Abstract
Background
Bone marrow stimulation techniques in cartilage repair such as drilling are limited by the formation of fibrous to hyaline-like repair tissue. It has been suggested such techniques can be enhanced by covering the defect with scaffolds. We present an innovative approach using a polyglycolic acid (PGA)-hyaluronan scaffold with platelet-rich-plasma (PRP) in drilling.
Questions/purposes
We asked whether (1) PRP immersed in a cell-free PGA-hyaluronan scaffold improves patient-reported 1-year outcomes for the Knee injury and Osteoarthritis Score (KOOS), and (2) implantation of the scaffold in combination with bone marrow stimulation leads to the formation of hyaline-like cartilage repair tissue.
Patients and Methods
We reviewed 52 patients who had arthroscopic implantation of the PGA-hyaluronan scaffold immersed with PRP in articular cartilage defects of the knee pretreated with Pridie drilling. Patients were assessed by KOOS. At 9 months followup, histologic staining was performed in specimens obtained from five patients to assess the repair tissue quality.
Results
The KOOS subscores improved for pain (55 to 91), symptoms (57 to 88), activities of daily living (69 to 86), sports and recreation (36 to 70), and quality of life (38 to 73). The histologic evaluation showed a homogeneous hyaline-like cartilage repair tissue.
Conclusions
The cell-free PGA-hyaluronan scaffold combined with PRP leads to cartilage repair and improved patient-reported outcomes (KOOS) during 12 months of followup. Histologic sections showed morphologic features of hyaline-like repair tissue. Long-term followup is needed to determine if the cartilage repair tissue is durable.
Level of Evidence
Level IV, therapeutic study. See the Guidelines for Authors for a complete description of levels of evidence.
Introduction
Knee cartilage lesions are diagnosed with increasing frequency [1–3], perhaps owing to widespread use of arthroscopy. Chondral or osteochondral defects have been reported in 60% [13] to 67% [37] of patients in studies reporting knee arthroscopies. The choice of treatment usually is based on the size, depth, and location of the defect. Bone marrow-stimulating techniques such as Pridie drilling [26] or microfracturing [34] frequently are used as the first-line surgical procedure. The Pridie drilling technique is a minimally invasive procedure that induces a healing response by establishing access to the bone marrow in areas of cartilage defects. Access to the subchondral bone is established by multiple drill holes although these typically are deeper than the defects created by microfracturing [5]. Limitations of these techniques are poor repair tissue quality, ranging from hyaline to fibrous cartilage [33], and a decrease of clinical scores in patients who are 40 years old or younger at 18 to 36 months [18]. To overcome these limitations, advanced minimally invasive one-step techniques are needed that would enhance the patient-reported outcome scores and matrix components of the cartilage repair tissue [10]. Advanced one-step cartilage repair techniques combine bone-marrow stimulation with absorbable scaffolds. These scaffolds may allow for homogenous three-dimensional cell distribution, initial mechanical stability, and easy surgical handling. In addition, compared with matrix-assisted chondrocyte implantation, they avoid biopsy harvest, reduce donor-site morbidity, and allow for long-term storage and on-demand use.
A wide range of biomaterials has been proposed [2, 22, 37, 38]. Polymers like PGA-hyaluronan for in vivo cartilage tissue repair reportedly gave rise to hyaline cartilage in animal studies [7, 8] and in clinical applications [22, 38]. PRP has been used in trauma and orthopaedic surgery [1, 27, 30] because it is an autologous source of various growth factors [21]. Injection of PRP in 115 knees with chronic degenerative symptoms reduced pain and improved knee function and quality of life in younger patients as assessed by International Knee Documentation Committee (IKDC) score and visual analog scale (VAS) score at 1 and 2 years of followup [6, 17]. One study suggested the importance of keeping PRP-derived growth factors and cytokines in the wound site and slowly releasing them as the wound site becomes infiltrated with repair cells [9]. Adding calcium chloride to gel PRP apparently prolongs the growth-factor secretion of up to 7 days [9]. In an ovine model, PRP was used as a gel stimulating cartilage repair after microfracture [19]. In a study of 20 patients with osteochondral lesions, PRP combined with a hyaluronan scaffold induced cartilaginous tissue regeneration and improved functional scores (IKDC and KOOS) at 2 years followup [4]. For chondral patellar defects, a pilot study reported five patients who had a PRP gel used to fill microfractured cartilage defects covered with a porcine type I/III collagen membrane [6]. Patients improved in VAS pain score and KOOS, however, MRI showed three of the five patients had incomplete filling [6]. None of the patients had isointense signal intensities on MRI [6], suggesting poor quality of the repair tissue. Therefore, we propose covering the defect with a cell-free PGA-hyaluronan scaffold immersed in PRP gel to induce hyaline-like cartilage repair tissue and improvement in knee function.
To confirm its utility, we therefore asked whether (1) PRP immersed in a PGA-hyaluronan scaffold improves the patient-reported 1-year outcome in KOOS and (2) implantation of the scaffold in combination with bone marrow stimulation leads to formation of hyaline-like cartilage repair tissue.
Patients and Methods
In this prospective study, we reviewed 52 patients with 1-year followup who underwent arthroscopic implantations of a cell-free PGA-hyaluronan scaffold immersed with PRP to treat full-thickness articular cartilage knee defects from August 2007 to January 2009 (Table 1). Defects were classified according to the International Cartilage Repair Society (ICRS) score [3]. Indications for the cartilage repair technique were symptomatic, traumatic, and degenerative chondral defects (Outerbridge Grades III–IV) in synovial joints. Contraindications for the technique were the presence of metabolic, inflammatory, neoplastic, or immunologic diseases and complex knee instability; these conditions should be investigated and managed before any treatment with reparative cartilage repair techniques. For this study, we included 25- to 65-year-old patients with symptomatic full-thickness chondral loss in the weightbearing surface of the femur, tibia, patella, or trochlear groove. Patients with partial-thickness defects, misalignment (mechanical axis deviation greater than 2°), knee infection, knee instability, prior treatment of the knee, concomitant surgery, nocturnal pain, systematic immune-mediated diseases, and systematic disease-induced arthritis were excluded (Table 2). All patients were recalled specifically for this study at followups of 3, 6, 9, and 12 months, and all data were obtained from medical records and radiographs. No patients were lost to followup.
