Abstract
Prohibitins are members of a highly conserved eukaryotic protein family containing the stomatin/prohibitin/flotillin/HflK/C (SPFH) domain [also known as the prohibitin (PHB) domain] found in divergent species from prokaryotes to eukaryotes. Prohibitins are found in unicellular eukaryotes, fungi, plants, animals and humans. Prohibitins are ubiquitously expressed and present in multiple cellular compartments including the mitochondria, nucleus, and the plasma membrane, and shuttles between the mitochondria, cytosol and nucleus. Multiple functions have been attributed to the mitochondrial and nuclear prohibitins, including cellular differentiation, anti-proliferation, and morphogenesis. In the present review, we focus on the recent developments in prohibitins research related to folliculogenesis. Based on current research findings, the data suggest that these molecules play important roles in modulating specific responses of granulose cells to follicle stimulating hormone (FSH) by acting at multiple levels of the FSH signal transduction pathway. Understanding the molecular mechanisms by which the intracellular signaling pathways utilize prohibitins in governing folliculogenesis is likely to result in development of strategies to overcome fertility disorders and suppress ovarian cancer growth.
Keywords: Prohibitins, PHB, PHB2, Repressor Of Estrogen Receptor Action, REA, mitochondria, nucleus, Review
2. INTRODUCTION
Understanding the mechanisms underlying the growth and development of a competent follicle with the capacity to release a fertilizable oocyte is a major goal of reproductive biology, particularly, since female infertility is clearly a major public health issue. Ovarian granulosa cells (GCs) play an important physiological role in supporting the development and selection of the dominant follicle by controlling oocyte maturation and by producing the steroid hormones, estrogen (E2) and progesterone (P4) that are critical for maintenance of the ovarian cycle. Mammalian ovarian follicular development is tightly regulated by crosstalk between cell death, survival and differentiation signals (1). Studies performed in our laboratory have demonstrated that survival/apoptotic events in GCs are mediated by a family of protein factors known as prohibitin (PHB; B-cell receptor associated protein-32, BAP-32; and prohibitin 2/repressor of estrogen receptor activity, PHB2, REA, BAP-37) (1-6). Based on current research findings, the data suggest that these molecules play important roles in modulating specific responses of GCs to follicle stimulating hormone (FSH) by acting at multiple levels of the FSH signal transduction pathway. Understanding the molecular mechanisms by which the intracellular signaling pathways utilize prohibitins in governing folliculogenesis is likely to result in development of strategies to overcome fertility disorders and suppress ovarian cancer growth. In the last decade, significant progress has been made in analyzing the expression, distribution and in determining the specific roles that the PHB and REA play in mammalian reproductive physiology. In the present review, we summarize some of the recent developments in PHB and REA research related to folliculogenesis.
3. PROHIBITINS
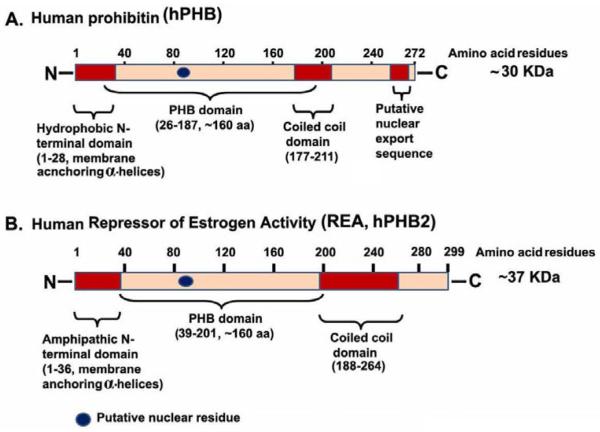
Prohibitins are members of an extensive evolutionarily conserved family of proteins which includes stomatins, plasma membrane proteins of Escherichia coli (HflKC), flotillins, the human insulin receptor (HIR) proteins and plant defense proteins (7). Two highly homologous members of prohibitins expressed in eukaryotes are PHB and REA. Although, initially associated with inhibition of cell proliferation, (hence the name “prohibitin”), PHB and REA appear to have an increasing array of functional roles (8). In humans, the PHB gene (hPhb) maps to chromosome 17q21.1 and spans ~11 kb with 7 exons (9). The human and rodent Phb genes are similar except for introns 2 and 3, which are approximately 1 kb larger in the rat gene (10). The human and rodent Phb gene encode approximately 30 kDa proteins that have a single amino acid difference (Figure 1) (8). The human Phb2 (11,12) is located on chromosome 12p13 (13) and spans approximately 5.3 kb with 10 exons, have smaller introns than Phb, and codes a protein of ~37 kDa (Figure 1). The PHB and REA proteins are 54% homologous (8) and found associated in a high molecular weight regulatory complex (14-21). Orthologues of the prohibitin gene have been identified in several organisms including bacteria (23-25), plants (26,27), Trypanosoma brucei (28), yeast (29-31), and Drosophila (32). While studies have shown that PHB and REA null yeast strains have reduced lifespan, it appears that higher eukaryotes are more dependent upon expression of their Phb and Phb2 genes, suggesting enhanced biological functions. The Phb orthologues in Drosophila are essential for normal development and differentiation during larvae to pupae metamorphosis (32). Genetic deletion of the Phb and Phb2/REA genes in mice are lethal before embryonic day 6.5, however, heterozygous mice show no appreciable defects in fertility (33), implying that these proteins play a critical role in the early stages of development in vertebrates. Furthermore, in C. elegans, Phb is required for differentiation in the larval gonad (34), indicating that both PHB and REA play essential reproductive roles that have yet to be elucidated.
Figure 1.
A. Schematic domain representation of human prohibitin (hPHB) protein. B. Schematic domain representation of human repressor of estrogen activity (REA/hPHB2) protein. N - amino terminal; C - carboxy terminal.
