenz LR, Vagge LM, Kelly JF, Bello LR, Michael J. The effect of depression on return to drinking: a prospective study. Arch Gen Psychiatry. 1998;55(3):259–265. doi: 10.1001/archpsyc.55.3.259. [DOI] [PubMed] [Google Scholar]
- Griffiths PJ, Littleton JM, Ortiz A. Changes in monoamine concentrations in mouse brain associated with ethanol dependence and withdrawal. Brit J Pharmacol. 1974;50:489–498. doi: 10.1111/j.1476-5381.1974.tb08582.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasin DS, Goodwin RD, Stinson FS, Grant BF. Epidemiology of major depressive disorder: results from the National Epidemiologic Survey on Alcoholism and Related Conditions. Arch Gen Psychiatry. 2005;62(10):1097–1106. doi: 10.1001/archpsyc.62.10.1097. [DOI] [PubMed] [Google Scholar]
- Heilig M, Egli M, Crabbe JC, Becker HC. Acute withdrawal, protracted abstinence and negative affect in alcoholism: are they linked? Addiction Biology. 2010;15(2):169–184. doi: 10.1111/j.1369-1600.2009.00194.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kliethermes CL, Cronise K, Crabbe JC. Anxiety-like behavior in mice in two apparatuses during withdrawal from chronic ethanol vapor inhalation. Alcohol Clin Exp Res. 2004;28(7):1012–1019. doi: 10.1097/01.alc.0000131976.40428.8f. [DOI] [PubMed] [Google Scholar]
- Kliethermes CL, Cronise K, Crabbe JC. Home cage activity and ingestive behaviors in mice following chronic ethanol vapor inhalation. Physiol Behav. 2005;85(4):479–488. doi: 10.1016/j.physbeh.2005.05.009. [DOI] [PubMed] [Google Scholar]
- Koob GF, Ahmed SH, Boutrel B, Chen SA, Kenny PJ, Markou A, O'Dell LE, Parsons LH, Sanna PP. Neurobiological mechanisms in the transition from drug use to drug dependence. Neurosci Biobehav Rev. 2004;27(8):739–749. doi: 10.1016/j.neubiorev.2003.11.007. [DOI] [PubMed] [Google Scholar]
- Koob GF, Le Moal M. Addiction and the brain antireward System. Ann Rev Psychol. 2008;59(1):29–53. doi: 10.1146/annurev.psych.59.103006.093548. [DOI] [PubMed] [Google Scholar]
- Landolt HP, Gillin JC. Sleep abnormalities during abstinence in alcohol-dependent patients. Aetiology and management. CNS Drugs. 2001;15(5):413–425. doi: 10.2165/00023210-200115050-00006. [DOI] [PubMed] [Google Scholar]
- Lett BT, Grant VL, Byrne MJ, Koh MT. Pairings of a distinctive chamber with the aftereffect of wheel running produce conditioned place preference. Appetite. 2000;34(1):87–94. doi: 10.1006/appe.1999.0274. [DOI] [PubMed] [Google Scholar]
- Logan RW, Seggio JA, Robinson SL, Richard GR, Rosenwasser AM. Circadian wheel-running activity during withdrawal from chronic intermittent ethanol exposure in mice. Alcohol. 2010;44(3):239–244. doi: 10.1016/j.alcohol.2010.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lopez M, Becker H. Effect of pattern and number of chronic ethanol exposures on subsequent voluntary ethanol intake in C57BL/6J mice. Psychopharmacol. 2005;181(4):688–696. doi: 10.1007/s00213-005-0026-3. [DOI] [PubMed] [Google Scholar]
- McElroy B, Zakaria A, Glass JD, Prosser RA. Ethanol modulates mammalian circadian clock phase resetting through extrasynaptic gaba receptor activation. Neurosci. 2009;164(2):842–848. doi: 10.1016/j.neuroscience.2009.08.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mehta AK, Ticku MK. Ethanol- and diazepam-withdrawal hyperlocomotion is not due to 5-HT3 receptor stimulation. Pharmacol Biochem Behav. 1993;45(3):755–757. doi: 10.1016/0091-3057(93)90537-4. [DOI] [PubMed] [Google Scholar]