Table 1.
Characteristics of the patients
| Characteristic | Patient data |
|---|---|
| Sex | Female (n = 32), Male (n = 20) |
| Age (years) | 44.3 (range, 31–65) |
| Body mass index (kg/m2) | 24.0 (range, 19.1–31.2) |
| Defect size of lesion (cm2) | 2.75 (1.5–5) |
| Outerbridge classification of first lesion | III (n = 16) –IV (n = 36) |
| Localization of first lesion | Medial femoral condyle (n = 12) |
| Medial tibial condyle (n = 31) | |
| Lateral tibial condyle (n = 9) | |
| Concomitant surgery | None |
| Previous surgical procedures | None |
Table 2.
Physical examinations and exclusion criteria for patients
| Instability side | Test | Exclusion criteria |
|---|---|---|
| Anterior | Lachman | Side-to-side laxity |
| Anterior drawer | Side-to-side laxity | |
| Pivot shift | Grade 2 | |
| Posterior | Posterior drawer | Posterior translation greater than 6 mm |
| Rotator | Jerk | Positive |
| Varus-valgus | Stress 0°–30° | 5 mm |
Forty-seven of the 52 patients (90%) had progressive articular degeneration with focal degenerative cartilage defects and the defects were classified using preoperative radiographs according to a modified Kellgren-Lawrence grading system described by Pearson et al. [23]. Of the 47 patients, 21 had Grade I osteoarthritis, 15 had Grade II osteoarthritis, and 11 had Grade III osteoarthritis. None of the patients had any contraindication for the technique. Five of 52 patients had a posttraumatic lesion and underwent surgery within 6 months after the injury. In these five patients no associated meniscus lesions were assessed.
All arthroscopies were performed by one surgeon (AS). For arthroscopic surgery, a one-step procedure with a standard high anterolateral portal was used. Arthroscopy was performed with the patient in the supine position. The defects ranged from 15 mm2 to 50 mm2 (mean defect size, 27.5 mm2). We performed the bone marrow-stimulating procedure and implantation as follows (Fig. 1). We exposed the subchondral bone of the defect area, preserving the calcified cartilage layer by removing the damaged cartilage with a shaver. Afterward, we drilled perforations of approximately 2 cm in depth into the subchondral bone using a K-wire. The PGA-hyaluronan scaffold (chondrotissue®; BioTissue AG, Zurich, Switzerland) then was immersed in 3 mL autologous PRP for 5 to 10 minutes, which one of us (GM) prepared previously from the patient’s blood samples (not conditioned PRP; mean concentration, 832.1 × 102 platelets/μL; 6.1 × 102 leukocytes/μL), as described by Zimmerman et al. [39]. The chondrotissue® scaffold is an absorbable nonwoven felt of pure PGA combined with fermentative and freeze-dried hyaluronic acid (average molecular weight, 1200 kDa) and a porosity of approximately 70%. According to the manufacturer, within 7 days, the scaffold loses 50% of the mechanical stability in liquids and generally shows complete absorption in as many as 60 days. We cut the scaffold to fit the size of the defect and implanted the PRP-enriched PGA-hyaluronan scaffold into the defect. For secure fixation, we fixed the implant in the femoral defect (Fig. 1A) using Smart Nails® (ConMed Linvatec Italy, Milano, Italy), as described previously [38]. In tibial defects, we fixed the PGA-hyaluronan scaffold using autologous PRP gelled by calcium gluconate and thrombin additives to achieve an adhesive fibrin-like glue (Fig. 1B). First, we glued the PGA-hyaluronan scaffold into the defect rim and then covered the whole implant with the autologous PRP glue. Patients followed a postoperative rehabilitation protocol (Table 3).
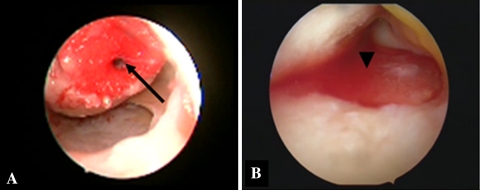
Fig. 1A–B.
The implanted PGA-hyaluronan scaffold is shown. (A) A PRP-immersed PGA-hyaluronan scaffold is placed into the femoral defect and fixed with a Smart Nail® (black arrow). (B) Fixation of the scaffold in the tibial defect is performed using fibrin-like autologous PRP glue. The PGA-hyaluronan scaffold was placed in the defect by an arthroscopic grasp and glued into the defect rim. Additionally the PGA-hyaluronan scaffold was covered with fibrin-like autologous PRP glue (black arrow).
Table 3.