4. GONADOTROPINS SYNERGIZE WITH ANDROGENS IN INDUCTION OF PROHIBITINS
The gonadotropins, FSH and luteinizing hormone (LH) play critical and complementary roles in the control of mammalian reproduction. In female mammals, FSH stimulates ovarian follicle growth and maturation, as well as E2 synthesis by GCs, whereas LH stimulates androgen production by theca cells and ovulation of the dominant follicle(s). The entire differentiation paradigm induced by FSH receptor activation in GCs in the intact animal can be duplicated by culturing primary GCs in the presence of FSH on an extracellular matrix under serum-free tissue culture conditions that mimick the avascular milieu within the follicle (35). Addition of FSH with testosterone (T) to rat GCs initiates a differentiation program which is completed within 48-72h (35). Our laboratory has taken advantage of these “physiological” culture conditions and has utilized a proteomic approach to identify and characterize PHB as a 28 kDa intracellular protein (p28) that is expressed during differentiation of rat GCs from preantral and early antral follicles (2-5). We have also demonstrated that increased PHB expression occurs during the transitional stages of rat ovarian follicular differentiation and have shown that PHB expression in GCs is not uniform across all stages of follicular development in vivo. These experimental observations suggest that PHB may be involved in GC proliferation as well as differentiation depending on the intensity of PHB signaling.
5. SUBCELLULAR LOCALIZATION OF PROHIBITINS
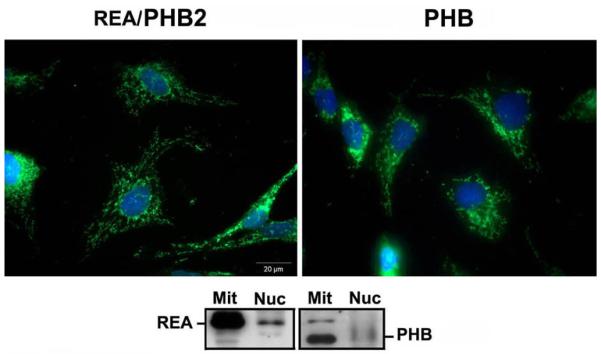
PHB and REA are present in multiple cellular compartments, suggesting that they play additional roles to those of chaperone proteins in the mitochondria (8,16,17), and their ability to target to lipid rafts. There is compelling evidence showing that both PHB and REA are localized in the nucleus and can modulate transcriptional activity by interacting with various transcription factors, either directly or through their interactions with chromatin remodeling proteins (18-22). In support of the above findings, recently published data have shown that both PHB and REA are located in both the mitochondria and nucleus of rat GCs (4,6,36, Figure 2).
Figure 2.
A. Localization by immunofluorescence of REA (left panel, green) and PHB (right panel, green) in GCs treated with FSH+T. B. GST-pull down assays for REA and PHB complexes. Mitochondrial and nuclear fractions of GCs treated with FSH+T (24h) were incubated with GST fusion proteins of REA and PHB, pre-conjugated to glutathione-Sepharose beads, and pull down, electrophoresed, blotted and probed with PHB and REA antibodies.
Experimental studies on the expression and distribution pattern of PHB and REA in various reproductive tissues of different species are limited and have been restricted to mice, rats, pigs and humans (8). Distinct differences in PHB and REA levels have been observed during mammalian folliculogenesis. Immunolocalization of PHB in rat ovarian sections showed that PHB is expressed differentially in GCs, theca interstitial cells and the oocyte (4). As follicles develop toward early or large antral stages, the theca-interstitial cells show more intense PHB expression, whereas, a varying pattern of PHB was observed in the GCs of early and large antral follicles. Interestingly, not all GCs from early antral follicle expressed PHB (4). Additionally, PHB was readily detectable in normal germinal vesicle (GV)- stage oocytes, albeit at low levels. A distinct, speckled pattern of PHB immunoreactivity was detected in the GV-stage oocytes that also showed abnormal condensation of chromatin, suggestive of atresia. Association of PHB with oocyte chromatin was not seen in metaphase II oocytes after in vitro or in vivo maturation. Similarly, neither the male nor the female pronuclei of zygotes fertilized in vitro displayed significant PHB immunoreactivity. However, the zygotes and embryos arrested at the 2-cell stage after the apoptosis-inducing heat shock treatment displayed the pattern of nuclear PHB accumulation similar to that seen in defective GV-stage oocytes. This nuclear expression pattern was more pronounced in the embryos that failed to develop normally after nulcear tranfer (NT). In contrast, low levels of nuclear immunoreactivity were observed in embryos that reached the blastocyst stage at Day 5–6 after NT or in vitro fertilization and displayed an acceptable morphology (4). Furthermore, in studies performed on rat testes, there is a pattern of strong expression for the Phb gene in the adult Leydig and Sertoli cells (37). In contrast, the PHB is not detected in rat spermatogonia and spermatocytes undergoing mitotic and meiotic divisions (37).
In contrast to Phb gene expression, only few studies have been performed to analyze the expression and distribution pattern of REA in reproductive tissues of humans and rodents, and non-mammalian species (38). Recently published reports have shown the expression of REA in mouse uteri and rainbow trout (Oncoryncus mykiss) eggs, suggesting the possibility that REA may play a fundamental role in these developmental processes (38,39). In fact, REA mRNA and protein levels in uteri of heterozygous animals were half that of the wild type, and studies with heterozygous animals revealed a greater uterine weight gain and epithelial hyperproliferation in response to E2 and a substantially greater stimulation by E2 of a number of estrogen up-regulated genes in the uterus (39).
Subcellular fractionation of rat ovarian GCs followed by two-dimensional (2-D) Western blot analyses performed in our laboratory showed that PHB is present as two isoforms (2,4,6). In the mitochondrial fraction, two PHB 30-kDa protein spots were detected with isoelectric points of 5.6 and 5.8, whereas only one 30-kDa protein spot with an isoelectric point of 5.8 was observed in the nuclear fraction. Interestingly, no detectable spots were identified in the cytosolic fraction in FSH and FSH + T stimulated GCs (2,4,6).
6. ROLE OF PROHIBITINS IN THE INHIBITION OF CELL PROLIFERATION
Cell proliferation in the ovary is critical for sustaining ovarian function since the release of germ cells (oocytes) and production of hormones are required for reproduction. During the development of oocytes, folliculogenesis is accompanied by significant proliferation that deletion of REA leads to the loss of both PHB and REA proteins and impairs cell proliferation of mouse embryonic fibroblasts (MEFs) (40). These studies suggest that prohibitins are required for the balance between proliferation vs differentiation, but whether similar molecular mechanisms exist in GCs is not currently known (18,19,41).