- Metten P, Crabbe JC. Alcohol withdrawal severity in inbred mouse (Mus musculus) strains. Behav Neurosci. 2005;119(4):911–925. doi: 10.1037/0735-7044.119.4.911. [DOI] [PubMed] [Google Scholar]
- Metten P, Sorensen ML, Cameron AJ, Yu CH, Crabbe JC. Withdrawal severity after chronic intermittent ethanol in inbred mouse strains. Alcohol Clin Exp Res. 2010;34(9):1552–1564. doi: 10.1111/j.1530-0277.2010.01240.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mistlberger RE, Nadeau J. Ethanol and circadian rhythms in the Syrian hamster: effects on entrained phase, reentrainment rate, and period. Pharmacol Biochem Behav. 1992;43(1):159–165. doi: 10.1016/0091-3057(92)90652-v. [DOI] [PubMed] [Google Scholar]
- O'Dell LE, Roberts AJ, Smith RT, Koob GF. Enhanced alcohol self-administration after intermittent versus continuous alcohol vapor exposure. Alcohol Clin Exp Res. 2004;28(11):1676–1682. doi: 10.1097/01.alc.0000145781.11923.4e. [DOI] [PubMed] [Google Scholar]
- Overstreet DH, Knapp DJ, Breese GR. Accentuated decrease in social interaction in rats subjected to repeated ethanol withdrawals. Alcohol Clin Exp Res. 2002;26(8):1259–1268. doi: 10.1097/01.ALC.0000023983.10615.D7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prosser RA, Glass JD. The mammalian circadian clock exhibits acute tolerance to ethanol. Alcohol Clin Exp Res. 2009;33(12):2088–2093. doi: 10.1111/j.1530-0277.2009.01048.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prosser RA, Mangrum CA, Glass JD. Acute ethanol modulates glutamatergic and serotonergic phase shifts of the mouse circadian clock in vitro. Neurosci. 2008;152(3):837–848. doi: 10.1016/j.neuroscience.2007.12.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenwasser AM, Pellowski MW, Hendley ED. Circadian timekeeping in hyperactive and hypertensive inbred rat strains. Am J Physiol. 1996;271:R787–R796. doi: 10.1152/ajpregu.1996.271.3.R787. [DOI] [PubMed] [Google Scholar]
- Rosenwasser AM, Fecteau ME, Logan RW, Reed JD, Cotter SJ, Seggio JA. Circadian activity rhythms in selectively bred ethanol-preferring and nonpreferring rats. Alcohol. 2005;36(2):69–81. doi: 10.1016/j.alcohol.2005.07.001. [DOI] [PubMed] [Google Scholar]
- Rosenwasser AM, Logan RW, Fecteau ME. Chronic ethanol intake alters circadian period-responses to brief light pulses in rats. Chronobiol Internat. 2005;22(2):227–236. doi: 10.1081/cbi-200053496. [DOI] [PubMed] [Google Scholar]
- Ruby CL, Prosser RA, Depaul MA, Roberts RJ, Glass JD. Acute ethanol impairs photic and nonphotic circadian phase resetting in the Syrian hamster. Am J Physiol Regul Integr Comp Physiol. 2009;296(2):R411–R418. doi: 10.1152/ajpregu.90782.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rylkova D, Shah HP, Small E, Bruijnzeel AW. Deficit in brain reward function and acute and protracted anxiety-like behavior after discontinuation of a chronic alcohol liquid diet in rats. Psychopharmacol (Berl) 2009;203(3):629–640. doi: 10.1007/s00213-008-1409-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salam JN, Fox JH, Detroy EM, Guignon MH, Wohl DF, Falls WA. Voluntary exercise in C57 mice is anxiolytic across several measures of anxiety. Behav Brain Res. 2009;197(1):31–40. doi: 10.1016/j.bbr.2008.07.036. [DOI] [PubMed] [Google Scholar]