Postoperative rehabilitation protocol
| Protocol | Weeks 0–2 | Week 3 | Weeks 4–6 | After 6 weeks |
|---|---|---|---|---|
| Mobilization/loading | Beginning 6 hours after surgery: active motion exercises without continuous passive motion: there is no expected pain after surgery, so the patient is requested to move the treated knee as much as possible | Foot sole contact with walking support/ braces at the beginning of the week up to free mobility at the end of the week | Free mobility | Free mobility |
| No load is permitted | Partial load at the beginning of the week, increase of loading to full body weight until the end of this week by patient’s own estimation | From Week 4 to Week 6, full body weight, complete load | Full body weight, complete load | |
| Walking, sports | No | Mobilization | Swimming, cycling to strengthen muscles, one day of rest after each sport day | Normal active life and daily activities resume work/ recreation |
| Physiotherapy | No | No | No | No |
One of us (AS) evaluated each patient clinically using the KOOS at 3, 6, 9, and 12 months after surgery [29]. The KOOS is divided into subcategories for pain, symptoms, activities of daily living, sports and recreation function, and knee-related quality of life. We scored the five subcategories of KOOS separately: pain (nine items); symptoms (seven items); activities of daily living (17 items); sports and recreation (five items), and quality of life (four items). We transformed scores to a zero- to 100-Likert scale, with zero representing extreme knee problems and 100 representing no knee problems. Each subcategory was calculated as the sum of all included items. Scores between zero and 100 represent the percentage of the total possible score of 100, with a score of 100 representing no knee problems [30].
All patients underwent MRI 6 months after surgery. All coronal and sagittal images were examined by one observer (DC) to assess defect filling, effusion, and bone marrow edema. No additional clinical grading was performed for any of the 52 patients. We examined 10 patients using second-look arthroscopy to assess repair tissue quality and tissue integration. These patients underwent a second surgery for a kissing lesion. In five of these 10 patients, we took repair tissue biopsy specimens at 9 months followup. Cartilage-bone cylinders of the newly formed cartilage repair tissue in the defect area (former chondrotissue® implant) of approximately 2.4 mm in diameter and 8.9 mm in length were obtained with a Jamshidi® needle (Nicolai GmbH, Langenhagen, Germany). One of us (CG) examined two slides from each specimen. The sections were stained (CG) with hematoxylin and eosin (H & E) (Sigma-Aldrich, St Louis, MO) to evaluate tissue morphology, cell distribution, and cartilage repair tissue integration, and with anti-Type II collagen monoclonal antibody to identify the presence of Type II collagen. Slides were stained with hematoxylin for 4 minutes and with eosin for 2 minutes to reveal morphologic features of the repair tissue and its integration. For the immunohistochemical analysis, sections were incubated with anti-collagen type II antibody (CIICI anti-COLII, DSHB University of Iowa, Iowa City, IA, USA) for 1 hour at room temperature. Afterward, sections were washed and incubated with biotinylated anti-mouse IgG, peroxidase-conjugated egg-white avidin, and 3-amino-9-ethylcarbazole substrate for 15 minutes (all from Jackson Laboratory Inc, West Grove, PA, USA). Staining was documented using a light microscope (Axiovert 10, Zeiss, Göttingen, Germany) equipped with a digital camera (Olympus DP10, Olympus Optical, Tokyo, Japan).
We tested for normal distribution of the KOOS using a Kolmogorov-Smirnov test. Owing to the nonparametric distribution of the data, we used the Friedman repeated measures analysis of variance by ranks to determine differences in preoperative and postoperative scores at 3, 6, 9, and 12 months. We performed pairwise multiple comparison procedures using the Student-Newman-Keuls method to determine differences in respective KOOS subcategories (pain, symptoms, activity of daily living, sports and recreation function, and knee-related quality of life) at each of the five times. All tests were performed using statistical software (SigmaStat 3.5, Statcon, Witzenhausen, Germany).
Results
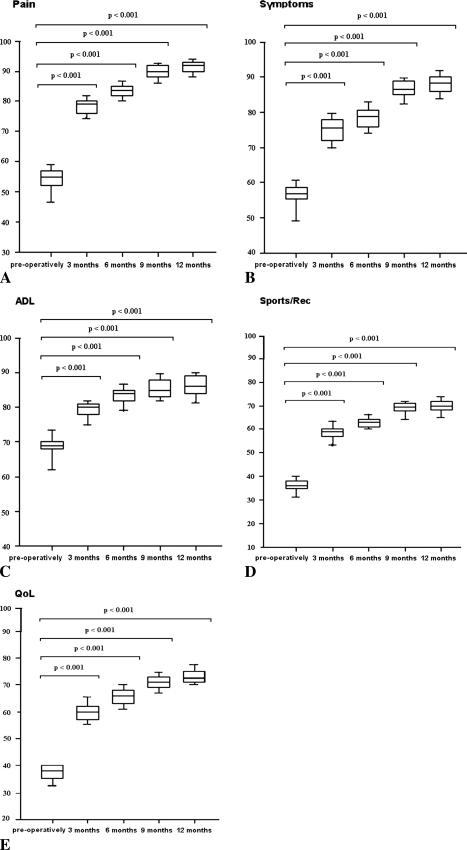
Patients who received PRP immersed in a cell-free PGA-hyaluronan scaffold had improved median KOOS as early as 3 months after surgery (p < 0.001) and at 6, 9, and 12 months (p < 0.001 each) postoperatively (Fig. 2). The median KOOS pain subcategory and symptom subcategory improved from preoperatively to the last followup: 55 to 91 (p < 0.001) and 57 to 88 (p < 0.001), respectively. Compared with the time before surgery, patients showed improvements in activities of daily living (69 to 86; p < 0.001), sports and recreation function (36 to 70; p < 0.001) and knee-related quality of life (38 to 73; p < 0.001) at 12 months postoperatively. All KOOS subcategories improved at 3 months postoperatively compared with the KOOS before surgery and this improvement increased continuously for up to 12 months.
Fig. 2A–E.