7. ROLE OF PROHIBITINS IN DIFFERENTIATION
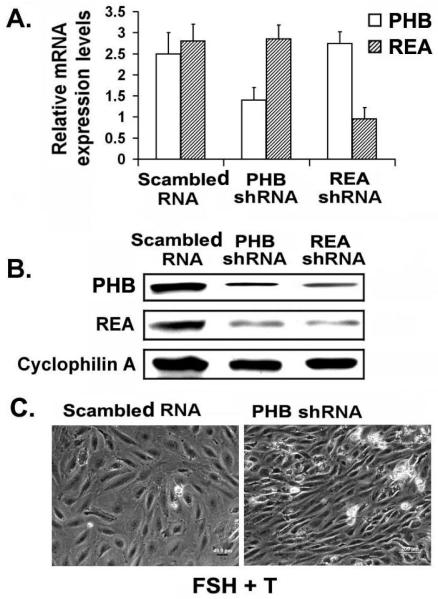
Studies from our laboratory have demonstrated that the increased expression of PHB that occurs during follicular development (3,4) correlates with GC differentiation and changes in mitochondrial structure and function (4-6). We found that PHB expression in ovarian tissue is age- and stage-regulated, suggesting a growth regulatory role for PHB in the ovary (3,4). PHB expression which is associated with differentiation, is up-regulated by gonadotropins, and is low during GC proliferation. In order to study the specific roles that the prohibitins play in GC differentiation, we employed adenoviral-mediated RNA interference methodology to knock-down endogenous prohibitins expression in the GC cells. Interestingly, although the small hairpin RNAs (shRNAs) were specific for PHB and REA as evidenced from the lack of effect of each on the mRNA levels of the other gene (Figure 3A), the protein levels (Figure 3B) of both REA and PHB were reduced by either shPhb or shREA, respectively. This observation is consistent with previous reports indicating that these two proteins are in a complex and that their respective levels/stability are interdependent as was evident in HeLa cells, human T cells, breast tissue, yeast and C. elegans since depletion of either one decreases the protein levels of the other (8,19). We found that shRNA treated GCs changed their shape from polygonal to elongated, when compared to the control (Figure 3C) suggesting that shRNA-mediated interference of PHB and/or REA inhibit GC differentiation. Our experimental observation indicates that PHB up-regulation in GCs treated with FSH+T is required to sustain the differentiation state. Taken together, the above observations and our results suggest that PHB is a critical intracellular mediator in GC differentiation.
Figure 3.
PHB and REA form a complex in primary GCs treated with FSH+T and their stability is interdependent. A. GCs were infected with Ad-scrambled (control), Ad-shPHB-GFP or Ad-shREA-GFP vectors (MOI=10) for 48h and the media replaced with serum-free medium for 24h. Semiquantitative RT-PCR revealed that PHB and REA mRNAs expression levels were efficiently and significantly reduced when compared to the scrambled control group. B. Forty-eight hours after infections as above, GCs were treated with FSH+T for 24h. Cell lysates (30 micro gram) were subjected to 12% SDS-PAGE and Western blot analysis with antibodies directed toward PHB, REA, and cyclophilin A as indicated. C. Live cell photographs taken under an inverted microscope uncovered that down-regulation of PHB in the presence of FSH+T results in a change from a polygonal to elongated morphology of GCs indicative of proliferation. Results are representative of 3 independent experiments.
8. PROHIBITINS PHOSPHORYLATION IN CELL DIFFERENTAITION
Our original experimental observation in 1997 suggested that PHB is phosporylated in GCs under in vitro physiological conditions, however, further studies to determine how PHB phosphorylation is regulated in GC have not been performed until recently (2,8). When the respective protein fractions of mitochondria and nucleus are incubated with alkaline phosphatase, the acidic mitochondrial isoform (isoelectric point 5.6) completely disappears. These results suggest that PHB within the mitochondrial fraction is phosphorylated (2,6). Recently, our studies have shown that when GCs are cultured in presence of FSH plus T, PHB phosphorylation was not inhibited by phosphoinositide 3-kinases (PI3Ks) inhibitor (LY294002) and protein kinase A (PKA) inhibitor (H89) suggesting that PHB is not a substrate for PKB/AKT or PKA under these experimental conditions. Moreover, using the selective p38MAPK (SB203580) and MEK1 (PD98059) inhibitor revealed that PHB is a likely substrate for MEK1 and possibly for p38MAPK during GC differentiation. However, the identification of the site of phosphorylation on PHB and REA during GC differentiation has not been determined. PHB/Raf interactions have been reported to be essential for activation of the pathway (43). In support of this observation, we have consistently observed that, depletion of PHB had a negative impact on FSH-induced phosphorylation of ERK1/2 without changes in its protein expression levels. These studies show that, in addition to PHB being required for MEK1 activity, PHB is also a potential target of MEK1, suggesting that a possible novel regulatory loop is activated during GC differentiation mediated by PHB that affects the Ras-Raf-MEK1-ERK1/2 pathway. These novel findings indicate that there is a mutual hierarchical relationship between PHB and the Ras-Raf-MEK1-ERK1/2 pathway. Studies have shown that PHB plays an indispensable role in the activation of the Ras-Raf-MEK-ERK pathway (43). Direct interaction with PHB is required for c-Raf activation, moreover, c-Raf kinase fails to interact with the active Ras induced by epidermal growth factor (EGF) in the absence of PHB. These studies strongly support the notion that transcription and translation of prohibitins are hormonally dependent in the ovary. The hormone dependent expression of PHB has also been demonstrated in other non-reproductive tissues (44). Nonetheless, the physiological roles that PHB isoforms play in GCs, and their detailed and relative subcellular localization as well as differential association to potential partners remain unknown. For instance, it is possible that the acidic isoform of the protein could well be the active form responsible for up-regulation of MEK/ERK activity in a feed-forward loop that will ensure potentiation of the signal during differentiation.