- Schulteis G, Liu J. Brain reward deficits accompany withdrawal (hangover) from acute ethanol in rats. Alcohol. 2006;39(1):21–28. doi: 10.1016/j.alcohol.2006.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schulteis G, Markou A, Cole M, Koob GF. Decreased brain reward produced by ethanol withdrawal. Proc Nat Acad Sci USA. 1995;92(13):5880–5884. doi: 10.1073/pnas.92.13.5880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seggio JA, Fixaris MC, Reed JD, Logan RW, Rosenwasser AM. Chronic ethanol intake alters circadian phase shifting and free-running period in mice. J Biol Rhythms. 2009;24(4):304–312. doi: 10.1177/0748730409338449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seggio JA, Logan RW, Rosenwasser AM. Chronic ethanol intake modulates photic and non-photic circadian phase responses in the Syrian hamster. Pharmacol Biochem Behav. 2007;87(3):297–305. doi: 10.1016/j.pbb.2007.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sherwin CM. Voluntary wheel running: a review and novel interpretation. Anim Behav. 1998;56(1):11–27. doi: 10.1006/anbe.1998.0836. [DOI] [PubMed] [Google Scholar]
- Slawecki CJ, Roth R. Comparison of the onset of hypoactivity and anxiety-like behavior during alcohol withdrawal in adolescent and adult rats. Alcohol Clin Exp Res. 2004;28(4):598–607. doi: 10.1097/01.alc.0000122767.69206.1b. [DOI] [PubMed] [Google Scholar]
- Stevenson JR, Schroeder JP, Nixon K, Besheer J, Crews FT, Hodge CW. Abstinence following alcohol drinking produces depression-like behavior and reduced hippocampal neurogenesis in mice. Neuropsychopharmacol. 2009;34(5):1209–1222. doi: 10.1038/npp.2008.90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uzbay IT, Kayaalp SO. A modified liquid diet of chronic ethanol administration: validation by ethanol withdrawal syndrome in rats. Pharmacological Res. 1995;31(1):37–42. doi: 10.1016/1043-6618(95)80045-x. [DOI] [PubMed] [Google Scholar]
- Valdez GR, Roberts AJ, Chan K, Davis H, Brennan M, Zorrilla EP, Koob GF. Increased ethanol self-administration and anxiety-like behavior during acute ethanol withdrawal and protracted abstinence: regulation by corticotropin-releasing factor. Alcohol Clin Exp Res. 2002;26(10):1494–1501. doi: 10.1097/01.ALC.0000033120.51856.F0. [DOI] [PubMed] [Google Scholar]
- Valdez GR, Zorrilla EP, Roberts AJ, Koob GF. Antagonism of corticotropin-releasing factor attenuates the enhanced responsiveness to stress observed during protracted ethanol abstinence. Alcohol. 2003;29(2):55–60. doi: 10.1016/s0741-8329(03)00020-x. [DOI] [PubMed] [Google Scholar]
- Walker BM, Drimmer DA, Walker JL, Liu T, Mathé AA, Ehlers CL. Effects of prolonged ethanol vapor exposure on forced swim behavior, and neuropeptide Y and corticotropin-releasing factor levels in rat brains. Alcohol. 2010;44(6):487–493. doi: 10.1016/j.alcohol.2010.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao Y, Weiss F, Zorrilla EP. Remission and resurgence of anxiety-like behavior across protracted withdrawal stages in ethanol-dependent rats. Alcohol Clin Exp Res. 2007;31(9):1505–1515. doi: 10.1111/j.1530-0277.2007.00456.x. [DOI] [PubMed] [Google Scholar]