Clinical evaluation was performed using the KOOS. PRP immersed in a cell-free PGA-hyaluronan scaffold for treatment of full-thickness cartilage defects improved patients’ reported outcome (KOOS) as early as 3 months postoperatively and at 6, 9, and 12 months after surgery. KOOS scores are based on (A) pain, (B) symptoms, (C) activities of daily living (ADL); (D) sports/recreation; and (E) quality of life (QoL). A KOOS of 100 indicates no knee problems and a KOOS of zero indicates severe knee problems.
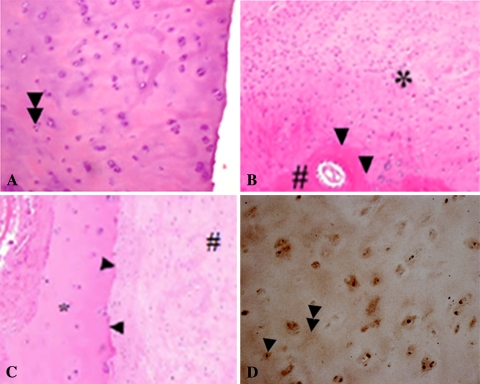
The repair tissue of the 10 patients examined by second-look arthroscopy 9 months after surgery seemed whiter than the normal surrounding cartilage and showed some small corrugations and an asymptomatic hypertrophy (Fig. 3). During palpation, the repair tissue appeared firmly attached to the bone and subjectively had a consistency similar to that of normal cartilage. Histologic evaluation of the five biopsy specimens showed homogeneous repair tissue with round chondrocytic cells (Fig. 4A), and good integration of the repair tissue to the underlying subchondral bone (Fig. 4B) and to the native adjacent cartilage (Fig. 4C). Immunohistochemistry of the central biopsy region showed intracellular formation of type II collagen (Fig. 4D) and cloudy type II collagen formation in the extracellular matrix of the repair tissue (Fig. 4D), indicating hyaline-like cartilage tissue. No residues of the PGA-hyaluronan scaffold were found, which indicated complete absorption of the biodegradable PGA fibers.
Fig. 3.
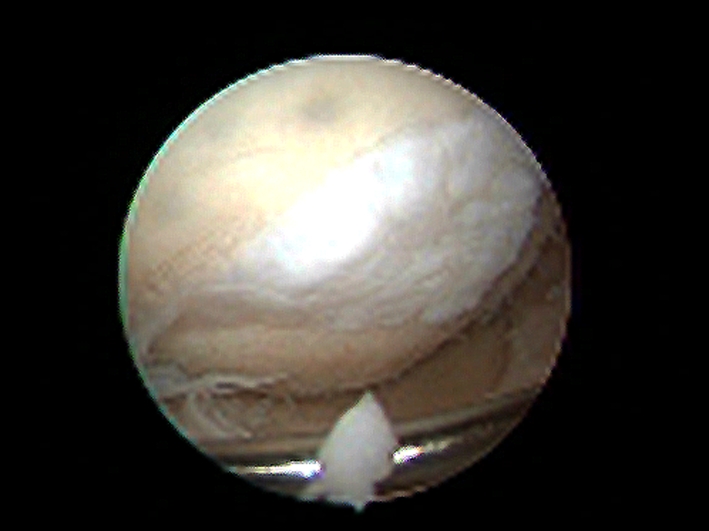
In a second-look arthroscopy of a femoral defect at the 9-month followup, the repair tissue appeared whiter than the normal surrounding cartilage and had some small corrugations and an asymptomatic hypertrophy. During palpation, it appeared firmly attached to the subchondral bone and subjectively had a tough consistency, similar to normal cartilage.
Fig. 4A–D.
Histologic and immunohistochemical evaluations of repair tissue biopsy specimens were performed 9 months after implantation of the PGA-hyaluronan scaffold immersed with PRP. (A) The central region of the specimen showed a repair tissue with multiple round chondrocytic cells and a single-cell distribution (double arrowheads) (Stain, hematoxylin & eosin; original magnification, ×200). (B) The cartilage-bone interface showed good bonding (arrowheads) of the repair tissue (asterisk) to the underlying subchondral bone (diamond) (Stain, hematoxylin & eosin; original magnification, ×200). (C) The cartilage repair tissue (diamond) adjacent to the native cartilage (asterisk) exhibited good integration of the repair tissue (Stain, hematoxylin & eosin; original magnification, ×200). (D) In the central biopsy region, immunohistochemical staining of type II collagen showed intracellular formation of type II collagen (single arrowhead) and cloudy type II collagen formation in the extracellular matrix of the repair tissue (double arrowheads) (Original magnification, ×200).
We observed effusion in 11 of the 52 patients at Day 7. The effusions were drained with no recurrence. During the year of followup we identified no patients with implant loosening, debonding, infections, inflammations, allergic reactions, thrombosis, or symptomatic hypertrophy. No patients required reoperation.
Discussion
Bone marrow-stimulating techniques in cartilage repair such as drilling and microfracture are limited by the formation of fibrous to hyaline-like repair tissue [33]. To enhance these techniques several authors have suggested covering defects with absorbable scaffolds [22, 38]. An advanced scaffold-based approach used a collagen membrane combined with PRP gel to cover microfractured chondral cartilage defects [6]. Despite patient-reported improvement in VAS pain and KOOS, none of the patients showed isointense signal intensities on MRI at 24 months followup [6], suggesting the formation of poor-quality cartilage repair tissue. In this study, we present an innovative approach using a cell-free PGA-hyaluronan scaffold immersed with PRP gel to investigate whether (1) PRP immersed in a PGA-hyaluronan scaffold improves patient-reported 1-year outcome in KOOS and (2) implantation of the scaffold in combination with bone marrow stimulation leads to the formation of hyaline-like cartilage repair tissue.