9. ROLE OF PROHIBITINS IN CELL APOPTOSIS AND ATRESIA
Only a small number of the primordial follicles present at birth in the mammalian ovary will reach the ovulatory stage, while the rest will succumb to follicular atresia. Follicular atresia is mediated by apoptosis, which initially starts in the GCs layer (45,46), followed by apoptosis of the theca cells (47). Ovarian follicular atresia is induced by activation of both the extrinsic (death receptor) and intrinsic (mitochondrial) pathways in GCs (47). The intrinsic pathway is activated within the cell, and is characterized by the permeabilization of the outer mitochondrial membrane (OMM) resulting in the release of pro-apoptotic factors, such as cytochrome c, Smac, and Omi, to the cytosol and loss of mitochondrial function (48). Published studies have demonstrated that Caspase-3 content is very high in the atretic preovulatory follicular GCs (45,49). However, follicles isolated from caspase-3 null ovaries do not show GCs apoptosis in response to serum starvation, suggesting that caspase-3 is the effector caspase in this process, and its activation leads to the final stages of cellular death (49).
From our recent studies (6) it appears that the action of PHB in GCs is dependent on the stage of differentiation. For instance, infection of undifferentiated GCs isolated from preantral follicles with a Phb adenoviral construct resulted in over-expression of PHB that markedly attenuated ceramide-, staurosporine (STS)-, campothecine- and serum withdrawal-induced apoptosis via the intrinsic apoptotic pathway (6; our unpublished data). Furthermore, we confirmed that over-expression of PHB maintained the mitochondrial transmembrane potential by inhibiting cytochrome c release and activation of caspase-3 (6). Moreover, over-expression of PHB in undifferentaited GCs isolated from preantral follicle delayed accelerated apoptosis by enhancing the transcription and translation of anti-apoptotic genes (Bcl2, Bclxl) in STS induced GCs. In contrast, silencing of PHB expression by adenoviral small interfering RNA (shRNA) or inhibiting ERK phosphorylation through MEK, sensitized GCs to STS-induced apoptosis (6,36). These studies indicate that PHB acts as a survival factor by regulating anti-apoptotic gene expression in ovarian preantral GCs. However, whether similar mechanism(s) exist in preovulatory GCs need further investigation.
Additionally, prohibitins regulate the morphology of mitochondria during apoptosis. Abnormal mitochondria accumulate in the nematode Caenorhabditis elegans after RNA-interference (RNAi)-mediated depletion of prohibitins (34). A similar observation was made in prohibitin-deficient yeast cells (29,50,51). In mammalian cells RNAi-mediated knockdown of either PHB or REA/PHB2 expression in HeLa cells or GCs resulted in fragmentation of the mitochondrial network (52, Chowdhury et al., unpublished data). In addition, fragmented mitochondria were also observed in prohibitin-deficient mouse embryonic fibroblasts (MEFs), which suggests that fusion of mitochondrial membranes are impaired in the absence of prohibitins (53, Chowdhury et al., unpublished data). Furthermore, prohibitins are required for cristae morphogenesis, as revealed by an ultrastructural analysis of mitochondria in prohibitin-deficient MEFs (53). Disrupted cristae morphology might facilitate the release of cytochrome c from the intracristal space, thereby explaining the increased sensitivity of prohibitin-deficient GCs to apoptotic stimuli.
The aberrant mitochondrial morphology observed in the absence of prohibitins can be explained by an altered processing of OPA1 (53), a large dynamin-like GTPase found in the mitochondrial intermembrane space (IMS) that regulates both mitochondrial fusion and cristae morphogenesis (54). Mutations in OPA1 gene cause degeneration of retinal ganglion cells in autosomal dominant optic atrophy (55,56). Proteolytic processing of OPA1 splice variants, which are expressed in a tissue-specific manner (57,58), results in the accumulation of long and short OPA1 isoforms (58-60). Normal mitochondrial fusion depends on expression of both long and short OPA1 isoforms (59,61). Indeed, ectopic expression of a non-cleavable long OPA1 isoform was able to restore tubular mitochondrial network, cristae morphogenesis and apoptotic resistance in Phb2–/– MEFs (53). Similar results were found when PHB was ectopically expressed in STS treated GCs (Chowdhury et al., unpublished data). These experiments confirm that interactions between OPA1 and prohibitins are likely the key events that govern the stabilization of mitochondria in GCs. Yet, the exact mechanisms by which prohibitins affect OPA1 processing in GCs remain to be determined.
10. FUTURE DIRECTIONS
Folliculogenesis is essential for providing both oocytes and the steroid hormones necessary for female physical development and reproduction. Failure in the normal processes of folliculogenesis is the major reason for ovarian dysfunction and fertility impairment in women. Dysfuntional folliculogenesis translates into several clinically relevant outcomes, for instance, polycystic ovarian syndrome (PCOS), premature ovarian failure, or complications from ensuing hormonal imbalances. It is also related to normal ageing and complications of menopause. Furthermore, in the clinical setting, folliculogenesis is a target in fertility interventions (control of ovulation, treatment of anovulatory conditions and in vitro fertilization, IVF).
While the initial stages of folliculogenesis are independent of gonadotropic hormones, antral follicles become responsive to and then dependent on FSH. We identified PHB as a potential mediator of FSH stimulation of GCs, and therefore of normal folliculogenesis, probably acting as a “molecular switch” between proliferation and differentiation. Despite advances in the application of novel molecular techniques and approaches, our grasp of the molecular action of prohibitins and its complexes in reproduction is still poorly understood, and many questions still remain to be answered. Prohibitins exist as both phosphorylated (acidic) and unphosphorylated (basic) forms (2,6), and they can differentially localize to a number of subcellular compartments, thus alluding to the multifunctional nature of these proteins. Among the questions to be addressed by future research: Does the “phosphorylation state” result in conformational changes in the prohibitins? Does it determine their subcellular localization and/or association with partners, thus regulating their activity? Which are the pathways responsible for prohibitin phosphorylation in response to different stimuli?, Since analyses of the PHB and PHB2/REA protein sequences reveal the presence of multiple kinase consensus sequences, which kinases are responsible for prohibitins phosphorylation in response to different stimuli (6,8,62, Chowdhury et al., unpublished data)? Whether PHB and REA individually interact with different kinases other than MEK-ERK pathway under diverse physiological conditions and what is the functional consequence of these phenomenons in folliculogenesis still remains to be investigated (63). To further our current understanding of the role of PHB in follicle development, it will be necessary to identify the factors regulating PHB expression and/or function in the follicle and ovary. It appears that PHB plays various roles at different stages of follicle development. Moreover, it is important to fully elucidate the funtions of prohibitins in controlling the fate of the follicular cells (pro-vs anti-apoptotic), and thus the destiny of the follicle (growth, differentiation, or atresia), particularly in follicles which do not rely on gonadotropin stimulation.
On the structural level, further investigations are required to determine whether prohibitins form complexes in the inner mitochondrial membrane (IMM) and whether prohibitins complexes regulate proliferation and differentiation of GCs by interacting with other proteins. Are these interactions with mitochondrial hydrophilic/hydrophobic proteins or nuclear-encoded protein substrates? Are prohibitins antiproliferative, anti-estrogen receptor (ER) or anti- androgen receptor (AR) actions converging or independent effects? The availability of techniques to manipulate the expression of PHB and PHB2/REA genes by forced expression or silencing at specific stages of cellular growth, development and differentiation in diverse physiological environments or in pathological conditions, would permit more comprehensive studies aimed at defining the critical role that prohibitins play in folliculogenesis. Although some published data has emerged regarding the functional role of prohibitins in other cell types, little is currently known about their normal role in proliferation and differentiation of GCs. Further studies to elucidate the interactions that prohibitins have with other intracellular and extracellular factors will provide new insights and clarify the roles that prohibitins play in diverse cellular activities regulating folliculogenesis.
11. CONCLUSIONS
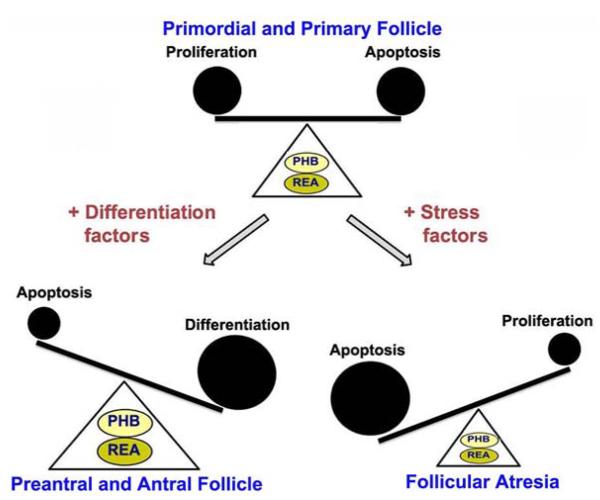
At present, we are just beginning to understand the critical roles that PHB and REA play in follicular development and regulation of ovarian function. However, a number of basic questions still remain to be answered, although current knowledge indicates that PHB is involved in regulating the fate of GCs, a fundamentally important finding that underscores the significance of elucidating prohibitin’s role in ovarian function. The studies on the prohibitins performed so far suggest that these gene products may function as a “molecular switch” that control cell fate and, in particular, that of the GCs and thereby determine the progress of follicular development in the ovary (Figure 4).
Figure 4.
Schematic representation of involvement of the prohibitins complex (PHB and REA/PHB2) in folliculogenesis in response to differentiation and stress inducing factors.
ACKNOWLEDGMENTS
This study was supported in part by National Institutes of Health Grants 1RO1HD057235, HD41749 and G12-RR03034. This investigation was conducted in a facility constructed with support from Research Facilities Improvement Grant #C06 RR18386 from NIH/NCRR.
13. REFERENCES
- 1.Craig J, Orisaka M, Wang H, Orisaka S, Thompson WE, Zhu C, Kotsuji F, Tsang BK. Gonadotropin and intra-ovarian signals regulating follicle development and atresia: the delicate balance between life and death. Front Biosci. 2007;12:3628–3639. doi: 10.2741/2339. [DOI] [PubMed] [Google Scholar]
- 2.Thompson WE, Sanbuissho A, Lee GY, Anderson E. Steroidogenic acute regulatory (StAR) protein (p25) and prohibitin (p28) from cultured rat ovarian granulosa cells. J Reprod Fertil. 1997;109:337–348. doi: 10.1530/jrf.0.1090337. [DOI] [PubMed] [Google Scholar]
- 3.Thompson WE, Powell JM, Whittaker JA, Sridaran R, Thomas KH. Immunolocalization and expression of prohibitin, a mitochondrial associated protein within the rat ovaries. Anat Rec. 1999;256:40–48. doi: 10.1002/(SICI)1097-0185(19990901)256:1<40::AID-AR6>3.0.CO;2-X. [DOI] [PubMed] [Google Scholar]
- 4.Thompson WE, Asselin E, Branch A, Stiles JK, Sutovsky P, Lai L, Im G-S, Prather RS, Isom SC, Rucker E, III, Tsang B. Regulation of prohibitin expression during follicular development and atresia in the mammalian ovary. Biol Reprod. 2004;71:282–290. doi: 10.1095/biolreprod.103.024125. [DOI] [PubMed] [Google Scholar]
- 5.Thompson WE, Branch A, Whittaker JA, Lyn D, Zilberstein M, Mayo KE, Thomas K. Characterization of prohibitin in a newly established rat ovarian granulosa cell line. Endocrinology. 2001;142:4076–4085. doi: 10.1210/endo.142.9.8354. [DOI] [PubMed] [Google Scholar]
- 6.Chowdhury I, Xu W, Stiles JK, Zeleznik A, Yao X, Matthews R, Thomas K, Thompson WE. Apoptosis of rat granulosa cells after staurosporine and serum withdrawal is suppressed by adenovirus directed overexpression of prohibitin. Endocrinology. 2006;148:206–217. doi: 10.1210/en.2006-0187. [DOI] [PubMed] [Google Scholar]
- 7.Nadimpalli R, Yalpani N, Johal GS, Simmons CR. Prohibitins, stomatins and plant desease response genes compose a superfamily that controls cell proliferation, ion channels regulation and death. J Biol Chem. 2000;275:29579–29586. doi: 10.1074/jbc.M002339200. [DOI] [PubMed] [Google Scholar]
- 8.Mishra S, Murphy LC, Murphy LJ. The Prohibitins: emerging roles in diverse functions. J Cell Mol Med. 2006;10:353–363. doi: 10.1111/j.1582-4934.2006.tb00404.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sato T, Saito H, Swensen J, Olifant A, Wood C, Danner D, Sakamoto T, Takita K, Kasumi F, Miki Y, Skolnick M, Nakamura Y. The human prohibitin gene located on chromosome 17q21 is mutated in sporadic breast cancer. Cancer Res. 1992;52:1643–1646. [PubMed] [Google Scholar]