Our prospective study has some limitations. First, it is an uncontrolled single-cohort observational case report study of 52 patients with focus on evaluation of clinical scores and potential complications. Thus we did not have a control group. Second, the KOOS data are limited to those obtained during a 12-month clinical followup, therefore lacking long-term evaluation. Therefore, we cannot say whether these implants will be durable. Third, the histologic evaluation of the repair tissue is limited to specimens obtained from five patients; we did not use MRI to evaluate defect filling or PCR to analyze repair tissue quality. Durability of the repaired cartilage and the potential to reduce subsequent osteoarthritis are unknown. However long-term, controlled studies addressing optimal defect size, age of patients, need for a PRP control group, inclusion of MRI (eg, 3-T delayed gadolinium-enhanced MRI) [25], and PCR analysis are suggested to further evaluate this promising treatment method. Finally, we did not examine the cost effectiveness of this approach. The implant costs are on average 2727/3409 US dollars depending on the defect size and fixation technique. Thus, the costs for the implant are approximately 70% lower than for an autologous chondrocyte implantation (ACI).
We used representative studies from the literature for our comparisons and synthesis (Table 4). Injection of PRP in knees with chronic degenerative symptoms reportedly reduced pain and improved knee function as assessed by VAS pain and IKDC scores in younger patients at 1 year followup [16]. Two randomized controlled studies have investigated whether cartilage regeneration after chondrocyte implantation is superior to microfracture treatment for cartilage defects of the knee [31, 32]. In the second study, at 36 months chondrocyte implantation was associated with greater improvement in the KOOS compared with the microfracture group: 21.25 versus 15.83, respectively [31]. We found similar improvements in KOOS (31) at 12 months followup compared with that reported by Saris et al. [31, 32]. Additional studies using one-step cartilage repair approaches showed improvements in KOOS of 46 (mean) at 24 months followup using a hyaluronan scaffold [4] and 28 at 2 years followup for a collagen type I/III membrane [6]. These findings are similar to those of our study (31) in which the patients improved in KOOS as early as 3 months after surgery and up to the 12 months followup, suggesting that our results are equivalent.
Table 4.
Literature comparison
| Clinical evaluation | Quality of cartilage repair tissue | Safety evaluation | ||||||
|---|---|---|---|---|---|---|---|---|
| Study | Followup | Findings | Study | Followup | Findings | Study | Followup | Findings |
| Saris et al. [31] | 18 months | Overall KOOS for ACI group (Δ KOOS = 15.5) and for MFX group (Δ KOOS = 18.4) comparable | Knutsen et al. [15] | 24 months | Predominantly hyaline-like cartilage repair tissue formation after MFX and ACI in 80 patients | Henderson et al. [12] | 3 months | Knee effusion rate of 5% after ACI for treatment of focal chondral defects of the knee |
| Saris et al. [30] | 36 months | Overall KOOS greater for ACI group (Δ KOOS = 21.25) than for MFX goup (Δ KOOS = 15.83) compared with preoperative | Erggelet et al. [8] | 3 months | PGA-hyaluronan scaffold immersed with autologous serum showed hyaline-like Type II collagen positive repair tissue in contrast to MFX control group in ovine model | Rodrigo et al. [27] | Postoperative | Knee effusion rate of 31% after using bone marrow-stimulating techniques in full-thickness knee defects |
| Kon et al. [16] | 12 months | PRP intraarticular injection in chronic degenerative condition of the knee (115 knees): pain reduction and improvement in knee function and quality of life in younger patients | Erggelet et al. [7] | 6 months | PGA-hyaluronan scaffold immersed with autologous serum showed hyaline-like Type II collagen positive repair tissue in contrast to MFX control group in ovine model | Tatari et al. [34] | 4–15 days | Drilling the osteochondral bone led to higher amounts of fluid and bleeding compared with nondrilling |
| Dhollander et al. [6] | 48 months | Type I/III collagen membrane with PRP gel: overall KOOS improved in 5 patients | Zantop & Petersen [37] | 12 months | PGA-hyaluronan scaffold immersed with autologous serum showed good defect filling | Saris et al. [30] | 36 months | Higher failure rates for MFX (7 = 11.5%) than for ACI (2 = 3.9%) |
| Buda et al. [4] | 24 months | Hyaluronan scaffold with PRP: improved condition as assessed by the International Knee Dokumentation Committee score and KOOS in 20 patients | Patrascu et al. [22] | 24 months | PGA-hyaluronan scaffold showed the formation of hyaline-like cartilage and patient’s improvement | Knutsen et al. [14] | 5 years | Same failure rates in ACI and MFX groups (9 failures = 23%) |
| Milano et al. [19] | 6 months | Ovine model: better histologic total scores for MFX used with PRP than for MFX control group | Peterson et al. [24] | 2–9 years | Failure rates of 5% for scaffold-based implants | |||
| Buda et al. [4] | 12 months | Hyaluronan scaffold with PRP: cartilage repair tissue biopsy showed proteoglycan-rich matrix and positivity for Type II collagen | Minas [20] | 24 months | Failure rates of 13% and revision surgery rate of 25% for scaffold-based implants | |||
| Current study | 12 months | PGA-hyaluronan scaffold immersed with PRP: overall KOOS improved compared with preoperative in 52 patients (Δ KOOS = 31) | Current study | 9 months | PGA-hyaluronan scaffold immersed with PRP: formation of hyaline-like cartilage repair tissue with Type II collagen positivity | Current study | 12 months | No failure rate and no reoperations |
| At Day 7 | Knee effusion in 11 of 52 patients with no recurrence after drainage | |||||||
KOOS: Knee injury and Osteoarthritis Score, ACI: Autologous Chondrocyte Implantation, MFX: microfracture, PRP: platelet-rich plasma, PGA: polyglycolic acid.