- 10.Altus MS, Wood CM, Stewart DA, Roskams AJ, Friedman V, Henderson T, Owens GA, Danner DB, Jupe ER, Dell’Orco RT, Keith McClung J. Regions of evolutionary conservation between the rat and human prohibitin-encoding genes. Gene. 1995;158:291–294. doi: 10.1016/0378-1119(95)00164-2. [DOI] [PubMed] [Google Scholar]
- 11.He B, Feng Q, Mukherjee A, Lonard DM, DeMayo FJ, Katzenellenbogen BS, Lydon JP, O’Malley BW. A repressive role for prohibitin in estrogen signaling. Mol Endocrinol. 2008;22:344–60. doi: 10.1210/me.2007-0400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Terashima M, Kim KM, Adachi T, Nielsen PJ, Reth KG, Lamers MC. The IgM antigen receptor of B lymphocytes is associated with prohibitin and a prohibitin-related protein. EMBO J. 1994;13:3782–3792. doi: 10.1002/j.1460-2075.1994.tb06689.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ansari-Lari MA, Shen Y, Muzny DM, Lee W, Gibbs RA. Large-scale sequencing in human chromosome 12p13: experimental and computational gene structure determination. Genome Res. 1997;7:268–280. doi: 10.1101/gr.7.3.268. [DOI] [PubMed] [Google Scholar]
- 14.Steglich G, Neupert W, Langer T. Prohibitins regulate membrane protein degradation by the m-AAA protease in mitochondria. Mol Cell Biol. 1999;19:3435–42. doi: 10.1128/mcb.19.5.3435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nuell MJ, Stewart DA, Walker L, Friedman V, Wood CM, Owens GA, Smith JR, Schneider EL, Dell’ Orco R, Lumpkin CK, Danner DB, McClung JK. Prohibitin, an evolutionarily conserved intracellular protein that blocks DNA synthesis in normal fibroblasts and HeLa cells. Mol. Cell. Biol. 1991;11:1372–1381. doi: 10.1128/mcb.11.3.1372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Nijtmans LG, de Jong L, Artal Sanz M, Coates PJ, Berden JA, Back JW, Muijsers AO, van der Spek H, Grivell LA. Prohibitins act as a membrane-bound chaperone for the stabilization of mitochondrial proteins. EMBO J. 2000;19:2444–51. doi: 10.1093/emboj/19.11.2444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nijtmans LG, Artal SM, Grivell LA, Coates PJ. The mitochondrial PHB complex: roles in mitochondrial respiratory complex assembly, ageing and degenerative disease. Cell Mol Life Sci. 2002;59:143–155. doi: 10.1007/s00018-002-8411-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wang S, Zhang B, Faller DV. Prohibitin requires Brg-1 and Brm for the repression of E2F and cell growth. EMBO J. 2002;21:3019–3028. doi: 10.1093/emboj/cdf302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wang S, Nath N, Fusaro G, Chellappan S. Rb and prohibitin target distinct regions of E2F1 for repression and respond to different upstream signals. Mol Cell Biol. 1999;19:7447–7460. doi: 10.1128/mcb.19.11.7447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Montano MM, Ekena K, Delage-Mourroux R, Chang W, Martini P, Katzenellenbogen BS. An estrogen receptor-selective coregulator that potentiates the effectiveness of antiestrogens and represses the activity of estrogens. Proc Natl Acad Sci U S A. 1999;96:6947–52. doi: 10.1073/pnas.96.12.6947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kurtev V, Margueron R, Kroboth K, Ogris E, Cavailles V, Seiser C. Transcriptional regulation by the repressor of estrogen receptor activity via recruitment of histone deacetylases. J Biol Chem. 2004;279:24834–43. doi: 10.1074/jbc.M312300200. [DOI] [PubMed] [Google Scholar]
- 22.Martini PG, Delage-Mourroux R, Kraichely DM, Katzenellenbogen BS. Prothymosin alpha selectively enhances estrogen receptor transcriptional activity by interacting with a repressor of estrogen receptor activity. 2000;20:6224–6232. doi: 10.1128/mcb.20.17.6224-6232.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Narasimhan S, Armstrong M, McClung JK, Richards FF, Spicer EK. Prohibitin, a putative negative control element present in Pneumocystis carinii. Infect Immun. 1997;65:5125–30. doi: 10.1128/iai.65.12.5125-5130.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Edman JC, Sogin ML. Molecular phylogeny of Pneumocystis carinnii. In: Walzer PD, editor. Pneumocystis carinii Pneumonia. Marcel Dekker, Inc.; New York, NY: 1994. pp. 91–107. [Google Scholar]
- 25.de Monbrison F, Picot S. Introducing antisense oligonucleotides into Pneumocystis carinii. J Microbiol Methods. 2002;50:211–213. doi: 10.1016/s0167-7012(02)00033-7. [DOI] [PubMed] [Google Scholar]
- 26.Snedden WA, Fromm H. Characterization of the plant homologue of prohibitin, a gene associated with antiproliferative activity in mammalian cells. Plant Mol Biol. 1997;33:753–6. doi: 10.1023/a:1005737026289. [DOI] [PubMed] [Google Scholar]
- 27.Takahashi A, Kawasaki T, Wong HL, Suharsono U, Hirano H, Shimamoto K. Hyperphosphorylation of a mitochondrial protein, prohibitin, is induced by calyculin A in a rice lesion-mimic mutant cdr1. Plant Physiol. 2003;132:1861–9. doi: 10.1104/pp.103.021733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Welburn SC, Murphy NB. Prohibitin and RACK homologues are up-regulated in trypanosomes induced to undergo apoptosis and in naturally occurring terminally differentiated forms. Cell Death Differ. 1998;5:615–22. doi: 10.1038/sj.cdd.4400393. [DOI] [PubMed] [Google Scholar]
- 29.Berger KH, Yaffe MP. Prohibitin family members interact genetically with mitochondrial inheritance components in Saccharomyces cerevisiae. Mol Cell Biol. 1998;8:4043–52. doi: 10.1128/mcb.18.7.4043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kirchman PA, Miceli MV, West RL, Jiang JC, Kim S, Jazwinski SM. Prohibitins and Ras2 protein cooperate in the maintenance of mitochondrial function during yeast aging. Acta Biochim Pol. 2003;50:1039–56. [PubMed] [Google Scholar]
- 31.Tatsuta T, Model K, Langer T. Formation of membrane-bound ring complexes by prohibitins in mitochondria. Mol Biol Cell. 2005;16:248–59. doi: 10.1091/mbc.E04-09-0807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Eveleth DD, Jr, Marsh JL. Sequence and expression of the Cc gene, a member of the dopa decarboxylase gene cluster of Drosophila: Possible translational regulation. Nucleic Acids Res. 1986;14:6169–6183. doi: 10.1093/nar/14.15.6169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Park SE, Xu J, Frolova A, Liao L, O’Malley BW, Katzenellenbogen BS. Genetic deletion of the repressor of estrogen receptor activity (REA) enhances the response to estrogen in target tissues in vivo. Mol Cell Biol. 2005;25:1989–99. doi: 10.1128/MCB.25.5.1989-1999.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Artal-Sanz M, Tsang WY, Willems EM, Grivell LA, Lemire BD, van der Spek H, Nijtmans LG. The mitochondrial prohibitin complex is essential for embryonic viability and germline function in Caenorhabditis elegans. J Biol Chem. 2003;278(34):32091–32099. doi: 10.1074/jbc.M304877200. [DOI] [PubMed] [Google Scholar]
- 35.Hsueh AJW, Adashi EY, Jones PBC, Welsh TH., Jr. Hormonal regulation of the differentiation of cultured ovarian granulosa cells. Endocrine Reviews. 1984;5:76–110. doi: 10.1210/edrv-5-1-76. [DOI] [PubMed] [Google Scholar]
- 36.Gregory-Bass RC, Olatinwo M, Xu W, Matthews R, Stiles JK, Thomas K, Liu D, Tsang B, Thompson WE. Prohibitin silencing reverses stabilization of mitochondrial integrity and chemoresistance in ovarian cancer cells by increasing their sensitivity to apoptosis. Int J Cancer. 2008;122(9):1923–30. doi: 10.1002/ijc.23351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Choongkittaworn NM, Kim KH, Danner DB, Griswold MD. Expression of prohibitin in rat seminiferous epithelium. Biol Reprod. 1993;49:300–310. doi: 10.1095/biolreprod49.2.300. [DOI] [PubMed] [Google Scholar]
- 38.Bonnet E, Fostier A, Bobe J. Microarray-based analysis of fish egg quality after natural or controlled ovulation. BMC Genomics. 2007;8:55. doi: 10.1186/1471-2164-8-55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Bukowski R, Hankins GD, Saade GR, Anderson GD, Thornton S. Labor-associated gene expression in the human uterine fundus, lower segment, and cervix. PLoS Med. 2006;3(6):e169. doi: 10.1371/journal.pmed.0030169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Merkwirth C, Dargazanli S, Tatsuta T, Geimer S, Löwer B, Wunderlich FT, von Kleist-Retzow JC, Waisman A, Westermann B, Langer T. Prohibitins control cell proliferation and apoptosis by regulating OPA1- dependent cristae morphogenesis in mitochondria. Genes Dev. 2008;22(4):476–88. doi: 10.1101/gad.460708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Choi D, Lee SJ, Hong S, Kim IH, Kang S. Prohibitin interacts with RNF2 and regulates E2F1 function via dual pathways. Oncogene. 2008;27:1716–25. doi: 10.1038/sj.onc.1210806. [DOI] [PubMed] [Google Scholar]
- 42.Rikova K, Guo A, Zeng Q, Possemato A, Yu J, Haack H, Nardone J, Lee K, Reeves C, Li Y, Hu Y, Tan Z, Stokes M, Sullivan L, Mitchell J, Wetzel R, Macneill J, Ren JM, Yuan J, Bakalarski CE, Villen J, Kornhauser JM, Smith B, Li D, Zhou X, Gygi SP, Gu TL, Polakiewicz RD, Rush J, Comb MJ. Global survey of phosphotyrosine signaling identifies oncogenic kinases in lung cancer. Cell. 2007;131(6):1190–203. doi: 10.1016/j.cell.2007.11.025. [DOI] [PubMed] [Google Scholar]
- 43.Rajalingam K, Wunder C, Brinkmann V, Churin Y, Hekman M, Sievers C, Rapp UR, Rudel T. Prohibitin is required for Ras-induced Raf-MEK-ERK activation and epithelial cell migration. Nat Cell Biol. 2005;7:837–843. doi: 10.1038/ncb1283. [DOI] [PubMed] [Google Scholar]
- 44.Dixit VD, Sridaran R, Edmonsond MA, Taub D, Thompson WE. Gonadotropin releasing hormone attenuates pregnancy-associated thymic involution and modulates the expression of antiproliferative gene product prohibitin. Endocrinology. 2003;144:1496–1505. doi: 10.1210/en.2002-220955. [DOI] [PubMed] [Google Scholar]