Hyaline-like cartilage is assumed to be more durable and smooth than fibrocartilage repair tissue [11]. In a previous study reporting 67 patients with 2 years followup comparing ACI with microfracture, none of the 55 of 67 patients with predominantly hyaline-like cartilage repair tissue had later failure [15]. Preclinical studies, covering microfractured ovine cartilage defects with a PGA-hyaluronan scaffold immersed with autologous serum, showed hyaline-like repair tissue formation in contrast to inferior fibrocartilage formation in a noncovered microfracture control group [7, 8]. Clinical case report studies suggest the use of this technique for cartilage repair resulted in defect filling, formation of hyaline-like cartilage, and improvement in patients′ pain and knee function at the 1- and 2-year followups [22, 38]. Previous scaffold application with a PRP gel showed better histologic total scores than for the microfracture-only group [19] and resulted in proteoglycan-enriched and type II collagen-positive repair tissue [4]. Our histologic findings at the 9-month followup are in line with those of previous studies [4, 7, 8] showing the formation of hyaline-like cartilage repair tissue with type II collagen.
We found knee effusions in 20% of the patients with no recurrence after drainage; presumably this is induced by Pridie drilling. Our rate is similar to those reported for other cartilage repair techniques such as microfracture and ACI, which show effusions from 5% to 31% [12, 28]. Tatari et al. found drilling of the osteochondral surface led to higher amounts of fluid and bleeding owing to the penetration of the subchondral bone in comparison to nondrilling [35]. They recommended suction drainage to prevent subsequent effusions in these arthroscopic procedures [35]. All of our patients had improvement at 12 months with no overt failures. Failure rates in cartilage and/or cartilage repair techniques are variable. Higher failure rates have been reported for microfracture (11.5%) than for ACI (3.9%) [31]. One long-term 5-year followup study documented a similar failure rate of 23% in patients having ACI and microfracture [14]. For ACI, previous studies described failure rates between 5% [24] and 13% [20] and the necessity for revision surgery of 25% [20]. In relation to these findings, we presume the use of PGA-hyaluronan scaffolds immersed with PRP are associated with low complication rates and allows durable repair.
Our observations suggest an arthroscopically applied PGA-hyaluronan scaffold immersed with PRP after bone marrow stimulation resulted in early improvement of the KOOS for patients and hyaline-like cartilage repair tissue in five patients who had the biopsy. Thus, short-term implantation of a PGA-hyaluronan scaffold in combination with PRP appears to be a reasonable one-step cartilage repair procedure. Long-term followup will be required to determine the durability of the repair tissue.
Acknowledgments
We thank D. Confalone (DC) MD for interpreting the MRIs to assess defect filling, effusion, and bone marrow edema.
Footnotes
Each author certifies that he or she has no commercial associations that might pose a conflict of interest in connection with the submitted article.
All ICMJE Conflict of Interest Forms for authors and Clinical Orthopedics and Related Research editors and board members are on file with the publication and can be reviewed upon request.
Each author certifies that his or her institution approved the human protocol for this investigation that all investigations were conducted in conformity with ethical principles of research and that informed consent for participation in the study was obtained.
This work was performed at Ospedale degli Infermi di Biella ASLBI.
References
- 1.Alsousou J, Thompson M, Hulley P, Noble A, Willett K. The biology of platelet-rich plasma and its application in trauma and orthopaedic surgery: a review of the literature. J Bone Joint Surg Br. 2009;91:987–996. doi: 10.1302/0301-620X.91B8.22546. [DOI] [PubMed] [Google Scholar]
- 2.Benthien JP, Behrens P. Autologous matrix-induced chondrogenesis (AMIC): a one-step procedure for retropatellar articular resurfacing. Acta Orthop Belg. 2010;76:260–263. [PubMed] [Google Scholar]
- 3.Brittberg M, Winalski CS. Evaluation of cartilage injuries and repair. J Bone Joint Surg Am. 2003;85(suppl 2):58–69. doi: 10.2106/00004623-200300002-00008. [DOI] [PubMed] [Google Scholar]
- 4.Buda R, Vannini F, Cavallo M, Grigolo B, Cenacchi A, Giannini S. Osteochondral lesions of the knee: a new one-step repair technique with bone-marrow-derived cells. J Bone Joint Surg Am. 2010;92(suppl 2):2–11. doi: 10.2106/JBJS.J.00813. [DOI] [PubMed] [Google Scholar]
- 5.Chen H, Hoemann CD, Sun J, Chevrier A, McKee MD, Shive MS, Hurtig M, Buschmann MD. Depth of subchondral perforation influences the outcome of bone marrow stimulation cartilage repair. J Orthop Res. 2011;29:1178–1184. doi: 10.1002/jor.21386. [DOI] [PubMed] [Google Scholar]