- 45.Boone DL, Carnegie JA, Rippstein PU, Tsang BK. Induction of apoptosis in equine chorionic gonadotropin (eCG)-primed rat ovaries by anti-eCG antibody. Biol Reprod. 1997;57:420–427. doi: 10.1095/biolreprod57.2.420. [DOI] [PubMed] [Google Scholar]
- 46.Palumbo A, Yeh J. In situ localization of apoptosis in the rat ovary during follicular atresia. Biol Reprod. 1994;51:888–895. doi: 10.1095/biolreprod51.5.888. [DOI] [PubMed] [Google Scholar]
- 47.Tilly JL, Kowalski KI, Johnson AL, Hsueh AJ. Involvement of apoptosis in ovarian follicular atresia and postovulatory regression. Endocrinology. 1991;129:2799–2801. doi: 10.1210/endo-129-5-2799. [DOI] [PubMed] [Google Scholar]
- 48.Jiang JY, Cheung CK, Wang Y, Tsang BK. Regulation of cell death and cell survival gene expression during ovarian follicular development and atresia. Front Biosci. 2003;8:222–237. doi: 10.2741/949. [DOI] [PubMed] [Google Scholar]
- 49.Jin Z, El-Deiry WS. Overview of cell death signaling pathways. Cancer Biol Ther. 2005;4:139–163. doi: 10.4161/cbt.4.2.1508. [DOI] [PubMed] [Google Scholar]
- 50.Matikainen T, Perez GI, Zheng TS, Kluzak TR, Rueda BR, Flavell RA, Tilly JL. Caspase-3 Gene Knockout Defines Cell Lineage Specificity for Programmed Cell Death Signaling in the Ovary. Endocrinol. 2001;142:2468–2480. doi: 10.1210/endo.142.6.8078. [DOI] [PubMed] [Google Scholar]
- 51.Osman C, Merkwirth C, Langer T. Prohibitins and the functional compartmentalization of mitochondrial membranes. J Cell Sci. 2009;122(Pt 21):3823–3830. doi: 10.1242/jcs.037655. [DOI] [PubMed] [Google Scholar]
- 52.Kasashima K, Ohta E, Kagawa Y, Endo H. Mitochondrial Functions and Estrogen Receptor-dependent Nuclear Translocation of Pleiotropic Human Prohibitin. J Biol Chem. 2006;281:36401–36410. doi: 10.1074/jbc.M605260200. [DOI] [PubMed] [Google Scholar]
- 53.Merkwirth C, Dargazanli S, Tatsuta T, Geimer S, Löwer B, Wunderlich FT, von Kleist-Retzow JC, Waisman A, Westermann B, Langer T. Prohibitins control cell proliferation and apoptosis by regulating OPA1- dependent cristae morphogenesis in mitochondria. Genes Dev. 2008;22(4):476–88. doi: 10.1101/gad.460708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Hoppins S, Lackner L, Nunnari J. The machines that divide and fuse mitochondria. Annu Rev Biochem. 2007;76:751–780. doi: 10.1146/annurev.biochem.76.071905.090048. [DOI] [PubMed] [Google Scholar]
- 55.Alexander C, Votruba M, Pesch UE, Thiselton DL, Mayer S, Moore A, Rodriguez M, Kellner U, Leo-Kottler B, Auburger G, Bhattacharya SS, Wissinger B. OPA1, encoding a dynamin-related GTPase, is mutated in autosomal dominant optic atrophy linked to chromosome 3q28. Nat Genet. 2000;26(2):211–5. doi: 10.1038/79944. [DOI] [PubMed] [Google Scholar]
- 56.Delettre C, Lenaers G, Griffoin JM, Gigarel N, Lorenzo C, Belenguer P, Pelloquin L, Grosgeorge J, Turc-Carel C, Perret E, Astarie-Dequeker C, Lasquellec L, Arnaud B, Ducommun B, Kaplan J, Hamel CP. Nuclear gene OPA1, encoding a mitochondrial dynamin-related protein, is mutated in dominant optic atrophy. Nat Genet. 2000;26(2):207–10. doi: 10.1038/79936. [DOI] [PubMed] [Google Scholar]
- 57.Akepati VR, Müller EC, Otto A, Strauss HM, Portwich M, Alexander C. Characterization of OPA1 isoforms isolated from mouse tissues. J Neurochem. 2008;106(1):372–83. doi: 10.1111/j.1471-4159.2008.05401.x. [DOI] [PubMed] [Google Scholar]
- 58.Delettre C, Griffoin JM, Kaplan J, Dollfus H, Lorenz B, Faivre L, Lenaers G, Belenguer P, Hamel CP. Mutation spectrum and splicing variants in the OPA1 gene. Hum Genet. 2001;109(6):584–91. doi: 10.1007/s00439-001-0633-y. [DOI] [PubMed] [Google Scholar]
- 59.Ishihara N, Fujita Y, Oka T, Mihara K. Regulation of mitochondrial morphology through proteolytic cleavage of OPA1. EMBO J. 2006;25(13):2966–2977. doi: 10.1038/sj.emboj.7601184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Satoh M, Hamamoto T, Seo N, Kagawa Y, Endo H. Differential sublocalization of the dynamin-related protein OPA1 isoforms in mitochondria. Biochem Biophys Res Commun. 2003;300(2):482–493. doi: 10.1016/s0006-291x(02)02874-7. [DOI] [PubMed] [Google Scholar]
- 61.Song Z, Chen H, Fiket M, Alexander C, Chan DC. OPA1 processing controls mitochondrial fusion and is regulated by mRNA splicing, membrane potential, and Yme1L. J Cell Biol. 2007;178(5):749–755. doi: 10.1083/jcb.200704110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Sun L, Liu L, Yang XJ, Wu Z. Akt binds prohibitin 2 and relieves its repression of MyoD and muscle differentiation. J Cell Sci. 2004;117:3021–3029. doi: 10.1242/jcs.01142. [DOI] [PubMed] [Google Scholar]
- 63.Song G, Ouyang G, Bao S. The activation of Akt / PKB signaling pathway and cell survival. J Cell Mol Med. 2005;9:59–71. doi: 10.1111/j.1582-4934.2005.tb00337.x. [DOI] [PMC free article] [PubMed] [Google Scholar]