- 6.Dhollander AA, Neve F, Almqvist KF, Verdonk R, Lambrecht S, Elewaut D, Verbruggen G, Verdonk PC. Autologous matrix-induced chondrogenesis combined with platelet-rich plasma gel: technical description and a five pilot patients report. Knee Surg Sports Traumatol Arthrosc. 2011;19:536–542. doi: 10.1007/s00167-010-1337-4. [DOI] [PubMed] [Google Scholar]
- 7.Erggelet C, Endres M, Neumann K, Morawietz L, Ringe J, Haberstroh K, Sittinger M, Kaps C. Formation of cartilage repair tissue in articular cartilage defects pretreated with microfracture and covered with cell-free polymer-based implants. J Orthop Res. 2009;27:1353–1360. doi: 10.1002/jor.20879. [DOI] [PubMed] [Google Scholar]
- 8.Erggelet C, Neumann K, Endres M, Haberstroh K, Sittinger M, Kaps C. Regeneration of ovine articular cartilage defects by cell-free polymer-based implants. Biomaterials. 2007;28:5570–5580. doi: 10.1016/j.biomaterials.2007.09.005. [DOI] [PubMed] [Google Scholar]
- 9.Foster TE, Puskas BL, Mandelbaum BR, Gerhardt MB, Rodeo SA. Platelet-rich plasma: From basic science to clinical applications. Am J Sports Med. 2009;37:2259–2272. doi: 10.1177/0363546509349921. [DOI] [PubMed] [Google Scholar]
- 10.Frisbie DD, Oxford JT, Southwood L, Trotter GW, Rodkey WG, Steadman JR, Goodnight JL, McIlwraith CW. Early events in cartilage repair after subchondral bone microfracture. Clin Orthop Relat Res. 2003;407:215–227. doi: 10.1097/00003086-200302000-00031. [DOI] [PubMed] [Google Scholar]
- 11.Hayes DW, Jr, Brower RL, John KJ. Articular cartilage: anatomy, injury, and repair. Clin Podiatr Med Surg. 2001;18:35–53. [PubMed] [Google Scholar]
- 12.Henderson I, Francisco R, Oakes B, Cameron J. Autologous chondrocyte implantation for treatment of focal chondral defects of the knee: a clinical, arthroscopic, MRI and histologic evaluation at 2 years. Knee. 2005;12:209–216. doi: 10.1016/j.knee.2004.07.002. [DOI] [PubMed] [Google Scholar]
- 13.Hjelle K, Solheim E, Strand T, Muri R, Brittberg M. Articular cartilage defects in 1, 000 knee arthroscopies. Arthroscopy. 2002;18:730–734. doi: 10.1053/jars.2002.32839. [DOI] [PubMed] [Google Scholar]
- 14.Knutsen G, Drogset JO, Engebretsen L, Grontvedt T, Isaksen V, Ludvigsen TC, Roberts S, Solheim E, Strand T, Johansen O. A randomized trial comparing autologous chondrocyte implantation with microfracture: findings at five years. J Bone Joint Surg Am. 2007;89:2105–2112. doi: 10.2106/JBJS.G.00003. [DOI] [PubMed] [Google Scholar]
- 15.Knutsen G, Engebretsen L, Ludvigsen TC, Drogset JO, Grontvedt T, Solheim E, Strand T, Roberts S, Isaksen V, Johansen O. Autologous chondrocyte implantation compared with microfracture in the knee: a randomized trial. J Bone Joint Surg Am. 2004;86:455–464. doi: 10.2106/00004623-200403000-00001. [DOI] [PubMed] [Google Scholar]
- 16.Kon E, Buda R, Filardo G, Di Martino A, Timoncini A, Cenacchi A, Fornasari PM, Giannini S, Marcacci M. Platelet-rich plasma: intra-articular knee injections produced favorable results on degenerative cartilage lesions. Knee Surg Sports Traumatol Arthrosc. 2010;18:472–479. doi: 10.1007/s00167-009-0940-8. [DOI] [PubMed] [Google Scholar]
- 17.Kon E, Gobbi A, Filardo G, Delcogliano M, Zaffagnini S, Marcacci M. Arthroscopic second-generation autologous chondrocyte implantation compared with microfracture for chondral lesions of the knee: prospective nonrandomized study at 5 years. Am J Sports Med. 2009;37:33–41. doi: 10.1177/0363546508323256. [DOI] [PubMed] [Google Scholar]
- 18.Kreuz PC, Erggelet C, Steinwachs MR, Krause SJ, Lahm A, Niemeyer P, Ghanem N, Uhl M, Sudkamp N. Is microfracture of chondral defects in the knee associated with different results in patients aged 40 years or younger? Arthroscopy. 2006;22:1180–1186. doi: 10.1016/j.arthro.2006.06.020. [DOI] [PubMed] [Google Scholar]
- 19.Milano G, Sanna Passino E, Deriu L, Careddu G, Manunta L, Manunta A, Saccomanno MF, Fabbriciani C. The effect of platelet rich plasma combined with microfractures on the treatment of chondral defects: an experimental study in a sheep model. Osteoarthritis Cartilage. 2010;18:971–980. doi: 10.1016/j.joca.2010.03.013. [DOI] [PubMed] [Google Scholar]
- 20.Minas T. Autologous chondrocyte implantation for focal chondral defects of the knee. Clin Orthop Relat Res. 2001;391(suppl):S349–S361. doi: 10.1097/00003086-200110001-00032. [DOI] [PubMed] [Google Scholar]
- 21.Nikolidakis D, Jansen JA. The biology of platelet-rich plasma and its application in oral surgery: literature review. Tissue Eng Part B Rev. 2008;14:249–258. doi: 10.1089/ten.teb.2008.0062. [DOI] [PubMed] [Google Scholar]
- 22.Patrascu JM, Freymann U, Kaps C, Poenaru DV. Repair of a post-traumatic cartilage defect with a cell-free polymer-based cartilage implant: a follow-up at two years by MRI and histological review. J Bone Joint Surg Br. 2010;92:1160–1163. doi: 10.1302/0301-620X.92B8.24341. [DOI] [PubMed] [Google Scholar]
- 23.Pearson RG, Kurien T, Shu KS, Scammell BE. Histopathology grading systems for characterisation of human knee osteoarthritis: reproducibility, variability, reliability, correlation, and validity. Osteoarthritis Cartilage. 2011;19:324–331. doi: 10.1016/j.joca.2010.12.005. [DOI] [PubMed] [Google Scholar]
- 24.Peterson L, Minas T, Brittberg M, Nilsson A, Sjogren-Jansson E, Lindahl A. Two- to 9-year outcome after autologous chondrocyte transplantation of the knee. Clin Orthop Relat Res. 2000;374:212–234. doi: 10.1097/00003086-200005000-00020. [DOI] [PubMed] [Google Scholar]
- 25.Pinker K, Szomolanyi P, Welsch GC, Mamisch TC, Marlovits S, Stadlbauer A, Trattnig S. Longitudinal evaluation of cartilage composition of matrix-associated autologous chondrocyte transplants with 3-T delayed gadolinium-enhanced MRI of cartilage. AJR Am J Roentgenol. 2008;191:1391–1396. doi: 10.2214/AJR.07.3930. [DOI] [PubMed] [Google Scholar]
- 26.Schmidt H, Hasse E. Arthroscopic surgical treatment of circumscribed cartilage damage with spongiolization or Pridie drilling. Beitr Orthop Traumatol. 1989;36:35–37. [PubMed]
- 27.Qi YY, Chen X, Jiang YZ, Cai HX, Wang LL, Song XH, Zou XH, Ouyang HW. Local delivery of autologous platelet in collagen matrix simulated in situ articular cartilage repair. Cell Transplant. 2009;18:1161–1169. doi: 10.3727/096368909X12483162197169. [DOI] [PubMed] [Google Scholar]
- 28.Rodrigo JJ, Steadman JR, Silliman JF, Fulstone HA. Improvement of full-thickness chondral defect healing in the human knee after debridement and microfracture using continuous passive motion. Am J Knee Surg. 1994;7:109–116. [Google Scholar]
- 29.Roos EM, Roos HP, Lohmander LS, Ekdahl C, Beynnon BD. Knee Injury and Osteoarthritis Outcome Score (KOOS): development of a self-administered outcome measure. J Orthop Sports Phys Ther. 1998;28:88–96. doi: 10.2519/jospt.1998.28.2.88. [DOI] [PubMed] [Google Scholar]
- 30.Saito M, Takahashi KA, Arai Y, Inoue A, Sakao K, Tonomura H, Honjo K, Nakagawa S, Inoue H, Tabata Y, Kubo T. Intraarticular administration of platelet-rich plasma with biodegradable gelatin hydrogel microspheres prevents osteoarthritis progression in the rabbit knee. Clin Exp Rheumatol. 2009;27:201–207. [PubMed] [Google Scholar]
- 31.Saris DB, Vanlauwe J, Victor J, Almqvist KF, Verdonk R, Bellemans J, Luyten FP, TIG/ACT/01/2000&EXT Study Group Treatment of symptomatic cartilage defects of the knee: characterized chondrocyte implantation results in better clinical outcome at 36 months in a randomized trial compared to microfracture. Am J Sports Med. 2009;37(suppl 1):10S–19S. doi: 10.1177/0363546509350694. [DOI] [PubMed] [Google Scholar]
- 32.Saris DB, Vanlauwe J, Victor J, Haspl M, Bohnsack M, Fortems Y, Vandekerckhove B, Almqvist KF, Claes T, Handelberg F, Lagae K, Bauwhede J, Vandenneucker H, Yang KG, Jelic M, Verdonk R, Veulemans N, Bellemans J, Luyten FP. Characterized chondrocyte implantation results in better structural repair when treating symptomatic cartilage defects of the knee in a randomized controlled trial versus microfracture. Am J Sports Med. 2008;36:235–246. doi: 10.1177/0363546507311095. [DOI] [PubMed] [Google Scholar]
- 33.Steadman JR, Briggs KK, Rodrigo JJ, Kocher MS, Gill TJ, Rodkey WG. Outcomes of microfracture for traumatic chondral defects of the knee: average 11-year follow-up. Arthroscopy. 2003;19:477–484. doi: 10.1053/jars.2003.50112. [DOI] [PubMed] [Google Scholar]
- 34.Steadman JR, Rodkey WG, Rodrigo JJ. Microfracture: surgical technique and rehabilitation to treat chondral defects. Clin Orthop Relat Res. 2001;391(suppl):S362–S369. doi: 10.1097/00003086-200110001-00033. [DOI] [PubMed] [Google Scholar]
- 35.Tatari H, Dervisbey M, Muratli K, Ergor A. Report of experience in 190 patients with the use of closed suction drainage in arthroscopic knee procedures. Knee Surg Sports Traumatol Arthrosc. 2005;13:458–462. doi: 10.1007/s00167-004-0537-1. [DOI] [PubMed] [Google Scholar]
- 36.Widuchowski W, Widuchowski J, Trzaska T. Articular cartilage defects: study of 25, 124 knee arthroscopies. Knee. 2007;14:177–182. doi: 10.1016/j.knee.2007.02.001. [DOI] [PubMed] [Google Scholar]
- 37.Williams CG, Kim TK, Taboas A, Malik A, Manson P, Elisseeff J. In vitro chondrogenesis of bone marrow-derived mesenchymal stem cells in a photopolymerizing hydrogel. Tissue Eng. 2003;9:679–688. doi: 10.1089/107632703768247377. [DOI] [PubMed] [Google Scholar]
- 38.Zantop T, Petersen W. Arthroscopic implantation of a matrix to cover large chondral defect during microfracture. Arthroscopy. 2009;25:1354–1360. doi: 10.1016/j.arthro.2009.04.077. [DOI] [PubMed] [Google Scholar]
- 39.Zimmermann R, Reske S, Metzler P, Schlegel A, Ringwald J, Eckstein R. Preparation of highly concentrated and white cell-poor platelet-rich plasma by plateletpheresis. Vox Sang. 2008;95:20–25. doi: 10.1111/j.1423-0410.2008.01062.x. [DOI] [PubMed] [Google Scholar]